Synthesis, Characterization and Thermal Studies of Zn(II), Cd(II) and Hg(II) Complexes of N-Methyl-N-Phenyldithiocarbamate: The Single Crystal Structure of [(C6H5)(CH3)NCS2]4Hg2
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis
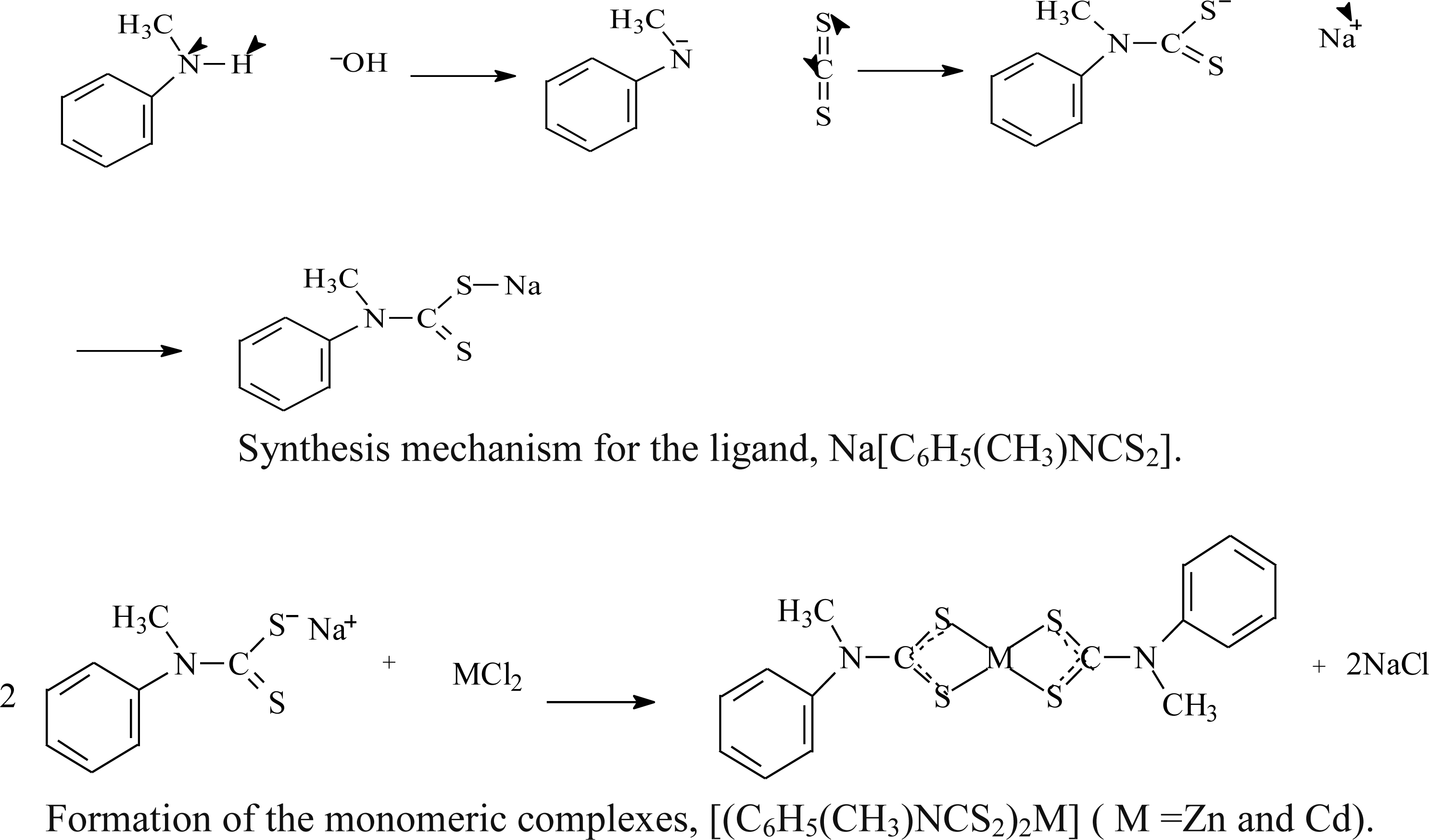
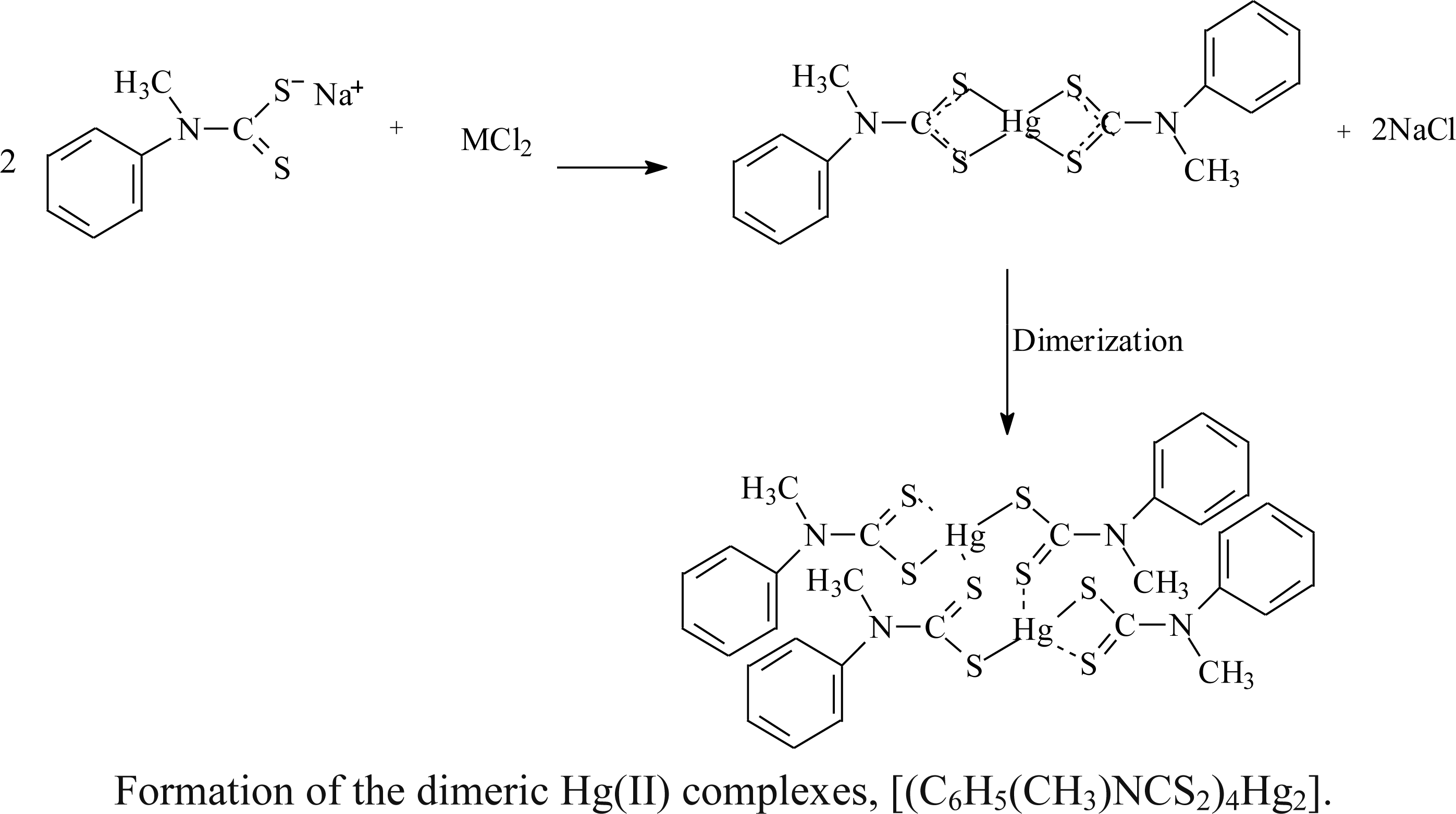
2.2. Spectroscopic Analysis
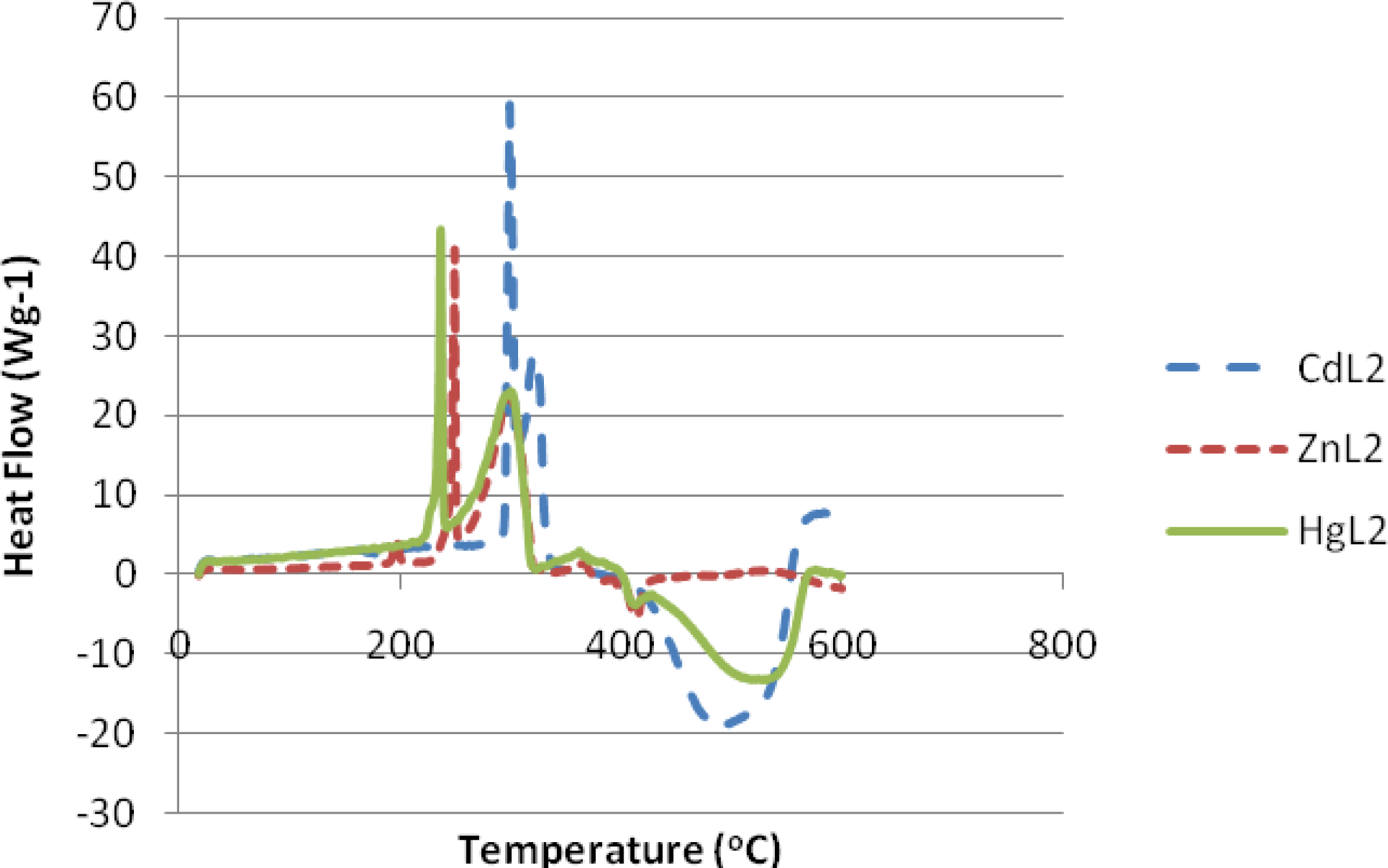
2.3. Thermal Analyses of the Complexes
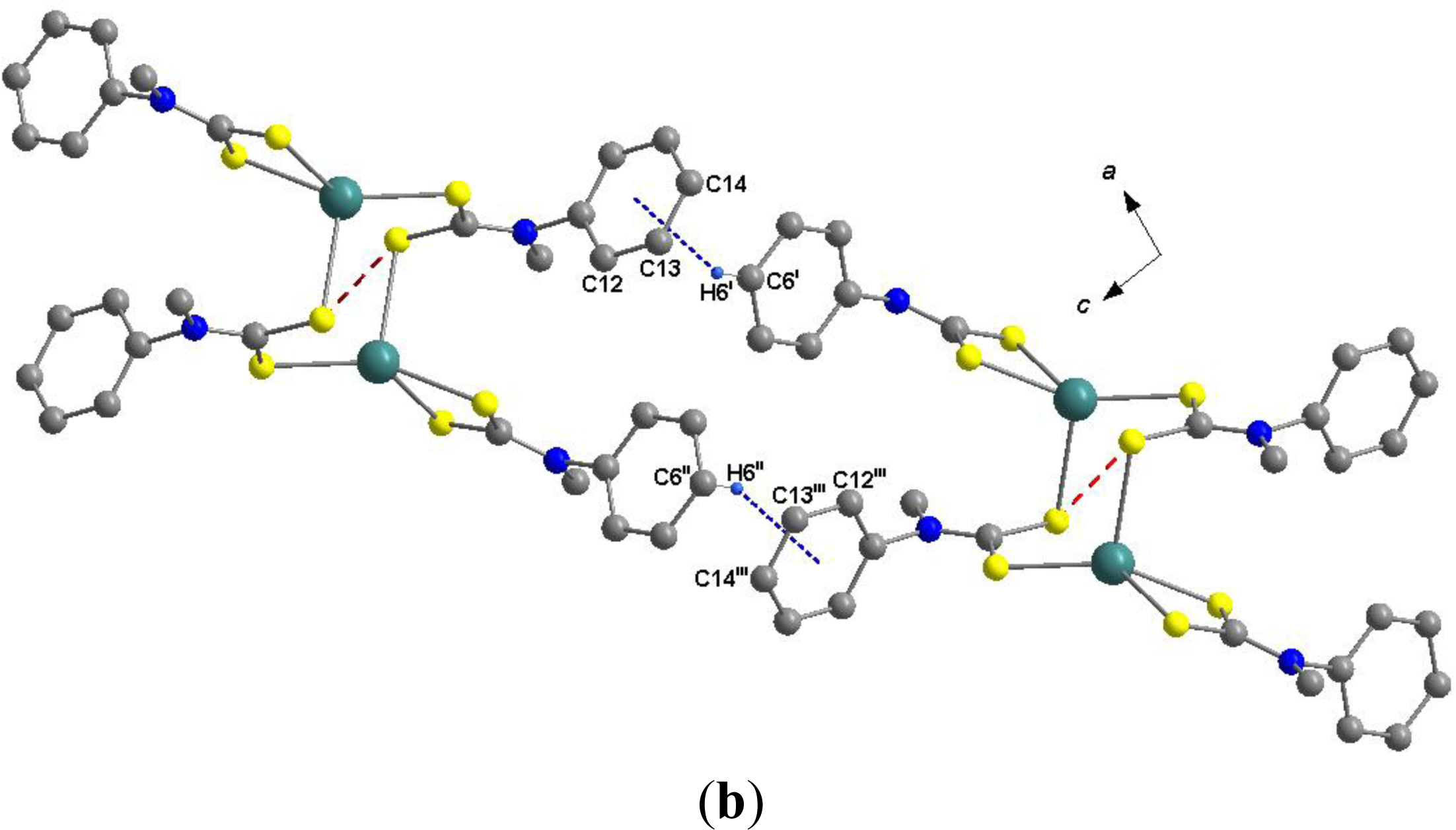
2.4. Molecular Structure of the Mercury Complex
3. Experimental Section
3.1. Materials and Methods
3.2. Synthesis of the Ligand: Sodium N-Methyl-N-phenyldithiocarbamate, Na[C6H5(CH3)NCS2]
3.3. Preparation of Complexes
3.3.1. Bis-(N-methyl-N-phenyldithiocarbamato)zinc(II): ZnL2 (1)
