A Novel Cold-Active Lipase from Candida albicans: Cloning, Expression and Characterization of the Recombinant Enzyme
Abstract
:1. Introduction
2. Results and Discussion
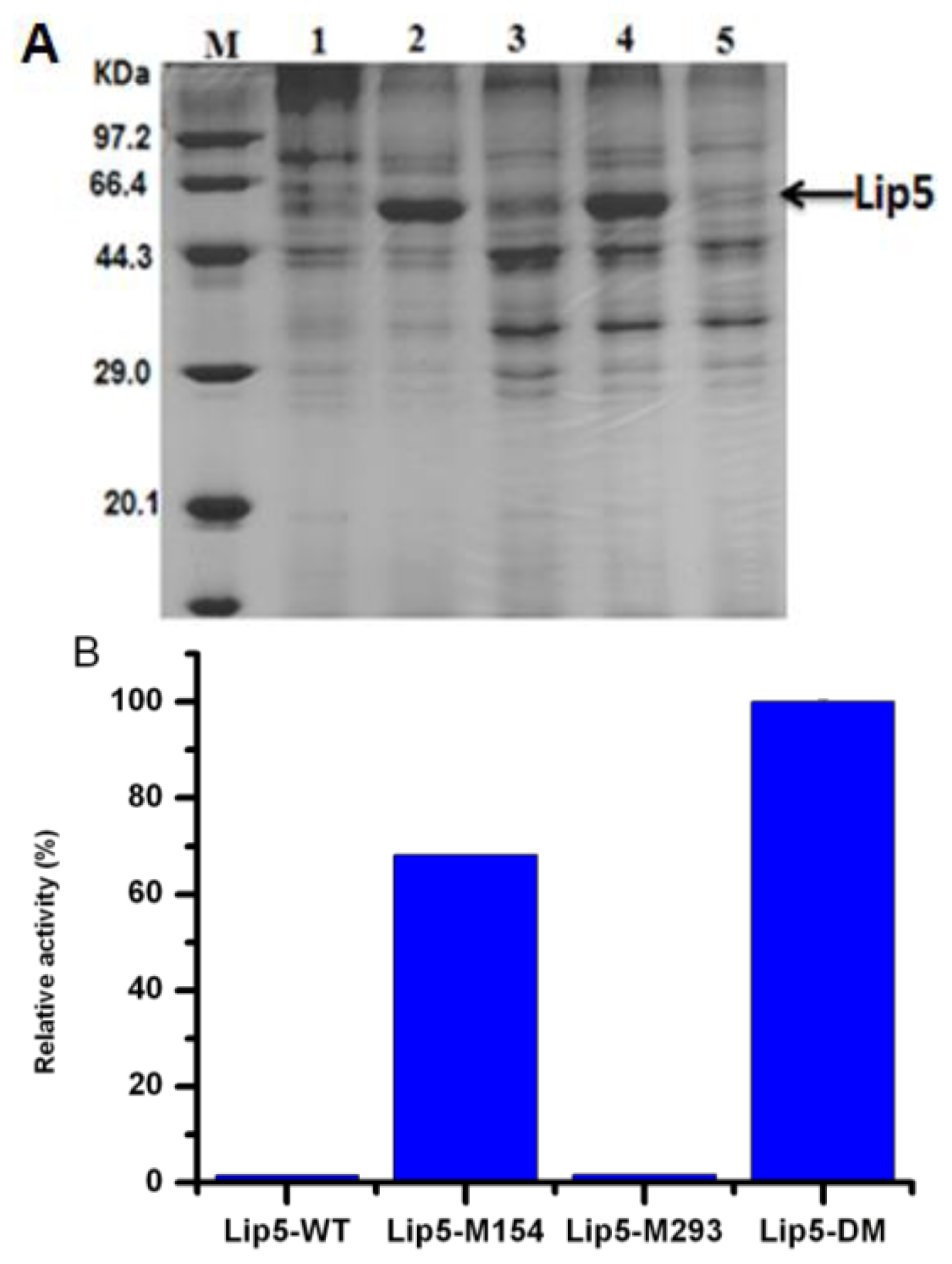
2.1. Cloning of lip5-wt Gene and Mutants from C. albicans
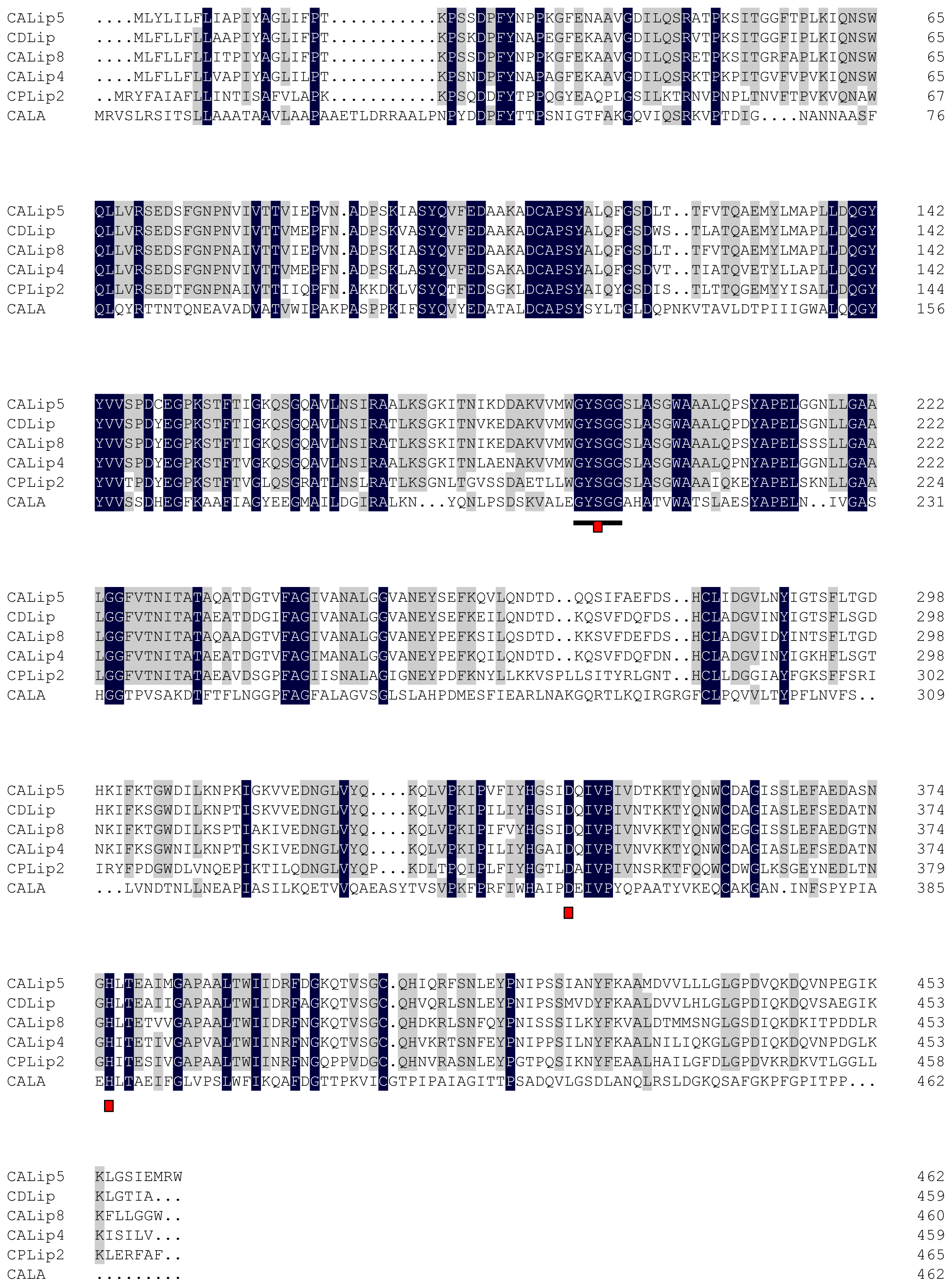
2.2. Amino Acid Sequence Analysis
2.3. Expression of lip5-wt and Mutants in P. pastoris
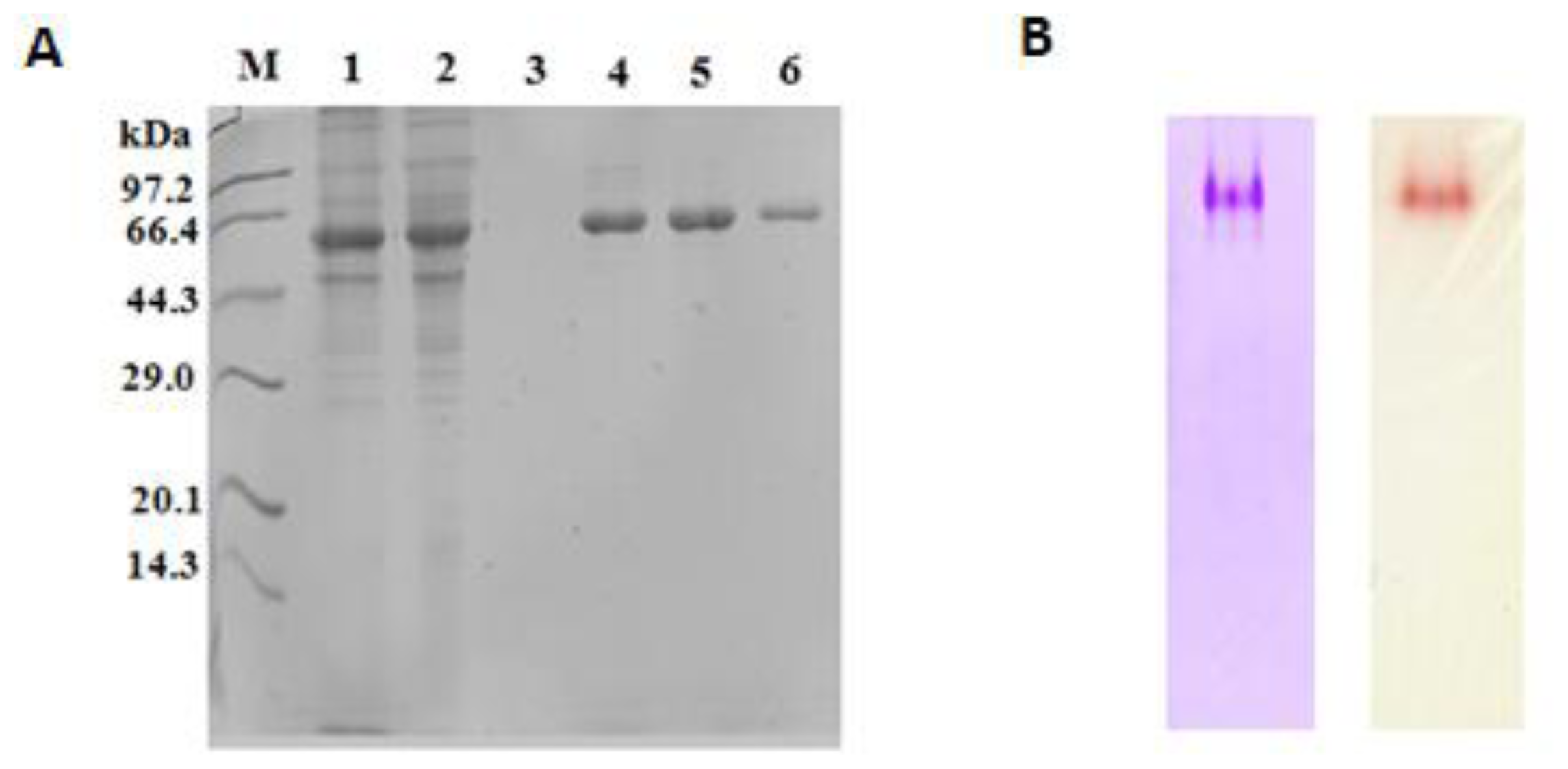
2.4. Purification of Recombinant Lip5-DM
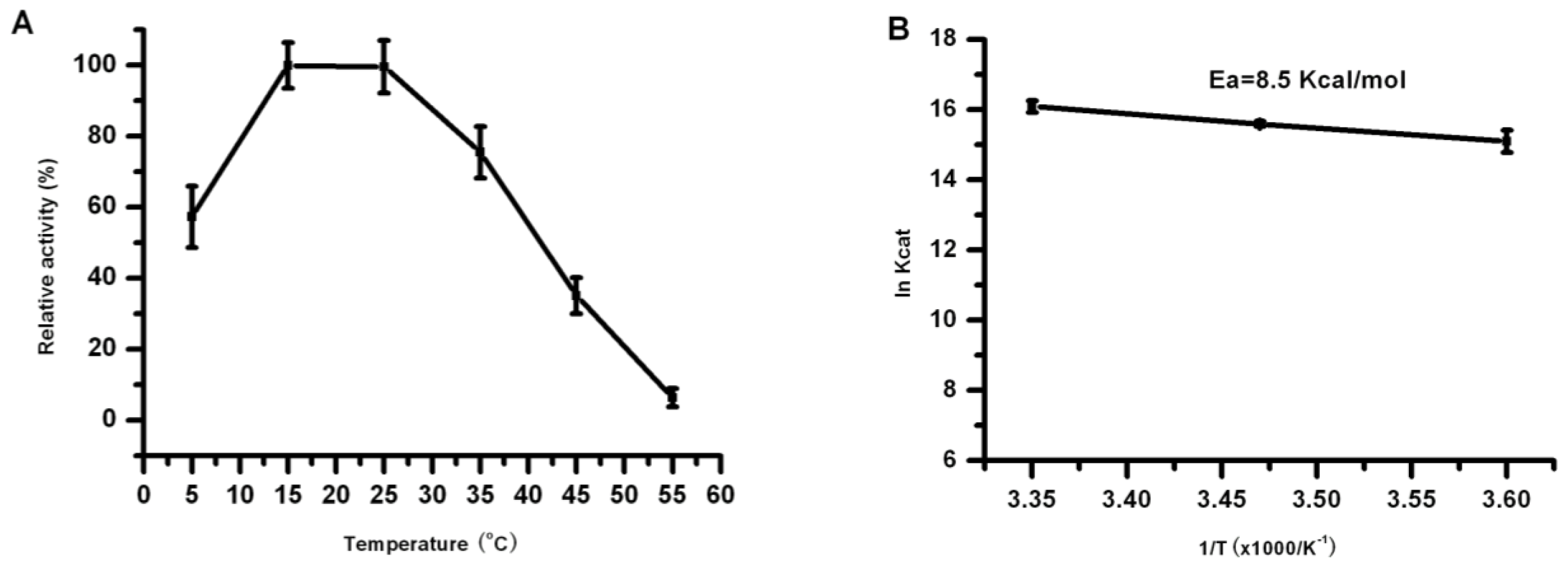
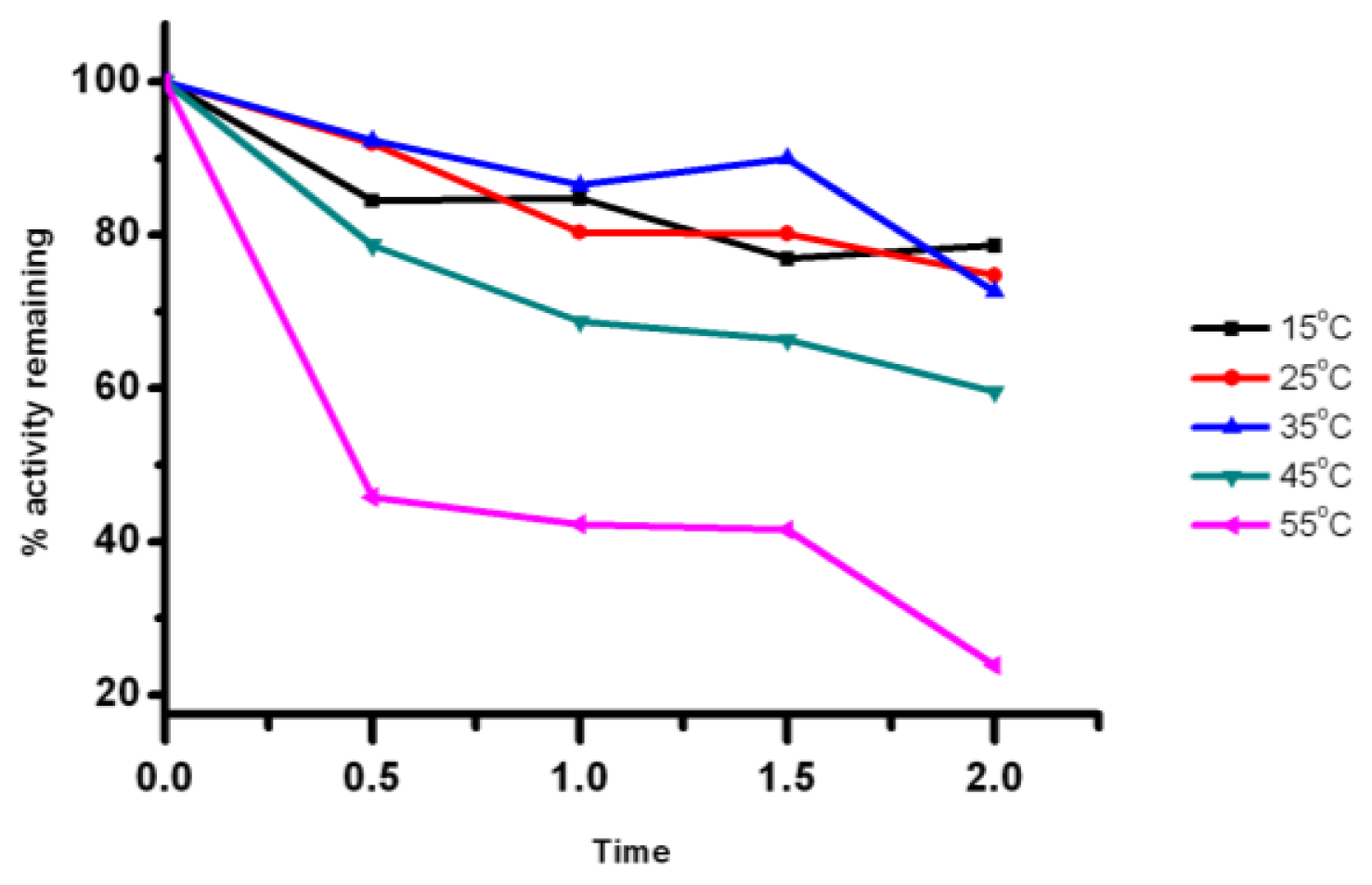
2.5. Effect of Temperature on the Lipase Activity and Stability
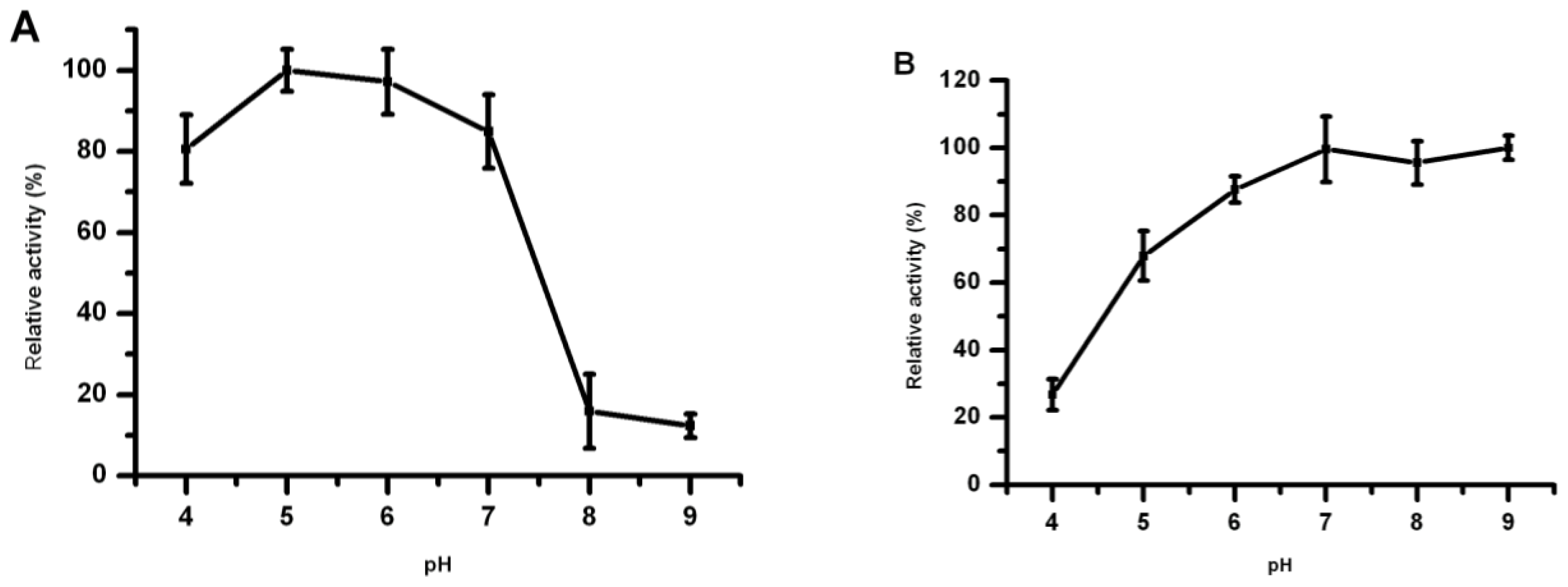
2.6. Effect of pH on Lip5-DM Activity and Stability
2.7. Effect of Metal Ions, Inhibitors, Detergents and Water-Miscible Solvent on the Activity of Lip5-DM
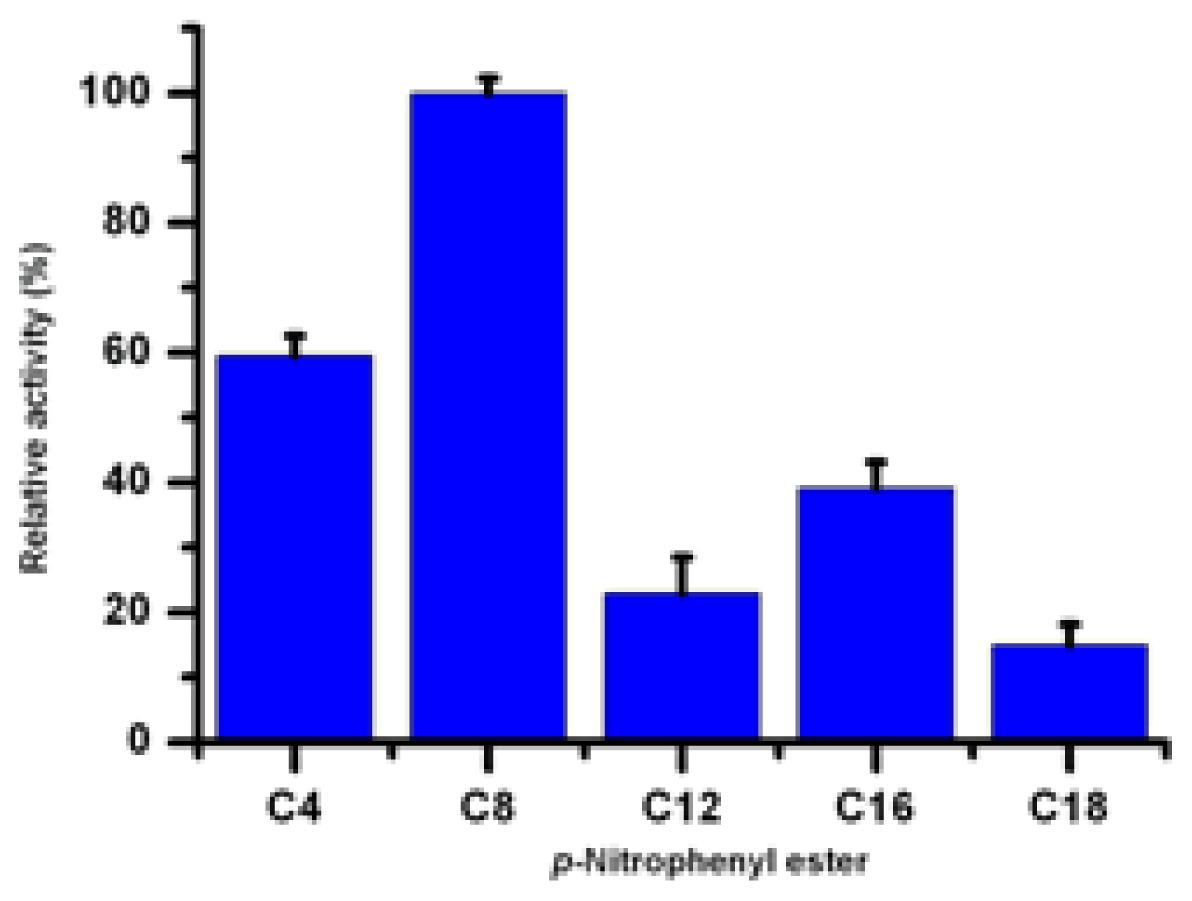
2.8. Substrate Specificity of Lip5-DM
3. Experimental Section
3.1. Strains, Plasmids and Materials
3.2. Cloning of lip5-wt and Sequence Analysis
3.3. Construction of the Mutant lip5 Gene Variants
3.4. Expression of lip5-wt, lip5-m154, lip5-m293 and lip5-dm
3.5. Purification of Recombinant Lip5-DM
3.6. Lipase Activity and Protein Analysis
3.7. Zymogram Analysis
3.8. Effect of Temperature on the Lip5-DM Activity and Stability
3.9. Effect of pH on the Lip5-DM Activity and Stability
3.10. Effect of Metal Ions, Inhibitor, Surfactants and Water-Miscible Solvent on the Lip5-DM Activity
3.11. Substrate Specificity
4. Conclusions
Acknowledgements
References
