YueF Overexpression Inhibits Cell Proliferation Partly through p21WAF1/Cip1 Upregulation in Renal Cell Carcinoma
Abstract
:1. Introduction
2. Materials and Methods
2.1. Human Tissue Preparation
2.2. Generation of Recombinant Lentivirus
2.3. Cell Culture and Lentivirus Transduction
2.4. Cell Proliferation Assay
2.5. p21 siRNA Transfection
2.6. Western Blot Analysis
2.7. Cell Cycle Analysis
2.8. Statistical Analysis
3. Results
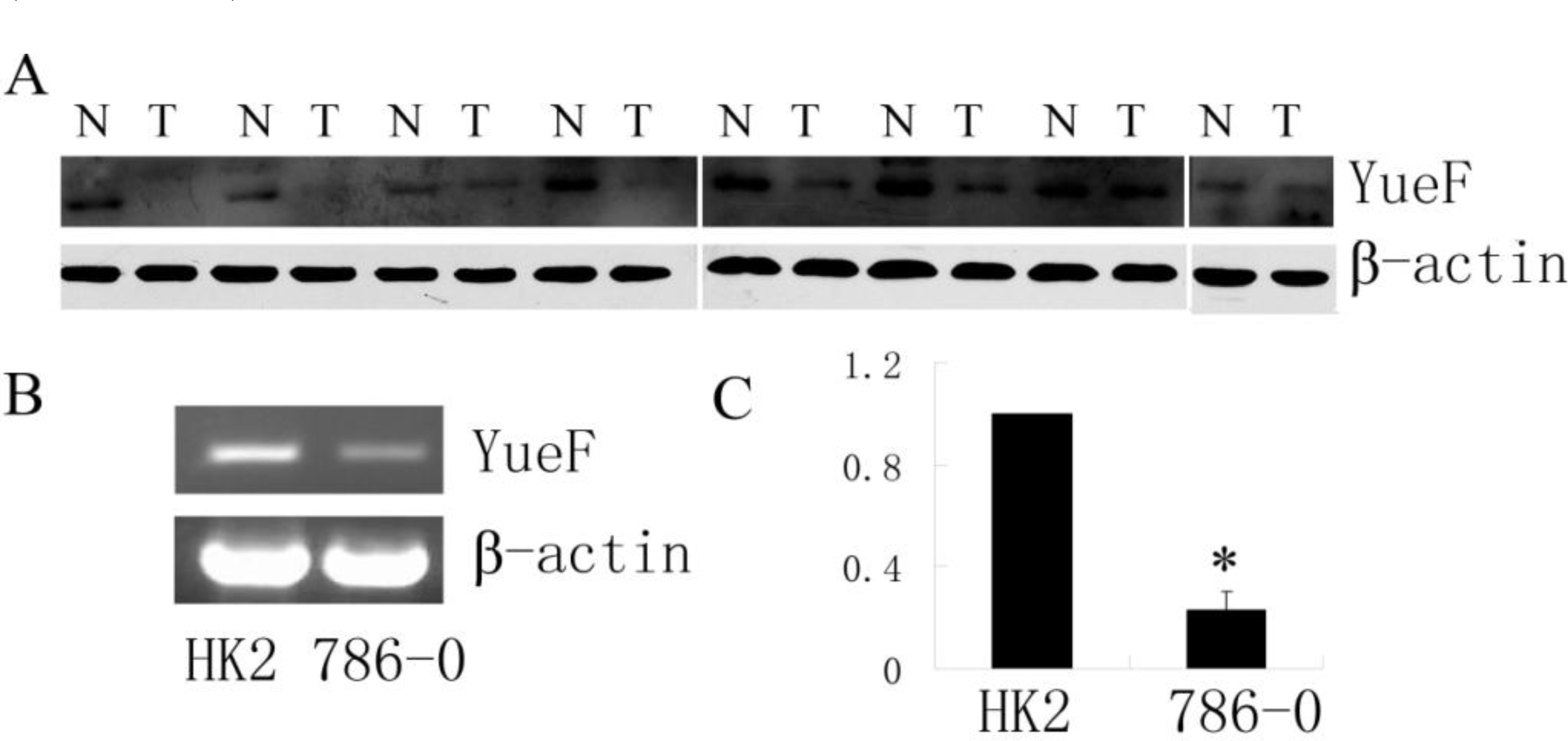
3.1. Downregulated Expression of YueF Protein in Clinical RCC Tissues and RCC 786-0 Cells
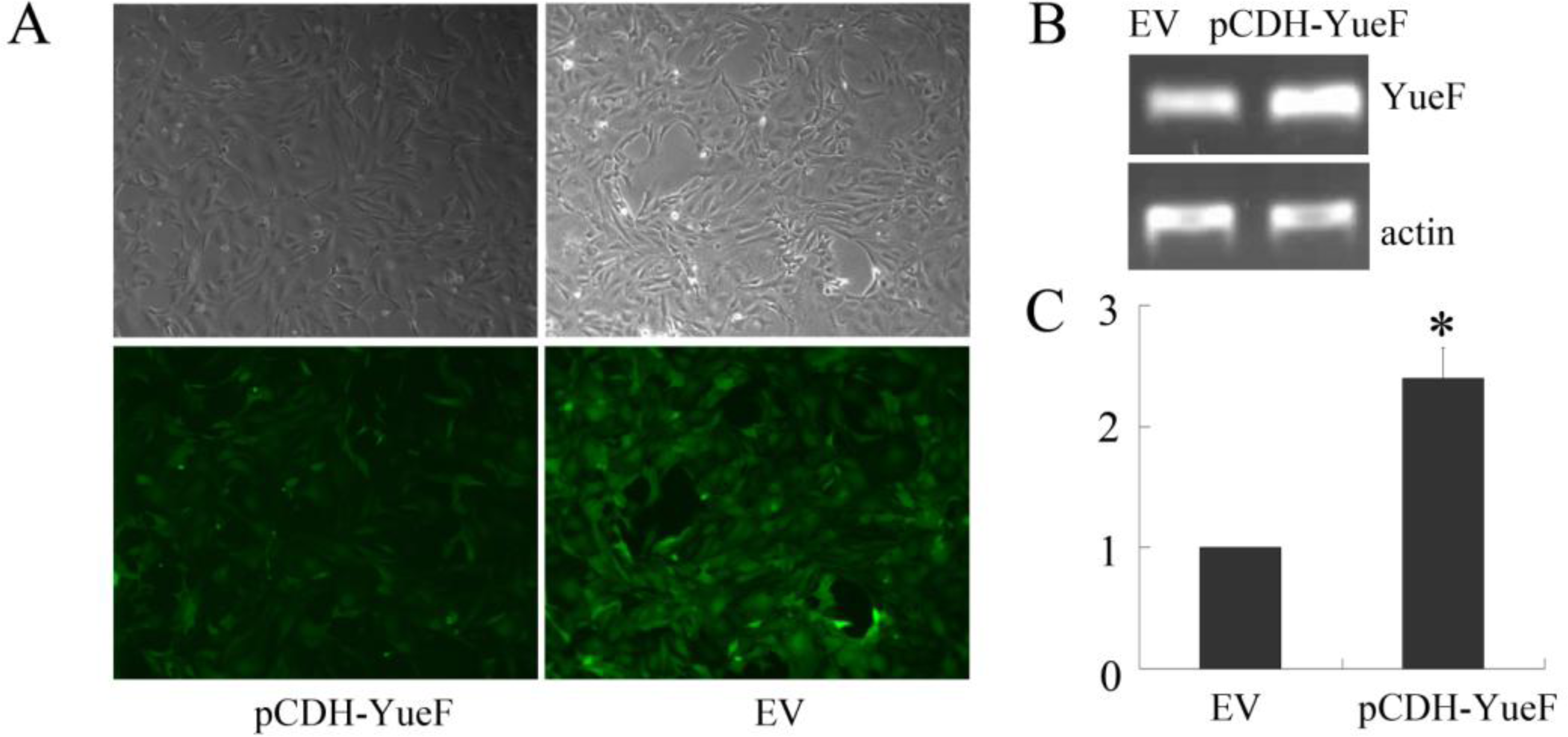
3.2. Overexpression of YueF in RCC 786-0 Cells with Lentivirus
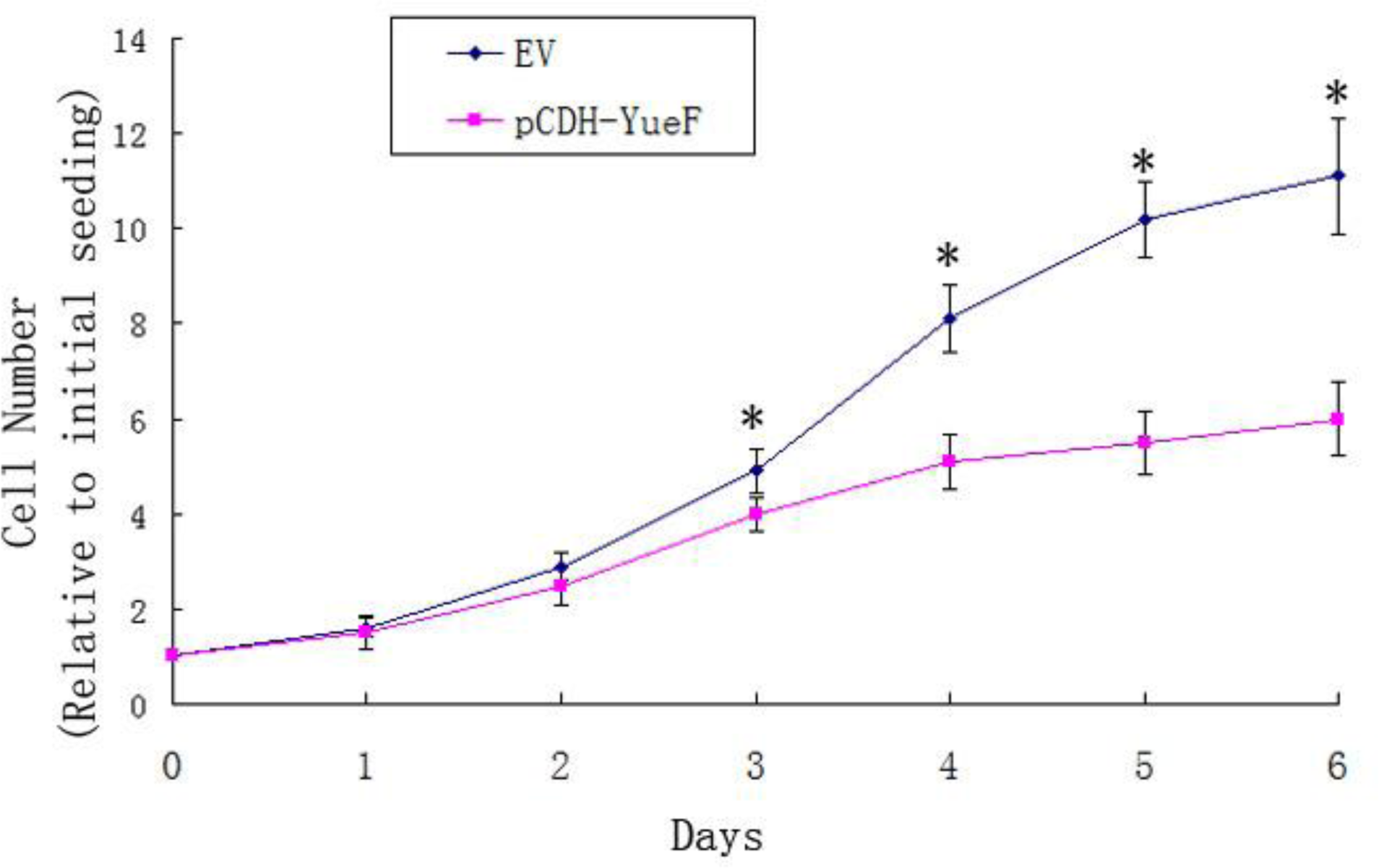
3.3. YueF Overexpression Inhibits the Proliferation of 786-0 Cells
3.4. YueF Overexpression in 786-0 Cells Causes Cell Cycle Arrest at the G1 Phase
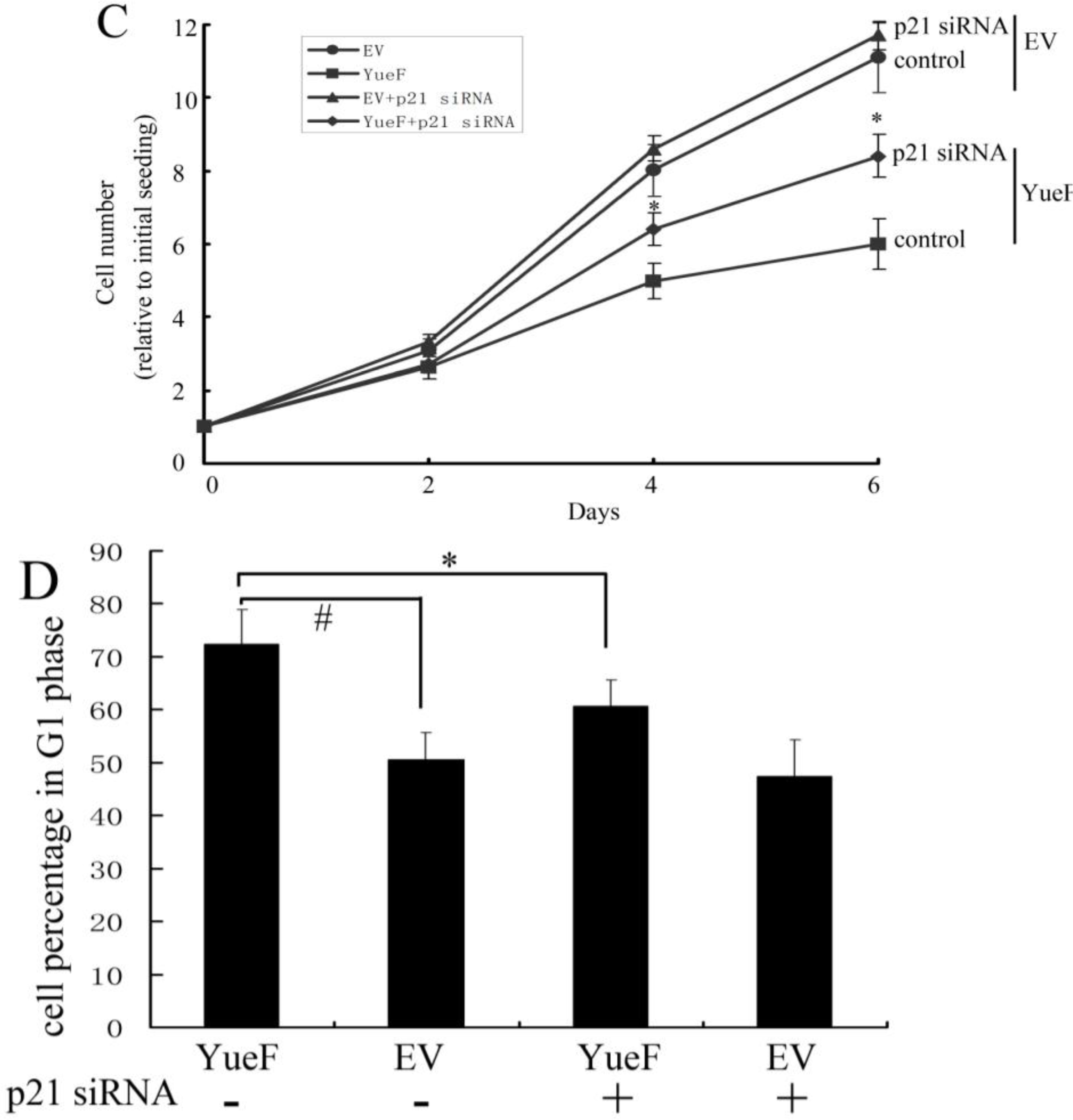
3.5. YueF Overexpression Inhibits the Proliferation of 786-0 Cells Partly through p21 Upregulation
4. Discussion
5. Conclusion
Acknowledgments
References
