Effect of High-Dose Vitamin D3 Intake on Ambulation, Muscular Pain and Bone Mineral Density in a Woman with Multiple Sclerosis: A 10-Year Longitudinal Case Report
Abstract
:1. Introduction
1.1. Endpoints
1.2. Biographical Data
2. Results
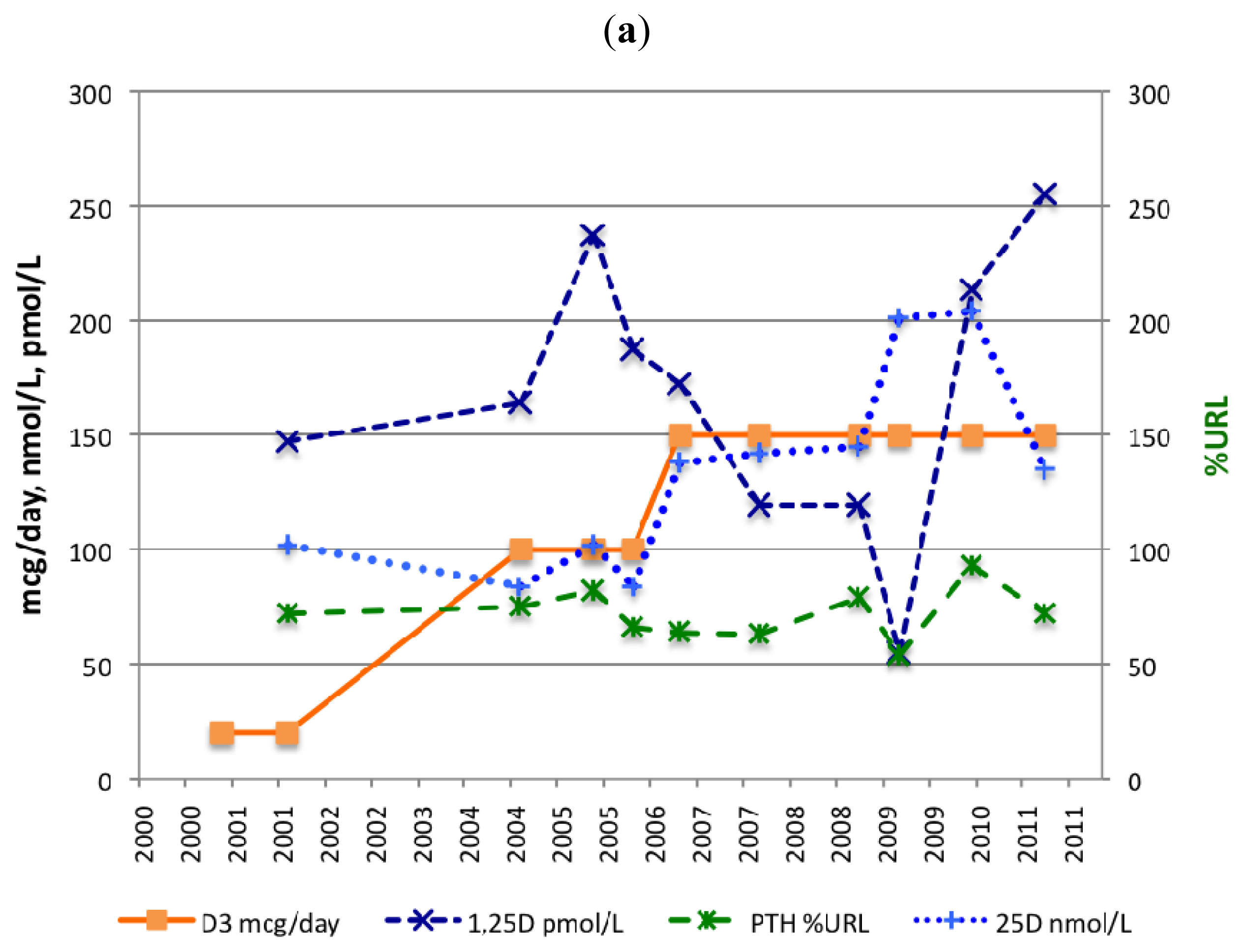
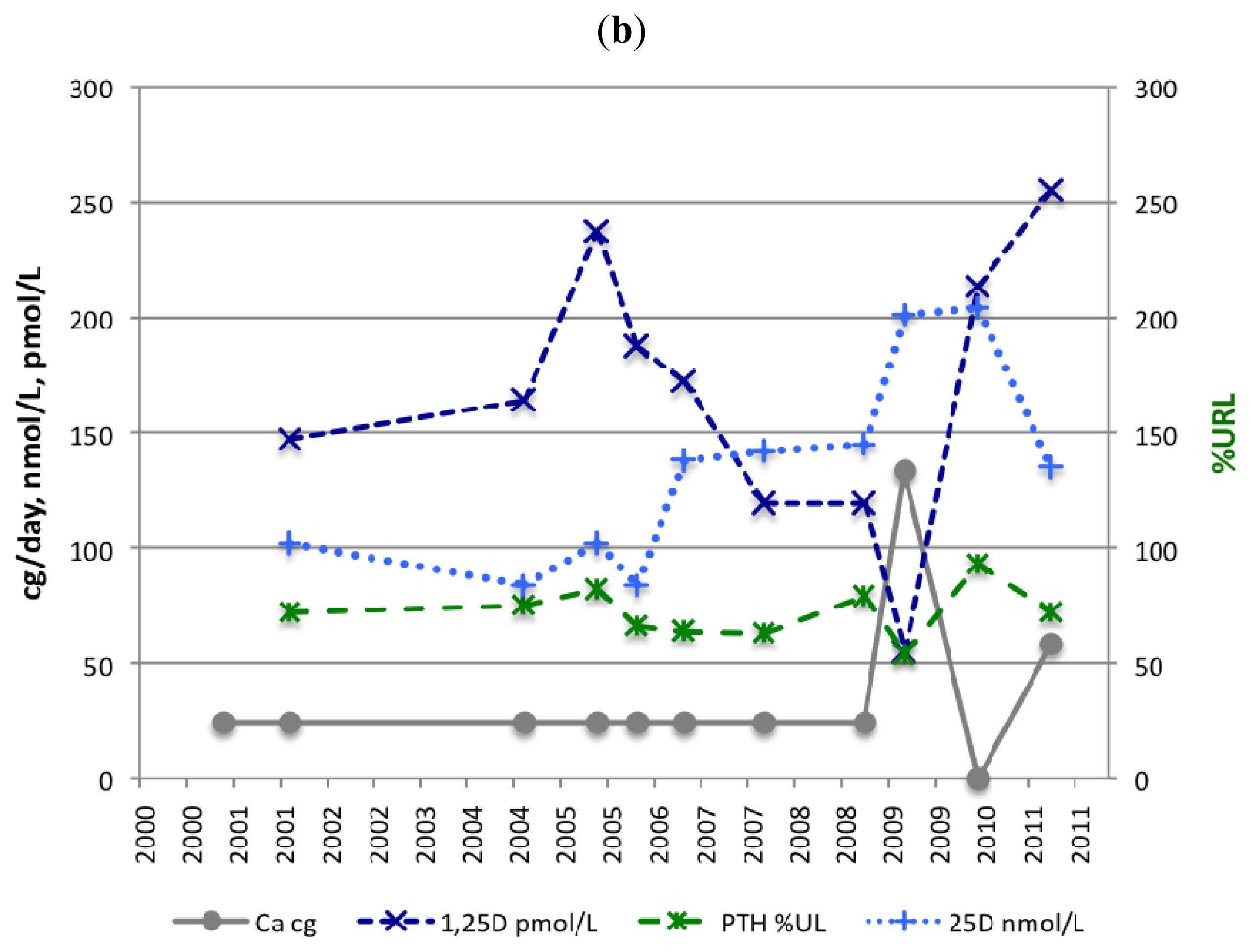
2.1. Biochemical Assessment
2.1.1. Serum 25(OH)D
2.1.2. Serum PTH
2.1.3. Serum 1,25(OH)2D
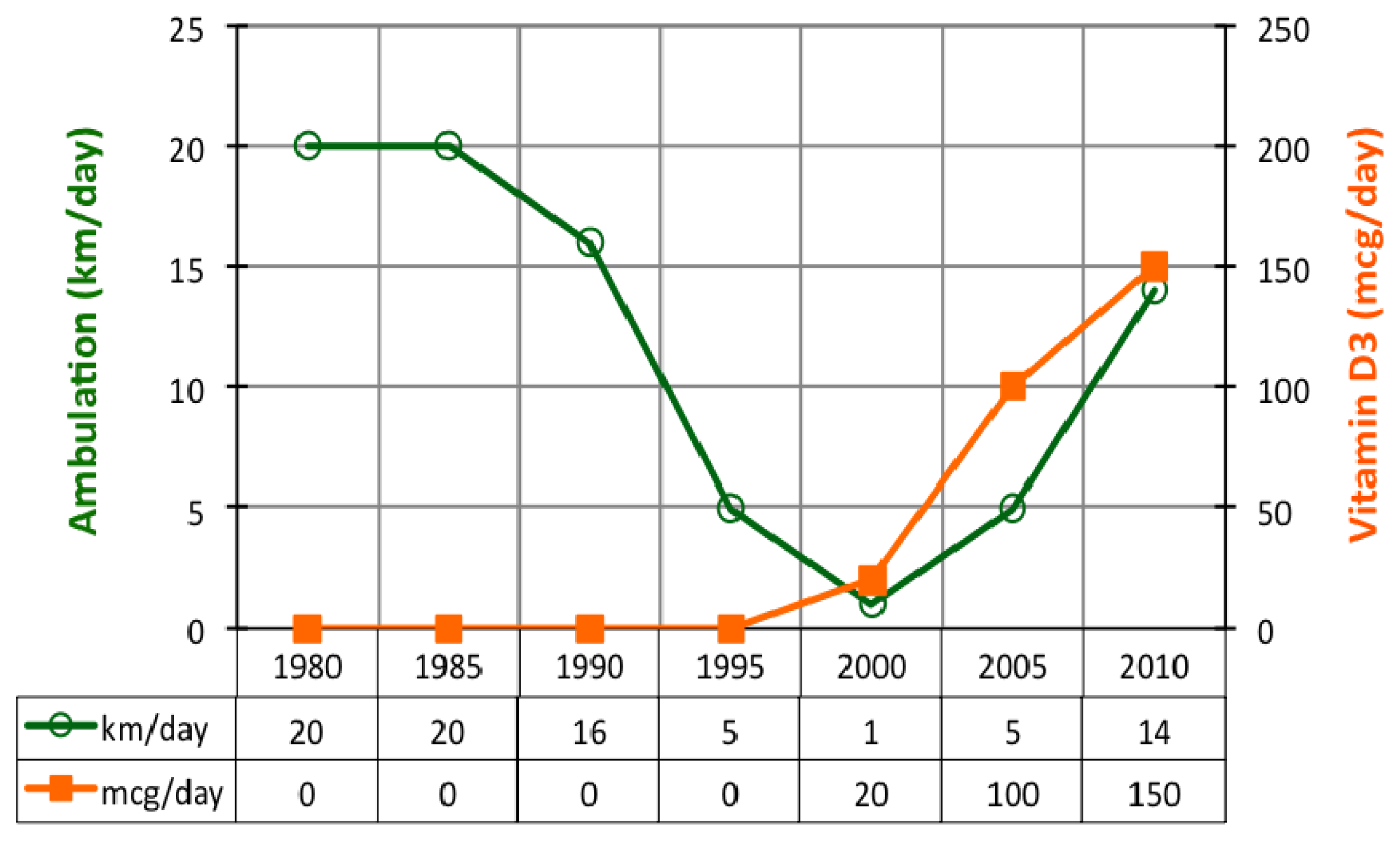
2.2. Ambulation
2.3. Muscular Pain
2.4. Bone Mineral Density
2.5. Adverse Events
3. Discussion
3.1. 25(OH)D, PTH and 1,25(OH)2D Levels
3.2. Calcium and Magnesium Supplementation
3.3. Ambulation
3.4. Muscular Pain
3.5. Bone Mineral Density
3.6. Limitations of the Study
3.7. Data Collection
3.8. One Person and Three Autoimmune Diseases
4. Experimental Section
4.1. Study Design
4.2. Vitamin D3 and Calcium Supplementation
4.3. Exercise
4.4. Blood Sampling
4.5. Bone Mineral Density
5. Conclusions
Acknowledgments
Abbreviations
| ALP | alkaline phosphatase |
| BMD | bone mineral density |
| BMI | body mass index |
| CD | coeliac disease |
| EDSS | expanded disability status scale |
| GI | gastrointestinal |
| GFD | gluten free diet |
| ITP | idiopathic thrombocytopenic purpura |
| MS | multiple sclerosis |
| NR | normal range |
| RRMS | relapsing remitting MS |
| PTH | parathyroid hormone |
| URL | upper reference limit |
| %URL | as a % of the corresponding upper reference limit |
References
- Moreno, L.A.; Valtueña, J.; Pérez-López, F.; González-Gross, M. Health effects related to low vitamin D concentrations: Beyond bone metabolism. Ann. Nutr. Metab 2011, 59, 22–27. [Google Scholar]
- Smolders, J.; Moen, S.M.; Damoiseaux, J.; Huitinga, I.; Holmøy, T. Vitamin D in the healthy and inflamed central nervous system: Access and function. J. Neurol. Sci 2011, 311, 37–43. [Google Scholar]
- Hayes, C.E.; Nashold, F.E.; Mayne, C.G.; Spanier, J.A.; Nelson, C.D. Vitamin D and Multiple Sclerosis. In Vitamin D, 3rd ed; Feldman, D., Pike, J.W., Adams, J.S., Eds.; Elsevier: Amsterdam, The Netherlands, 2011; Volume II, pp. 1843–1878. [Google Scholar]
- Van Amerongen, B.M.; Dijkstra, C.D.; Lips, P.; Polman, C.H. Multiple sclerosis and vitamin D: An update. Eur. J. Clin. Nutr 2004, 58, 1095–1109. [Google Scholar]
