Antioxidant Activities of Fractions of Polymeric Procyanidins from Stem Bark of Acacia confusa
Abstract
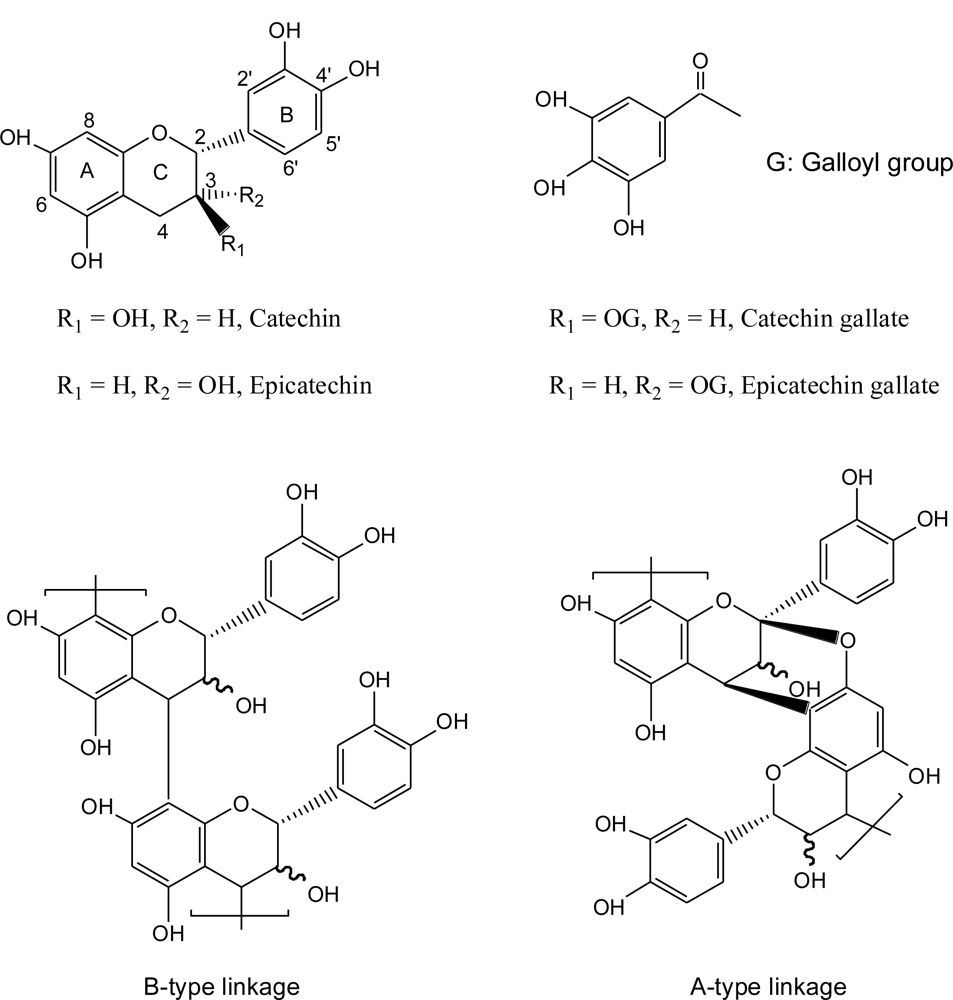
:1. Introduction
2. Results and Discussion
2.1. Antioxidant Activities of Fractions F1-F11
2.2. MALDI-TOF MS Analysis
2.3. Thiolysis of Fractions F2–F11
3. Experimental Section
3.1. Chemicals and Plant Materials
3.2. Extraction and Fractionation of Procyanidin Polymers
3.3. DPPH Radical Scavenging Activity
3.4. Ferric Reducing/Antioxidant Power (FRAP) Assay
3.5. MALDI-TOF MS Analysis
3.6. Thiolysis Reaction and HPLC Analysis
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Fuleki, T; Ricardo da Silva, JM. Catechin and procyanidin composition of seeds from grape cultivars grown in Ontario. J. Agric. Food Chem 1997, 45, 1156–1160. [Google Scholar]
- Prieur, C; Rigaud, J; Cheynier, V; Moutounet, M. Oligomeric and polymeric procyanidins from grape seeds. Phytochemistry 1994, 36, 781–784. [Google Scholar]
- de Freitas, VAP; Glories, Y; Laguerres, M. Incidence of molecular structure in oxidation of grape seed procyanidins. J. Agric. Food Chem 1998, 46, 376–382. [Google Scholar]
- Ricardo da Silva, JM; Rigaud, J; Cheynier, V; Cheminat, A; Moutounet, M. Procyanidin dimers and trimers from grape seeds. Phytochemistry 1991, 30, 1259–1264. [Google Scholar]
- Gu, L; Kelm, M; Hammerstone, JF; Beecher, G; Cunningham, D; Vannozzi, S; Prior, RL. Fractionation of polymeric procyanidins from lowbush blueberry and quantification of procyanidins in selected foods with an optimized normal-phase HPLC-MS fluorescent detection method. J. Agric. Food Chem 2002, 50, 4852–4860. [Google Scholar]
- Vivas, N; Glories Isabelle, Y. A complete structural and conformational investigation of procyanidin A2 dimer. Tetrahedron Lett 1996, 37, 2015–2018. [Google Scholar]
- Esatbeyoglu, T; Wray, V; Winterhalter, P. Dimeric procyanidins: Screening for B1 to B8 and semisynthetic preparation of B3, B4, B6, and B8 from a polymeric procyanidin fraction of White Willow Bark (Salix alba). J. Agric. Food Chem 2010, 58, 7820–7830. [Google Scholar]
- Santos-Buelga, C; Scalbert, A. Proanthocyanidins and tannin-like compounds-nature, occurrence, dietary intake and effects on nutrition and health. J. Sci. Food Agric 2000, 80, 1094–1117. [Google Scholar]
- Ariga, T; Koshiyama, I; Fukushima, D. Antioxidative properties of procyanidins B-1 and B-3 from azuki beans in aqueous systems. Agric. Biol. Chem 1988, 52, 2717–2722. [Google Scholar]
- Landrault, N; Poucheret, P; Ravel, P; Gasc, F; Cros, G; Teissedre, PL. Antioxidant capacities and phenolics levels of French wines from different varieties and vintages. J. Agric. Food Chem 2001, 49, 3341–3348. [Google Scholar]