- Noon, AP; Vlatkovic, N; Polanski, R; Maguire, M; Shawki, H; Parsons, K; Boyd, MT. p53 and MDM2 in renal cell carcinoma: biomarkers for disease progression and future therapeutic targets? Cancer 2010, 116, 780–790. [Google Scholar]
- Balabanov, S; Zimmermann, U; Protzel, C; Scharf, C; Klebingat, KJ; Walther, R. Tumour-related enzyme alterations in the clear cell type of human renal cell carcinoma identified by two-dimensional gel electrophoresis. Eur. J. Biochem 2001, 268, 5977–5980. [Google Scholar]
- Sandim, V; Pereira, DA; Ornellas, AA; Alves, G. Renal cell carcinoma and proteomics. Urol. Int 2010, 84, 373–377. [Google Scholar]
- Zhang, JL; Zhao, WG; Wu, KL; Wang, K; Zhang, X; Gu, CF; Li, Y; Zhu, Y; Wu, JG. Human hepatitis B virus X protein promotes cell proliferation and inhibits cell apoptosis through interacting with a serine protease Hepsin. Arch. Virol 2005, 150, 721–741. [Google Scholar]
- Huang, J; Wu, K; Zhang, J; Si, W; Zhu, Y; Wu, J. Putative tumor suppressor YueF affects the functions of hepatitis B virus X protein in hepatoma cell apoptosis and p53 expression. Biotechnol. Lett 2008, 30, 235–242. [Google Scholar]