- 1H NMR (DMSO) δ = 7.46 – 7.19 (m, 10H, –C6H5), 3.64 (s, 6H, –CH3).
- 13C NMR (DMSO) δ 147.17, 129.55, 127.81, 126.31(−C6H5), 47.24(−CH3), 190.51(−CS2).
- Selected IR, υ (cm−1): 1491 (C=N), 1261 (C2-N), 969 (C=S). Anal. Calc. for C16H16N2S4Zn (429.94): C, 44.70; H, 3.75; N, 6.52; S, 29.83. Found: C, 44.69; H, 3.80; N, 6.90; S, 29.59.
- The product was recrystallized in dichloromethane.
3.3.2. Bis-(N-methyl-N-phenyldithiocarbamato)cadmium(II): CdL2 (2)
- Complex was obtained as white solid. Yield: 0.559 g (93.95 %), M.p. 296–298 °C.
- 1H NMR (DMSO) δ = 7.46–7.19 (m, 10H, −C6H5), 3.64 (s, 6H, −CH3).
- 13C NMR (DMSO) δ 148.55, 129.39, 128.79, 126.44 (−C6H5), 45.40 (−CH3), 202.10 (−CS2).
- Selected IR, υ (cm−1): 1490 (C=N), 1255 (C2-N), 963 (C=S). Anal. Calc. for C16H16N2S4Cd (476.97): C, 40.29; H, 3.38; N, 5.87; S, 26.89. Found: C, 39.96; H, 3.36; N, 6.05; S, 27.31
3.3.3. Bis(μ-N-methyl-N-phenyldithiocarbamato-S:S’)bis-[(N-methyl-N-henyldithiocarbamato) Mercury(II)]: Hg2L4 (3)
- Complex was obtained as yellow solid. Yield: 0.600 g (84.51%), M.p. 235–236 °C.
- 1H NMR (CHCl3) δ = 7.53–7.29 (m, 20H, −C6H5), 3.82 (s, 12H, −CH3).
- 13C NMR (CHCl3) δ 129.65, 128.35, 125.38 (−C6H5), 48.78 (−CH3), 201.20 (−CS2).
- Selected IR, υ (cm−1): 1491 (C=N), 1255 (C2-N), 959 (C=S). Anal. Calc. for C32H32N4S8Hg2 (565.15): C, 34.00; H, 2.85; N, 4.96; S, 22.69. Found: C, 34.02; H, 2.95; N, 5.04; S, 22.81.
- The product was recrystallized from a dichloromethane/ethyl acetate (3:1 v/v) mixture to afford yellow crystals suitable for X-ray crystallography.