- Pinent, M; Blade, C; Salvadó, MJ; Blay, M; Pujadas, G; Fernandez-Larrea, J; Arola, L; Ardévol, A. Procyanidin effects on adipocyte-related pathologies. Crit. Rev. Food Sci. Nutr 2006, 46, 543–550. [Google Scholar]
- Yilmaz, Y; Toledo, RT. Health aspects of functional grape seed constituents. Trends Food Sci. Technol 2004, 15, 422–433. [Google Scholar]
- Kolodziej, H; Haberland, C; Woerdenbag, HJ; Konings, AWT. Moderate cytotoxicity of proanthocyanidins to human tumour cell lines. Phytother. Res 1995, 9, 410–415. [Google Scholar]
- Wu, JH; Tung, YT; Wang, SY; Shyur, LF; Kuo, YH; Chang, ST. Phenolic antioxidants from the heartwood of Acacia confusa. J. Agric. Food Chem 2005, 53, 5917–5921. [Google Scholar]
- Kan, WS. Leguminosae. In Manual of Medicinal Plants in Taiwan; Kan, WS, Ed.; National Research Institute of Chinese Medicine: Taipei, Taiwan, 1978; Volume 2, pp. 239–240. [Google Scholar]
- Wei, SD; Zhou, HC; Lin, YM; Liao, MM; Chai, WM. MALDI-TOF MS analysis of condensed tannins with potent antioxidant activity from the leaf, stem bark and root bark of Acacia confusa. Molecules 2010, 15, 4369–4381. [Google Scholar]
- Chang, ST; Wu, JH; Wang, SY; Kang, PL; Yang, NS; Shyur, LF. Antioxidant activity of extracts from Acacia confusa bark and heartwood. J. Agric. Food Chem 2001, 49, 3420–3424. [Google Scholar]
- Tung, YT; Wu, JH; Kuo, YH; Chang, ST. Antioxidant activities of natural phenolic compounds from Acacia confusa bark. Bioresour. Technol 2007, 98, 1120–1123. [Google Scholar]
- de Gaulejac, NSC; Provost, C; Vivas, N. Comparative study of polyphenol scavenging activities assessed by different methods. J. Agric. Food Chem 1999, 47, 425–431. [Google Scholar]
- Hagerman, AE; Riedl, KM; Jones, GA; Sovik, KN; Ritchard, NT; Hartzfeld, PW; Riechel, TL. High molecular weight plant polyphenolics (tannins) as biological antioxidants. J. Agric. Food Chem 1998, 46, 1887–1892. [Google Scholar]
- Es-Safi, NE; Guyot, S; Ducrot, PH. NMR, ESI/MS, and MALDI-TOF/MS analysis of pear juice polymeric proanthocyanidins with potent free radical scavenging activity. J. Agric. Food Chem 2006, 54, 6969–6977. [Google Scholar]
- Hümmer, W; Schreier, P. Analysis of proanthocyanidins. Mol. Nutr. Food Res 2008, 52, 1381–1398. [Google Scholar]
- Rigaud, J; Escribano-Bailon, MT; Prieur, C; Souquet, JM; Cheynier, V. Normal-phase high-performance liquid chromatographic separation of procyanidins from cacao beans and grape seeds. J. Chromatogr. A 1993, 654, 255–260. [Google Scholar]