- Tong, Z; Chakraborty, S; Sung, B; Koolwal, P; Kaur, S; Aggarwal, BB; Mani, SA; Bresalier, RS; Batra, SK; Guha, S. Epidermal growth factor down-regulates the expression of neutrophil gelatinase-associated lipocalin (NGAL) through E-cadherin in pancreatic cancer cells. Cancer, 2010. [Google Scholar] [CrossRef]
- Volakaki, AA; Lafkas, D; Kassi, E; Schally, AV; Papavassiliou, AG; Kiaris, H. Essential role of p21/waf1 in the mediation of the anti-proliferative effects of GHRH antagonist JMR-132. J. Mol. Endocrinol 2008, 41, 389–392. [Google Scholar]
- el-Deiry, WS; Tokino, T; Velculescu, VE; Levy, DB; Parsons, R; Trent, JM; Lin, D; Mercer, WE; Kinzler, KW; Vogelstein, B. WAF1, a potential mediator of p53 tumor suppression. Cell 1993, 75, 817–825. [Google Scholar]
- Giono, LE; Manfredi, JJ. The p53 tumor suppressor participates in multiple cell cycle checkpoints. J. Cell. Physiol 2006, 209, 13–20. [Google Scholar]
- Innocente, SA; Abrahamson, JL; Cogswell, JP; Lee, JM. p53 regulates a G2 checkpoint through cyclin B1. Proc. Natl. Acad. Sci. USA 1999, 96, 2147–2152. [Google Scholar]
