Use of a Reflectance Spectroscopy Accessory for Optical Characterization of ZnO-Bi2O3-TiO2 Ceramics
Abstract
:1. Introduction
2. Experimental
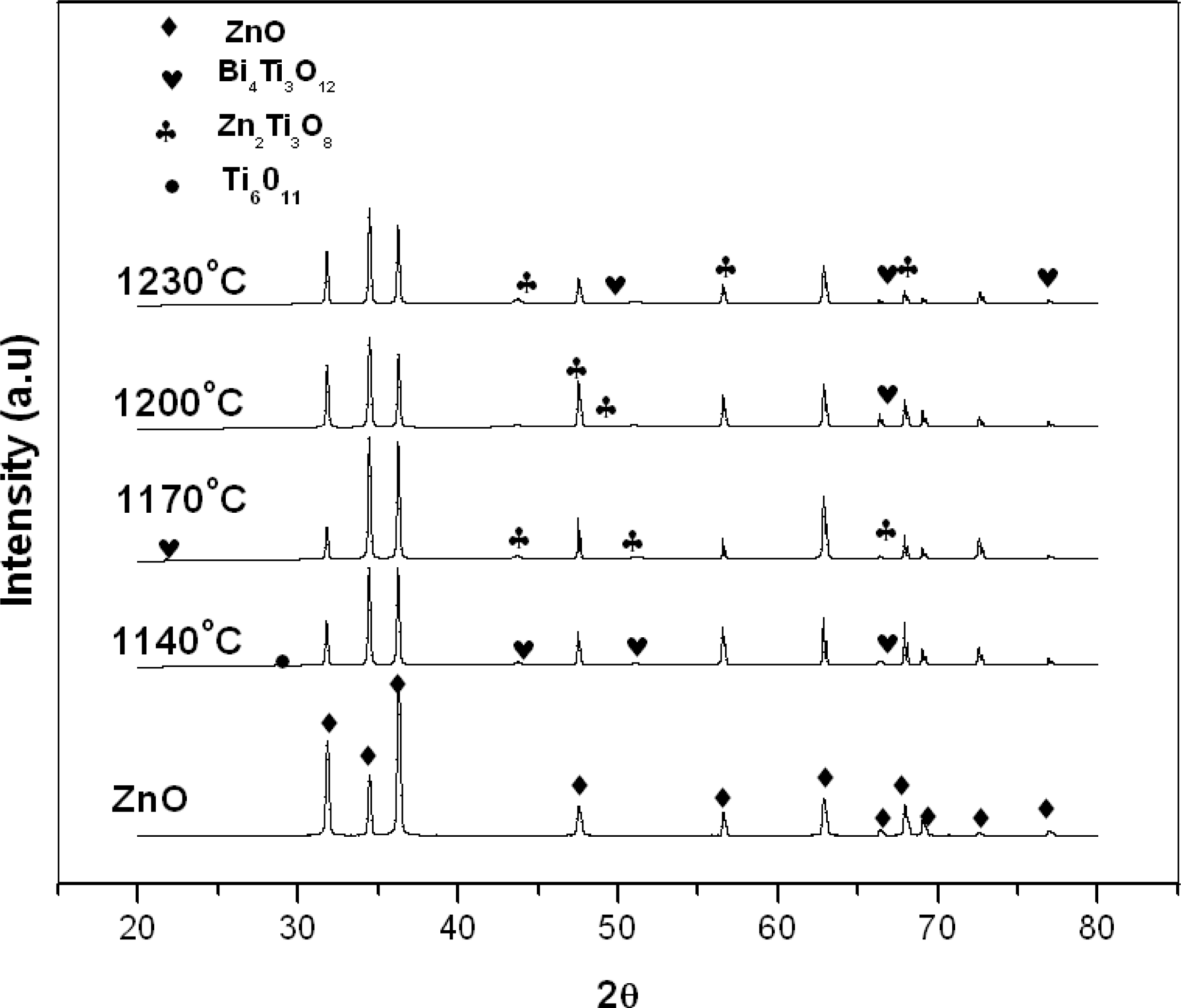
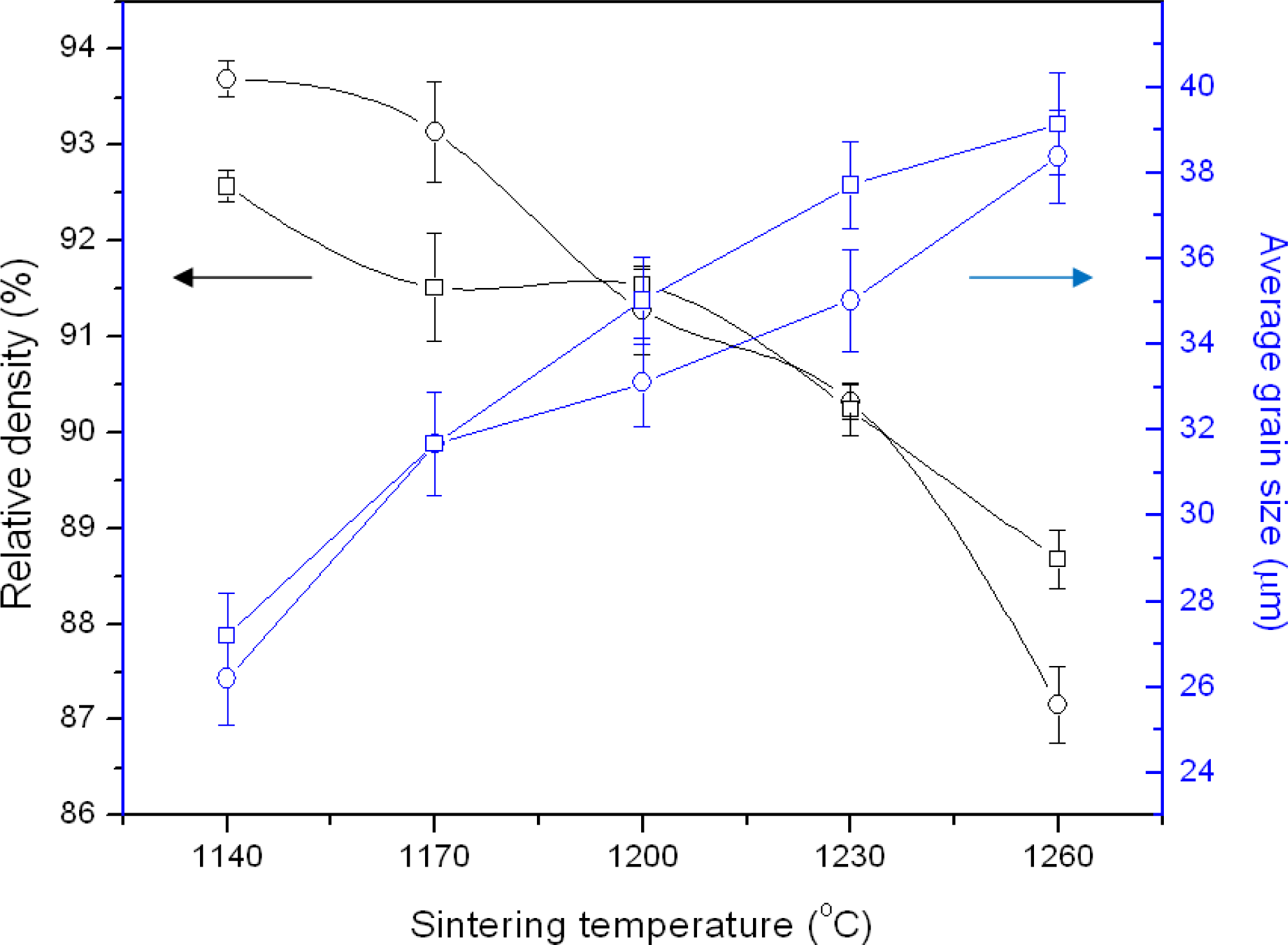
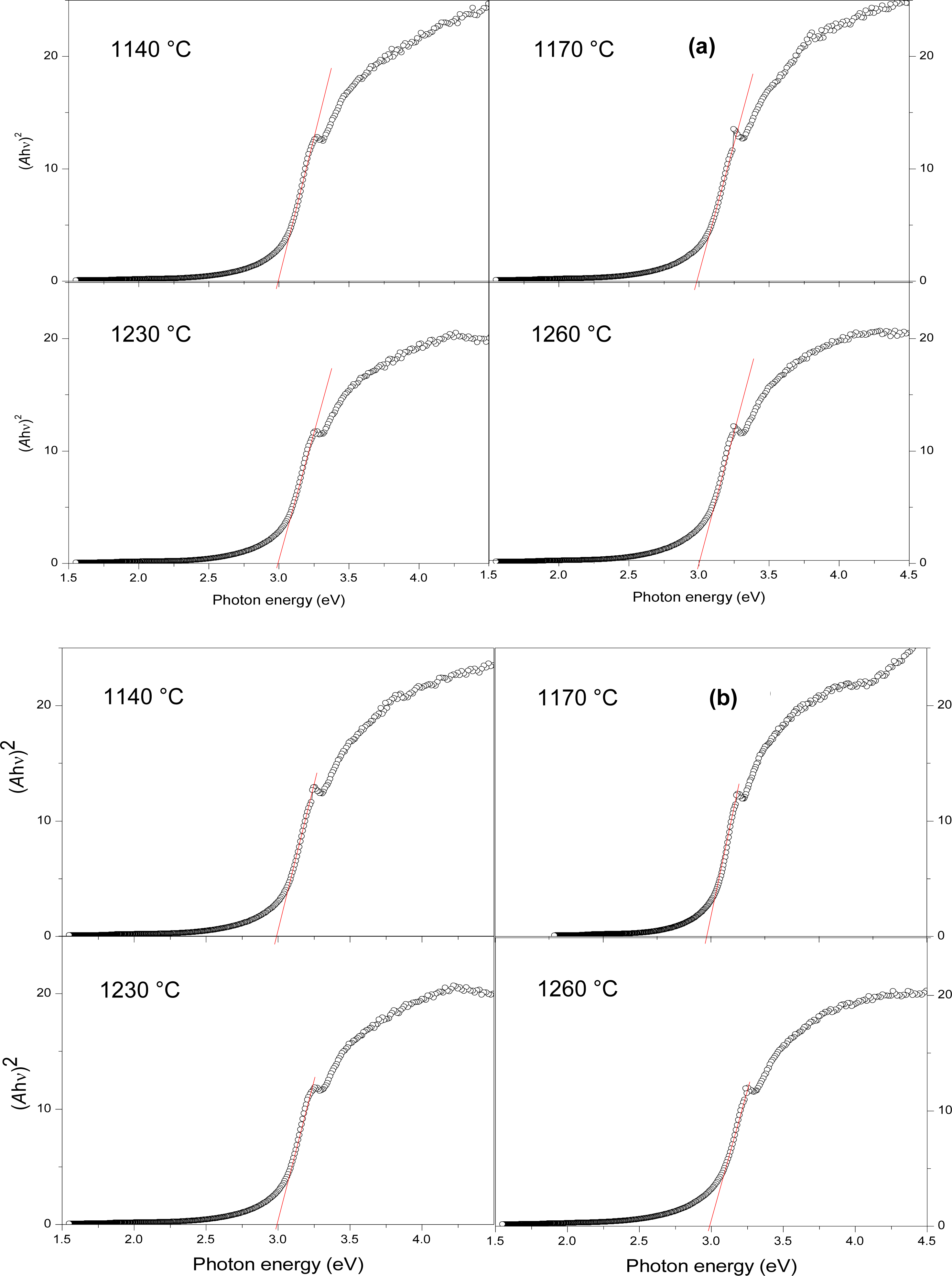
3. Results and Discussion
4. Conclusions
Acknowledgments
References
- Clarke, DR. Varistor ceramics. J. Am. Ceram. Soc 1999, 82, 485–502. [Google Scholar]
- Souza, FL; Gomez, JW; Bueno, PR; Cassia-Santos, MR; Araujo, AL; Leiti, ER; Longo, E; Varela, AJ. Effect of the addition of ZnO seeds on the electrical properties of ZnO-based varistors. Mater. Chem. Phys 2003, 80, 512–516. [Google Scholar]
- Matsuoka, M. Nonohmic properties of zinc oxide ceramics. Jpn. J. Appl. Phys 1971, 10, 736–746. [Google Scholar]
- Snow, GS; White, SS; Cooper, RA; Armijo, JR. Characterization of high field varistors in the system ZnO-CoO-PbO-Bi2O3. Am. Ceram. Soc. Bulletin 1980, 59, 617–622. [Google Scholar]
- Eda, K. Zinc oxide varistor. IEEE Electr. Insul. Mag 1989, 5, 28–41. [Google Scholar]
- Bai, SN; Shieh, JS; Tseng, TY. Characteristic analysis of ZnO varistors made with spherical precipitation powders. Mater. Chem. Phys 1995, 41, 104–109. [Google Scholar]