- Rohr, GE; Meier, B; Sticher, O. Quantitative reversed-phase high-performance liquid chromatography of procyanidins in Crataegus leaves and flowers. J. Chromatogr. A 1999, 835, 59–65. [Google Scholar]
- Yanagida, A; Kanda, T; Shoji, T; Ohnishi-Kameyama, M; Nagata, T. Fractionation of apple procyanidins by size-exclusion chromatography. J. Chromatogr. A 1999, 855, 181–190. [Google Scholar]
- Hammerstone, JF; Lazarus, SA; Mitchell, AE; Rucker, R; Schmitz, HH. Identification of procyanidins in cocoa (Theobroma cacao) and chocolate using high-performance liquid chromatography/mass spectrometry. J. Agric. Food Chem 1999, 47, 490–496. [Google Scholar]
- Lea, AGH; Timberlake, CF. The phenolics of ciders. 1. Procyanidins. J. Sci. Food Agric 1974, 25, 1537–1545. [Google Scholar]
- Taylor, AW; Barofsky, E; Kennedy, JA; Deinzer, ML. Hop (Humulus lupulus L.) proanthocyanidins characterized by mass spectrometry, acid catalysis, and gel permeation chromatography. J. Agric. Food Chem 2003, 51, 4101–4110. [Google Scholar]
- Hsieh, CY; Chang, ST. Antioxidant activities and xanthine oxidase inhibitory effects of phenolic phytochemicals from Acacia confusa twigs and branches. J. Agric. Food Chem 2010, 58, 1578–1583. [Google Scholar]
- Tung, YT; Chang, ST. Variation in antioxidant activity of extracts of Acacia confusa of different ages. Nat. Prod. Commun 2010, 5, 73–76. [Google Scholar]
- Tung, YT; Wu, JH; Hsieh, CY; Chen, PS; Chang, ST. Free radical-scavenging phytochemicals of hot water extracts of Acacia confusa leaves detected by an on-line screening method. Food Chem 2009, 115, 1019–1024. [Google Scholar]
- Molyneux, P. The use of the stable free radical diphenylpicrylhydrazyl (DPPH) for estimating antioxidant activity. Songklanakarin J. Sci. Technol 2004, 26, 211–219. [Google Scholar]
- Loo, AY; Jain, K; Darah, I. Antioxidant and radical scavenging activities of the pyroligneous acid from a mangrove plant, Rhizophora apiculata. Food Chem 2007, 104, 300–307. [Google Scholar]
- Wei, SD; Zhou, HC; Lin, YM. Antioxidant activities of extract and fractions from the hypocotyls of the mangrove plant Kandelia candel. Int. J. Mol. Sci 2010, 11, 4080–4093. [Google Scholar]
- Zhang, LL; Lin, YM; Zhou, HC; Wei, SD; Chen, JH. Condensed tannins from mangrove species Kandelia candel and Rhizophora mangle and their antioxidant activity. Molecules 2010, 15, 420–431. [Google Scholar]
- Zhang, LL; Lin, YM. Tannins from Canarium album with potent antioxidant activity. J. Zhejiang Univ. Sci. B 2008, 9, 407–415. [Google Scholar]
