Expression of an Endo-β-1,4-glucanase Gene from Orpinomyces PC-2 in Pichia pastoris
Abstract
:1. Introduction
2. Results and Discussion
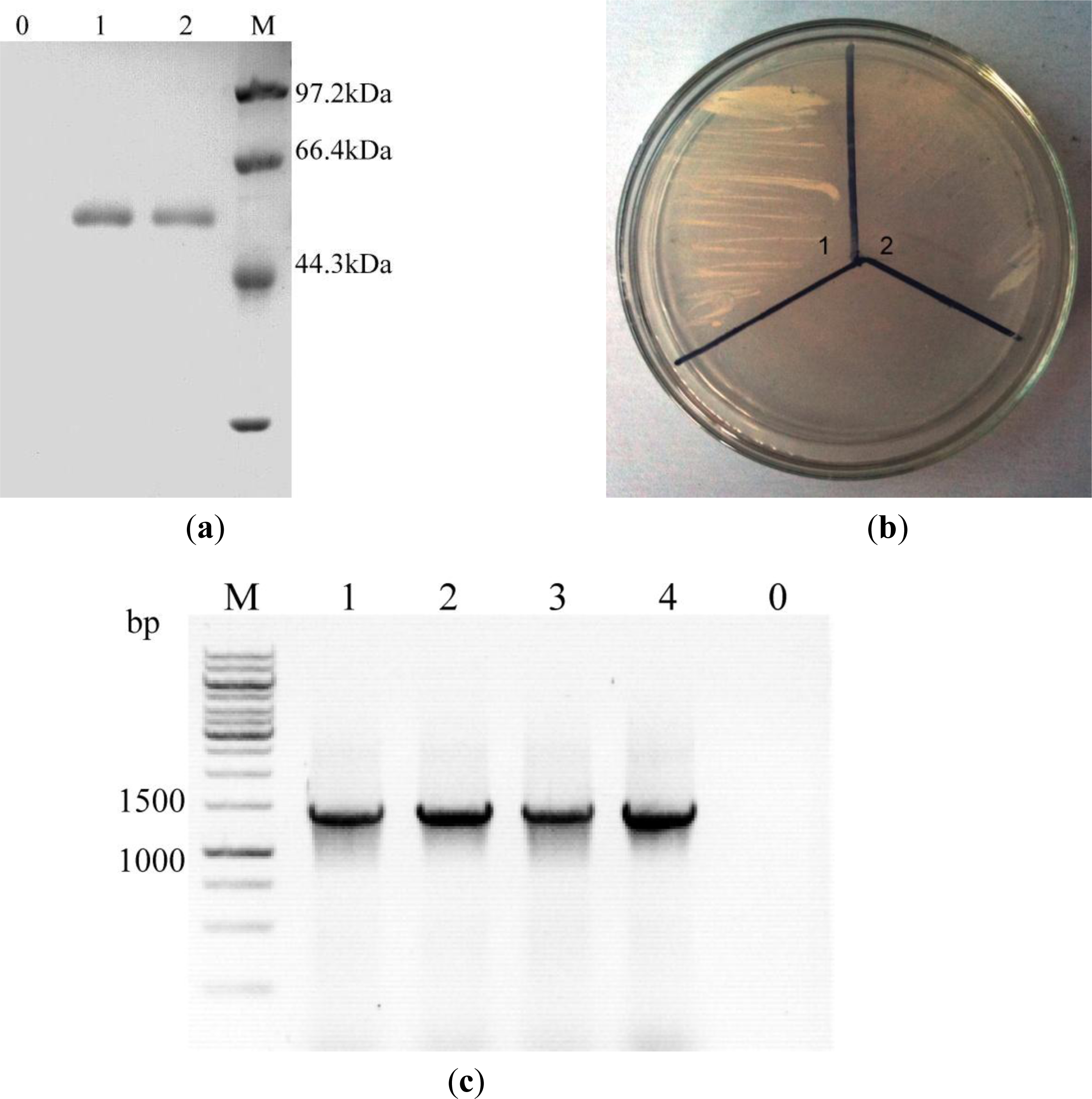
2.1. Expression of celE Gene in Recombinant P. Pastoris
2.2. Properties of Expressed Endo-β-1,4-glucanase (EG)
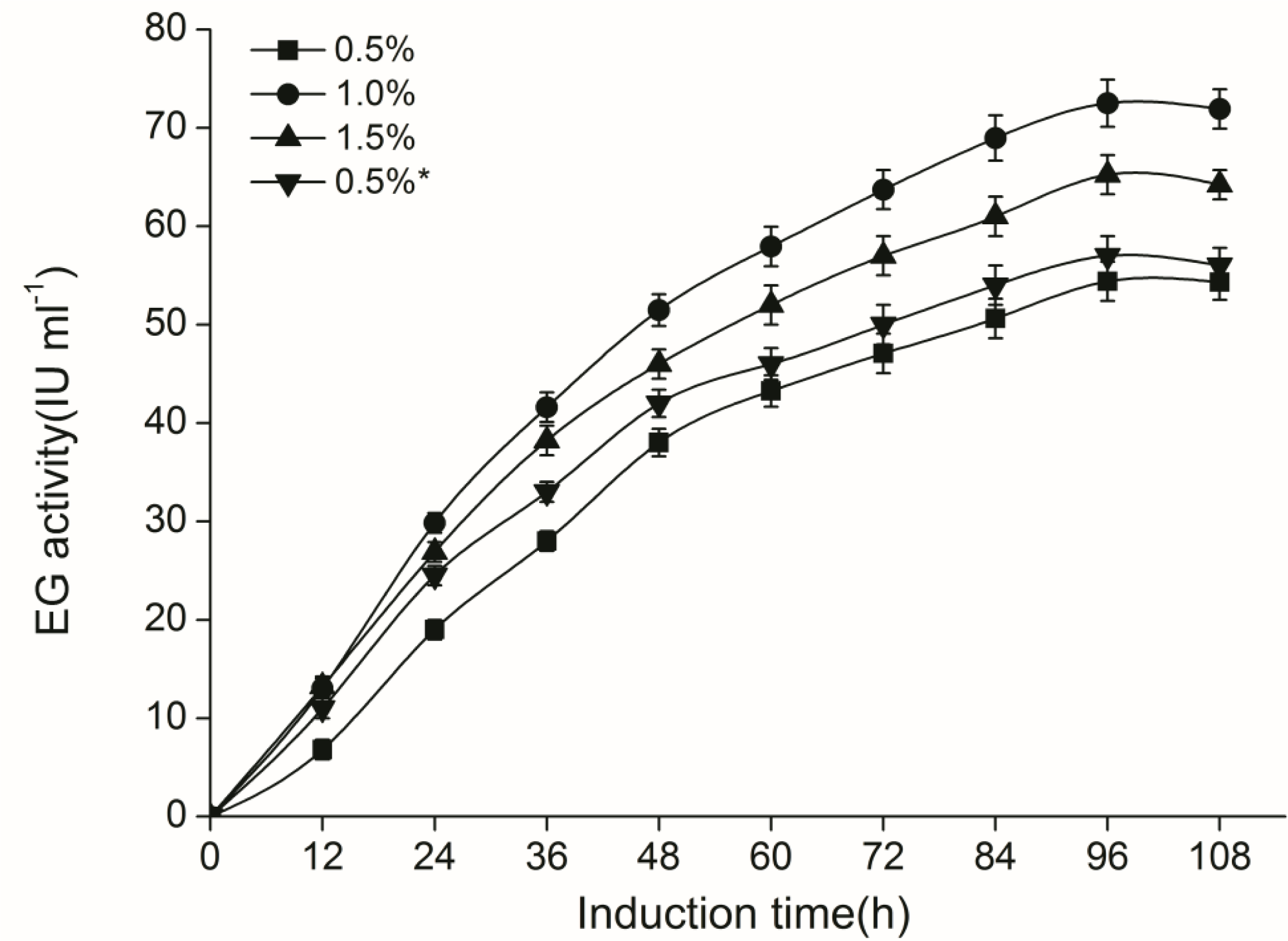
2.3. CelE Production by Transgenic P. Pastoris in Shaking Flasks
3. Experimental Section
3.1. Strains, Plasmids and Culture Medium
3.2. Chemicals and Enzymes
3.3. Recombinant DNA Techniques
3.4. Transformation and Transformants Screening
3.5. Sodium Dodecyl Sulphate-Polyacrylamide Gel Electrophoresis (SDS-PAGE)
3.6. Endo-β-1,4-glucanase Activity Assay
3.7. Optimal pH and Temperature Condition
3.8. Enzyme Production in Shaking Flask Culture
4. Conclusions
Acknowledgments
References
- Gu, FY; Chen, CY; Shi, JJ; Qian, SJ. Advances in cellulase and its development tendency. J. Microbiol 2008, 28, 83–87. [Google Scholar]
- Koga, J; Baba, Y; Shimonaka, A; Nishimura, T; Hanamura, S; Kono, T. Purification and characterization of a new family 45 endoglucanase, STCE1, from Staphylotrichum coccosporum and its overproduction in Humicola insolens. Appl. Environ. Microbiol 2008, 74, 4210–4217. [Google Scholar]
- Schulein, M. Enzymatic properties of cellulases from Humicola insolens. J. Biotechnol 1997, 57, 71–81. [Google Scholar]
- Ljungdahl, LG. The cellulase/hemicellulase system of the anaerobic fungus Orpinomyces PC-2 and aspects of its applied use. Ann. N.Y. Acad. Sci 2008, 1125, 308–321. [Google Scholar]
- Uhlig, H. Industrial Enzymes and Their Applications; John Wiley & Sons, Inc: New York, NY, USA, 1998. [Google Scholar]
- Zhang, YHP; Himmel, ME; Mielenz, JR. Outlook for cellulase improvement: Screening and selection strategies. Biotechnol. Adv 2006, 24, 452–481. [Google Scholar]
- Lenting, HBM; Warmoeskerken, MMCG. Guidelines to come to minimized tensile strength loss upon cellulase application. J. Biotechnol 2001, 89, 227–232. [Google Scholar]
- Saloheimo, A; Henrissat, B; Hoffren, AM; Teleman, O; Penttila, M. A novel, small endoglucanase gene, egl5, from Trichoderma reesei isolated by expression in yeast. Mol. Microbiol 1994, 13, 219–228. [Google Scholar]
- Okada, H; Tada, K; Sekiya, T; Yokoyama, K; Takahashi, A; Tohda, H; Kumagai, H; Morikawa, Y. Molecular characterization and heterologous expression of the gene encoding a low-molecular-mass endoglucanase from Trichoderma reesei QM9414. Appl. Environ. Microbiol 1998, 64, 555–563. [Google Scholar]