- Van der Mei, I.A.; Ponsonby, A.L.; Dwyer, T.; Blizzard, L.; Simmons, R.; Taylor, B.V.; Butzkueven, H.; Kilpatrick, T. Past exposure to sun, skin phenotype, and risk of multiple sclerosis: Case-control study. Br. Med. J 2003, 327, 316. [Google Scholar]
- Van der Mei, I.A.; Ponsonby, A.L.; Dwyer, T.; Blizzard, L.; Taylor, B.V.; Kilpatrick, T.; Butzkueven, H.; McMichael, A.J. Vitamin D levels in people with multiple sclerosis and community controls in Tasmania, Australia. J. Neurol 2007, 254, 581–590. [Google Scholar]
- Lucas, R.M.; Ponsonby, A.L.; Dear, K.; Valery, P.C.; Pender, M.P.; Taylor, B.V.; Kilpatrick, T.J.; Dwyer, T.; Coulthard, A.; Chapman, C.; et al. Sun exposure and vitamin D are independent risk factors for CNS demyelination. Neurology 2011, 76, 540–548. [Google Scholar]
- Munger, K.L.; Zhang, S.M.; O’Reilly, E.; Hernán, M.A.; Olek, M.J.; Willett, W.C.; Ascherio, A. Vitamin D intake and incidence of multiple sclerosis. Neurology 2004, 62, 60–65. [Google Scholar]
- Munger, K.L.; Levin, L.I.; Hollis, B.W.; Howard, N.S.; Ascherio, A. Serum 25-hydroxyvitamin D levels and risk of multiple sclerosis. J. Am. Med. Assoc 2006, 296, 2832–2838. [Google Scholar]
- Soilu-Hänninen, M.; Airas, L.; Mononen, I.; Heikkilä, A.; Viljanen, M.; Hänninen, A. 25-Hydroxyvitamin D levels in serum at the onset of multiple sclerosis. Mult. Scler 2005, 11, 266–271. [Google Scholar]
- Soilu-Hänninen, M.; Laaksonen, M.; Laitinen, I.; Erälinna, J.P.; Lilius, E.M.; Mononen, I. A longitudinal study of serum levels of 25-hydroxyvitamin D and intact parathyroid hormone indicate the importance of vitamin D and calcium homeostasis regulation in multiple sclerosis. J. Neurol. Neurosurg. Psychiatry 2008, 79, 152–157. [Google Scholar]
- Smolders, J.; Menheere, P.; Kessels, A.; Damoiseaux, J.; Hupperts, R. Association of vitamin D metabolite levels with relapse rate and disability in multiple sclerosis. Mult. Scler 2008, 14, 1220–1224. [Google Scholar]
- Kragt, J.; van Amerongen, B.; Killestein, J.; Dijkstra, C.; Uitdehaag, B.; Polman, C.H.; Lips, P. Higher levels of 25-hydroxyvitamin D are associated with a lower incidence of multiple sclerosis only in women. Mult. Scler 2009, 15, 9–15. [Google Scholar]