- Toplan, O; Gunay, V; Ozkan, OT. Grain growth in the MnO added ZnO-6 wt% Sb2O3 ceramic system. Ceram. Int 1997, 23, 251–255. [Google Scholar]
- Fah, CP; Wang, J. Effect of high-energy mechanical activation on the microstructure and electrical properties of ZnO-based varistors. Solid State Ionics 2000, 132, 107–117. [Google Scholar]
- Linsebigler, AL; Lu, GQ; Yates, JT. Photocatalysis on TiO2 surfaces-principles, mechanisms, and selected results. Chem. Rev 1995, 95, 735–758. [Google Scholar]
- Nahm, C-W; Shin, B-C. Highly stable nonlinear properties of ZnO-Pr6O11-CoO-Cr2O3-Y2O3-based varistor ceramics. Mater. Lett 2003, 57, 1322–1326. [Google Scholar]
- Ghoosh, AK; Som, KK; Chatterjee, S; Chaudhuri, BK. Photoacoustic spectroscopic study of energy gap, optical absorption, and thermal diffusivity of polycrystalline ZnSexTe1−x (0 ≤ x ≤ 1) alloys. Phys. Rev. B 1995, 51, 4842–4848. [Google Scholar]
- Zelaya-Angel, O; Alvarado-Gil, JJ; Lozada-Morales, R. Band-gap shift in CdS semiconductor by photoacoustic spectroscopy: Evidence of a cubic to hexagonal lattice transition. Appl. Phys. Lett 1994, 64, 291–293. [Google Scholar]