- Karlsson, J; Saloheimo, M; Siika-aho, M; Tenkanen, M; Penttila, M; Tjerneld, F. Homologous expression and characterization of Cel61A (EG IV) of Trichoderma reesei. Eur. J. Biochem 2001, 268, 6498–6507. [Google Scholar]
- Nakazawa, H; Okada, K; Kobayashi, R; Kubota, T; Onodera, T; Ochiai, N; Omata, N; Ogasawara, W; Okada, H; Morikawa, Y. Characterization of the catalytic domains of Trichoderma reesei endoglucanase I, II, and III, expressed in Escherichia coli. Appl. Microbiol. Biotechnol 2008, 81, 681–689. [Google Scholar]
- Fujii, T; Murakami, K; Sawayama, S. Cellulase hyperproducing mutants derived from the fungus Trichoderma reesei QM9414 produced large amounts of cellulase at the enzymatic and transcriptional levels. Biosci. Biotechnol. Biochem 2010, 74, 419–422. [Google Scholar]
- Anish, R; Rahman, MS; Rao, MA. Application of cellulases from an alkalothermophilic Thermomonospora sp in biopolishing of denims. Biotechnol. Bioeng 2007, 96, 48–56. [Google Scholar]
- Orpin, CG. Studies on rumen flagellate Neocallimastix frontalis. J. Gen. Microbiol 1975, 91, 249–262. [Google Scholar]
- Qiu, X; Selinger, B; Yanke, LJ; Cheng, KJ. Isolation and analysis of two cellulase cDNAs from Orpinomyces joyonii. Gene 2000, 245, 119–126. [Google Scholar]
- Comlekcioglu, U; Akyol, I; Ozkose, E; Kar, B; Ekinci, MS. Carboxymethylcellulase production by the anaerobic rumen fungus Neocallimastix sp GMLF7. Ann. Microbiol 2008, 58, 115–119. [Google Scholar]
- Tsai, CF; Qiu, X; Liu, JH. A comparative analysis of two cDNA clones of the cellulase gene family from anaerobic fungus Piromyces rhizinflata. Anaerobe 2003, 9, 131–140. [Google Scholar]
- Li, XL; Chen, HZ; Ljungdahl, LG. Two cellulases, CelA and CelC, from the polycentric anaerobic fungus Orpinomyces strain PC-2 contain N-terminal docking domains for a cellulase-hemicellulase complex. Appl. Environ. Microbiol 1997, 63, 4721–4728. [Google Scholar]
- Liu, JH; Selinger, LB; Hu, YJ; Moloney, MM; Cheng, KJ; Beauchemin, KA. An endoglucanase from the anaerobic fungus Orpinomyces joyonii: Characterization of the gene and its product. Can. J. Microbiol 1997, 43, 477–485. [Google Scholar]
- Chen, HZ; Li, XL; Blum, DL; Ljungdahl, LG. Two genes of the anaerobic fungus Orpinomyces sp. strain PC-2 encoding cellulases with endoglucanase activities may have arisen by gene duplication. FEMS Microbiol. Lett 1998, 159, 63–68. [Google Scholar]
