New Approaches to Clarify Antinociceptive and Anti-Inflammatory Effects of the Ethanol Extract from Vernonia condensata Leaves
Abstract
:1. Introduction
2. Results and Discussion
2.1. Phytochemistry Screening
2.2. Acute Toxicity
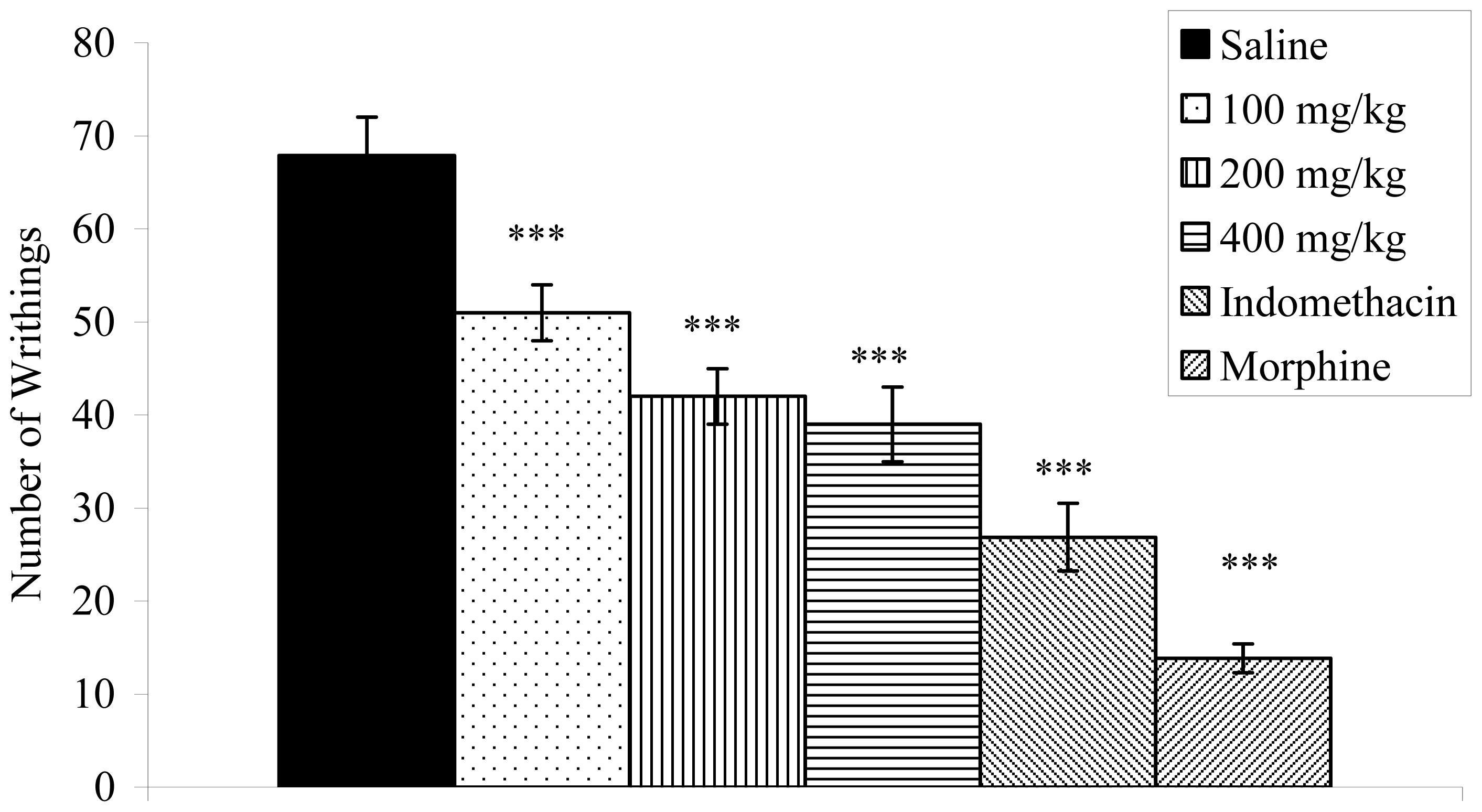
2.3. Writhing Response Induced by Acetic Acid in Mice
2.4. Effects on Formalin-Induced Nociception in Mice
2.5. Effects on Hot-Plate Latency Assay in Mice
2.6. Effect on Sleeping Time
2.7. Effects on Carrageenan-Induced Edema in Rats
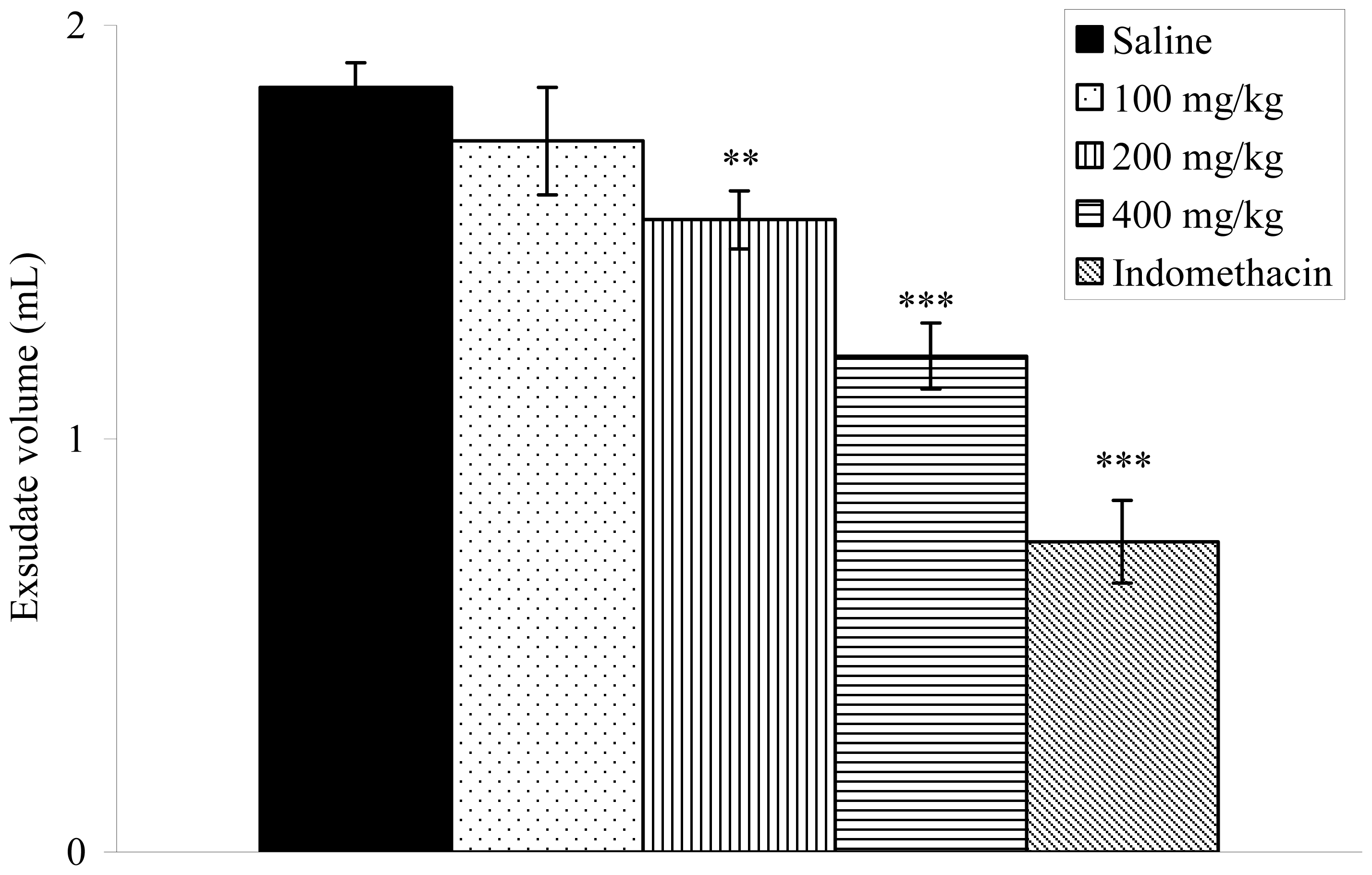
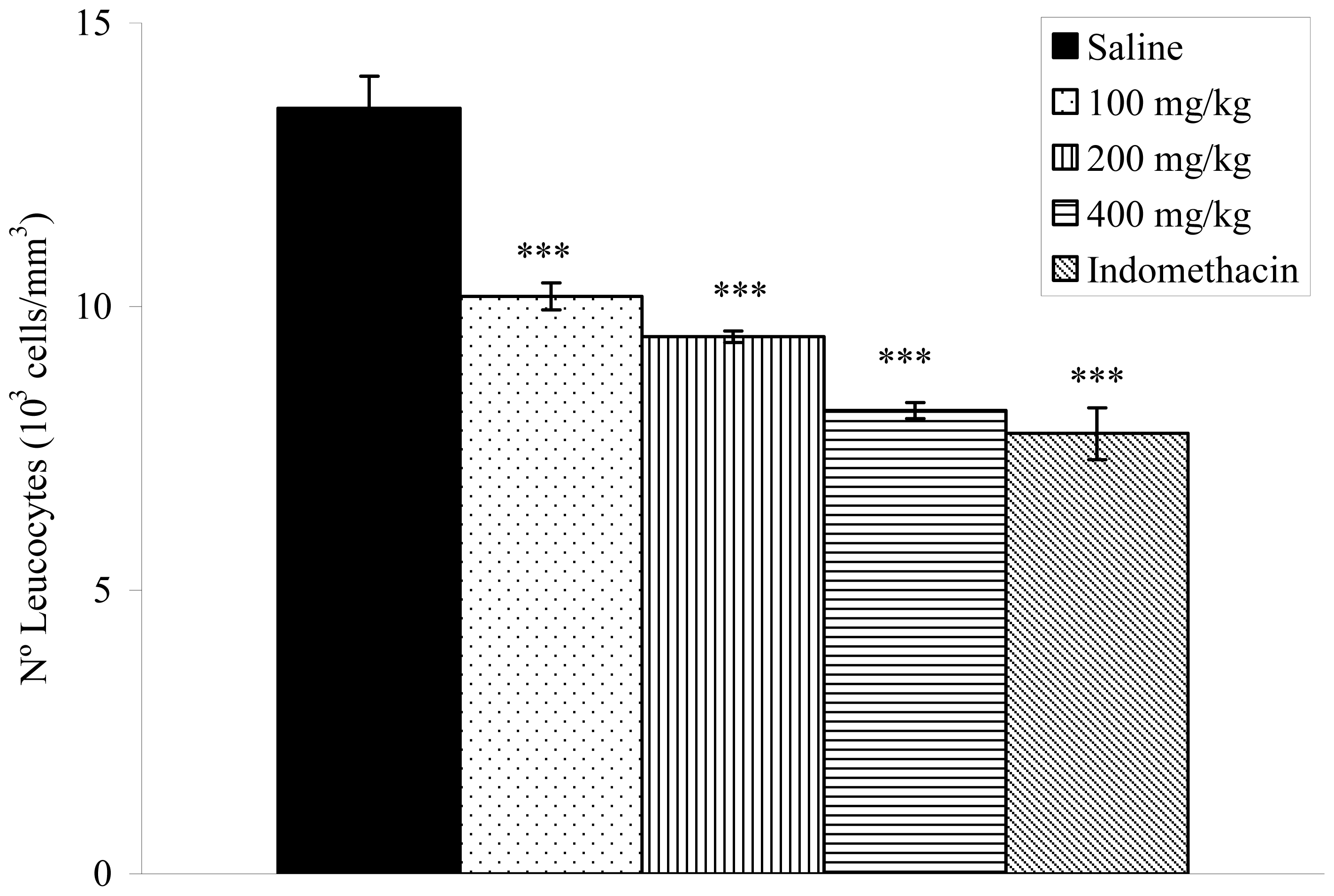
2.8. Effects on Carrageenan-Induced Pleurisy in Rats
3. Experimental Section
3.1. Plant Material and Extraction
3.2. Chemicals
3.3. Phytochemical Screening of the Ethanol Extract
3.4. Animals
3.5. Acute Toxicity
3.6. Acetic Acid-Induced Writhing Test
3.7. Formalin Test
3.8. Hot Plate Test
3.9. Effect on Sleeping Time in Mice
3.10. Carrageenan-Induced Rat Paw Edema
3.11. Carrageenan-Induced Pleurisy in Rats
3.12. Calculus and Statistical Analysis
4. Conclusions
Acknowledgments
References
- White, F.A.; Bhangoo, S.K.; Miller, R.J. Chemokines: Integrators of pain and inflammation. Nat. Rev. Drug Discov 2005, 4, 834–844. [Google Scholar]
- Cunha, T.M.; Verri, W.A., Jr; Schivo, I.R.; Napimoga, M.H.; Parada, C.A.; Poole, S.; Teixeira, M.M.; Ferreira, S.H.; Cunha, F.Q. Crucial role of neutrophils in the development of mechanical inflammatory hypernociception. J. Leukoc. Biol. 2008, 83, 824–832. [Google Scholar]
