Intracluster Ion Molecule Reactions Following the Generation of Mg+ Within Polar Clusters
Abstract
:1. Introduction
2. Results and Discussion
2.1. Experimental Results
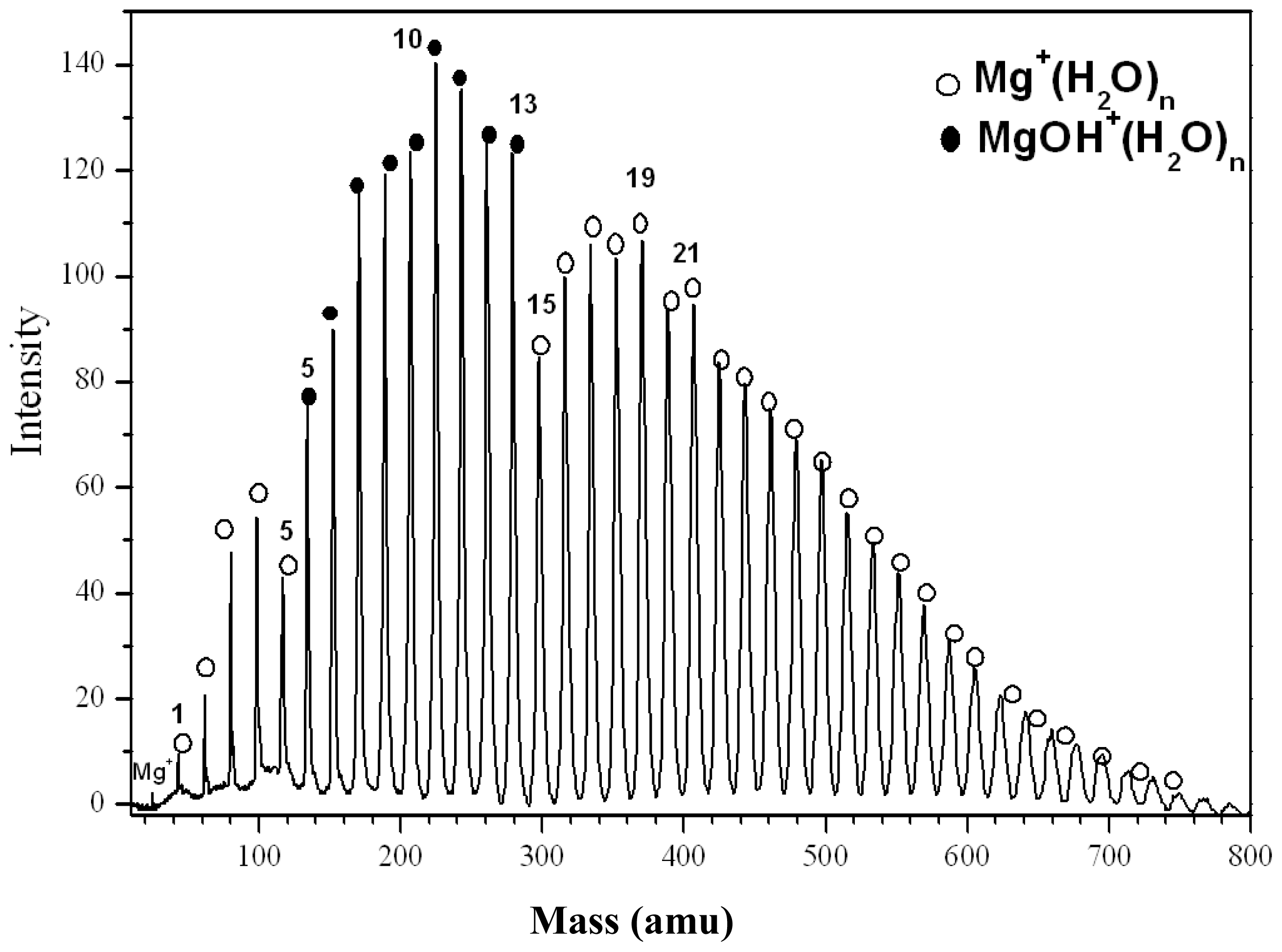
2.1.1. Mg+(H2O)n
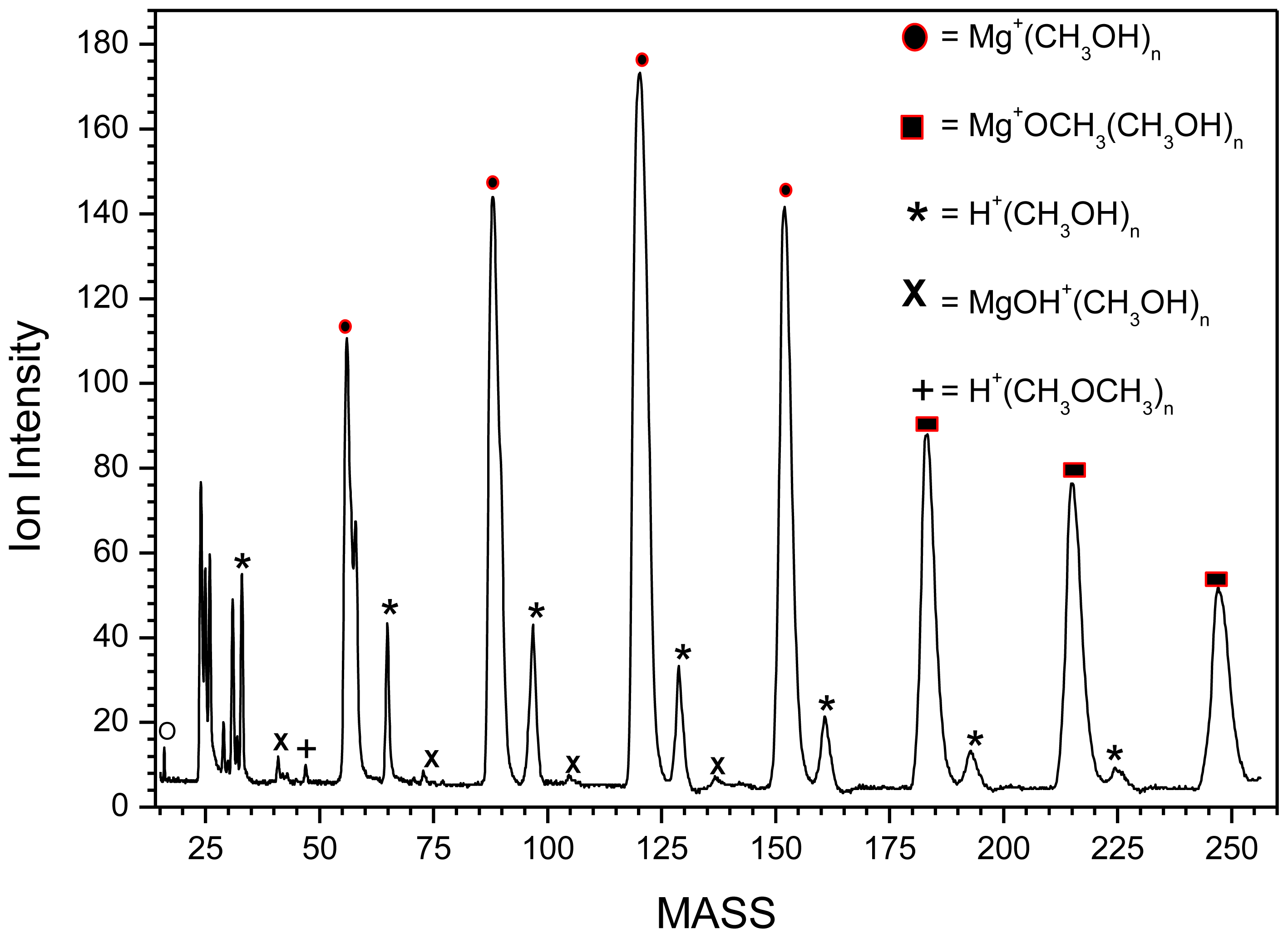
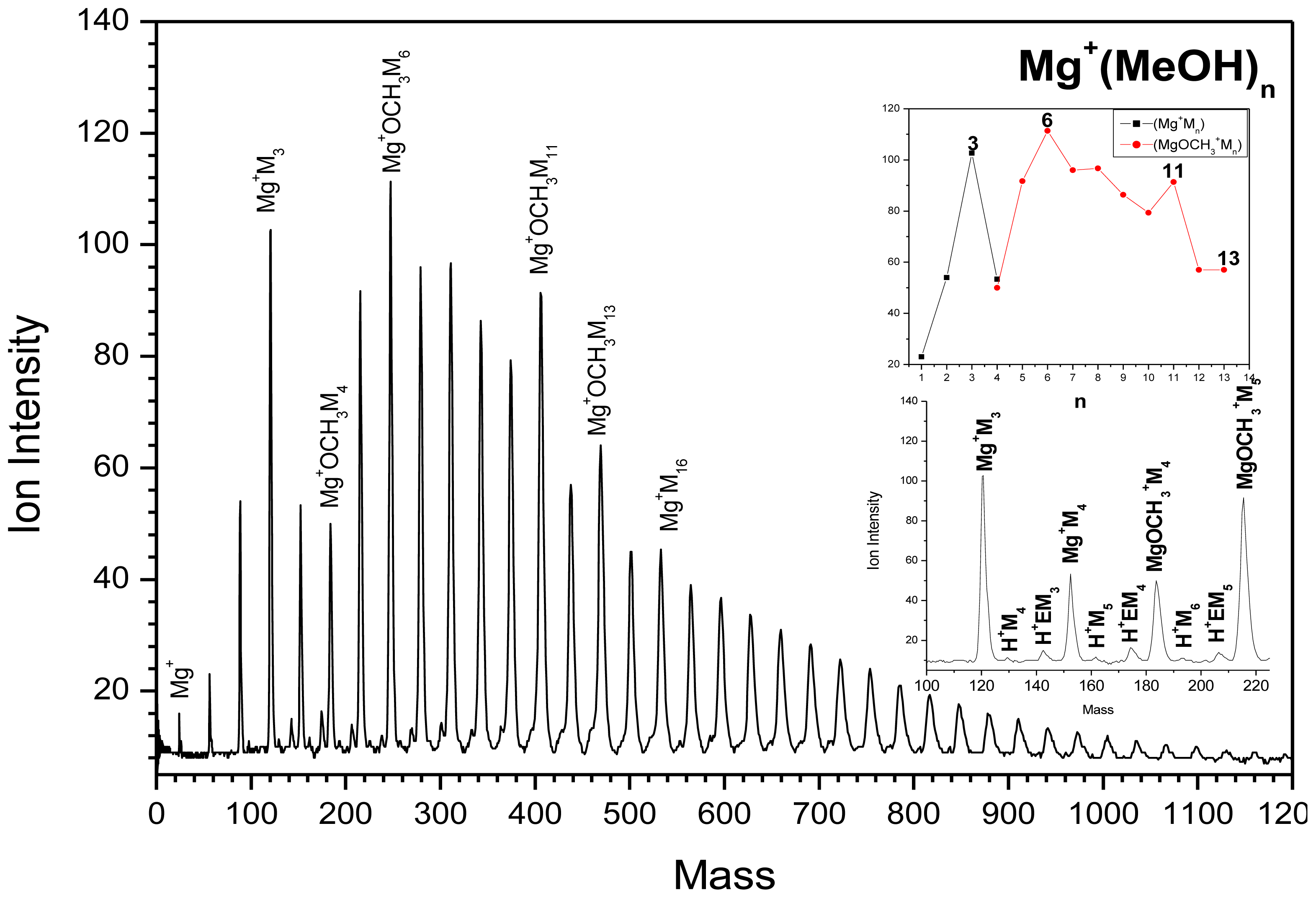
2.1.2. Mg+(CH3OH)n
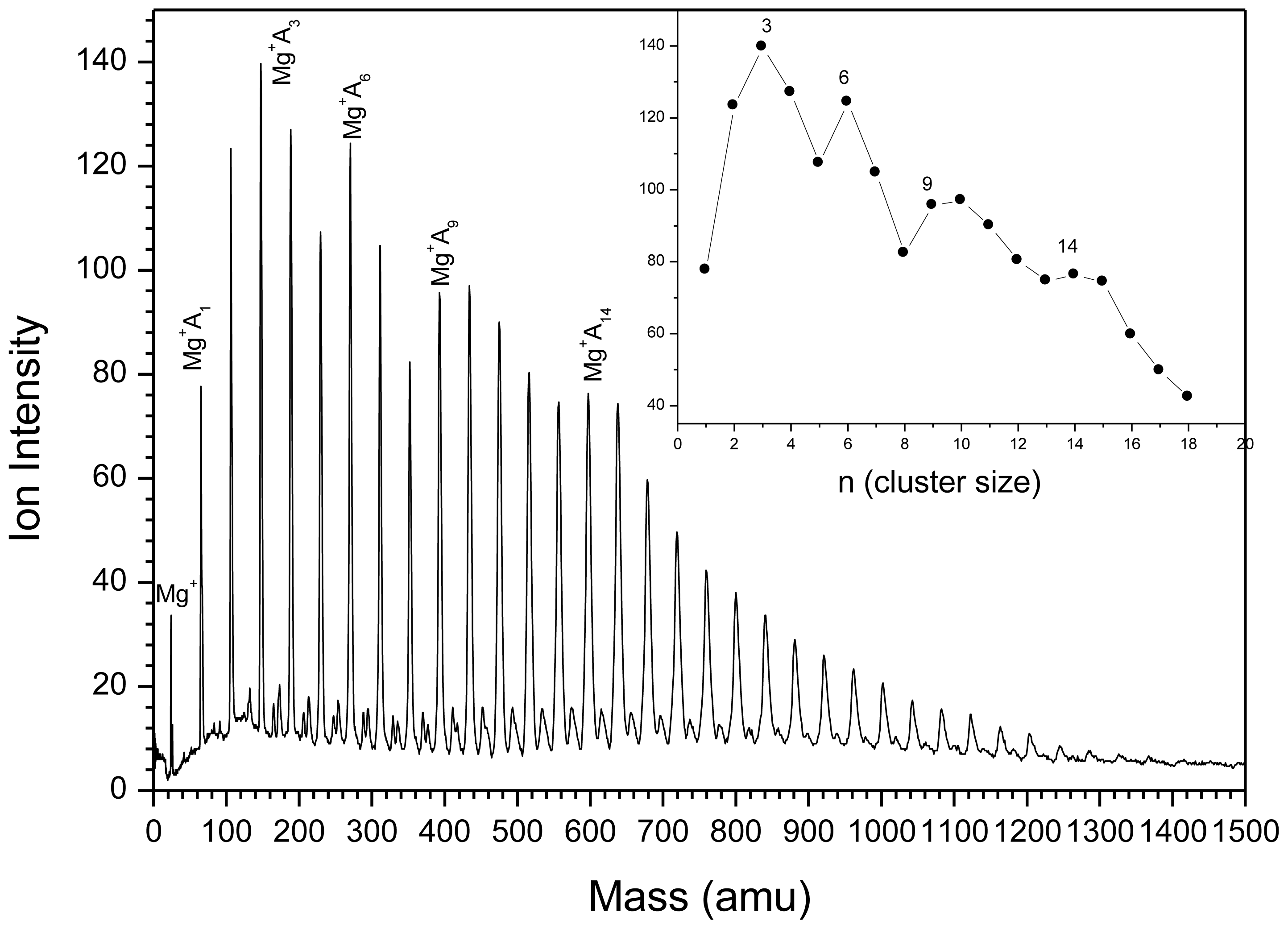
2.1.3. Mg+(CH3OCH3)n
2.1.4. Mg+(CH3CN)n
2.2. Discussion
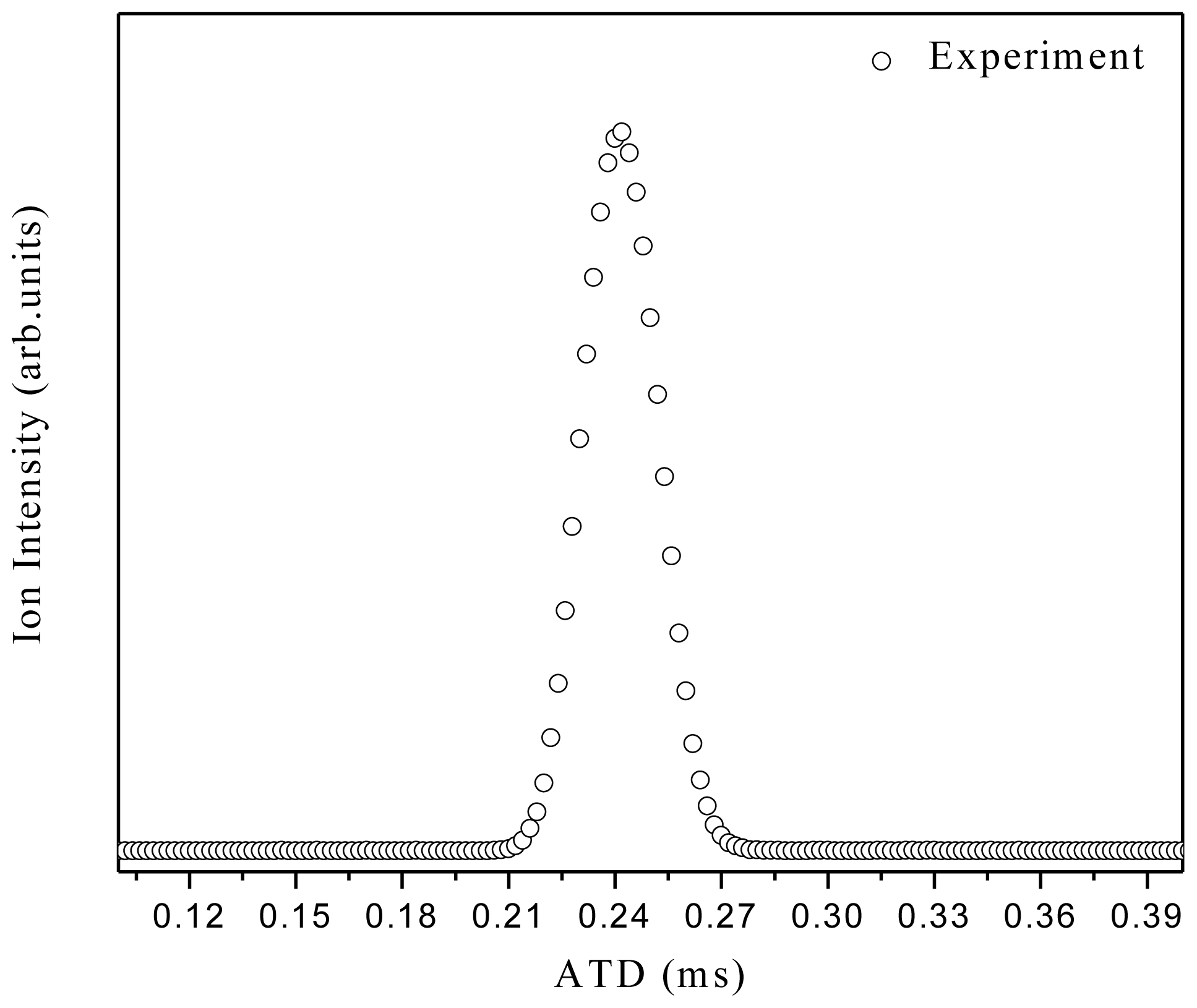
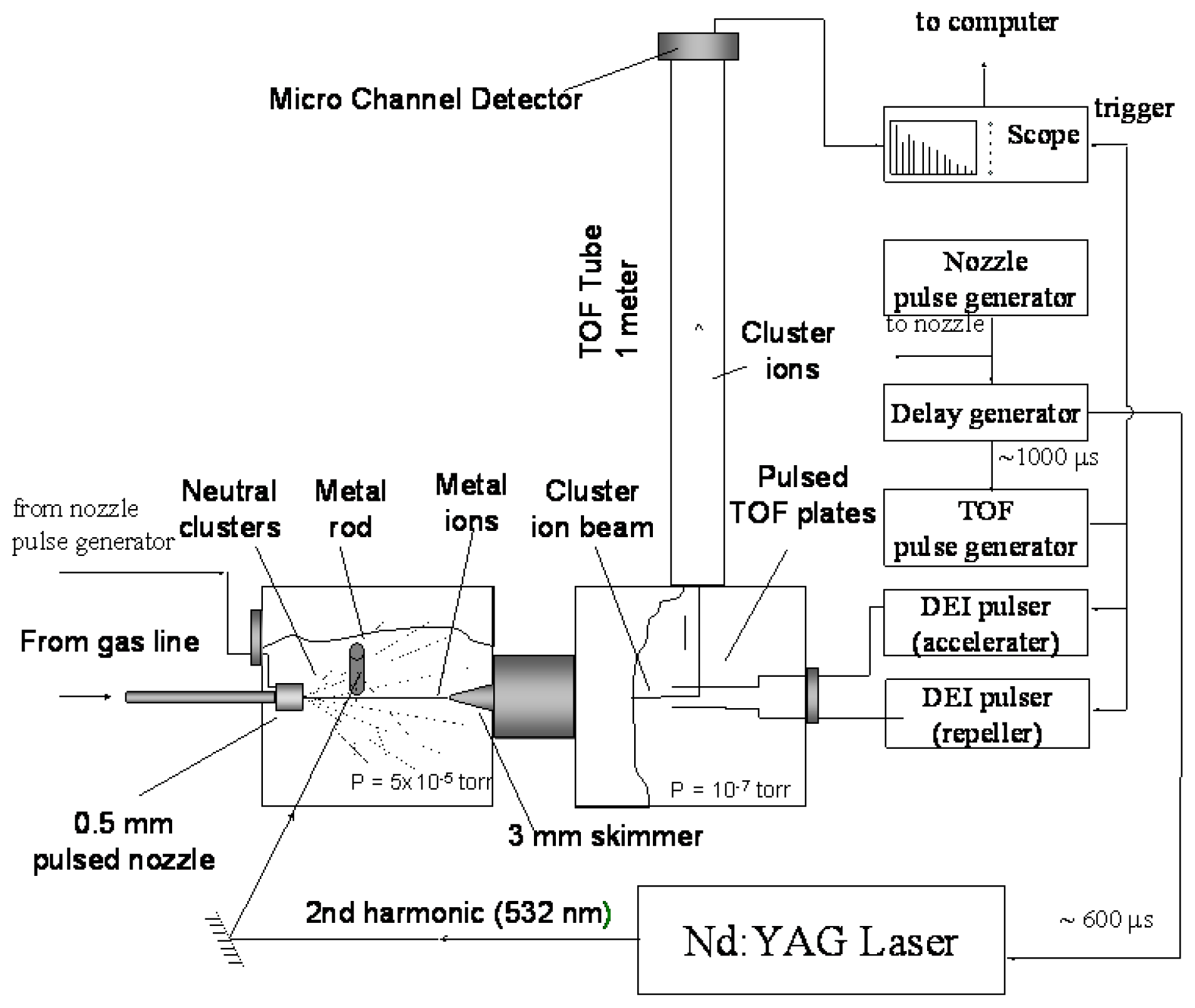
3. Experimental Section
4. Conclusions
Acknowledgments
References
- Soep, B.; Elhanine, M.; Schulz, C.P. Intracluster reactions of singly ionised magnesium atoms with dimethyl ether. Chem. Phys. Lett 2000, 327, 365–367. [Google Scholar]
