Lead, Cadmium and Cobalt (Pb, Cd, and Co) Leaching of Glass-Clay Containers by pH Effect of Food
Abstract
:1. Introduction
2. Experimental Section
2.1. Samples
2.2. Preparation of Food
2.3. Leaching of Heavy Metals into Food
2.4. Heavy-Metal Leaching Effect in Glass-Clay Containers with Acetic Acid
2.5. Extraction of Metals in Clays and Enamels Used in the Manufacturing of the Containers
2.6. Digesting of Samples
2.7. Quantification of Cd, Co, and Pb
2.8. Data Analysis
3. Results
3.1. Quantifying Cd, Co, and Pb in Enamels and Clays Used for the Manufacture of Glass-Clay Containers
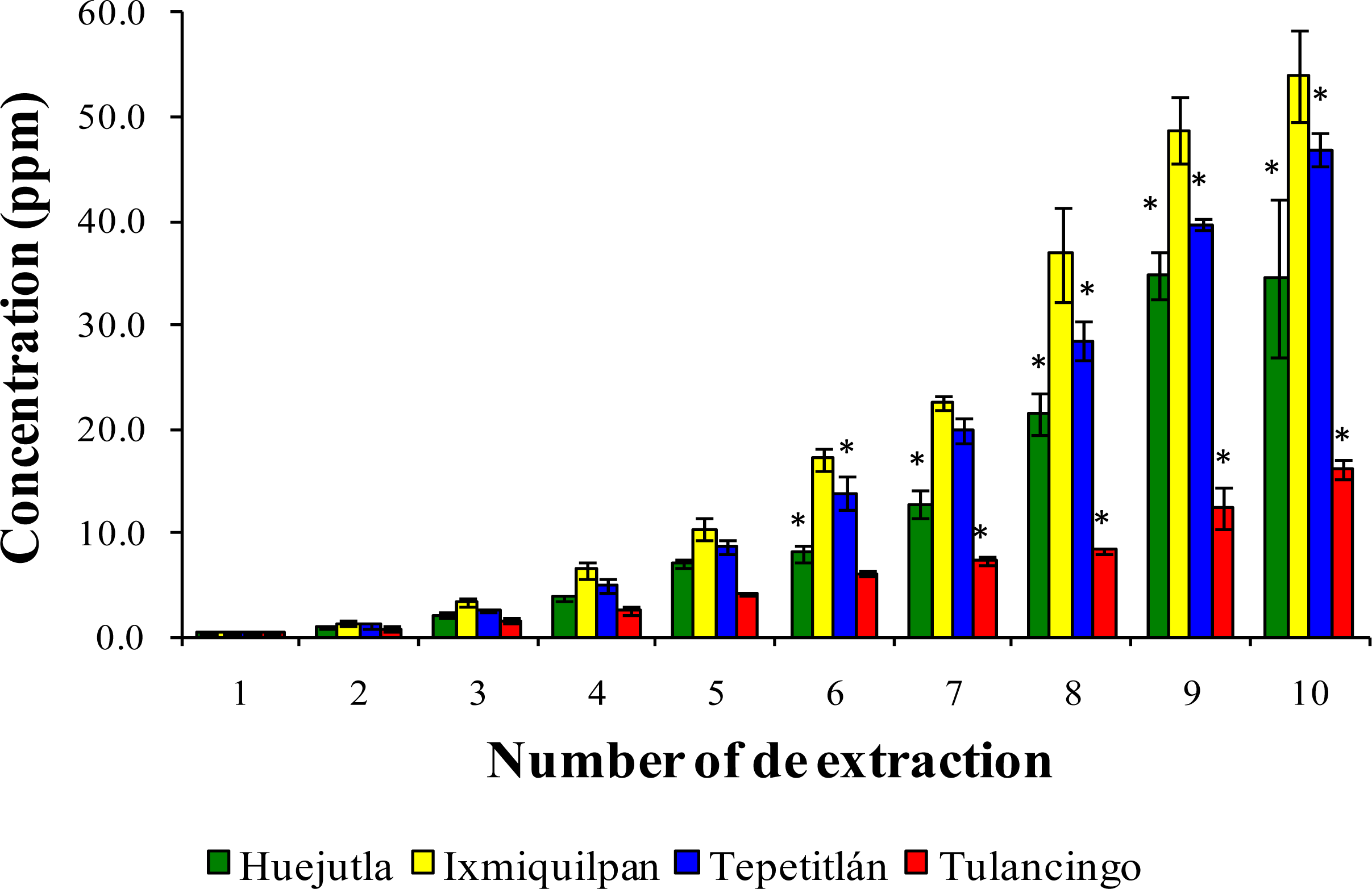
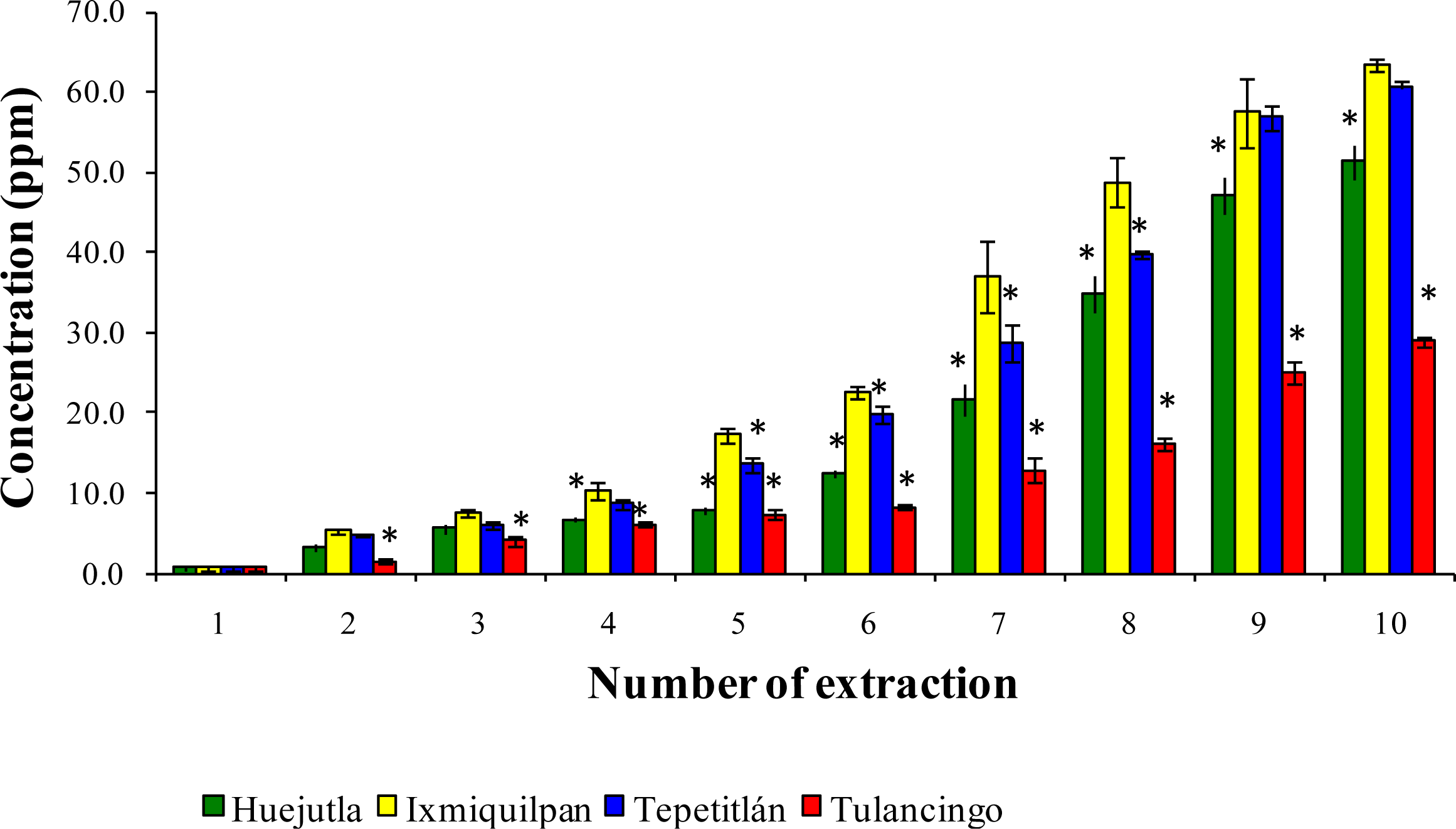
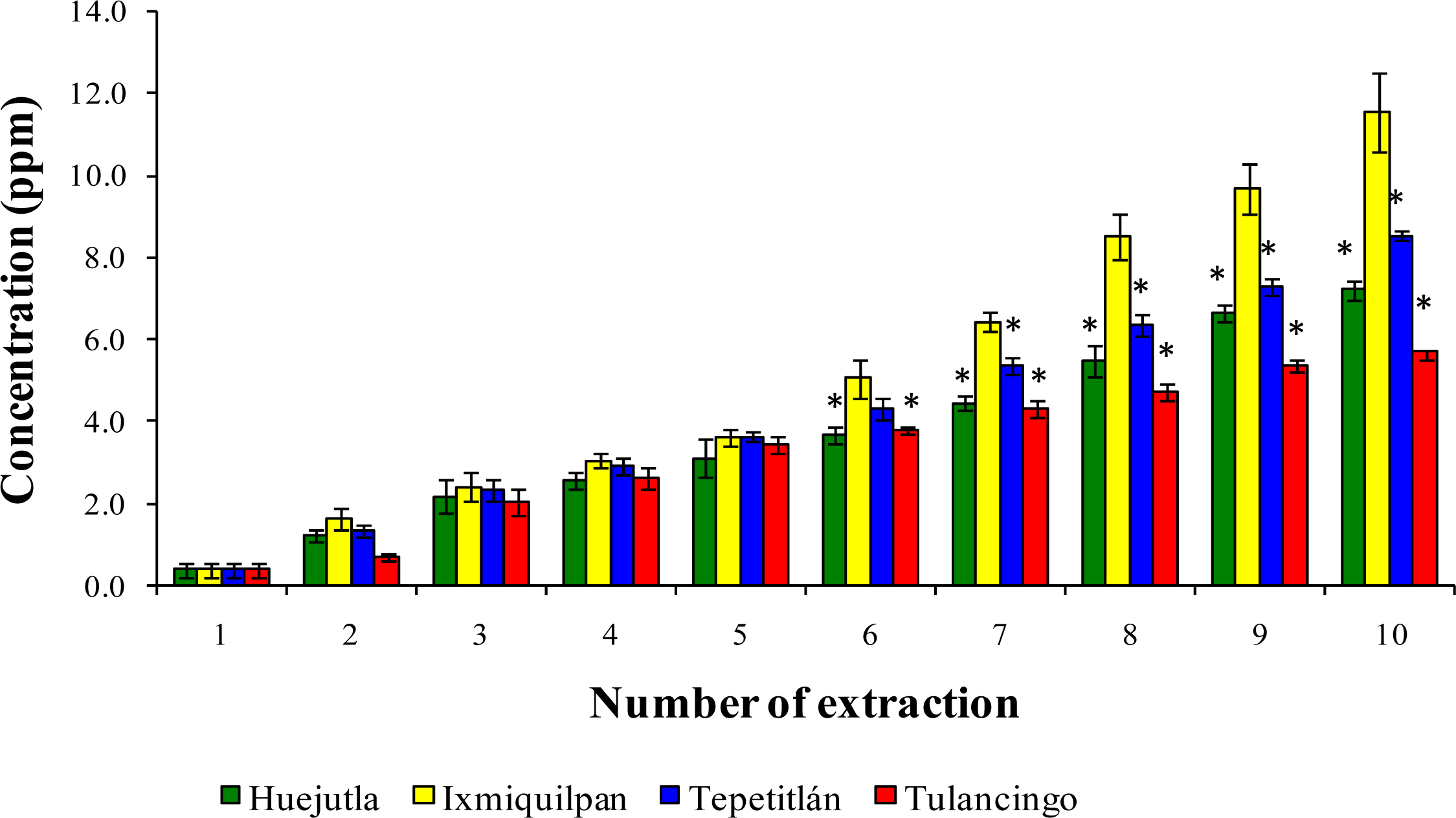
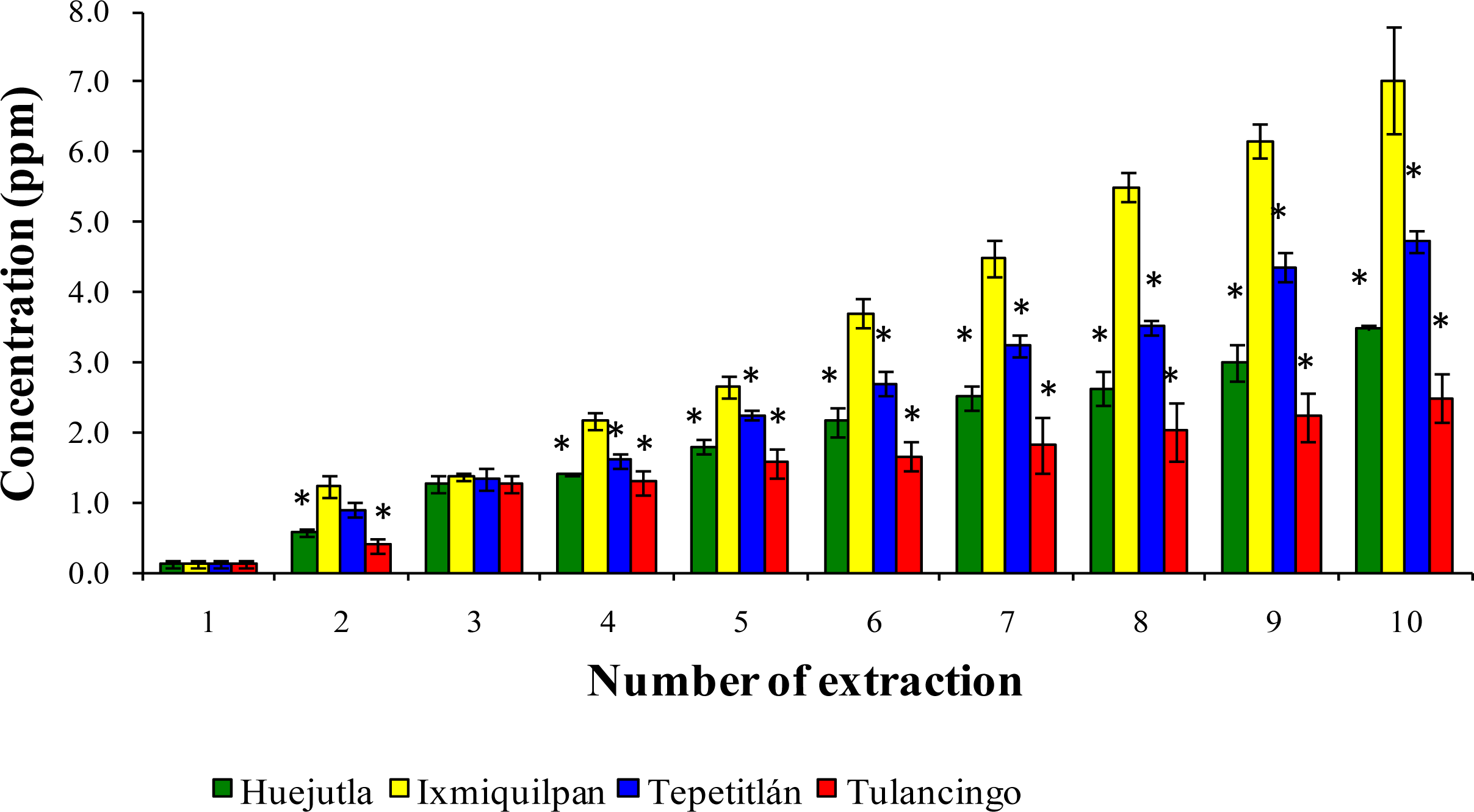
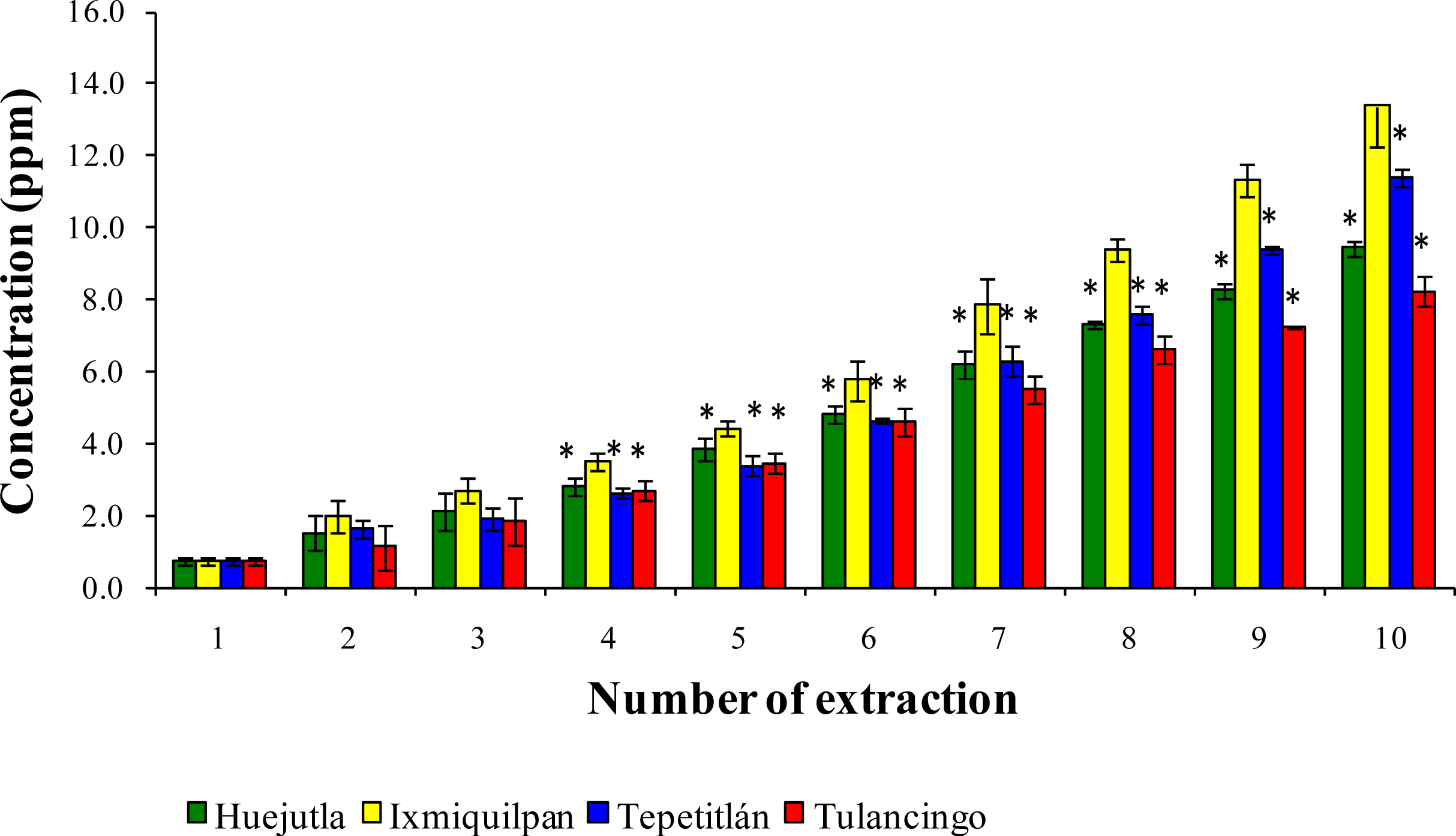
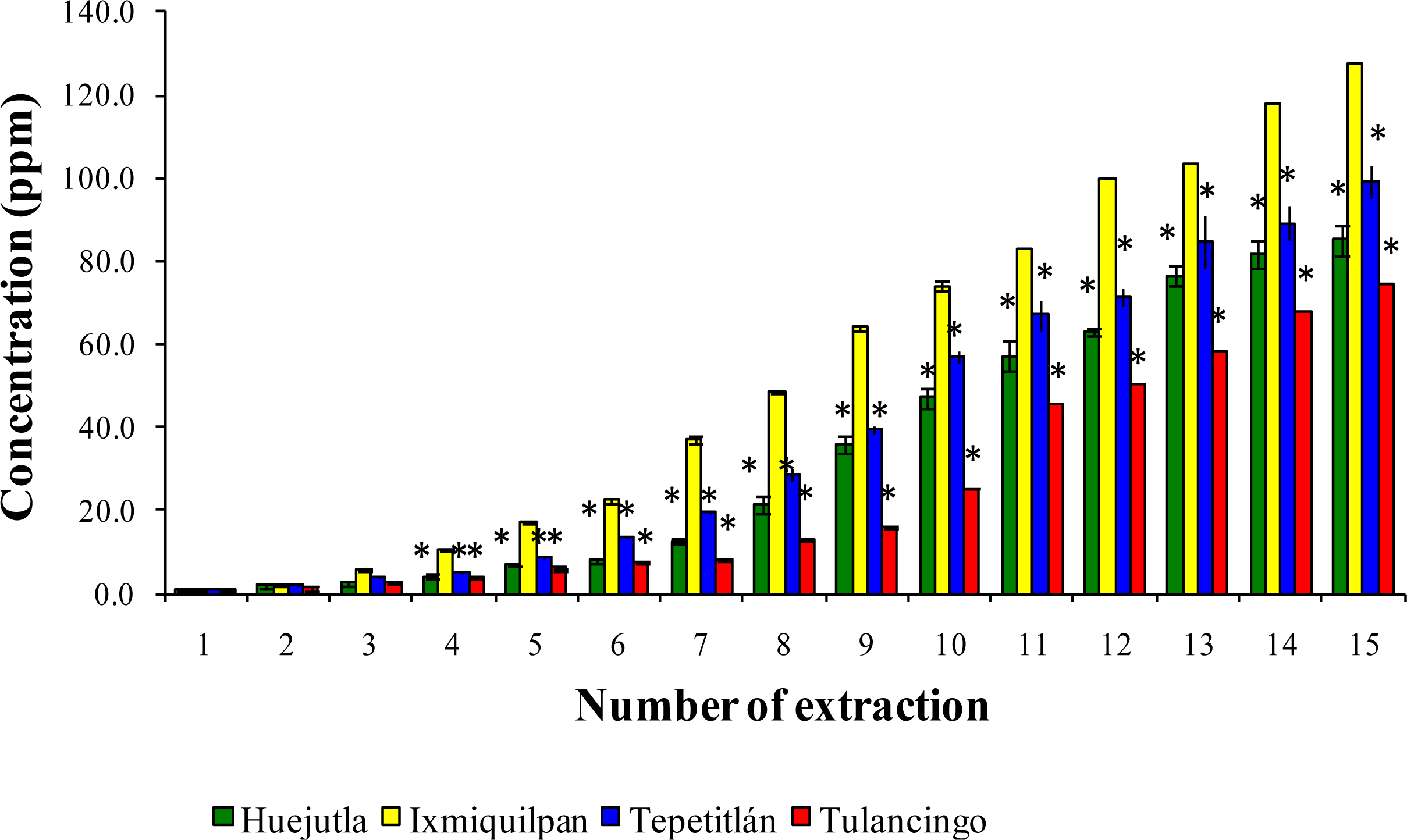
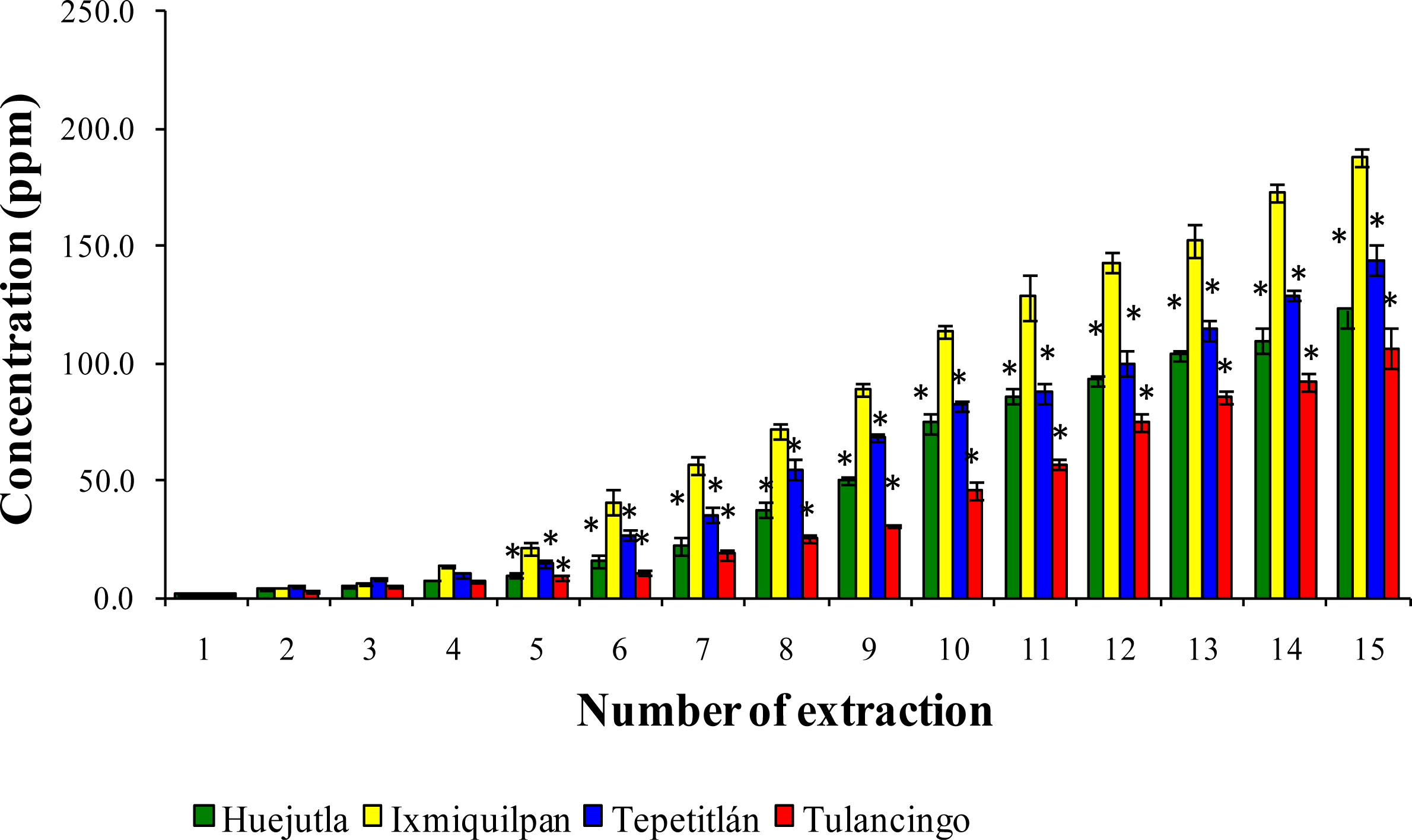
3.2. Leaching of Cd, Co, and Pb into Green Tomato Sauce (Salsa) at pH 4.2
3.3. Leaching of Cd, Co, and Pb in Chickpea Puree at pH 6.5
3.4. Leaching of Cd, Co, and Pb with Acetic Acid at pH 2.75
4. Discussion
5. Conclusions
Acknowledgments
References
- Picado, F; Mendoza, A; Cuadra, S; Barmen, G; Jakobsson, K; Bengtsson, G. Ecological, groundwater, and human health risk assessment in a mining region of Nicaragua. Risk Anal 2010, 30, 916–933. [Google Scholar]