- Kaye, A.D.; Baluch, A.; Scott, J. Pain management in the elderly population: A review. Ochsner J 2010, 10, 179–187. [Google Scholar]
- Macfarlane, T.V.; Glenny, A.-M.; Worthington, H.V. Systematic review of population-based epidemiological studies of oro-facial pain. J. Dentist 2001, 29, 451–467. [Google Scholar]
- Bovill, J.G. Mechanisms of actions of opioids and non-steroidal anti-inflammatory drugs. Eur. J. Anaesthesiol 1997, 15, 9S–15S. [Google Scholar]
- Sousa, O.V.; Del-Vechio-Vieira, G.; Pinho, J.J.R.G.; Yamamoto, C.H.; Alves, M.S. Antinociceptive and anti-inflammatory activities of the ethanol extract of Annona muricata L. leaves in animal models. Int. J. Mol. Sci 2010, 11, 2067–2078. [Google Scholar]
- Fagundes, L.L.; Del-Vechio-Vieira, G.; Pinho, J.J.R.G.; Yamamoto, C.H.; Alves, M.S.; Stringheta, P.C.; Sousa, O.V. Pharmacological proprieties of the ethanol extract of Muehlenbeckia platyclada (F. Muell.) Meisn. leaves. Int. J. Mol. Sci 2010, 11, 3942–3953. [Google Scholar]
- Vázquez, A.I.F.; Sánchez, C.M.D.; Delgado, N.G.; Alfonso, A.M.S.; Ortega, Y.S.; Sánchez, H.C. Anti-inflammatory and analgesic activities of red seaweed Dichotomaria obtusata. Braz. J. Pharm. Sci 2011, 47, 111–118. [Google Scholar]
- King, B.L.; Jones, S.B. Chemosystematics of Vernonia series flexuosae (Vernonieae: Compositae). Bull. Torrey Bot. Club 1982, 109, 279–286. [Google Scholar]