- Yu, J; Zhang, L; Hwang, PM; Rago, C; Kinzler, KW; Vogelstein, B. Identification and classification of p53-regulated genes. Proc. Natl. Acad. Sci. USA 1999, 96, 14517–14522. [Google Scholar]
- Oda, E; Ohki, R; Murasawa, H; Nemoto, J; Shibue, T; Yamashita, T; Tokino, T; Taniguchi, T; Tanaka, N. Noxa, a BH3-only member of the Bcl-2 family and candidate mediator of p53-induced apoptosis. Science 2000, 288, 1053–1058. [Google Scholar]
- Vousden, KH. p53: death star. Cell 2000, 103, 691–694. [Google Scholar]
- Guo, Y; Schoell, MC; Freeman, RS. The von Hippel-Lindau protein sensitizes renal carcinoma cells to apoptotic stimuli through stabilization of BIM(EL). Oncogene 2009, 28, 1864–1874. [Google Scholar]
- Qi, H; Ohh, M. The von Hippel-Lindau tumor suppressor protein sensitizes renal cell carcinoma cells to tumor necrosis factor-induced cytotoxicity by suppressing the nuclear factor-kappaB-dependent antiapoptotic pathway. Cancer Res 2003, 63, 7076–7080. [Google Scholar]
- Cheng, WL; Lin, TY; Tseng, YH; Chu, FH; Chueh, PJ; Kuo, YH; Wang, SY. Inhibitory Effect of Human Breast Cancer Cell Proliferation via p21-Mediated G1 Cell Cycle Arrest by Araliadiol Isolated from Aralia cordata Thunb. Planta Med 2010, 77, 164–168. [Google Scholar]