- Youns, M; Hoheisel, JD; Efferth, T. Toxicogenomics for the prediction of toxicity related to herbs from traditional chinese medicine. Planta Med 2010, 76, 2019–2025. [Google Scholar]
- Cook, AG; Weinstein, P; Centeno, JA. Health effects of natural dust: Role of trace elements and compounds. Biol. Trace Elem. Res 2005, 103, 1–15. [Google Scholar]
- Massadeh, A; Al-Momani, F; Elbetieha, A. Assessment of heavy metals concentrations in soil samples from the vicinity of busy roads: Influence on Drosophila melanogaster life cycle. Biol. Trace. Elem. Res 2007, 122, 1–8. [Google Scholar]
- Nobuntou, W; Parkpian, P; Oanh, NT; Noomhorm, A; Delaune, RD; Jugsujinda, A. Lead distribution and its potential risk to the environment: Lesson learned from environmental monitoring of abandon mine. J. Environ. Sci. Health A Tox. Hazard. Subst. Environ. Eng 2010, 45, 1702–1714. [Google Scholar]
- Chang, L. Toxicology of Metals; CRC Lewis Publishers: Boca Raton, FL, USA, 1996; pp. 225–232. [Google Scholar]
- Abou-Arab, AK; Abou Donia, MA. Heavy metals in egyptian spices and medicinal plants and the effect of processing on their levels. J. Agric. Food Chem 2000, 48, 2300–2304. [Google Scholar]
- Bocio, A; Nadal, M; Domingo, JL. Human exposure to metals through the diet in Tarragona, Spain: Temporal trend. Biol. Trace Elem. Res 2005, 104, 193–201. [Google Scholar]