- Jaeger, KE; Eggert, T. Lipases for biotechnology. Curr. Opin. Biotechnol 2002, 13, 390–397. [Google Scholar]
- Schmitt-Rozieres, M; Deyris, V; Comeau, LC. Enrichment of polyunsaturated fatty acids from sardine cannery effluents by enzymatic selective esterification. J. Am. Oil Chem. Soc 2000, 77, 329–332. [Google Scholar]
- Lumor, SE; Jones, KC; Ashby, R; Strahan, GD; Kim, BH; Lee, GC; Shaw, JF; Kays, SE; Chang, SW; Foglia, TA; Akoh, CC. Synthesis and characterization of canola oil-stearic acid-based trans-free structured lipids for possible margarine application. J. Agric. Food Chem 2007, 55, 10692–10702. [Google Scholar]
- Kheadr, EE; Vuillemard, JC; El-Deeb, SA. Impact of liposome-encapsulated enzyme cocktails on cheddar cheese ripening. Food Res. Int 2003, 36, 241–252. [Google Scholar]
- Jaeger, KE; Reetz, MT. Microbial lipases form versatile tools for biotechnology. Trends Biotechnol 1998, 16, 396–403. [Google Scholar]
- Marshall, CJ. Cold-adapted enzymes. Trends Biotechnol 1997, 15, 359–364. [Google Scholar]
- Joseph, B; Ramteke, PW; Thomas, G. Cold active microbial lipases: Some hot issues and recent developments. Biotechnol. Adv 2008, 26, 457–470. [Google Scholar]
- Ryu, HS; Kim, HK; Choi, WC; Kim, MH; Park, SY; Han, NS; Oh, TK; Lee, JK. New cold-adapted lipase from Photobacterium lipolyticum sp nov that is closely related to filamentous fungal lipases. Appl. Microbiol. Biotechnol 2006, 70, 321–326. [Google Scholar]
- Jeon, JH; Kim, JT; Kim, YJ; Kim, HK; Lee, HS; Kang, SG; Kim, SJ; Lee, JH. Cloning and characterization of a new cold-active lipase from a deep-sea sediment metagenome. Appl. Microbiol. Biotechnol 2009, 81, 865–874. [Google Scholar]