- Fujino, Y; Ogata, K; Nagamine, T; Ushida, K. Cloning, sequencing, and expression of an endoglucanase gene from the rumen anaerobic fungus Neocallimastix frontalis MCH3. Biosci. Biotech. Bioch 1998, 62, 1795–1798. [Google Scholar]
- Chen, HZ; Li, XL; Blum, DL; Ximenes, EA; Ljungdahl, LG. CelF of Orpinomyces PC-2 has an intron and encodes a cellulase (CelF) containing a carbohydrate-binding module. Appl. Biochem. Biotech 2003, 105, 775–785. [Google Scholar]
- Krause, DO; Denman, SE; Mackie, RI; Morrison, M; Rae, AL; Attwood, GT; McSweeney, CS. Opportunities to improve fiber degradation in the rumen: Microbiology, ecology, and genomics. FEMS Microbiol. Rev 2003, 27, 663–693. [Google Scholar]
- Borneman, WS; Akin, DE; Ljungdahl, LG. Fermentation products and plant cell wall-degrading enzymes produced by monocentric and polycentric anaerobic ruminal fungi. Appl. Environ. Microbiol 1989, 55, 10–73. [Google Scholar]
- Gilbert, HJ; Hazlewood, GP; Laurie, JI; Orpin, CG; Xue, GP. Homologous catalytic domains in a rumen fungal xylanase: Evidence for gene duplication and prokaryotic origin. Mol. Microbiol 1992, 6, 2065–2072. [Google Scholar]
- Li, XL; Chen, HZ; Ljungdahl, LG. Monocentric and polycentric anaerobic fungi produce structurally related cellulases and xylanases. Appl. Environ. Microbiol 1997, 63, 628–635. [Google Scholar]
- Cereghino, JL; Cregg, JM. Heterologous protein expression in the methylotrophic yeast Pichia pastoris. FEMS Microbiol. Rev 2000, 24, 45–66. [Google Scholar]
- Cregg, JM; Cereghino, JL; Shi, JY; Higgins, DR. Recombinant protein expression in Pichia pastoris. Mol. Biotechnol 2000, 16, 23–52. [Google Scholar]
- Wonganu, B; Pootanakit, K; Boonyapakron, K; Champreda, V; Tanapongpipat, S; Eurwilaichitr, L. Cloning, expression and characterization of a thermotolerant endoglucanase from Syncephalastrum racemosum (BCC18080) in Pichia pastoris. Protein Expr. Purif 2008, 58, 78–86. [Google Scholar]
- Ding, SJ; Ge, W; Buswell, JA. Secretion, purification and characterisation of a recombinant Volvariella volvacea endoglucanase expressed in the yeast Pichia pastoris. Enzyme Microb. Technol 2002, 31, 621–626. [Google Scholar]
- Hong, J; Tamaki, H; Kumagai, H. Cloning and functional expression of thermostable beta-glucosidase gene from Thermoascus aurantiacus. Appl. Microbiol. Biotechnol 2007, 73, 1331–1339. [Google Scholar]