- Yun, Z; Hui, Y; Junli, W; Wei, F; Jiangang, Y; Zhongyi, Y. Heavy metal accumulations of 24 asparagus bean cultivars grown in soil contaminated with cd alone and with multiple metals (Cd, Pb, and Zn). J. Agric. Food Chem 2007, 55, 1045–1052. [Google Scholar]
- Zukowska, J; Biziuk, M. Methodological evaluation of method for dietary heavy metal intake. J. Food Sci 2008, 73, R21–R29. [Google Scholar]
- Schütz, A; Olsson, M; Jensen, A; Gerhardsson, L; Börjesson, J; Mattsson, S; Skerfving, S. Lead in finger bone, whole blood, plasma and urine in lead-smelter workers: Extended exposure range. Int. Arch. Occup. Environ. Health 2005, 78, 35–43. [Google Scholar]
- Solliway, BM; Schaffer, A; Pratt, H. Effects of exposure to lead on selected biochemical and haematological variables. Pharmacol. Toxicol 1996, 78, 18–22. [Google Scholar]
- Hengstler, JG; Bolm-Auorff, U; Faldum, A; Janssen, K; Reifenrath, M; Gotte, W; Jung, D; Mayer-Popken, O; Fuchs, J; Gebhard, S; Bienfait, HG; Schlink, K; Dietrich, C; Faust, D; Epe, B; Oesch, F. Occupational exposure to heavy metals: DNA damage induction and DNA repair inhibition prove co-exposures to cadmium, cobalt and lead as more dangerous than hitherto expected. Carcinogenesis 2003, 24, 63–73. [Google Scholar]