- Parra, LP; Reyes, F; Acevedo, JP; Salazar, O; Andrews, BA; Asenjo, JA. Cloning and fusion expression of a cold-active lipase from marine Antarctic origin. Enzym. Microb. Tech 2008, 42, 371–377. [Google Scholar]
- Arpigny, JL; Lamotte, J; Gerday, C. Molecular adaptation to cold of an Antarctic bacterial lipase. J. Mol. Catal. B: Enzym 1997, 3, 29–35. [Google Scholar]
- Hube, B; Stehr, F; Bossenz, M; Mazur, A; Kretschmar, M; Schafer, W. Secreted lipases of Candida albicans: cloning, characterisation and expression analysis of a new gene family with at least ten members. Arch. Microbiol 2000, 174, 362–374. [Google Scholar]
- Santos, MAS; Keith, G; Tuite, MF. Nonstandard Translational Events in Candida-Albicans Mediated by an Unusual Seryl-Transfer Rna with a 5′-Cag-3′ (Leucine) Anticodon. EMBO J 1993, 12, 607–616. [Google Scholar]
- Ohama, T; Suzuki, T; Mori, M; Osawa, S; Ueda, T; Watanabe, K; Nakase, T. Nonuniversal decoding of the leucine codon cug in several candida species. Nucleic Acids Res 1993, 21, 4039–4045. [Google Scholar]
- Roustan, JL; Chu, AR; Moulin, G; Bigey, F. A novel lipase/acyltransferase from the yeast Candida albicans: Expression and characterisation of the recombinant enzyme. Appl. Microbiol. Biotechnol 2005, 68, 203–212. [Google Scholar]
- Gacser, A; Stehr, F; Kroger, C; Kredics, L; Schafer, W; Nosanchuk, JD. Lipase 8 affects the pathogenesis of Candida albicans. Infect. Immun 2007, 75, 4710–4718. [Google Scholar]