3.4. Thermal Studies
3.5. Crystal Structure Determination
4. Conclusions
Supplementary Material
CCDC 762809 contains the supplementary crystallographic data for this paper. These data can be obtained free of charge via http://www.ccdc.cam.ac.uk/conts/retrieving.html, or from the Cambridge Crystallographic Data Centre, 12 Union Road, Cambridge, CB2 1EZ, UK; Fax: (+44)-1223-336-033 or E-Mail: [email protected].Acknowledgments
References
- Thammakan, N; Somsook, E. Synthesis and thermal decomposition of cadmium dithiocarbamate complexes. Mater. Lett 2006, 60, 1161–1165. [Google Scholar]
- Wong, WWH; Crokson, J; Evans, EAL; Mclnnes, EJL; Wolowska, J; Maher, JP; Bishop, P; Beer, PD. Heteropolymetallic copper(II)–gold(III) dithiocarbamate [2] catenanes via magic ring synthesis. Chem. Commun 2005, 8, 2214–2216. [Google Scholar]
- Fox, OD; Drew, MGB; Beer, PD. Resorcarene-based nanoarchitectures: Metal-directed assembly of a molecular loop and tetrahedron. Angew. Chem. Int. Ed 2000, 39, 136–140. [Google Scholar]
- Beer, PD; Berry, N; Drew, MGB; Fox, OD; Padilla-Tosta, ME; Patell, S. Self-assembled dithiocarbamate–copper(II) macrocycles for electrochemical anion recognition. Chem. Commun 2001, 4, 199–200. [Google Scholar]
- Berry, NG; Pratt, MD; Fox, OD; Beer, PD. Transition metal self-assembly of dithiocarbamate based anion receptors. Supramol. Chem 2001, 13, 677–682. [Google Scholar]
- Lon, W; Chen, M; Wang, X; Liu, W. Synthesis of high-luminescent cadmium sulfide nanocrystallites by a novel single-source precursor route. Mater. Lett 2007, 61, 3612–3615. [Google Scholar]
- Hursthouse, MB; Malik, MA; Motevalli, M; O’Brien, P. The crystal and molecular structure of N,N-diethyldiselenocarbamatocadmium(II): Cadmium and zinc diethyldiselenocarbamates as precursors for selenides. Polyhedron 1992, 11, 45–48. [Google Scholar]
- Monoussakis, G; Bolos, C; Ecateriniadou, L; Sarris, C. Synthesis, characterization and anti-bacterial studies of mixed-ligand complexes of dithiocarbamato—Thiocyanato and iron(III), nickel(II), copper(II) and zinc(II). Eur. J. Med. Chem 1987, 22, 421–425. [Google Scholar]
- Pazukhina, YE; Isakova, NV; Nagy, V; Petrukhi, OM. Influence of the nature of metal ions on the extraction constants of their dithiocarbamates. Solvent Extr. Ion. Exch 1997, 15, 777–790. [Google Scholar]
- Stary, J; Kratzer, K. Stability and distribution constants of thallium (I) dithiocarbamates. J. Radioanal. Nucl. Chem. Lett 1992, 165, 137–143. [Google Scholar]