- Escobedo Morales, A; Sànchez Mora, E; Pal, U. Use of diffuse reflectance spectroscopy for optical characterization of un-supported nanostructures. Revista Mexicana de Física S 2007, 53, 18–22. [Google Scholar]
- Wang, JF; Su, WB; Chen, HC; Wang, WX; Zang, GZ. (Pr, Co, Nb)-Doped SnO2 Varistor Ceramics. J. Am. Ceram. Soc 2005, 88, 331–334. [Google Scholar]
- Wurst, JC; Nelson, JA. Lineal intercept technique for measuring grain size in two-phase polycrystalline ceramics. J. Am. Ceram. Soc 1972, 55, 109–111. [Google Scholar]
- Gonzalez-Hernandez, J; Gorley, PM; Horley, PP; Vartsabyuk, OM; Vorobiev Yu, V. X-ray, kinetic and optical properties of thin CuInS2 films. Thin Solid Films 2002, 403–404, 471–475. [Google Scholar]
- Smith, RA. Semiconductors, 2nd ed; Cambridge University Press: Cambridge, UK, 1978. [Google Scholar]
- Sung, GY; Kim, CH. Anisotropic grain growth of ZnO grain in the varistor system ZnO-Bi2O3-MnO-TiO2. Adv. Ceram. Mater 1988, 3, 604–606. [Google Scholar]
- Suzuki, H; Bradt, RC. Grain growth of ZnO in ZnO-Bi2O3 ceramics with TiO2 additions. J. Am. Ceram. Soc 1995, 78, 1354–1360. [Google Scholar]
- Sabri, MGM; Azmi, BZ; Rizwan, Z; Halimah, MK; Hashim, M; Sidek, HAA. Application of direct current and temperature stresses of low-voltage ZnO based varistor ceramics. Am. J. Appl. Sci 2009, 6, 1591–1595. [Google Scholar]