- Weinstock-Guttman, B.; Zivadinov, R.; Qu, J.; Cookfair, D.; Duan, X.; Bang, E.; Bergsland, N.; Hussein, S.; Cherneva, M.; Willis, L.; et al. Vitamin D metabolites are associated with clinical and MRI outcomes in multiple sclerosis patients. J. Neurol. Neurosurg. Psychiatry 2011, 82, 189–195. [Google Scholar]
- Mowry, E.M.; Waubant, E.; McCulloch, C.E.; Okuda, D.T.; Evangelista, A.A.; Lincoln, R.R.; Gourraud, P.A.; Brenneman, D.; Owen, M.C.; Qualley, P.; et al. Vitamin D status predicts new brain MRI activity in multiple sclerosis. Ann. Neurol 2012, in press. [Google Scholar]
- Mowry, E.M.; Krupp, L.B.; Milazzo, M.; Chabas, D.; Strober, J.B.; Belman, A.L.; McDonald, J.C.; Oksenberg, J.R.; Bacchetti, P.; Waubant, E. Vitamin D status is associated with relapse rate in pediatric-onset multiple sclerosis. Ann. Neurol 2010, 67, 618–624. [Google Scholar]
- Simpson, S., Jr; Taylor, B.; Blizzard, L.; Ponsonby, A.L.; Pittas, F.; Tremlett, H.; Dwyer, T.; Gies, P.; van der Mei, I. Higher 25-hydroxyvitamin D is associated with lower relapse risk in multiple sclerosis. Ann. Neurol 2010, 68, 193–203. [Google Scholar]
- Goldberg, P.; Fleming, M.C.; Picard, E.H. Multiple sclerosis: Decreased relapse rate through dietary supplementation with calcium, magnesium and vitamin D. Med. Hypotheses 1986, 21, 193–200. [Google Scholar]
- Nordvik, I.; Myhr, K.M.; Nyland, H.; Bjerve, K.S. Effect of dietary advice and n-3 supplementation in newly diagnosed MS patients. Acta Neurol. Scand 2000, 102, 143–149. [Google Scholar]
- Mahon, B.D.; Gordon, S.A.; Cruz, J.; Cosman, F.; Cantorna, M.T. Cytokine profile in patients with multiple sclerosis following vitamin D supplementation. J. Neuroimmunol 2003, 134, 128–132. [Google Scholar]
- Kimball, S.M.; Ursell, M.R.; O’Connor, P.; Vieth, R. Safety of vitamin D3 in adults with multiple sclerosis. Am. J. Clin. Nutr 2007, 86, 645–651. [Google Scholar]
- Burton, J.M.; Kimball, S.; Vieth, R.; Bar-Or, A.; Dosch, H.M.; Cheung, R.; Gagne, D.; D’Souza, C.; Ursell, M.; O’Connor, P. A phase I/II dose-escalation trial of vitamin D3 and calcium in multiple sclerosis. Neurology 2010, 74, 1852–1859. [Google Scholar]
- Kimball, S.; Vieth, R.; Dosch, H.M.; Bar-Or, A.; Cheung, R.; Gagne, D.; O’Connor, P.; D’Souza, C.; Ursell, M.; Burton, J.M. Cholecalciferol plus calcium suppresses abnormal PBMC reactivity in patients with multiple sclerosis. J. Clin. Endocrinol. Metab 2011, 96, 2826–2834. [Google Scholar]