- Jeya, M; Thiagarajan, S; Lee, JK; Gunasekaran, P. Cloning and expression of GH11 xylanase gene from Aspergillus fumigatus MKU1 in Pichia pastoris. J. Biosci. Bioeng 2009, 108, 22–29. [Google Scholar]
- Qiao, JY; Rao, ZH; Dong, B; Cao, YH. Expression of Bacillus subtilis MA139 beta-mannanase in Pichia pastoris and the Enzyme Characterization. Appl. Biochem. Biotechnol 2010, 160, 1362–1370. [Google Scholar]
- Shi, BH; Zeng, LH; Song, HL; Shi, QQ; Wu, SG. Cloning and expression of Aspergillus tamarii FS132 lipase gene in Pichia pastoris. Int. J. Mol. Sci 2010, 11, 2373–2382. [Google Scholar]
- Cai, X; Wang, JF; Wang, YY; Yang, YC; Gao, J; Fu, WL; Wang, JX; Xu, DG. Expression, purification and characterization of recombinant human interleukin-22 in Pichia pastoris. Mol. Biol. Rep 2010, 37, 2609–2613. [Google Scholar]
- Fanutti, C; Ponyi, T; Black, GW; Hazlewood, GP; Gilbert, HJ. The conserved noncatalytic 40-residue sequence in cellulases and hemicellulases from anaerobic fungi functions as a protein docking domain. J. Biol. Chem 1995, 270, 29314–29322. [Google Scholar]
- Steenbakkers, PJM; Li, XL; Ximenes, EA; Arts, JG; Chen, HZ; Ljungdahl, LG; Den-Camp, H. Noncatalytic docking domains of cellulosomes of anaerobic fungi. J. Bacteriol 2001, 183, 5325–5333. [Google Scholar]
- Lo-Leggio, L; Larsen, S. The 1.62 angstrom structure of Thermoascus aurantiacus endoglucanase: Completing the structural picture of subfamilies in glycoside hydrolase family 5. FEBS Lett 2002, 523, 103–108. [Google Scholar]
- Zhang, W; Inan, M; Meagher, MM. Fermentation strategies for recombinant protein expression in the methylotrophic yeast Pichia pastoris. Biotechnol. Bioprocess Eng 2000, 5, 275–287. [Google Scholar]
- Minning, S; Serrano, A; Ferrer, P; Sola, C; Schmid, RD; Valero, F. Optimization of the high-level production of Rhizopus oryzae lipase in Pichia pastoris. J. Biotechnol 2001, 86, 59–70. [Google Scholar]
- Ding, SJ; Song, MJ; Yang, HJ; Xing, ZT; Zhou, R; Cao, J. High-level production of neutral endoglucanase 1 in Pichia pastoris. Sheng Wu Gong Cheng Xue Bao 2006, 22, 71–76. [Google Scholar]
- Faber, KN; Elgersma, Y; Heyman, JA; Koller, A; Luers, GH; Nuttley, WM; Terlecky, SR; Wenzel, TJ; Subramani, S. Use of Pichia pastoris as a model eukaryotic system. Peroxisome biogenesis. Methods. Mol. Biol 1998, 103, 121–147. [Google Scholar]
- Cos, O; Ramon, R; Montesinos, JL; Valero, F. Operational strategies, monitoring and control of heterologous protein production in the methylotrophic yeast Pichia pastoris under different promoters: A review. Microb. Cell. Fact 2006, 5, 17. [Google Scholar]