- Neugnot, V; Moulin, G; Dubreucq, E; Bigey, F. The lipase/acyltransferase from Candida parapsilosis-Molecular cloning and characterization of purified recombinant enzymes. Eur. J. Biochem 2002, 269, 1734–1745. [Google Scholar]
- Hegh, I; Patkar, S; Halkier, T; Hansen, MT. Two lipases from Candida antarctica: Cloning and expression in Aspergillus oryzae. Can. J. Bot 1995, 73, 869–875. [Google Scholar]
- Zhang, JW; Zeng, RY. Molecular cloning and expression of a cold-adapted lipase gene from an Antarctic deep sea psychrotrophic bacterium Pseudomonas sp. 7323. Mar. Biotechnol 2008, 10, 612–621. [Google Scholar]
- Santarossa, G; Lafranconi, PG; Alquati, C; DeGioia, L; Alberghina, L; Fantucci, P; Lotti, M. Mutations in the “lid” region affect chain length specificity and thermostability of a Pseudomonas fragi lipase. FEBS Lett 2005, 579, 2383–2386. [Google Scholar]
- Zhang, J; Lin, S; Zeng, RY. Cloning, expression, and characterization of a cold-adapted lipase gene from an Antarctic deep-sea psychrotrophic bacterium, Psychrobacter sp. 7195. J. Microbiol. Biotechnol 2007, 17, 604–610. [Google Scholar]
- Brady, L; Brzozowski, AM; Derewenda, ZS; Dodson, E; Dodson, G; Tolley, S; Turkenburg, JP; Christiansen, L; Hugejensen, B; Norskov, L; Thim, L; Menge, U. A Serine protease triad forms the catalytic center of a triacylglycerol lipase. Nature 1990, 343, 767–770. [Google Scholar]
- Tang, SJ; Sun, KH; Sun, GH; Chang, TY; Wu, WL; Lee, GC. A transformation system for the nonuniversal CUG(Ser) codon usage species Candida rugosa. J. Microbiol. Methods 2003, 52, 231–238. [Google Scholar]