- Smolders, J.; Peelen, E.; Thewissen, M.; Cohen Tervaert, J.W.; Menheere, P.; Hupperts, R.; Damoiseaux, J. Safety and T cell modulating effects of high dose vitamin D3 supplementation in multiple sclerosis. PLoS One 2010, 5, e15235. [Google Scholar]
- Mosayebi, G.; Ghazavi, A.; Ghasami, K.; Jand, Y.; Kokhaei, P. Therapeutic effect of vitamin D3 in multiple sclerosis patients. Immunol. Invest 2011, 40, 627–639. [Google Scholar]
- Kampman, M.T.; Steffensen, L.H.; Mellgren, S.I.; Jørgensen, L. Effect of vitamin D3 supplementation on relapses, disease progression and measures of function in persons with multiple sclerosis: Exploratory outcomes from a double-blind randomised controlled trial. Mult. Scler 2012, 18, 1144–1151. [Google Scholar]
- Soilu-Hänninen, M.; Aivo, J.; Lindström, B.M.; Elovaara, I.; Sumelahti, M.L.; Färkkilä, M.; Tienari, P.; Atula, S.; Sarasoja, T.; Herrala, L.; et al. A randomised, double blind, placebo controlled trial with vitamin D3 as an add on treatment to interferon β-1b in patients with multiple sclerosis. J. Neurol. Neurosurg. Psychiatry 2012, 83, 565–571. [Google Scholar]
- A Multicentre Study of the Efficacy and Safety of Supplementary Treatment With Cholecalciferol in Patients With Relapsing Multiple Sclerosis Treated With Subcutaneous Interferon Beta-1a 44 μg 3 Times Weekly (CHOLINE). 2010. Available online: http://clinicaltrials.gov/ct2/show/NCT01198132 accessed on 26 September 2012.
- Supplementation of VigantOL® Oil Versus Placebo as Add-on in Patients With Relapsing Remitting Multiple Sclerosis Receiving Rebif® Treatment (SOLAR). 2011. Available online: http://clinicaltrials.gov/ct2/show/NCT01285401 accessed on 26 September 2012.
- Smolders, J.; Hupperts, R.; Barkhof, F.; Grimaldi, L.M.; Holmoy, T.; Killestein, J.; Rieckmann, P.; Schluep, M.; Vieth, R.; Hostalek, U.; et al. Efficacy of vitamin D(3) as add-on therapy in patients with relapsing-remitting multiple sclerosis receiving subcutaneous interferon beta-1a: A Phase II, multicenter, double-blind, randomized, placebo-controlled trial. J. Neurol. Sci 2011, 311, 44–49. [Google Scholar]
- Vitamin D Supplementation in Multiple Sclerosis. 2011. Available online: http://clinicaltrials.gov/ct2/show/NCT01490502 accessed on 26 September 2012.