- García-Lestón, J; Méndez, J; Pásaro, E; Laffon, B. Genotoxic effects of lead: An updated review. Environ. Int 2010, 36, 623–636. [Google Scholar]
- Lepper, TW; Oliveira, E; Koch, GD; Berlese, DB; Feksa, LR. Lead inhibits in vitro creatine kinase and pyruvate kinase activity in brain cortex of rats. Toxicol. In Vitro 2010, 24, 1045–1051. [Google Scholar]
- Sorahan, T; Esmen, N. Lung cancer mortality in UK nickel-cadmium battery workers, 1947–2000. Occup. Env. Med 2004, 61, 108–116. [Google Scholar]
- Drebler, J; Schulz, K; Klemm, M; Schuttig, R; Beuthin, A; Felscher, D. Lethal manganese-cadmium intoxication. A case report. Arch. Toxicol 2002, 76, 449–451. [Google Scholar]
- Cvjetko, P; Tolić, S; Sikić, S; Balen, B; Tkalec, M; Vidaković-Cifrek, Z; Pavlica, M. Effect of copper on the toxicity and genotoxicity of cadmium in duckweed (Lemna Minor L). Arh. Hig. Rada. Toksikol 2010, 61, 287–296. [Google Scholar]
- Lewis, R. Occupational exposures. In Occupational and Environmental Medicine; LaDou, J, Ed.; Appleton y Lange: Stamford, CT, USA, 1997; pp. 142–154. [Google Scholar]
- Dai, W; Du, H; Fu, L; Jin, C; Xu, Z; Liu, H. Effects of dietary Pb on accumulation, histopathology, and digestive enzyme activities in the digestive system of tilapia (Oreochromis niloticus). Biol. Trace Elem. Res 2009, 127, 124–131. [Google Scholar]