- Tang, SJ; Shaw, JF; Sun, KH; Sun, GH; Chang, TY; Lin, CK; Lo, YC; Lee, GC. Recombinant expression and characterization of the Candida rugosa lip4 lipase in Pichia pastoris: Comparison of glycosylation, activity, and stability. Arch. Biochem. Biophys 2001, 387, 93–98. [Google Scholar]
- Tang, SJ; Sun, KH; Sun, GH; Chang, TY; Lee, GC. Recombinant expression of the Candida rugosa lip4 lipase in Escherichia coli. Protein Expr. Purif 2000, 20, 308–313. [Google Scholar]
- Brocca, S; Schmidt-Dannert, C; Lotti, M; Alberghina, L; Schmid, RD. Design, total synthesis, and functional overexpression of the Candida rugosa lip1 gene coding for a major industrial lipase. Protein Sci 1998, 7, 1415–1422. [Google Scholar]
- Chang, SW; Lee, GC; Shaw, JF. Efficient production of active recombinant Candida rugosa LIP3 lipase in Pichia pastoris and biochemical characterization of the purified enzyme. J. Agric. Food Chem 2006, 54, 5831–5838. [Google Scholar]
- Pfeffer, J; Richter, S; Nieveler, J; Hansen, CE; Rhlid, RB; Schmid, RD; Rusnak, M. High yield expression of lipase a from Candida antarctica in the methylotrophic yeast Pichia pastoris and its purification and characterisation. Appl. Microbiol. Biotechnol 2006, 72, 931–938. [Google Scholar]
- Gerday, C; Aittaleb, M; Arpigny, JL; Baise, E; Chessa, JP; Garsoux, G; Petrescu, I; Feller, G. Psychrophilic enzymes: A thermodynamic challenge. Biochim. Biophys. Acta Protein Struct. Mol. Enzymol 1997, 1342, 119–131. [Google Scholar]