- Danis, PO; Karr, DE. A facile sample preparation for the analysis of synthetic organic polymers by matrix-assisted laser desorption/ionization. Org. Mass Spectrom 1993, 28, 923–925. [Google Scholar]
- Ehring, H; Karas, M; Hillenkamp, F. Role of photoionization and photochemistry in ionization processes of organic molecules and relevance for matrix-assisted laser desorption lonization mass spectrometry. Org. Mass Spectrom 1992, 27, 472–480. [Google Scholar]
- Ku, CS; Mun, SP. Characterization of proanthocyanidin in hot water extract isolated from Pinus radiata bark. Wood Sci. Technol 2007, 41, 235–247. [Google Scholar]
- Pasch, H; Pizzi, A; Rode, K. MALDI-TOF mass spectrometry of polyflavonoid tannins. Polymer 2001, 42, 7531–7539. [Google Scholar]
- Montaudo, G; Montaudo, MS; Samperi, F. Martix-assisted laser desorption/ionization mass spectrometry of polymers (MALDI-MS). In Mass Spectrometry of Polymers; Montaudo, G, Lattimer, RP, Eds.; CRC Press: Boca Raton, FL, USA, 2002; pp. 419–521. [Google Scholar]
- Yang, Y; Chien, M. Characterization of grape procyanidins using high-performance liquid chromatography/mass spectrometry and matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. J. Agric. Food Chem 2000, 48, 3990–3996. [Google Scholar]
- Ohnishi-Kameyama, M; Yanagida, A; Kanda, T; Nagata, T. Identification of catechin oligomers from apple (Malus pumila cv. Fuji) in matrix-assisted laser desorption/ionization time-of-flight mass spectrometry and fast-atom bombardment mass spectrometry. Rap. Comm. Mass Spectrom 1997, 11, 31–36. [Google Scholar]
- Mané, C; Sommerer, N; Yalcin, T; Cheynier, V; Cole, RB; Fulcrand, H. Assessment of the molecular weight distribution of tannin fractions through MALDI-TOF MS analysis of protein-tannin complexes. Anal. Chem 2007, 79, 2239–2248. [Google Scholar]
- Twerenbold, D; Gerber, D; Gritti, D; Gonin, Y; Netuschill, A; Rossel, F; Schenker, D; Vuilleumier, JL. Single molecule detector for mass spectrometry with mass independent detection efficiency. Proteomics 2001, 1, 66–69. [Google Scholar]
- Perret, C; Pezet, R; Tabacchi, R. Fractionation of grape tannins and analysis by matrix-assisted laser desorption/ionisation time-of-flight mass spectrometry. Phytochem. Anal 2003, 14, 202–208. [Google Scholar]
- Behrens, A; Maie, N; Knicker, H; Kögel-Knabner, I. MALDI-TOF mass spectrometry and PSD fragmentation as means for the analysis of condensed tannins in plant leaves and needles. Phytochemistry 2003, 62, 1159–1170. [Google Scholar]
- Foo, LY; Lu, Y; Howell, AB; Vorsa, N. The structure of cranberry proanthocyanidins which inhibit adherence of uropathogenic P-fimbriated Escherichia coli in vitro. Phytochemistry 2000, 54, 173–181. [Google Scholar]