- Abraham, KS; Abdel-Gawad, NB; Mahmoud, AM; El-Gowaily, MM; Emara, AM; Hwaihy, MM. Genotoxic effect of occupational exposure to cadmium. Toxicol. Ind. Health 2011, 27, 123–127. [Google Scholar]
- Buchko, GW; Hess, NJ; Kennedy, MA. Cadmium mutagenicity and human nucleotide excision repair protein XPA: CD, EXAFS and (1)H/(15)N-NMR spectroscopic studies on the zinc(II)- and cadmium(II)-associated minimal DNA-binding domain (M98-F219). Carcinogenesis 2000, 21, 1051–1057. [Google Scholar]
- Hartwig, A; Asmuss, M; Blessing, H. Interference by toxic metal ions with zinc-dependent proteins involved in maintaining genomic stability. Food Chem. Toxicol 2002, 40, 1179–1184. [Google Scholar]
- Punsar, S; Erämetsä, O; Karvonen, MJ; Ryhänen, A; Hilska, P; Vornamo, H. Coronary heart disease and drinking water. A search in two Finnish male cohorts for epidemiologic evidence of a water factor. J. Chronic. Dis 1975, 28, 259–287. [Google Scholar]
- Hamilton, EI. The geobiochemistry of cobalt. Sci. Total Environ 1994, 150, 7–39. [Google Scholar]
- Delpeux, S; Szostak, K; Frackowiak, EJ. High yield of pure multiwalled carbon nanotubes from the catalytic decomposition of acetylene on in-situ formed cobalt nanoparticles. Nanosci. Nanotechnol 2002, 2, 481–484. [Google Scholar]