- Lorenzi, H.; Matos, F.J.A. Plantas Medicinais no Brasil: Nativas e Exóticas, 2nd ed; Instituto Plantarum: Nova Odessa, Brazil, 2008; pp. 165–166. [Google Scholar]
- Alves, V.F.G.; Neves, L.J. Anatomia foliar de Vernonia polyanthes Less (Asteraceae). Rev. Univ. Rural Sér. Ciên. da Vida 2003, 22, 1–8. [Google Scholar]
- Achola, K.J.; Mwangi, J.W.; Munenge, R.W.; Mwaura, A.M. Pharmacological activities of Vernonia glabra. Pharm. Biol 1996, 34, 141–144. [Google Scholar]
- Pérez-Amador, M.C.; Ocotero, V.M.; Benitez, S.P.; Jiménez, F.G. Vernonia patens Kunth, an Asteraceae species with phototoxic and pharmacological activity. Phyton 2008, 77, 275–282. [Google Scholar]
- Nergard, C.S.; Diallo, D.; Michaelsen, T.E.; Malterud, K.E.; Kiyohara, H.; Matsumoto, T.; Yamada, H.; Paulsen, B.S. Isolation, partial characterisation and immunomodulating activities of polysaccharides from Vernonia kotschyana Sch. Bip. ex Walp. J. Ethnopharmacol 2004, 91, 141–152. [Google Scholar]
- Laekeman, G.M.; Mertens, J.; Totté, J.; Bult, H.; Vlietinck, A.J.; Herman, A.G. Isolation and pharmacological characterization of vernolepin. J. Nat. Prod 1983, 46, 161–169. [Google Scholar]
- Ojiako, O.A.; Nwanjo, H.U. Is Vernonia amygdalina hepatotoxic or hepatoprotective? Response from biochemical and toxicity studies in rats. Afr. J. Biotechnol 2006, 5, 1648–1651. [Google Scholar]