- Feller, G; Narinx, E; Arpigny, JL; Aittaleb, M; Baise, E; Genicot, S; Gerday, C. Enzymes from psychrophilic organisms. FEMS Microbiol. Rev 1996, 18, 189–202. [Google Scholar]
- Choo, DW; Kurihara, T; Suzuki, T; Soda, K; Esaki, N. A cold-adapted lipase of an Alaskan psychrotroph, Pseudomonas sp. strain B11-1: Gene cloning and enzyme purification and characterization. Appl. Environ. Microbiol 1998, 64, 486–491. [Google Scholar]
- Mayordomo, I; Randez-Gil, F; Prieto, JA. Isolation, purification, and characterization of a cold-active lipase from Aspergillus nidulans. J. Agric. Food Chem 2000, 48, 105–109. [Google Scholar]
- Choi, WC; Kim, MH; Ro, HS; Ryu, SR; Oh, TK; Lee, JK. Zinc in lipase L1 from Geobacillus stearothermophilus L1 and structural implications on thermal stability. FEBS Lett 2005, 579, 3461–3466. [Google Scholar]
- Kiran, GS; Shanmughapriya, S; Jayalakshmi, J; Selvin, J; Gandhimathi, R; Sivaramakrishnan, S; Arunkumar, M; Thangavelu, T; Natarajaseenivasan, K. Optimization of extracellular psychrophilic alkaline lipase produced by marine Pseudomonas sp. (MSI057). Bioprocess Biosystems Eng 2008, 31, 483–492. [Google Scholar]
- Dong, H; Gao, SJ; Han, SP; Cao, SG. Purification and characterization of a Pseudomonas sp. lipase and its properties in non-aqueous media. Biotechnol. Appl. Biochem 1999, 30, 251–256. [Google Scholar]
- Kojima, Y; Shimizu, S. Purification and characterization of the lipase from Pseudomonas fluorescens HU380. J. Biosci. Bioeng 2003, 96, 219–226. [Google Scholar]