- Coates, EO; Watson, JHL. Diffuse interstitial lung disease in tungsten carbide workers. Ann. Intern. Med 1971, 75, 709–716. [Google Scholar]
- Pier, SM. The role of heavy metals in human health. Tex. Rep. Biol. Med 1975, 33, 85–106. [Google Scholar]
- Bucher, JR; Elwell, MR; Thompson, MB. Inhalation toxicity studies of cobalt sulfate in F344/N rats and B6C3F1 mice. Fundam. Appl. Toxicol 1990, 15, 357–372. [Google Scholar]
- Gulson, BL; Mizon, KJ; Korsch, MJ; Howarth, D. Non-orebody sources are significant contributors to blood lead of some children with low to moderate lead exposure in a major lead mining community. Sci. Total Environ 1996, 181, 223–230. [Google Scholar]
- Mariné, AF. Influencia del Medio Ambiente en Las Relaciones Entre la Alimentación y Salud; Departamento de Nutrición y Bromatología, Universidad de Barcelona: Barcelona, Spain, 2000; pp. 354–377. [Google Scholar]
- Garcia, M; Quintero, R; López-Munguia, A. Biotecnología Alimentaria; Goldberg, I, Ed.; Chapman & Hall: Limusa, Mexico, 2000; pp. 48–64. [Google Scholar]
- Wallace, DM; Kalman, DA; Bird, TD. Hazardous lead release from glazed dinnerware: A cautionary note. Sci. Total Environ 1985, 44, 289–292. [Google Scholar]
- Landa, ER; Councell, TB. Leaching of uranium from glass and ceramic foodware and decorative items. Health. Phys 1992, 63, 343–348. [Google Scholar]