- Fan, D; Afzaal, M; Malik, MA; Nguyen, CQ; O’Brien, P; Thomas, PJ. Using coordination chemistry to develop new routes to semiconductor and other materials. Coord. Chem. Rev 2007, 251, 1878–1888. [Google Scholar]
- Romano, R; Alves, OL. Semiconductor/porous glass nanocomposites via the single-source precursor approach. Mater. Res. Bull 2006, 41, 376–386. [Google Scholar]
- Green, M; Prince, P; Gardener, M; Steed, J. Mercury(II) N,N methyl-phenylethyldithiocarbamates and its use as a precursor for the room-temperature solution deposition of β-HgS thin films. Adv. Mater 2004, 16, 994–996. [Google Scholar]
- Rogach, AL; Koktysh, DS; Harrsion, M; Kotov, NA. Layer-by-layer assembled films of HgTe nanocrystals with strong infrared emission. Chem. Mater 2000, 12, 1526–1528. [Google Scholar]
- Yu, Z; Ren, J; Cook, JW; Schetzina, JF. Blue/green laser diodes and light emitting diodes. Phys. B Condens. Matter 1993, 191, 119–123. [Google Scholar]
- Monteiro, OC; Trindade, T; Park, JH; O’Brien, P. The use of bismuth(III) dithiocarbamato complexes as precursors for the low-pressure. MOCVD of Bi2S3. Chem. Vap. Depos 2000, 6, 230–232. [Google Scholar]
- Manav, N; Mishra, AK; Kaushik, NK. In vitro antitumour and antibacterial studies of some Pt (IV) dithiocarbamate complexes. Spectrochim. Acta Part A 2006, 65, 32–35. [Google Scholar]
- Shahid, M; Ruffer, T; Lang, H; Awan, SA; Ahmad, S. Synthesis and crystal structure of a dinuclear zinc(II)-dithiocarbamate complex, bis{[(μ2-pyrrolidinedithiocarbamato-S,S’) (pyrrolidinedithiocarbamato-S,S’)zinc(II)]}. J. Coord. Chem 2009, 62, 440–445. [Google Scholar]
- Faraglia, G; Sitran, S; Montagner, D. Pyrrolidine dithiocarbamates of Pd(II). Inorg. Chim. Acta 2005, 358, 971–980. [Google Scholar]
- Gossage, A; Jenkins, HA. The crystal structure of bis-(μ-N-ethyl-phenyldithiocarbamato-S,S’)-bis[(N-ethyl-Nphenyldithiocarbamato-κ2S,S’)zinc(II)]. Acta Chim. Slov 2009, 56, 329–333. [Google Scholar]
- Howie, RA; Tiekink, ERT; Wardell, JL; Wardell, SMSV. Complementary supramolecular aggregation via O–H….O hydrogen-bonding and Hg…..S interactions in bis[N,N’-di(2-hydroxyethyl)-dithiocarbamato-S,S’]mercury(II): Hg[S2CN(CH2CH2OH)2]2. J. Chem. Crystallogr 2009, 39, 293–298. [Google Scholar]
- Ondrušova, D; Pajtášova, M; Jóna, E; Koman, M. Structural properties of Co(III), Hg(II) and Pb(II) N-Ethyl-N-Phenyl-Dithiocarbamates and their application in the rubber industry. Solid State Phenom 2003, 90–91, 383–388. [Google Scholar]
- Yin, H-D; Zhai, J; Sun, Y-Y; Wang, D-Q. Synthesis, characterizations and crystal structures of new antimony (III) complexes with dithiocarbamate ligands. Polyhedron 2008, 27, 663–670. [Google Scholar]