- Krueger, C; Dopke, N; Treichel, P; Folts, J; Reed, J. Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry of polygalloyl polyflavan-3-ols in grape seed extract. J. Agric. Food Chem 2000, 48, 1663–1667. [Google Scholar]
- Jerez, M; Sineiro, J; Guitián, E; Núñez, MJ. Identification of polymeric procyanidins from pine bark by mass spectrometry. Rap. Comm. Mass Spectrom 2009, 23, 4013–4018. [Google Scholar]
- Torres, JL; Selga, A. Procyanidin size and composition by thiolysis with cysteamine hydrochloride and chromatography. Chromatographia 2003, 57, 441–445. [Google Scholar]
- Svedström, U; Vuorela, H; Kostiainen, R; Huovinen, K; Laakso, I; Hiltunen, R. High-performance liquid chromatographic determination of oligomeric procyanidins from dimers up to the hexamer in hawthorn. J. Chromatogr. A 2002, 968, 53–60. [Google Scholar]
- Touriño, S; Selga, A; Jiménez, A; Juliá, L; Lozano, C; Lizárraga, D; Cascante, M; Torres, JL. Procyanidin fractions from pine (Pinus pinaster) bark: Radical scavenging power in solution, antioxidant activity in emulsion, and antiproliferative effect in melanoma cells. J. Agric. Food Chem 2005, 53, 4728–4735. [Google Scholar]
- Jerez, M; Touriño, S; Sineiro, J; Torres, JL; Núñez, MJ. Procyanidins from pine bark: Relationships between structure, composition and antiradical activity. Food Chem 2007, 104, 518–527. [Google Scholar]
- Braca, A; de Tommasi, N; Di Bari, L; Pizza, C; Politi, M; Morelli, I. Antioxidant principles from Bauhinia tarapotensis. J. Nat. Prod 2001, 64, 892–895. [Google Scholar]
- Benzie, IFF; Strain, JJ. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem 1996, 239, 70–76. [Google Scholar]
- Xiang, P; Lin, YM; Lin, P; Xiang, C. Effects of adduct ions on matrix-assisted laser desorption/ionization time of flight mass spectrometry of condensed tannins: A prerequisite knowledge. Chin. J. Anal. Chem 2006, 34, 1019–1022. [Google Scholar]
- Torres, JL; Lozano, C. Chromatographic characterization of proanthocyanidins after thiolysis with cysteamine. Chromatographia 2001, 54, 523–526. [Google Scholar]


| Fractions | Antioxidant Activity | |
|---|---|---|
| IC50/DPPH (μg/mL) a | FRAP (mmol AAE/g) b | |
| F1 | 450.24 ± 5.21 a | 1.33 ± 0.06 g |
| F2 | 136.38 ± 4.01 b | 4.20 ± 0.09 e |
| F3 | 116.64 ± 1.83 d | 5.55 ± 0.12 c |
| F4 | 99.12 ± 0.77 e | 6.09 ± 0.06 b |
| F5 | 90.74 ± 0.86 g | 6.13 ± 0.02 b |
| F6 | 93.88 ± 0.44 f | 5.34 ± 0.06 d |
| F7 | 89.84 ± 0.74 g | 5.52 ± 0.07 c |
| F8 | 87.42 ± 1.82 g | 5.50 ± 0.06 c |
| F9 | 81.91 ± 0.60 h | 6.33 ± 0.04 a |
| F10 | 115.23 ± 0.94 d | 4.29 ± 0.05 e |