- Hight, SC. Determination of lead and cadmium in ceramicware leach solutions by graphite furnace atomic absorption spectroscopy: Method development and interlaboratory trial. J. AOAC Int 2001, 84, 861–872. [Google Scholar]
- Villalobos, M; Merino-Sánchez, C; Hall, C; Grieshop, J; Gutiérrez-Ruiz, ME; Handley, MA. Lead (II) detection and contamination routes in environmental sources, cookware and home-prepared foods from Zimatlán, Oaxaca, Mexico. Sci. Total Environ 2009, 407, 2836–2844. [Google Scholar]
- González de Mejía, E; Craigmill, AL. Transfer of lead from lead-glazed ceramics to food. Arch. Environ. Contam. Toxicol 1996, 31, 581–584. [Google Scholar]
- Alper-Baba, A; Gulbin, G; Sengunalp, F; Ozay, O. Effects of leachant temperature and pH on leachability of metals from fly ash. A case study: Can thermal power plant, province of Canakkale, Turkey. Environ. Monit. Assess 2008, 139, 287–298. [Google Scholar]
- Karen, W; Barnes, ED; Zhang, L. The Nutritional Analysis of Corn Products; The Perkin-Elmer Corporation, ICP Aplication Study Number: 72; 1995; pp. 2–4. [Google Scholar]
- Glazed pottery, glazed pottery and porcelain. Limits soluble lead and cadmium. Test method. Norma Official Mexicana NOM-231-SSA1-2002, 1 August 2003.