- Noseworthy, J.H.; Lucchinetti, C.; Rodriguez, M.; Weinshenker, B.G. Multiple sclerosis. N. Engl. J. Med 2000, 343, 938–952. [Google Scholar]
- Mellanby, E. Nutritional Influences on the Nervous System-Experimental Work on Animals. In Nutrition and Disease. The Interaction of Clinical and Experimental Work on Animals; Oliver and Boyd: London, UK, 1934; Volume V, pp. 98–118. [Google Scholar]
- Pfeifer, M.; Begerow, B.; Minne, H.W. Vitamin D and muscle function. Osteoporos. Int 2002, 13, 187–194. [Google Scholar]
- Birge, S.J.; Haddad, J.G. 25-hydroxycholecalciferol stimulation of muscle metabolism. J. Clin. Invest 1975, 56, 1100–1107. [Google Scholar]
- Wassner, S.J.; Li, J.B.; Sperduto, A.; Norman, M.E. Vitamin D Deficiency, hypocalcemia, and increased skeletal muscle degradation in rats. J. Clin. Invest 1983, 72, 102–112. [Google Scholar]
- Chabas, J.F.; Alluin, O.; Rao, G.; Garcia, S.; Lavaut, M.N.; Risso, J.J.; Legre, R.; Magalon, G.; Khrestchatisky, M.; Marqueste, T.; et al. Vitamin D2 potentiates axon regeneration. J. Neurotrauma 2008, 25, 1247–1256. [Google Scholar]
- Sørensen, O.H.; Lund, B.; Saltin, B.; Lund, B.; Andersen, R.B.; Hjorth, L.; Melsen, F.; Mosekilde, L. Myopathy in bone loss of ageing: improvement by treatment with 1α-hydroxycholecalciferol and calcium. Clin. Sci. (Lond) 1979, 56, 157–161. [Google Scholar]
- Sato, Y.; Iwamoto, J.; Kanoko, T.; Satoh, K. Low-dose vitamin D prevents muscular atrophy and reduces falls and hip fractures in women after stroke: A randomized controlled trial. Cerebrovasc. Dis 2005, 20, 187–192. [Google Scholar]
- Moreira-Pfrimer, L.D.; Pedrosa, M.A.; Teixeira, L.; Lazaretti-Castro, M. Treatment of vitamin D deficiency increases lower limb muscle strength in institutionalized older people independently of regular physical activity: A randomized double-blind controlled trial. Ann. Nutr. Metab 2009, 54, 291–300. [Google Scholar]
- Dawson-Hughes, B.; Heaney, R.P.; Holick, M.F.; Lips, P.; Meunier, P.J.; Vieth, R. Estimates of optimal vitamin D status. Osteoporos. Int 2005, 16, 713–716. [Google Scholar]
- Geller, J.L.; Adams, J.S. Vitamin D therapy. Curr. Osteoporos. Rep 2008, 6, 5–11. [Google Scholar]
- Lips, P. Vitamin D deficiency and secondary hyperparathyroidism in the elderly: Consequences for bone loss and fractures and therapeutic implications. Endocr. Rev 2001, 22, 477–501. [Google Scholar]
- Nieves, J.; Cosman, F.; Herbert, J.; Shen, V.; Lindsay, R. High prevalence of vitamin D deficiency and reduced bone mass in multiple sclerosis. Neurology 1994, 44, 1687–1692. [Google Scholar]
- Cosman, F.; Nieves, J.; Komar, L.; Ferrer, G.; Herbert, J.; Formica, C.; Shen, V.; Lindsay, R. Fracture history and bone loss in patients with MS. Neurology 1998, 51, 1161–1165. [Google Scholar]
- Weinstock-Guttman, B.; Gallagher, E.; Baier, M.; Green, L.; Feichter, J.; Patrick, K.; Miller, C.; Wrest, K.; Ramanathan, M. Risk of bone loss in men with multiple sclerosis. Mult. Scler 2004, 10, 170–175. [Google Scholar]
- Steffensen, L.H.; Mellgren, S.I.; Kampman, M.T. Predictors and prevalence of low bone mineral density in fully ambulatory persons with multiple sclerosis. J. Neurol 2010, 257, 410–418. [Google Scholar]
- Steffensen, L.H.; Jørgensen, L.; Straume, B.; Mellgren, S.I.; Kampman, M.T. Can vitamin D3 supplementation prevent bone loss in persons with MS? A placebo-controlled trial. J. Neurol 2011, 258, 1624–1631. [Google Scholar]
- Marrie, R.A.; Cutter, G.; Tyry, T.; Vollmer, T. A cross-sectional study of bone health in multiple sclerosis. Neurology 2009, 73, 1394–1398. [Google Scholar]
- Moen, S.M.; Celius, E.G.; Sandvik, L.; Nordsletten, L.; Eriksen, E.F.; Holmøy, T. Low bone mass in newly diagnosed multiple sclerosis and clinically isolated syndrome. Neurology 2011, 77, 151–157. [Google Scholar]
- Kampman, M.T.; Eriksen, E.F.; Holmøy, T. Multiple sclerosis, a cause of secondary osteoporosis? What is the evidence and what are the clinical implications? Acta Neurol. Scand. Suppl 2011, 191, 44–49. [Google Scholar]
- Kimball, S.M.; Burton, J.M.; O’Connor, P.G.; Vieth, R. Urinary calcium response to high dose vitamin D3 with calcium supplementation in patients with multiple sclerosis. Clin. Biochem 2011, 44, 930–932. [Google Scholar]
- Kurtzke, J.F. Geography in multiple sclerosis. J. Neurol 1977, 215, 1–26. [Google Scholar]
- Hoogervorst, E.L.; van Winsen, L.M.; Eikelenboom, M.J.; Kalkers, N.F.; Uitdehaag, B.M.; Polman, C.H. Comparisons of patient self-report, neurologic examination, and functional impairment in MS. Neurology 2001, 56, 934–937. [Google Scholar]
- Kalkers, N.F.; de Groot, V.; Lazeron, R.H.; Killestein, J.; Adèr, H.J.; Barkhof, F.; Lankhorst, G.J.; Polman, C.H. MS functional composite: relation to disease phenotype and disability strata. Neurology 2000, 54, 1233–1239. [Google Scholar]
- Kimball, S.; Vieth, R. Self-prescribed high-dose vitamin D3: Effects on biochemical parameters in two men. Ann. Clin. Biochem 2008, 45, 106–110. [Google Scholar]