- Carvalho, M.G.; Costa, P.M.; Abreu, H.S. Flavanones from Vernonia diffusa. J. Braz. Chem. Soc 1999, 10, 163–166. [Google Scholar]
- Frutuoso, V.S.; Gurjão, M.R.R.; Cordeiro, R.S.B.; Martins, M.A. Analgesic and anti-ulcerogenic effects of a polar extract from leaves of Vernonia condensata. Planta Med 1994, 60, 21–25. [Google Scholar]
- Monteiro, M.H.; Gomes-Carneiro, M.R.; Felzenszwalb, I.; Chahoud, I.; Paumgartten, F.J. Toxicological evaluation of a tea from leaves of Vernonia condensata. J. Ethnopharmacol 2001, 74, 149–157. [Google Scholar]
- Valverde, A.L.; Cardoso, G.L.C.; Pereira, N.A.; Silva, A.J.R.; Kuster, R.M. Analgesic and antiinflammatory activities of vernonioside B2 from Vernonia condensata. Phytother. Res 2001, 15, 263–264. [Google Scholar]
- Risso, W.E.; Scarminio, I.S.; Moreira, E.G. Antinociceptive and acute toxicity evaluation of Vernonia condensata Baker leaves extracted with different solvents and their mixtures. Indian J. Exp. Biol 2010, 48, 811–816. [Google Scholar]
- Matta, C.B.B.; Souza, É.T.; Queiroz, A.C.; Lira, D.P.; Araújo, M.V.; Cavalcante-Silva, L.H.A.; Miranda, G.E.C.; Araújo-Júnior, J.X.; Barbosa-Filho, J.M.; Santos, B.V.O.; et al. Antinociceptive and anti-inflammatory activity from algae of the Genus Caulerpa. Mar. Drugs 2011, 9, 307–318. [Google Scholar]
- Yuan, G.; Wahlgvist, M.L.; He, G.; Yang, M.; Li, D. Natural products and anti-inflammatory activity. Asia Pac. J. Clin. Nutr 2006, 15, 143–152. [Google Scholar]