- Gould, JH; Butler, SW; Boyer, KW; Steele, EA. Hot leaching of ceramic and enameled cookware: Collaborative study. J. Assoc. Anal. Chem 1983, 66, 610–619. [Google Scholar]
- Azcona-Cruz, MI; Rothenberg, SJ; Schnaas, L; Zamora-Munoz, JS; Romero-Placeres, M. Lead-glazed ceramic ware and blood lead levels of children in the city of Oaxaca, Mexico. Arch. Environ. Health 2000, 55, 217–222. [Google Scholar]
- Gould, JH; Hight, SC; Alvarez, GH; Nelson, CE; Capar, SG. Influence of automatic dishwashings and scrubbings on release of lead from glazed ceramicware. J. Assoc. Anal. Chem 1990, 73, 401–404. [Google Scholar]
- Sheets, RW. Extraction of lead, cadmium and zinc from overglaze decorations on ceramic dinnerware by acidic and basic food substances. Sci. Total Environ 1997, 197, 167–175. [Google Scholar]
- Matte, TD; Proops, D; Palazuelos, E; Graef, J; Hernandez-Avila, M. Acute high-dose lead exposure from beverage contaminated by traditional Mexican pottery. Lancet 1994, 344, 1064–1075. [Google Scholar]
- Hernandez-Avila, M; Romieu, I; Rios, C; Rivero, A; Palazuelos, E. Lead-glazed ceramics as major determinants of blood lead levels in Mexican women. Environ. Health Perspect 1991, 94, 117–120. [Google Scholar]
- Belgaied, JE. Release of heavy metals from Tunisian traditional earthenware. Food Chem. Toxicol 2003, 41, 95–98. [Google Scholar]
- Ajmal, M; Khan, A; Nomani, AA; Ahmed, S. Heavy metals: Leaching from glazed surfaces of tea mugs. Sci. Total Environ 1997, 207, 49–54. [Google Scholar]
- Linna, A; Oksa, P; Groundstroem, K; Halkosaari, M; Palmroos, P; Huikko, S; Uitti, J. Exposure to cobalt in the production of cobalt and cobalt compounds and its effect on the heart. Occup. Environ. Med 2004, 61, 877–885. [Google Scholar]
- Naura, AS; Sharma, R. Toxic effects of hexaammine cobalt (III) chloride on liver and kidney in mice: Implication of oxidative stress. Drug. Chem. Toxicol 2009, 32, 293–299. [Google Scholar]
- Wild, P; Bourgkard, E; Paris, C. Lung cancer and exposure to metals: The epidemiological evidence. Method. Mol. Biol 2009, 472, 139–167. [Google Scholar]
- Stefaniak, AB; Harvey, CJ; Virji, MA; Day, GA. Dissolution of cemented carbide powders in artificial sweat: Implications for cobalt sensitization and contact dermatitis. J. Environ. Monit 2010, 12, 1815–1822. [Google Scholar]


| Region | Huejutla | Ixmiquilpan | Tepetitlán | Tulancingo |
|---|---|---|---|---|
| Metal | Concentration (ppm) | |||
| Cd | 65.95 ± 5.55 | 63.32 ± 2.90 | 64.77 ± 0.37 | 64.46 ± 0.94 |
| Co | 43.20 ± 0.45 | 43.24 ± 1.69 | 43.20 ± 0.45 | 44.00 ± 1.98 |
| Pb | 64.75 ± 3.05 | 63.01 ± 3.38 | 62.60 ± 3.45 | 63.44 ± 4.20 |
| Region | Huejutla | Ixmiquilpan | Tepetitlán | Tulancingo |
|---|---|---|---|---|
| Metal | Concentration (ppm) | |||
| Cd | 559.56 ± 3.16a | 757.99 ± 4.16 | 685.53 ± 7.60a | 557.99 ± 4.16a |
| Co | 472.43 ± 2.77b | 571.39 ± 3.72 | 562.82 ± 3.84 | 468.24 ± 6.18b |
| Pb | 621.90 ± 0.52c | 805.33 ± 1.98 | 704.40 ± 1.25 | 607.59 ± 3.48c |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Valadez-Vega, C.; Zúñiga-Pérez, C.; Quintanar-Gómez, S.; Morales-González, J.A.; Madrigal-Santillán, E.; Villagómez-Ibarra, J.R.; Sumaya-Martínez, M.T.; García-Paredes, J.D. Lead, Cadmium and Cobalt (Pb, Cd, and Co) Leaching of Glass-Clay Containers by pH Effect of Food. Int. J. Mol. Sci. 2011, 12, 2336-2350. https://doi.org/10.3390/ijms12042336
Valadez-Vega C, Zúñiga-Pérez C, Quintanar-Gómez S, Morales-González JA, Madrigal-Santillán E, Villagómez-Ibarra JR, Sumaya-Martínez MT, García-Paredes JD. Lead, Cadmium and Cobalt (Pb, Cd, and Co) Leaching of Glass-Clay Containers by pH Effect of Food. International Journal of Molecular Sciences. 2011; 12(4):2336-2350. https://doi.org/10.3390/ijms12042336
Chicago/Turabian StyleValadez-Vega, Carmen, Clara Zúñiga-Pérez, Samuel Quintanar-Gómez, José A. Morales-González, Eduardo Madrigal-Santillán, José Roberto Villagómez-Ibarra, María Teresa Sumaya-Martínez, and Juan Diego García-Paredes. 2011. "Lead, Cadmium and Cobalt (Pb, Cd, and Co) Leaching of Glass-Clay Containers by pH Effect of Food" International Journal of Molecular Sciences 12, no. 4: 2336-2350. https://doi.org/10.3390/ijms12042336