- Vieth, R. The Pharmacology of Vitamin D. In Vitamin D, 3rd ed; Feldman, D., Pike, J.W., Adams, J.S., Eds.; Elsevier: Amsterdam, The Netherlands, 2011; Volume I, pp. 1041–1076. [Google Scholar]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med 2007, 357, 266–281. [Google Scholar]
- Roder, J.D.; Stair, E.L. An overview of cholecalciferol toxicosis. Vet. Hum. Toxicol 1999, 343. [Google Scholar]
- Barger-Lux, M.J.; Heaney, R.P.; Dowell, S.; Chen, T.C.; Holick, M.F. Vitamin D and its major metabolites: serum levels after graded oral dosing in healthy men. Osteoporos. Int 1998, 8, 222–230. [Google Scholar]
- Sanders, K.M.; Stuart, A.L.; Williamson, E.J.; Simpson, J.A.; Kotowicz, M.A.; Young, D.; Nicholson, G.C. Annual high-dose oral vitamin D and falls and fractures in older women: A randomized controlled trial. J. Am. Med. Assoc 2010, 303, 1815–1822. [Google Scholar]
- Reusch, J.; Ackermann, H.; Badenhoop, K. Cyclic changes of vitamin D and PTH are primarily regulated by solar radiation: 5-year analysis of a German (50 degrees N) population. Horm. Metab. Res 2009, 41, 402–407. [Google Scholar]
- Karaguzel, G.; Holick, M.F. Diagnosis and treatment of osteopenia. Rev. Endocr. Metab. Disord 2010, 11, 237–251. [Google Scholar]
- Lieben, L.; Masuyama, R.; Torrekens, S.; van Looveren, R.; Schrooten, J.; Baatsen, P.; Lafage-Proust, M.H.; Dresselaers, T.; Feng, J.Q.; Bonewald, L.F.; et al. Normocalcemia is maintained in mice under conditions of calcium malabsorption by vitamin D-induced inhibition of bone mineralization. J. Clin. Invest 2012, 122, 1803–1815. [Google Scholar]
- Mocanu, V.; Stitt, P.A.; Costan, A.R.; Voroniuc, O.; Zbranca, E.; Luca, V.; Vieth, R. Long-term effects of giving nursing home residents bread fortified with 125 microg (5000 IU) vitamin D(3) per daily serving. Am. J. Clin. Nutr 2009, 89, 1132–1137. [Google Scholar]
- Ginde, A.A.; Wolfe, P.; Camargo, C.A., Jr; Schwartz, R.S. Defining Vitamin D Status by Secondary Hyperparathyroidism in the U.S. Population. J. Endocrinol. Invest 2012, 35, 42–48. [Google Scholar]
- Ledger, G.A.; Burritt, M.F.; Kao, P.C.; O’Fallon, W.M.; Riggs, B.L.; Khosla, S. Abnormalities of parathyroid hormone secretion in elderly women that are reversible by short term therapy with 1,25-dihydroxyvitamin D3. J. Clin. Endocrinol. Metab 1994, 79, 211–216. [Google Scholar]
- McKane, W.R.; Khosla, S.; Egan, K.S.; Robins, S.P.; Burritt, M.F.; Riggs, B.L. Role of calcium intake in modulating age-related increases in parathyroid function and bone resorption. J. Clin. Endocrinol. Metab 1996, 81, 1699–1703. [Google Scholar]
- Kärkkäinen, M.U.; Lamberg-Allardt, C.J.; Ahonen, S.; Valimaki, M. Does it make a difference how and when you take your calcium? The acute effects of calcium on calcium and bone metabolism. Am. J. Clin. Nutr 2001, 74, 335–342. [Google Scholar]
- Aydin, H.; Deyneli, O.; Yavuz, D.; Gözü, H.; Mutlu, N.; Kaygusuz, I.; Akalın, S. Short-Term oral magnesium supplementation suppresses bone turnover in postmenopausal osteoporotic women. Biol. Trace Elem. Res 2010, 133, 136–143. [Google Scholar]
- Corazza, G.R.; di Sario, A.; Cecchetti, L.; Tarozzi, C.; Corrao, G.; Bernardi, M.; Gasbarrini, G. Bone mass and metabolism in patients with celiac disease. Gastroenterology 1995, 109, 122–128. [Google Scholar]
- Selby, P.L.; Davies, M.; Adams, J.E.; Mawer, E.B. Bone loss in celiac disease is related to secondary hyperparathyroidism. J. Bone Miner. Res 1999, 14, 652–657. [Google Scholar]
- Rude, R.K.; Olerich, M. Magnesium deficiency: possible role in osteoporosis associated with gluten-sensitive enteropathy. Osteoporos. Int 1996, 6, 453–461. [Google Scholar]
- Ramsaransing, G.S.; Mellema, S.A.; de Keyser, J. Dietary patterns in clinical subtypes of multiple sclerosis: An exploratory study. Nutr. J 2009, 8, 36. [Google Scholar]
- Sinert, R.; Zehtabchi, S.; Desai, S.; Peacock, P.; Altura, B.T.; Altura, B.M. Serum ionized magnesium and calcium levels in adult patients with seizures. Scand. J. Clin. Lab Invest 2007, 67, 317–326. [Google Scholar]
- Plum, L.A.; DeLuca, H.F. Vitamin D, disease and therapeutic opportunities. Nat. Rev. Drug Discov 2010, 9, 941–955. [Google Scholar]
- Smolders, J.; Damoiseaux, J. Vitamin D as a T-cell modulator in multiple sclerosis. Vitam Horm 2011, 86, 401–428. [Google Scholar]
- Fernandes de Abreu, D.A.; Eyles, D.; Féron, F. Vitamin D, a neuro-immunomodulator: Implications for neurodegenerative and autoimmune diseases. Psychoneuroendocrinology 2009, 34, S265–S277. [Google Scholar]
- Fernandes de Abreu, D.A.; Landel, V.; Féron, F. Seasonal, gestational and postnatal influences on multiple sclerosis: the beneficial role of a vitamin D supplementation during early life. J. Neurol. Sci 2011, 311, 64–68. [Google Scholar]
- Bianco, J.; Gueye, Y.; Marqueste, T.; Alluin, O.; Risso, J.J.; Garcia, S.; Lavault, M.N.; Khrestchatisky, M.; Feron, F.; Decherchi, P. Vitamin D(3) improves respiratory adjustment to fatigue and H-reflex responses in paraplegic adult rats. Neuroscience 2011, 188C, 182–192. [Google Scholar]