| F11 | 129.72 ± 1.77 c | 3.97 ± 0.04 f |
| Ascorbic acid | 101.96 ± 1.84 e | - |
| BHA | 116.91 ± 0.97 d | 5.18 ± 0.11 d |
| Polymer | Observed [M+Cs]+ | |||||||
|---|---|---|---|---|---|---|---|---|
| F2 | F3 | F4 | F5 | F6 | F7 | F8 | F9 | |
| DP2 | -- | 711.21 | 711.13 | -- | -- | -- | -- | -- |
| DP3 | 999.25 | 999.29 | 999.20 | 999.24 | 999.26 | 999.43 | 999.37 | 999.36 |
| DP4 | -- | 1287.41 | 1287.26 | 1287.30 | 1287.33 | 1287.40 | 1287.33 | 1287.38 |
| DP5 | -- | -- | 1575.35 | 1575.36 | 1575.38 | 1575.53 | 1575.51 | 1575.62 |
| DP6 | -- | -- | -- | 1864.46 | 1864.45 | 1864.63 | 1863.59 | 1863.57 |
| DP7 | -- | -- | -- | -- | 2152.54 | 2152.70 | 2152.60 | 2152.60 |
| DP8 | -- | -- | -- | -- | 2440.63 | 2440.78 | 2440.70 | 2440.63 |
| DP9 | -- | -- | -- | -- | -- | 2728.87 | 2728.76 | 2728.71 |
| DP10 | -- | -- | -- | -- | -- | 3016.93 | 3017.69 | 3017.10 |
| DP11 | -- | -- | -- | -- | -- | 3304.86 | 3305.39 | 3305.81 |
| DP12 | -- | -- | -- | -- | -- | -- | 3593.99 | 3593.81 |
| DP13 | -- | -- | -- | -- | -- | -- | 3881.56 | 3881.59 |
| DP14 | -- | -- | -- | -- | -- | -- | -- | 4169.72 |
| DP15 | -- | -- | -- | -- | -- | -- | -- | 4457.63 |
| Fractions | mDP by Thiolysis | Polymer Range in MALDI | Most Intense Polymer in MALDI |
|---|---|---|---|
| F2 | 1.68 ± 0.05 j | 3 | 3 |
| F3 | 2.13 ± 0.16 i | 2–4 | 3 |
| F4 | 2.71 ± 0.06 h | 2–5 | 2 |
| F5 | 5.46 ± 0.09 g | 3–6 | 4 |
| F6 | 7.37 ± 0.08 f | 3–8 | 5 |
| F7 | 9.49 ± 0.11 e | 3–11 | 6 |
| F8 | 9.80 ± 0.09 d | 3–13 | 8 |
| F9 | 10.65 ± 0.13 c | 3–15 | 9 |
| F10 | 15.44 ± 0.06 b | - | - |
| F11 | 17.31 ± 0.16 a | - | - |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wei, S.-D.; Zhou, H.-C.; Lin, Y.-M. Antioxidant Activities of Fractions of Polymeric Procyanidins from Stem Bark of Acacia confusa. Int. J. Mol. Sci. 2011, 12, 1146-1160. https://doi.org/10.3390/ijms12021146
Wei S-D, Zhou H-C, Lin Y-M. Antioxidant Activities of Fractions of Polymeric Procyanidins from Stem Bark of Acacia confusa. International Journal of Molecular Sciences. 2011; 12(2):1146-1160. https://doi.org/10.3390/ijms12021146
Chicago/Turabian StyleWei, Shu-Dong, Hai-Chao Zhou, and Yi-Ming Lin. 2011. "Antioxidant Activities of Fractions of Polymeric Procyanidins from Stem Bark of Acacia confusa" International Journal of Molecular Sciences 12, no. 2: 1146-1160. https://doi.org/10.3390/ijms12021146
APA StyleWei, S.-D., Zhou, H.-C., & Lin, Y.-M. (2011). Antioxidant Activities of Fractions of Polymeric Procyanidins from Stem Bark of Acacia confusa. International Journal of Molecular Sciences, 12(2), 1146-1160. https://doi.org/10.3390/ijms12021146