- Hong, F; Meinander, NQ; Jonsson, LJ. Fermentation strategies for improved heterologous expression of laccase in Pichia pastoris. Biotechnol. Bioeng 2002, 79, 438–449. [Google Scholar]
- Karhunen, T; Mantyla, A; Nevalainen, KM; Suominen, PL. High frequency one-step gene replacement in Trichoderma reesei. I. Endoglucanase I overproduction. Mol. Gen. Genet 1993, 241, 515–522. [Google Scholar]
- Miettinen-Oinonen, A; Suominen, P. Enhanced production of Trichoderma reesei endoglucanases and use of the new cellulase preparations in producing the stonewashed effect on denim fabric. Appl. Environ. Microbiol 2002, 68, 3956–3964. [Google Scholar]
- Qiao, Y; Mao, AJ; He, YZ; Liu, WF; Dong, ZY. Secrted expression of Trichoderma reesei endo-β-glucanase II gene in Pichia pastoris and analysis of enzymic properties. Mycosystema 2004, 23, 388–396. [Google Scholar]
- Liu, G; Tang, X; Tian, SL; Deng, X; Xing, M. Improvement of the cellulolytic activity of Trichoderma reesei endoglucanase IV with an additional catalytic domain. World J. Microb. Biot 2006, 22, 1301–1305. [Google Scholar]
- Thongekkaew, J; Ikeda, H; Masaki, K; Iefuji, H. An acidic and thermostable carboxymethyl cellulase from the yeast Cryptococcus sp S-2: Purification, characterization and improvement of its recombinant enzyme production by high cell-density fermentation of Pichia pastoris. Protein Expr. Purif 2008, 60, 140–146. [Google Scholar]
- Ding, SJ; Ge, W; Buswell, JA. Endoglucanase I from the edible straw mushroom, Volvariella volvacea–Purification, characterization, cloning and expression. Eur. J. Biochem 2001, 268, 5687–5695. [Google Scholar]
- Chen, H; Li, XL; Ljungdahl, LG. Sequencing of a 1,3-1,4-beta-D-glucanase (lichenase) from the anaerobic fungus Orpinomyces strain PC-2: Properties of the enzyme expressed in Escherichia coli and evidence that the gene has a bacterial origin. J. Bacteriol 1997, 179, 6028–6034. [Google Scholar]
- Chen, CC; Wu, PH; Huang, CT; Cheng, KJ. A Pichia pastoris fermentation strategy for enhancing the heterologous expression of an Escherichia coli phytase. Enzyme Microb. Technol 2004, 35, 315–320. [Google Scholar]
- Rozman, D; Komel, R. Isolation of genomic DNA from filamentous fungi with high glucan level. BioTechniques 1994, 16, 382–384. [Google Scholar]
- Miller, GL. Use of dinitrosalicylic acid reagent for the determination of reducing sugar. Anal. Chem 1959, 31, 426–428. [Google Scholar]