- Toyoda, T; Shimamoto, S. Effect of Bi2O3 in ceramic ZnO on photoacoustic spectra and current voltage characteristics. Jpn. J. Appl. Phys 1998, 37, 2827–2831. [Google Scholar]



© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ghazali, M.S.M.; Zakaria, A.; Rizwan, Z.; Kamari, H.M.; Hashim, M.; Zaid, M.H.M.; Zamiri, R. Use of a Reflectance Spectroscopy Accessory for Optical Characterization of ZnO-Bi2O3-TiO2 Ceramics. Int. J. Mol. Sci. 2011, 12, 1496-1504. https://doi.org/10.3390/ijms12031496
Ghazali MSM, Zakaria A, Rizwan Z, Kamari HM, Hashim M, Zaid MHM, Zamiri R. Use of a Reflectance Spectroscopy Accessory for Optical Characterization of ZnO-Bi2O3-TiO2 Ceramics. International Journal of Molecular Sciences. 2011; 12(3):1496-1504. https://doi.org/10.3390/ijms12031496
Chicago/Turabian StyleGhazali, Mohd Sabri Mohd, Azmi Zakaria, Zahid Rizwan, Halimah Mohamed Kamari, Mansor Hashim, Mohd Hafiz Mohd Zaid, and Reza Zamiri. 2011. "Use of a Reflectance Spectroscopy Accessory for Optical Characterization of ZnO-Bi2O3-TiO2 Ceramics" International Journal of Molecular Sciences 12, no. 3: 1496-1504. https://doi.org/10.3390/ijms12031496