- Shimada, Y; Koga, C; Sugihara, A; Nagao, T; Takada, N; Tsunasawa, S; Tominaga, Y. Purification and characterization of a novel solvent-tolerant lipase from Fusarium heterosporum. J. Ferment. Bioeng 1993, 75, 349–352. [Google Scholar]
| Primers | Sequences a (5′-3′) |
|---|---|
| Lip5-FP | GGGGTACCGGCCTTATTTTCCCTACCAA |
| Lip5-RP | CCGCTCGAGTTATAACCACCTCATTTCAATTG |
| Lip5-CTG154-RP | GTAGATTTAGGTCCTTCATAATCAG |
| Lip5-CTG154-FP | GATTATGAAGGACCTAAATCTACATTCACTATTGGTAAACAATCAGG |
| Lip5-CTG293-RP | CTTATGATCACCAGTTAAGAAAGAAGTACCGATATAGTTGAGCACAC |
| Lip5-CTG293-FP | TCTTTCTTAACTGGTGATCATAAGA |
| Reagents | Relative activity (%)
| |
|---|---|---|
| 1 mM | 5 mM | |
| Control | 100 | 119.22 ± 6.61 |
| ZnSO4 | 100 | 115.19 ± 7.44 |
| CuSO4 | 95.02 ± 2.61 | 78.35 ± 8.35 |
| MgSO4 | 94.49 ± 7.85 | 97.66 ± 9.64 |
| FeCl3 | 64.01 ± 8.18 | 35.11 ± 0.51 |
| CaCl2 | 89.57 ± 8.32 | 72.04 ± 1.85 |
| MnSO4 | 111.44 ± 5.85 | 90.66 ± 2.38 |
| LiCl | 96.56 ± 8.31 | 92.24 ± 4.84 |
| Fe SO4 | 72.99 ± 4.89 | 20.27 ± 2.98 |
| HgCl2 | 18.67 ± 3.04 | 13.19 ± 6.60 |
| BaCl2 | 80.36 ± 6.84 | 77.22 ± 5.74 |
| NiCl2 | 96.56 ± 8.31 | 92.24 ± 4.84 |
| CoCl2 | 100.86 ± 9.73 | 89.10 ± 3.67 |
| EDTA | 71.86 ± 10.09 | 65.41 ± 2.58 |
| PMSF | 52.14 ± 2.63 | 38.54 ± 2.15 |
| Reagents | Relative activity (%)
| |
|---|---|---|
| 0.1% (w/v) | 30% (v/v) | |
| Control | 100 | 100 |
| Tween 20 | 82.52 ± 9.57 | |
| Triton X-100 | 48.55 ± 1.57 | |
| SDS | 15.10 ± 4.72 | |
| Tween 80 | 57.24 ± 4.17 | |
| Methanol | 48.23 ± 3.17 | |
| Ethanol | 24.32 ± 2.42 | |
| Acetone | 44.76 ± 3.59 | |
| Isopropanol | 4.59 ± 1.09 | |