- Johnson, BFG; Al-Obalidi, KH; Mecleverty, JA. Transition-metal nitrosyl compounds. Part III. (NN-dialkyldithiocarbamato)nitrosyl compounds of molybdenum and tungsten. J. Am. Chem. Soc. A 1969, 19, 1668–1670. [Google Scholar]
- Nami, SAA; Siddiqi, KS. Convenient one-pot synthesis of symmetrical dithiocarbamates. Synth. React. Inorg. Met.-Org. Chem 2004, 34, 1581–1590. [Google Scholar]
- Sykes, AG. Comprehensive Coordination Chemistry; Wilkinson, G, Gillard, RD, McCleverty, JA, Eds.; Pergamon Press: Oxford, UK, 1987; 229. [Google Scholar]
- Riveros, PC; Perilla, IC; Poveda, A; Keller, HJ; Pritzkow, H. Tris(dialkyldithiocarbamato) diazenido(1-) and hydrazido(2-) molybdenum complexes: Synthesis and reactivity in acid medium. Polyhedron 2000, 19, 2327–2335. [Google Scholar]
- Anderson, RJ; Bendell, DJ; Groundwater, PW. Organic Spectroscopic Analysis; RSC: Cambridge, UK, 2004. [Google Scholar]
- Prakasam, BA; Ramalingam, K; Bocelli, G; Cantoni, A. NMR and fluorescence spectral studies on bisdithiocarbamates of divalent Zn, Cd and their nitrogenous adducts: Single crystal X-ray structure of (1,10-phenanthroline)bis(4-methylpiperazinecarbodithioato) zinc(II). Polyhedron 2007, 26, 4489–4493. [Google Scholar]
- Khan, S; Nami, SAA; Siddiqi, KS. Piperazine pivoted transition metal dithiocarbamates. J. Mol. Struct 2008, 875, 478–485. [Google Scholar]
- Khan, S; Nami, SAA; Siddiqi, KS. Mononuclear indolyldithiocarbamates of SnCl4 and R2SnCl2: Spectroscopic, thermal characterizations and cytotoxicity assays in vitro. J. Organomet. Chem 2008, 693, 1049–1057. [Google Scholar]
- Sovil, SP; Babic-Samardzija, K; Minic, DM. Thermal stability and kinetic studies of new dinuclear copper(II) complexes with octaazamacrocyclic and multidonor bidentate ligands. Thermochim. Acta 2001, 370, 29–36. [Google Scholar]
- Kumar, S; Kaushik, NK. Preparation, characterization and thermal studies on bis-[N-(ethyl, m-tolyl)-dithiocarbamato] complexes of zinc(II), cadmium(II) and mercury(II). J. Therm. Anal 1981, 21, 3–7. [Google Scholar]
- Hill, JO; Murray, JP; Patil, KC. The thermochemistry of the metal dithiocarbamate and xanthate complexes: A review up-date. Rev. Inorg. Chem 1994, 14, 363–387. [Google Scholar]
- Botelho, JR; Souza, AG; Gondim, AD; Athayde-Filho, PF; Dunstan, PO; Pinheiro, CD; Longo, E; Carvalho, LH. Thermochemical parameters and calculations ab-initio of the bisdimethyldithiocarbamate zinc(II) complex. J. Therm. Anal. Cal 2005, 79, 309–312. [Google Scholar]