- Castleman, A.W., Jr; Bowen, K.H., Jr. Clusters: Structure, energetics, and dynamics of intermediate states of matter. J. Phys. Chem. 1996, 100, 12911–12944. [Google Scholar]
- Sanekata, M.; Misaizu, F.; Fuke, K.; Iwata, S.; Hashimoto, K. Reactions of singly charged alkaline-earth metal ions with water clusters: Characteristic size distribution of product ions. J. Am. Chem. Soc 1995, 117, 747–754. [Google Scholar]
- Misaizu, F.; Sanekata, M.; Fuke, K.; Iwata, S. Photodissociation study on Mg+(H2O)n, n = 1–5: Electronic structure and photoinduced intracluster reaction. J. Chem. Phys 1994, 100, 1161– 1171. [Google Scholar]
- Harms, A.C.; Khanna, S.N.; Chen, B.; Castleman, A.W., Jr. Dehydrogenation reactions in Mg+(H2O)n clusters. J. Chem. Phys. 1994, 100, 3540–3544. [Google Scholar]
- Berg, C.; Beyer, M.; Achatz, U.; Joos, S.; Niedner-Schatteburg, G.; Bondybey, V.E. Stability and reactivity of hydrated magnesium cations. Chem. Phys 1998, 239, 379–392. [Google Scholar]
- Uppal, J.S.; Staley, R.H. Reactions of aluminum(+) and magnesium(+) ions with alcohols and organic halides in the gas phase. J. Am. Chem. Soc 1982, 104, 1229–1234. [Google Scholar]
- Kaya, T.; Horiki, Y.; Kobayashi, M.; Shinohara, H.; Sato, H. Intensity gaps in M+(CH3OH)n as studied by laser ablation—Molecular beam method. Chem. Phys. Lett 1992, 200, 435–439. [Google Scholar]
- Woodward, C.A.; Dobson, M.P.; Stace, A.J. Chemistry of Mg+ and Mg2+ in association with methanol clusters. J. Phys. Chem. A 1997, 101, 2279–2285. [Google Scholar]