- Wergeland, S.; Torkildsen, Ø.; Myhr, K.M.; Aksnes, L.; Mørk, S.J.; Bø, L. Dietary vitamin D3 supplements reduce demyelination in the cuprizone model. PLoS One 2011, 6, e26262. [Google Scholar]
- Cannell, J.J.; Hollis, B.W.; Sorenson, M.B.; Taft, T.N.; Anderson, J.J. Athletic performance and vitamin D. Med. Sci. Sports Exerc 2009, 41, 1102–1110. [Google Scholar]
- Alexander, F.M. The Use of the Self. In Its Conscious Direction in Relation to Diagnosis, Functioning, and the Control of Reaction, 3rd ed; Richard Clay and Company, Ltd: Bungay, UK, 1946. [Google Scholar]
- Carrière, B. The Swiss Ball: Theory, Basic Exercises and Clinical Application; Springer: Berlin, Germany, 1998. [Google Scholar]
- Gloth, F.M., III; Lindsay, J.M.; Zelesnick, L.B.; Greenough, W.B., III. Can vitamin D deficiency produce an unusual pain syndrome? Arch. Intern. Med 1991, 151, 1662–1664. [Google Scholar]
- Glerup, H.; Eriksen, E.F. Acroparaesthesia—A typical finding in vitamin D deficiency. Rheumatology 1999, 38, 482. [Google Scholar]
- De Torrenté de la Jara, G.; Pécoud, A.; Favrat, B. Female asylum seekers with musculoskeletal pain: The importance of diagnosis and treatment of hypovitaminosis D. BioMed Central Fam. Pract 2006, 7, 4. [Google Scholar]
- Tague, S.E.; Smith, P.G. Vitamin D receptor and enzyme expression in dorsal root ganglia of adult female rats: Modulation by ovarian hormones. J. Chem. Neuroanat 2011, 41, 1–12. [Google Scholar]
- Tague, S.E.; Clarke, G.L.; Winter, M.K.; McCarson, K.E.; Wright, D.E.; Smith, P.G. Vitamin D deficiency promotes skeletal muscle hypersensitivity and sensory hyperinnervation. J. Neurosci 2011, 31, 13728–13738. [Google Scholar]
- PatientsLikeMe. Available online: http://www.patientslikeme.com accessed on 7 July 2012.
- Katz, S.; Weinerman, S. Osteoporosis and gastrointestinal disease. Gastroenterol. Hepatol 2010, 6, 506–517. [Google Scholar]
- Ludvigsson, J.F.; Leffler, D.A.; Bai, J.C.; Biagi, F.; Fasano, A.; Green, P.H.; Hadjivassiliou, M.; Kaukinen, K.; Kelly, C.P.; Leonard, J.N.; et al. The Oslo definitions for coeliac disease and related terms. Gut 2012, in press. [Google Scholar]
- Pazianas, M.; Butcher, G.P.; Subhani, J.M.; Finch, P.J.; Ang, L.; Collins, C.; Heaney, R.P.; Zaidi, M.; Maxwell, J.D. Calcium absorption and bone mineral density in celiacs after long term treatment with gluten-free diet and adequate calcium intake. Osteoporos. Int 2005, 16, 56–63. [Google Scholar]
- Wingerchuk, D.M. Smoking: Effects on multiple sclerosis susceptibility and disease progression. Ther. Adv. Neurol. Disord 2012, 5, 13–22. [Google Scholar]
- Robinson, D.P.; Klein, S.L. Pregnancy and pregnancy-associated hormones alter immune responses and disease pathogenesis. Horm. Behav 2012, in press. [Google Scholar]
- Rietberg, M.B.; Brooks, D.; Uitdehaag, B.M.; Kwakkel, G. Exercise therapy for multiple sclerosis. Cochrane Database Syst. Rev 2005, 25, CD003980. [Google Scholar]
- Segal, J.B.; Powe, N.R. Prevalence of immune thrombocytopenia: analyses of administrative data. J. Thromb. Haemost 2006, 4, 2377–2383. [Google Scholar]
- Horstman, L.L.; Jy, W.; Ahn, Y.S.; Zivadinov, R.; Maghzi, A.H.; Etemadifar, M.; Steven Alexander, J.; Minagar, A. Role of platelets in neuroinflammation: A wide-angle perspective. J. Neuroinflamm 2010, 7, 10. [Google Scholar]
- Schwalfenberg, G.K. Solar radiation and vitamin D: Mitigating environmental factors in autoimmune disease. J. Environ. Public Health 2012, 2012, 619381. [Google Scholar]
- Rodrigo, L.; Hernández-Lahoz, C.; Fuentes, D.; Alvarez, N.; López-Vázquez, A.; González, S. Prevalence of celiac disease in multiple sclerosis. BioMed Central Neurol 2011, 11, 31. [Google Scholar]
- Cotsapas, C.; Voight, B.F.; Rossin, E.; Lage, K.; Neale, B.M.; Wallace, C.; Abecasis, G.R.; Barrett, J.C.; Behrens, T.; Cho, J.; et al. Pervasive sharing of genetic effects in autoimmune disease. PLoS Genet 2011, 7, e1002254. [Google Scholar]
- Gutierrez-Achury, J.; Coutinho de Almeida, R.; Wijmenga, C. Shared genetics in coeliac disease and other immune-mediated diseases. J. Intern. Med 2011, 269, 591–603. [Google Scholar]
- Olén, O.; Montgomery, S.M.; Elinder, G.; Ekbom, A.; Ludvigsson, J.F. Increased risk of immune thrombocytopenic purpura among inpatients with coeliac disease. Scand. J. Gastroenterol 2008, 43, 416–422. [Google Scholar]
- Hadjivassiliou, M.; Sanders, D.S.; Grünewald, R.A.; Woodroofe, N.; Boscolo, S.; Aeschlimann, D. Gluten sensitivity: From gut to brain. Lancet Neurol 2010, 9, 318–330. [Google Scholar]
- Hernández-Lahoz, C.; Rodrigo, L. Gluten sensitivity and the CNS: Diagnosis and treatment. Lancet Neurol 2010, 9, 653. [Google Scholar]
- Phan-Ba, R.; Lambinet, N.; Louis, E.; Delvenne, P.; Tshibanda, L.; Boverie, J.; Moonen, G.; Belachew, S. Natalizumab to kill two birds with one stone: A case of celiac disease and multiple sclerosis. Inflamm. Bowel. Dis 2011, 17, E62–E63. [Google Scholar]
- Health Council of the Netherlands, Towards an Adequate Intake of Vitamin D; Publication no. 2008/15E; Health Council of the Netherlands: The Hague, The Netherlands, September 2008.