- Gartel, AL; Radhakrishnan, SK. Lost in transcription: p21 repression, mechanisms, and consequences. Cancer Res 2005, 65, 3980–3985. [Google Scholar]
- Korenjak, M; Brehm, A. E2F-Rb complexes regulating transcription of genes important for differentiation and development. Curr. Opin. Genet. Dev 2005, 15, 520–527. [Google Scholar]
- Harbour, JW; Dean, DC. Rb function in cell-cycle regulation and apoptosis. Nat. Cell Biol 2000, 2, E65–E67. [Google Scholar]
- Vermeulen, K; van Bockstaele, DR; Berneman, ZN. The cell cycle: a review of regulation, deregulation and therapeutic targets in cancer. Cell Prolif 2003, 36, 131–149. [Google Scholar]
- Morgan, DO. Principles of CDK regulation. Nature 1995, 374, 131–134. [Google Scholar]
- Ishino, K; Ohba, M; Kashiwagi, M; Kawabe, S; Chida, K; Kuroki, T. Phorbol ester-induced G1 arrest in BALB/MK-2 mouse keratinocytes is mediated by delta and eta isoforms of protein kinase C. Jpn. J. Cancer Res 1998, 89, 1126–1133. [Google Scholar]
- Guo, K; Andres, V; Walsh, K. Nitric oxide-induced downregulation of Cdk2 activity and cyclin A gene transcription in vascular smooth muscle cells. Circulation 1998, 97, 2066–2072. [Google Scholar]
- Thut, CJ; Goodrich, JA; Tjian, R. Repression of p53-mediated transcription by MDM2: a dual mechanism. Genes Dev 1997, 11, 1974–1986. [Google Scholar]


© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Huang, H.-W.; Peng, J.-P.; Zhang, J. YueF Overexpression Inhibits Cell Proliferation Partly through p21WAF1/Cip1 Upregulation in Renal Cell Carcinoma. Int. J. Mol. Sci. 2011, 12, 2477-2487. https://doi.org/10.3390/ijms12042477
Huang H-W, Peng J-P, Zhang J. YueF Overexpression Inhibits Cell Proliferation Partly through p21WAF1/Cip1 Upregulation in Renal Cell Carcinoma. International Journal of Molecular Sciences. 2011; 12(4):2477-2487. https://doi.org/10.3390/ijms12042477
Chicago/Turabian StyleHuang, Hsuan-Wei, Jian-Ping Peng, and Jie Zhang. 2011. "YueF Overexpression Inhibits Cell Proliferation Partly through p21WAF1/Cip1 Upregulation in Renal Cell Carcinoma" International Journal of Molecular Sciences 12, no. 4: 2477-2487. https://doi.org/10.3390/ijms12042477