- Watanabe, H.; Iwata, S.; Hashimoto, K.; Misaizu, F.; Fuke, K. Molecular orbital studies of the structures and reactions of singly charged magnesium ion with water clusters, Mg+(H2O)n. J. Am. Chem. Soc 1995, 117, 755–763. [Google Scholar]
- Lu, W.; Yang, S. Reactions of alkaline earth metal ions with methanol clusters. J. Phys. Chem. A 1998, 102, 825–840. [Google Scholar]
- Castleman, A.W.; Wei, S. Cluster reaction. Annu. Rev. Phys. Chem 1994, 45, 685–719. [Google Scholar]
- Operti, L.; Tews, E.C.; MacMahon, T.J.; Freiser, B.S. Thermochemical properties of gas-phase MgOH and MgO determined by Fourier transform mass spectrometry. J. Am. Chem. Soc 1989, 111, 9152–9156. [Google Scholar]
- Operti, L.; Tews, E.C.; Freiser, B.S. Determination of gas-phase ligand binding energies to magnesium(+) by FTMS techniques. J. Am. Chem. Soc 1988, 110, 3847–3853. [Google Scholar]
- Moore, C.E. Lecture notes in physics. US Natl. Bur. Standards Circ 1949, 467, 309–312. [Google Scholar]
- Andersen, A.; Muntean, F.; Walter, D.; Rue, C.; Armentrout, P.B. Collision-induced dissociation and theoretical studies of Mg+ complexes with CO, CO2, NH3, CH4, CH3OH, and C6H6. J. Phys. Chem. A 2000, 104, 692–699. [Google Scholar]
- Donnelly, S.G.; Schmuttenmaer, C.A.; Qian, J.; Farrar, J.M. Frequency- and time-resolved cluster photodissociation dynamics in Sr+(H2O)n, Sr+(NH3)n and Sr+(CH3OH)n. J. Chem. Soc. Faraday Trans 1993, 89, 1457–1465. [Google Scholar]