- Cox, MJ; Tiekink, ERT. Structural variations in the mercury(II) bis(1,1-dithiolate)s the crystal and molecular structure of [Hg(S2CNMe2)2]. Z. Kristallogr 1997, 212, 542–544. [Google Scholar]
- Cox, MJ; Tiekink, ERT. Structural diversity in the mercury(II) bis(N,N-dialkyldithiocarbamate) compounds: An example of the importance of considering crystal structure when rationalising molecular structure. Z. Kristallogr 1999, 214, 571–579. [Google Scholar]
- Ito, M; Iwasaki, H. The structure of the monomeric form of mercury(II) N,N-diisopropyldithiocarbamate [bis(N,N-diisopropyldithiocarbamato)mercury(II)]. Acta Cryst 1979, B35, 2720–2721. [Google Scholar]
- Ivanov, AV; Korneeva, EV; Bukvetskii, BV; Goryan, AS; Antzutkin, ON; Forsling, W. Structural organization of mercury(II) and copper(II) dithiocarbamates from EPR and 13C and 15N MAS NMR spectra and X-ray diffraction analysis. Russ. J. Coord. Chem 2008, 34, 59–69. [Google Scholar]
- Iwasaki, H. The crystal structure of dimeric and monomeric forms of mercury(II) N,N-diethyldithiocarbamate, Hg2(S2CNEt2)4 and Hg(S2CNEt2)2. Acta Cryst 1973, B29, 2115–2124. [Google Scholar]
- Bondi, A. Van der Waals volumes and radii. J. Phys. Chem 1964, 68, 441–451. [Google Scholar]
- Tiekink, ERT. Influence of crystal structure on molecular structure: Syntactic structural chemistry. Rigaku J 2002, 19, 14–24. [Google Scholar]
- APEX2, SADABS, and SAINT Software Reference Manuals; Bruker-AXS: Madison, WI, USA, 2007.
- Sheldrick, GM. A short history of SHELX. Acta Cryst 2008, A64, 112–122. [Google Scholar]
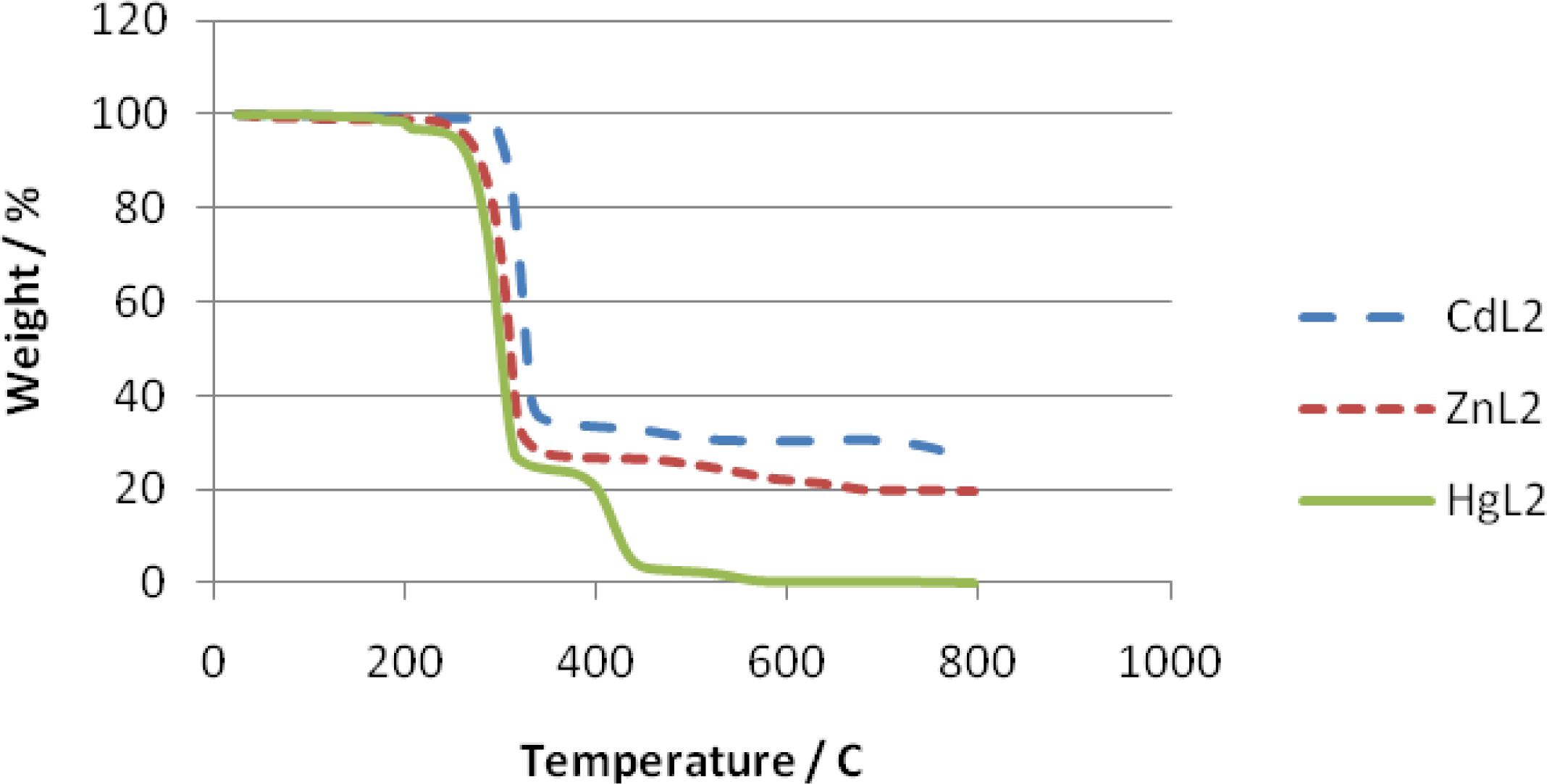
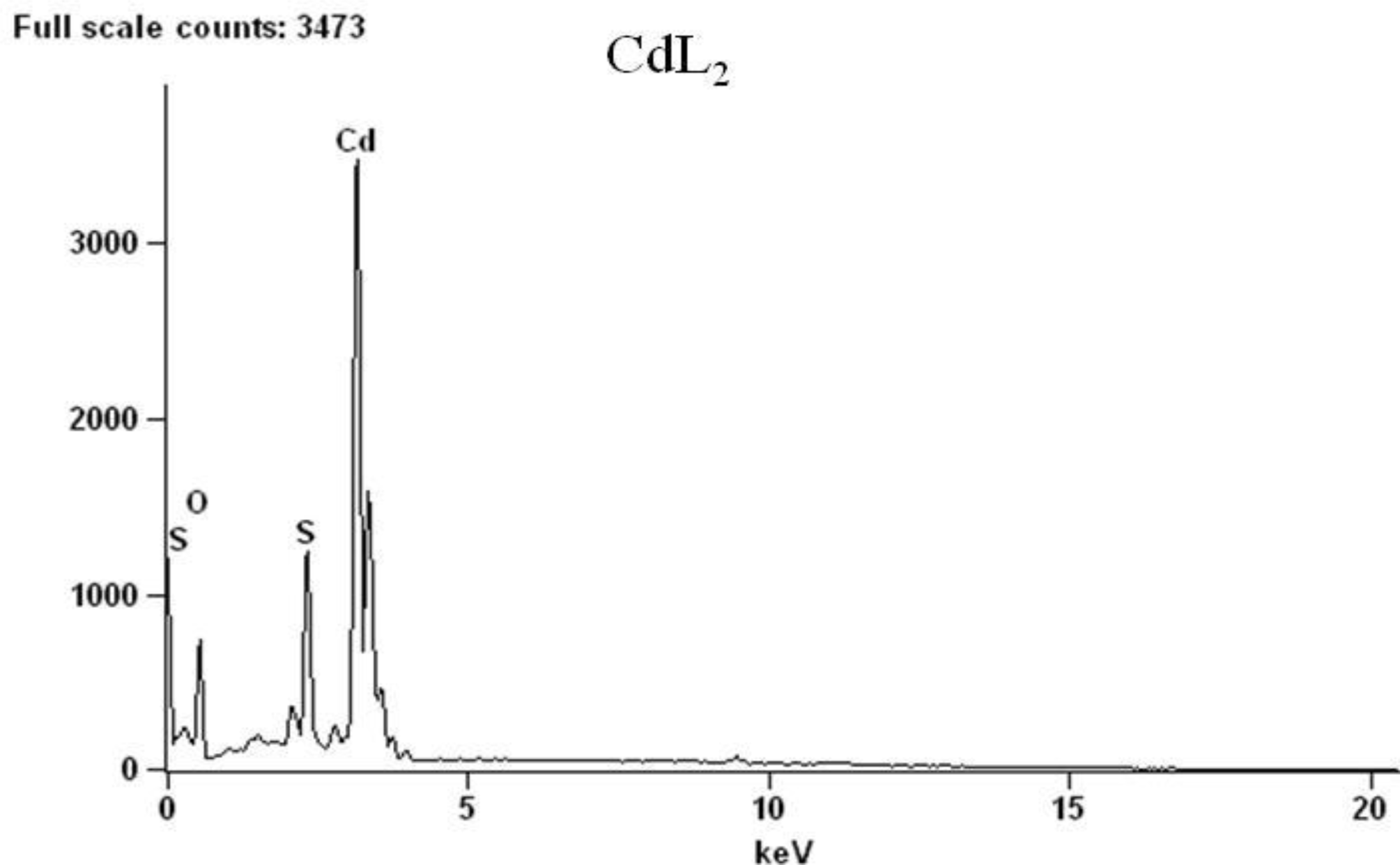
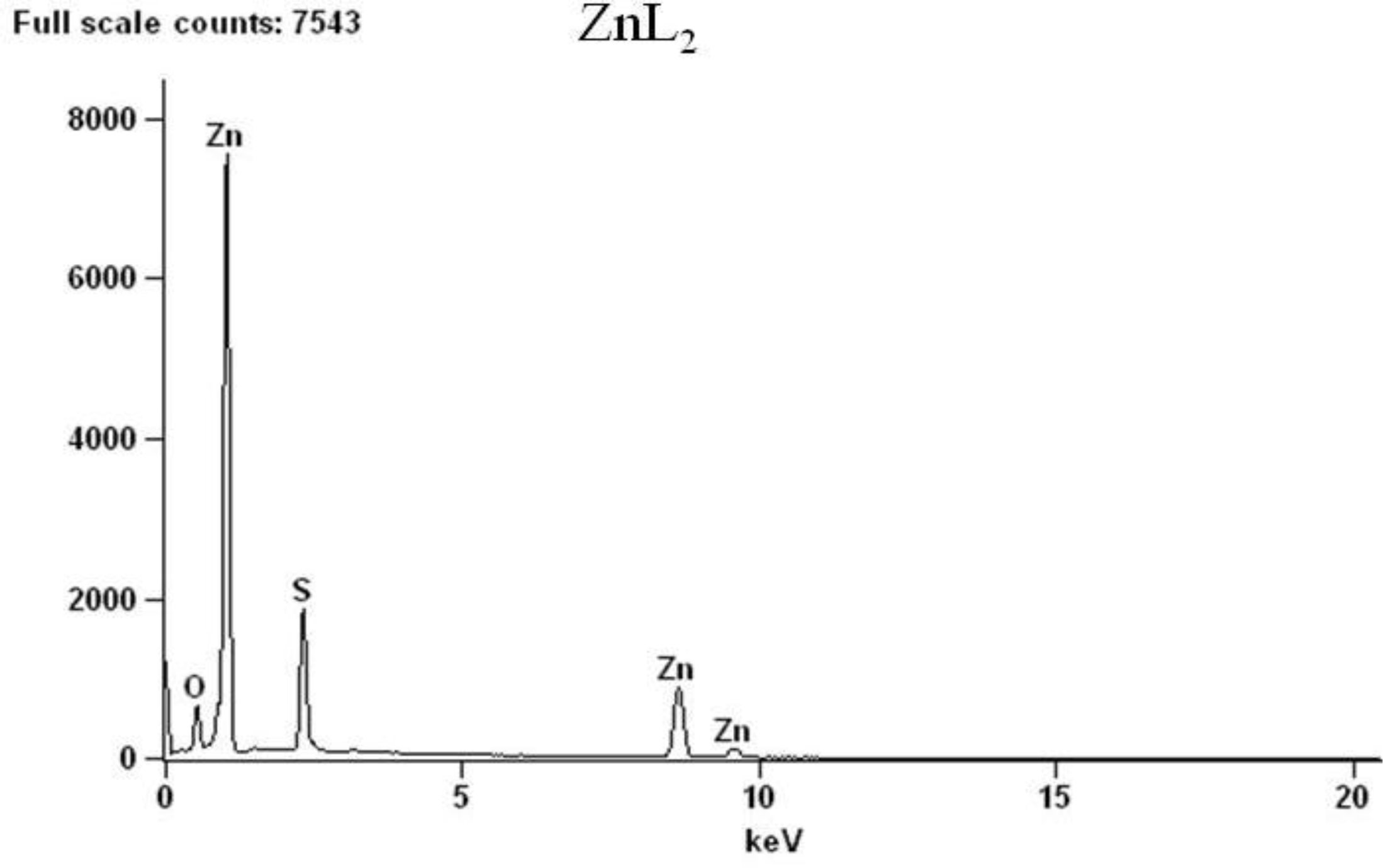
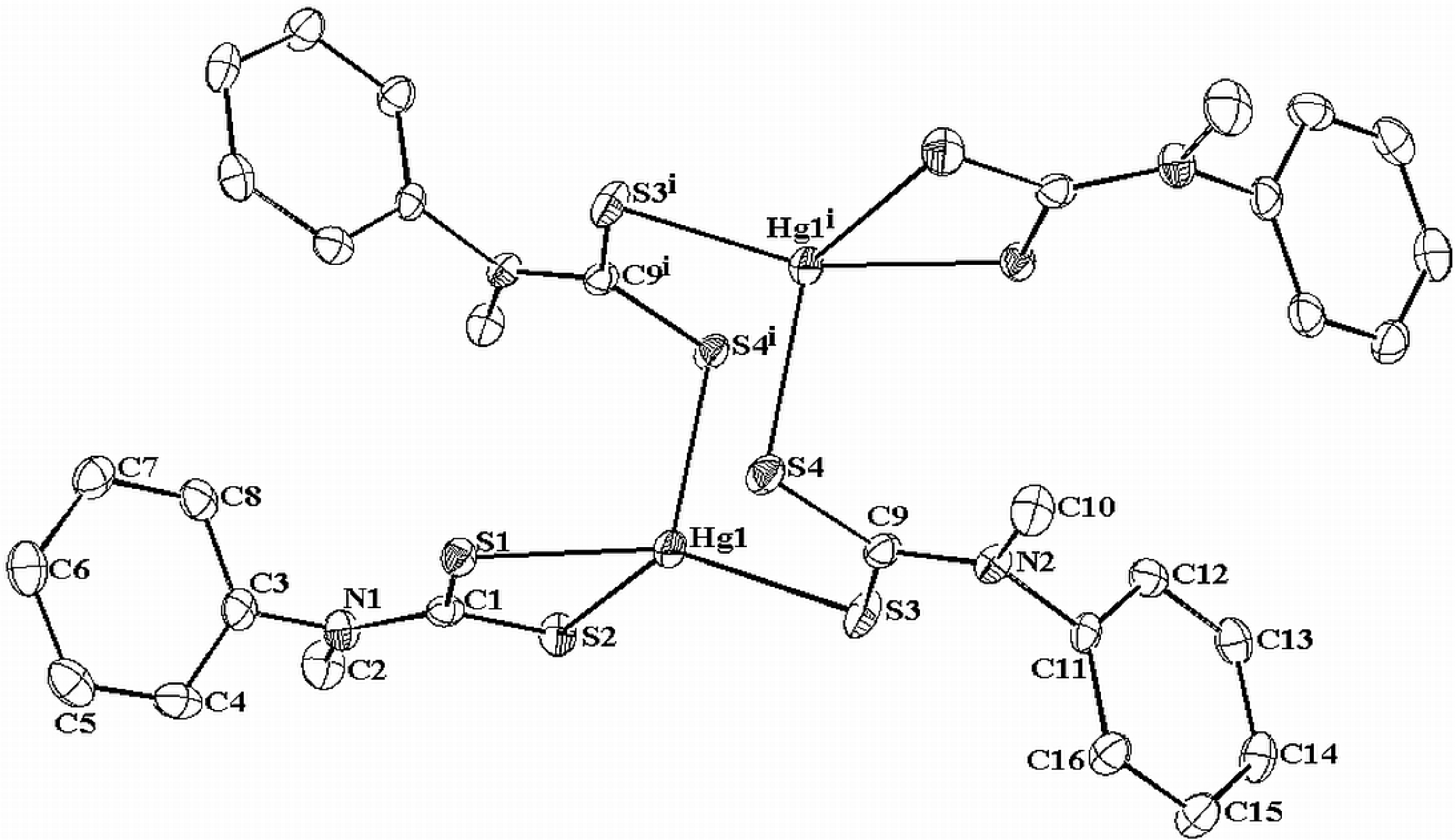
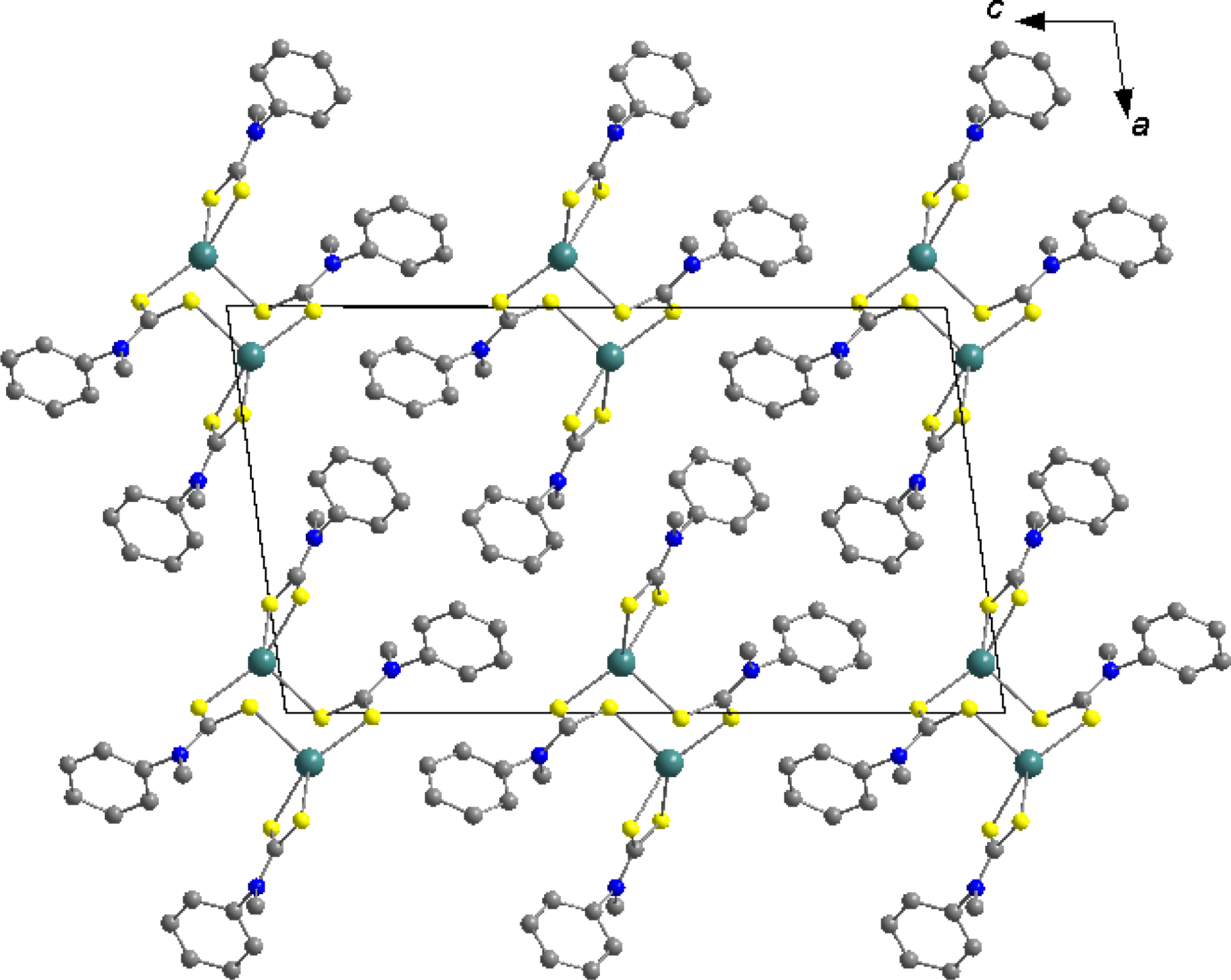

| Complex | Decomposition temperature | DTG max. value | Decomposition reaction | Mass changes expet. found | |
|---|---|---|---|---|---|
| Zn(S2CNmeC6H5)2 | 219–375 | 310 | Zn(S2CNmeC6H5)2→ZnS ZnS→ZnO | 3.05 | 3.60 |
| 450–683 | 2.61 | 2.67 | |||
| Cd(S2CNmeC6H5)2 | 270–372 | 324 | Cd(S2CNmeC6H5)2→CdS CdS→CdO | 2.92 | 3.25 |
| 430–640 | 2.59 | 2.65 | |||
| Hg(S2CNmeC6H5)2 | 168–361 | 299 | Hg(S2CNmeC6H5)2→HgS Volatilization | 4.16 | 2.50 |
| 361–585 | - | - | |||
| Compound | [(C6H5)(CH3)NCS2]4Hg2 |
| Empirical formula | C32H32Hg2N4S8 |
| Formula weight | 1130.28 |
| Temperature | 100(2) K |
| Wavelength | 1.54178 |
| Crystal system | Monoclinic |
| Space group | P21/C |
| Unit cell dimensions | |
| a (Å) | 12.7168(10) |
| b (Å) | 6.5198(6) |
| c (Å) | 22.2612(19) |
| β (°) | 98.341(3) |
| γ (°) | 90 |
| Volume (A3) | 1826.2(3) |
| Z | 2 |