- Lee, KM; Jeya, M; Joo, AR; Singh, R; Kim, IW; Lee, JK. Purification and characterization of a thermostable endo-beta-1,4-glucanase from a novel strain of Penicillium purpurogenum. Enzyme Microb. Technol 2010, 46, 206–211. [Google Scholar]
- Fu, XY; Liu, PF; Lin, L; Hong, YZ; Huang, XL; Meng, X; Liu, ZD. A Novel Endoglucanase (Cel9P) From a marine bacterium Paenibacillus sp BME-14. Appl. Biochem. Biotechnol 2010, 160, 1627–1636. [Google Scholar]
- Eberhardt, RY; Gilbert, HJ; Hazlewood, GP. Primary sequence and enzymic properties of two modular endoglucanases, Cel5A and Cel45A, from the anaerobic fungus Piromyces equi. Microbiology 2000, 146, 1999–2008. [Google Scholar]


| Media | Composition |
|---|---|
| BMGY | 1% Glycerol, 1% yeast extract (YE), 2% peptone, 0.34% yeast nitrogen base without amino acids (YNB), 1% (NH4)2SO4 and 4 × 10−5% biotin in pH 6.0 100 mM potassium phosphate buffer |
| BMMY | 0.5% Methanol, 1% YE, 2% peptone, 0.34% YNB, 1% (NH4)2SO4 and 4 × 10−5% biotin in 100 mM potassium phosphate buffer |
| FBSH | 26.7 mL/L H3PO4 (85% stock), 0.93 g/L CaSO4, 18.2 g/L K2SO4, 14.9 g/L MgSO4·7H2O, 4.13 g/L KOH and adjusted to pH 5.0 by NH4OH. Add 0.004% histidine and 0.435 mL of PTM1 trace metal solution (6 g/L CuSO4·5H2O, 0.08 g/L KI, 3 g/L MgSO4·H2O, 0.2 g/L Na2MoO4, 0.02 g/L H3BO3, 0.5 g/L CoCl2, 20 g/L ZnCl2, 65 g/L FeSO4·7H2O, 0.2 g/L biotin and 5 mL H2SO4) |
| BCG | 1% corn steep liquor, 2% glucose in 100 mM potassium phosphate buffer |
| YPD | 1% yeast extract, 2% peptone, 2% glucose, add 2% agar powder when YPD plate prepared |
| YSM | 0.67% YNB, 2% Sodium carboxymethyl cellulose (CMC-Na), 0.5% methanol, 2% agar powder, pH 6.0 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jin, X.; Meng, N.; Xia, L.-m. Expression of an Endo-β-1,4-glucanase Gene from Orpinomyces PC-2 in Pichia pastoris. Int. J. Mol. Sci. 2011, 12, 3366-3380. https://doi.org/10.3390/ijms12053366
Jin X, Meng N, Xia L-m. Expression of an Endo-β-1,4-glucanase Gene from Orpinomyces PC-2 in Pichia pastoris. International Journal of Molecular Sciences. 2011; 12(5):3366-3380. https://doi.org/10.3390/ijms12053366
Chicago/Turabian StyleJin, Xin, Nan Meng, and Li-ming Xia. 2011. "Expression of an Endo-β-1,4-glucanase Gene from Orpinomyces PC-2 in Pichia pastoris" International Journal of Molecular Sciences 12, no. 5: 3366-3380. https://doi.org/10.3390/ijms12053366