- Donnelly, S.G.; Farrar, J.M. Size-dependent photodissociation cross sections for Sr+(NH3)n, n = 3–6: Rydberg state formation and electron transfer. J. Chem. Phys 1993, 98, 5450–5460. [Google Scholar]
- Dalleska, N.F.; Tjelta, B.L.; Armentrout, P.B. Sequential bond energies of water to Na+ (3s0), Mg+ (3s1), and Al+ (3s2). J. Phys. Chem 1994, 98, 4191–4195. [Google Scholar]
- Lide, D.R. CRC Editor Handbook of Chemistry and Physics, 77th ed; CRC-Press: New York, NY, USA, 1996. [Google Scholar]
- Ibrahim, Y.; Alsharaeh, E.; Mabrouki, R.; Momoh, P.; Xie, E.; El-Shall, M.S. Ion mobility of ground and excited states of laser-generated transition metal cations. J. Phys. Chem. A 2008, 112, 1112–1124. [Google Scholar]
- Ibrahim, Y.M.; Alsharaeh, E.H.; El-Shall, M.S. Evidence for penning ionization in the generation of electronically excited states of transition metal cations by laser vaporization. J. Phys. Chem. B 2004, 108, 3959–3962. [Google Scholar]
- Alsharaeh, E.H.; Samy El-Shall, M. Ion mobility study of the mechanism of the gas phase thermal polymerization of styrene and the structures of the early oligomers. Polymer 2011, 52, 5551–5559. [Google Scholar]
- Wiley, W.C.; McLaren, I.H. Time-of-flight mass spectrometer with improved resolution. Rev. Sci. Instrum 1955, 26, 1150–1157. [Google Scholar]J. Mass Spectrom 1997, 32, 1–11.


| Solvated Cluster ions | Magic Numbers | |||
|---|---|---|---|---|
| Mg+(H2O)n | 4 | 17 | 19 | 21 |
| MgOH+(H2O)n | 5 | 10 | 13 | - |
| Mg+(CH3OH)n | 3 | 16 | - | - |
| MgOCH3+(CH3OH)n | 6 | 11 | 13 | - |
| Mg+(CH3OCH3)n | 3 | 15 | 21 | - |
| Mg+(CH3CN)n | 3 | 6 | 9 | 14 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Alsharaeh, E.H. Intracluster Ion Molecule Reactions Following the Generation of Mg+ Within Polar Clusters. Int. J. Mol. Sci. 2011, 12, 9095-9107. https://doi.org/10.3390/ijms12129095
Alsharaeh EH. Intracluster Ion Molecule Reactions Following the Generation of Mg+ Within Polar Clusters. International Journal of Molecular Sciences. 2011; 12(12):9095-9107. https://doi.org/10.3390/ijms12129095
Chicago/Turabian StyleAlsharaeh, Edreese H. 2011. "Intracluster Ion Molecule Reactions Following the Generation of Mg+ Within Polar Clusters" International Journal of Molecular Sciences 12, no. 12: 9095-9107. https://doi.org/10.3390/ijms12129095