| Date | 5 December 2001 | 4 October 2004 | 29 September 2005 | 24 March 2006 | 5 October 2006 | 17 October 2007 | 28 January 2009 | 28 July 2009 | 23 June 2010 | 7 June 2011 | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hospital | a | b | b | b | b | b | c | b | b | d | ||
| Blood test | Unit | NR | ||||||||||
| 25(OH)D | nmol/L | >50 | 102 | 84 | 102 | 84 | 138 | 142 | 145 | 201 | 204 | 135 |
| 1,25(OH)2D | pmol/L | 50–180 | 147 | 164 | 237 | 187 | 172 | 119 | nd | 55 | 213 | 255 |
| PTH | pmol/L | 7.9 | 4.1 | 4.5 | 3.6 | 3.5 | 4.3 | 5.4 | 3.7 | 6.3 | 3.2 | |
| PTH | %URL | < 30 | 72 | 75 | 82 | 66 | 64 | 63 | 79 | 54 | 93 | 72 |
| Calcium | mmol/L | 2.2–2.6 | 2.25 | 2.34 | 2.34 | 2.29 | 2.32 | 2.38 | 2.37 | 2.42 | 2.32 | 2.34 |
| Magnesium | mmol/L | 0.7–1.0 | nd | 0.9 | 0.8 | 0.8 | 0.9 | 0,8 | nd | nd | 0.9 | 0.8 |
| Phosphate | mmol/L | 0.7–1.4 | 0.87 | 1.20 | 1.10 | 1.00 | 1.00 | 1.00 | 1.30 | nd | 1.20 | 1.17 |
| Creatinine | mmol/L | 60–110 | 85 | 75 | 74 | 64 | 70 | 76 | 70 | nd | 73 | 67 |
| Albumin | g/L | 35–50 | nd | 40 | 41 | 39 | 40 | 38 | 42 | nd | 39 | 44 |
| ALP | IU/L | 40–120 | 65 | 79 | 74 | 79 | 80 | 63 | nd | nd | 73 | 47 |
| Date | Supplementation | Hospital | BMI | Location | BMD g/cm2 | T-Score (SD) | Z-Score (SD) | ||
|---|---|---|---|---|---|---|---|---|---|
| VD3 mcg/day | Ca mg/day | Mg mg/day | |||||||
| 1 January 1980 | 0 | 0 | - | - | - | - | - | - | - |
| 22 November 1999 | 0 | 0 | - | a | 21.6 | LS | 0.990 | -0.5 | nd |
| 9 January 2001 | 20 | 240/1day | - | - | - | - | - | - | - |
| 4 September 2004 | 100 | 240/1day | - | - | - | - | - | - | - |
| 21 December 2005 | 150 | 240/1day | - | - | - | - | - | - | - |
| 28 January 2009 | 150 | 240/1day | - | b | 22.8 | LS LPF | 0.873 0.788 | −1.6 −1.3 | −0.2 −0.4 |
| 10 March 2009 | 150 | 333/4day | 133/4day | - | - | - | - | - | - |
| 27 July 2009 | 150 | 0 | - | - | - | - | - | - | - |
| 22 June 2010 | 150 | 0 | - | c | 20.8 | LS L2 t/m 4 LPF | 0.962 0.755 | −2.0 −1.9 | −0.7 −0.7 |
| 16 May 2011 | 150 | 0 | - | c | 20.5 | LS L2 t/m 4 LPF | 0.966 0.738 | −2.0 −2.2 | −0.6 −0.8 |
| 5 July 2011 | 150 | 145/4day | 237/4day | - | - | - | - | - | - |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Van Amerongen, B.M.; Feron, F. Effect of High-Dose Vitamin D3 Intake on Ambulation, Muscular Pain and Bone Mineral Density in a Woman with Multiple Sclerosis: A 10-Year Longitudinal Case Report. Int. J. Mol. Sci. 2012, 13, 13461-13483. https://doi.org/10.3390/ijms131013461
Van Amerongen BM, Feron F. Effect of High-Dose Vitamin D3 Intake on Ambulation, Muscular Pain and Bone Mineral Density in a Woman with Multiple Sclerosis: A 10-Year Longitudinal Case Report. International Journal of Molecular Sciences. 2012; 13(10):13461-13483. https://doi.org/10.3390/ijms131013461
Chicago/Turabian StyleVan Amerongen, Barbara M., and François Feron. 2012. "Effect of High-Dose Vitamin D3 Intake on Ambulation, Muscular Pain and Bone Mineral Density in a Woman with Multiple Sclerosis: A 10-Year Longitudinal Case Report" International Journal of Molecular Sciences 13, no. 10: 13461-13483. https://doi.org/10.3390/ijms131013461
APA StyleVan Amerongen, B. M., & Feron, F. (2012). Effect of High-Dose Vitamin D3 Intake on Ambulation, Muscular Pain and Bone Mineral Density in a Woman with Multiple Sclerosis: A 10-Year Longitudinal Case Report. International Journal of Molecular Sciences, 13(10), 13461-13483. https://doi.org/10.3390/ijms131013461