| Substrate | Km (mM) | Vmax (mM/min) | Kcat (s−1) | Kcat/Km (s−1·mM−1) |
|---|---|---|---|---|
| p-NP-butyrate | 0.41 | 1.20 | 54.18 | 131.00 |
| p-NP-caprylate | 0.27 | 1.22 | 55.12 | 203.41 |
| p-NP-laurate | 0.50 | 0.20 | 9.41 | 18.58 |
| p-NP-palmitate | 0.36 | 0.70 | 31.59 | 86.86 |
© 2011 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lan, D.-M.; Yang, N.; Wang, W.-K.; Shen, Y.-F.; Yang, B.; Wang, Y.-H. A Novel Cold-Active Lipase from Candida albicans: Cloning, Expression and Characterization of the Recombinant Enzyme. Int. J. Mol. Sci. 2011, 12, 3950-3965. https://doi.org/10.3390/ijms12063950
Lan D-M, Yang N, Wang W-K, Shen Y-F, Yang B, Wang Y-H. A Novel Cold-Active Lipase from Candida albicans: Cloning, Expression and Characterization of the Recombinant Enzyme. International Journal of Molecular Sciences. 2011; 12(6):3950-3965. https://doi.org/10.3390/ijms12063950
Chicago/Turabian StyleLan, Dong-Ming, Ning Yang, Wen-Kai Wang, Yan-Fei Shen, Bo Yang, and Yong-Hua Wang. 2011. "A Novel Cold-Active Lipase from Candida albicans: Cloning, Expression and Characterization of the Recombinant Enzyme" International Journal of Molecular Sciences 12, no. 6: 3950-3965. https://doi.org/10.3390/ijms12063950
APA StyleLan, D.-M., Yang, N., Wang, W.-K., Shen, Y.-F., Yang, B., & Wang, Y.-H. (2011). A Novel Cold-Active Lipase from Candida albicans: Cloning, Expression and Characterization of the Recombinant Enzyme. International Journal of Molecular Sciences, 12(6), 3950-3965. https://doi.org/10.3390/ijms12063950