| Dcalc Mg/m3 | 2.056 Mg/m3 |
| Absorption coefficient (mm−1) | 19.379 |
| F(000) | 1080 |
| Crystal size (mm) | 0.27 × 0.18 ×0.17 |
| Theta range (°) | 3.51 to 69.82 |
| Limiting indices | −15 ≤ h ≤ 15, −7 ≤ k ≤ 7, −27 < +1 ≤ 26 |
| Reflections collected | 27791 |
| Independent reflection | 3412 [R(int) =0.0343] |
| Refinement method | Full-matrix least-squares on F2 |
| Completeness to θ = 67.00 | 99.8 % |
| Data/restraints/parameters/ | 3412/0/210 |
| Goodness-of-fit on F2 | 1.022 |
| Final R indices [I > 2sigma(I)] | R1 = 0.0262, wR2 = 0.0716 |
| R indices (all data) | R1 = 0.0265, wR2 = 0.0718 |
| Largest diff. Peak and hole e. Å−3 | 1.836 and −1.181 |
| Bond length (Å) | Bond Angle (°) | ||
|---|---|---|---|
| Hg(1)—S(3) | 2.4114(9) | S(3)—Hg(1)—S(1) | 146.42(3) |
| Hg(1)—S(1) | 2.4810(9) | S(3)—Hg(1)—S(4)#1 | 102.35(3) |
| Hg(1)—S(4)#1 | 2.6955(9) | S(1)—Hg(1)—S(4)#1 | 107.67(3) |
| Hg(1)—S(2) | 2.7327(9) | S(3)—Hg(1)—S(2) | 122.15(3) |
| S(1)—C(1) | 1.7404(4) | S(1)—Hg(1)—S(1) | 69.91(3) |
| S(3)—C(9) | 1.702(4) | S(4)#1—Hg(1)—S(2) | 95.46(3) |
| S(4)—C(9) | 1.732(4) | C(1)—S(1)—Hg(1) | 88.17(13) |
| S(4)—Hg(1)#1 | 1.716(4) | C(1)—S(2)—Hg(1) | 80.98(13) |
| N(1)—C(1) | 1.347(5) | C(9)—S(3)—Hg(1) | 99.34(13) |
| N(1)—C(3) | 1.444(5) | C(9)—S(4)—Hg(1)#1 | 95.89(13) |
| N(1)—C(2) | 1.469(5) | S(2)—C(1)—S(1) | 120.9(2) |
| N(2)—C(9) | 1.332(5) | N(1)—C(1)—S(2) | 121.7(3) |
| N(2)—C(11) | 1.454(5) | N(1)—C(1)—S(1) | 117.4(3) |
| N(2)—C(10) | 1.462(5) | N(2)—C(9)—S(4) | 122.0(3) |
| N(2)—C(9)—S(3) | 116.2(3) | ||
| S(4)—C(9)—S(3) | 121.8(2) | ||
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Onwudiwe, D.C.; Ajibade, P.A. Synthesis, Characterization and Thermal Studies of Zn(II), Cd(II) and Hg(II) Complexes of N-Methyl-N-Phenyldithiocarbamate: The Single Crystal Structure of [(C6H5)(CH3)NCS2]4Hg2. Int. J. Mol. Sci. 2011, 12, 1964-1978. https://doi.org/10.3390/ijms12031964
Onwudiwe DC, Ajibade PA. Synthesis, Characterization and Thermal Studies of Zn(II), Cd(II) and Hg(II) Complexes of N-Methyl-N-Phenyldithiocarbamate: The Single Crystal Structure of [(C6H5)(CH3)NCS2]4Hg2. International Journal of Molecular Sciences. 2011; 12(3):1964-1978. https://doi.org/10.3390/ijms12031964
Chicago/Turabian StyleOnwudiwe, Damian C., and Peter A. Ajibade. 2011. "Synthesis, Characterization and Thermal Studies of Zn(II), Cd(II) and Hg(II) Complexes of N-Methyl-N-Phenyldithiocarbamate: The Single Crystal Structure of [(C6H5)(CH3)NCS2]4Hg2" International Journal of Molecular Sciences 12, no. 3: 1964-1978. https://doi.org/10.3390/ijms12031964