- Salminen, A.; Lehtonen, M.; Suuronena, T.; Kaarnirantad, K.; Huuskonen, J. Terpenoids: Natural inhibitors of NF-B signaling with anti-inflammatory and anticancer potential. Cell. Mol. Life Sci 2008, 65, 2979–2999. [Google Scholar]
- García-Lafuente, A.; Guillamón, E.; Villares, A.; Rostagno, M.A.; Martínez, J.A. Flavonoids as anti-inflammatory agents: Implications in cancer and cardiovascular disease. Inflamm. Res 2009, 58, 537–552. [Google Scholar]
- Collier, H.D.J.; Dinnin, L.C.; Johnson, C.A.; Schneider, C. The abdominal response and its suppression by analgesic drugs in the mouse. Brit. J. Pharm 1968, 32, 295–310. [Google Scholar]
- Nemirovsky, A.; Chen, L.; Zelma, V.; Jurna, I. The antinociceptive effect of the combination of spinal morphine with systemic morphine or buprenorphine. Anesth. Analg 2001, 93, 197–203. [Google Scholar]
- Dubuisson, D.; Dennis, S.G. The formalin test: A quantitative study of the analgesic affects of morphine, meperidine, and brain stem stimulation in rats and cats. Pain 1977, 21, 161–174. [Google Scholar]
- Hunskaar, S.; Fasmer, O.B.; Hole, K. Formalin test in mice, a useful technique for evaluating mild analgesics. J. Neurosci. Meth 1985, 14, 69–79. [Google Scholar]
- Campo-Soria, C.; Chang, Y.; Weiss, D.S. Mechanism of action of benzodiazepines on GABAA receptors. Br. J. Pharmacol 2006, 148, 984–990. [Google Scholar]
- Ho, I.K.; Harris, R.A. Mechanism of action of barbiturates. Ann. Rev. Pharmacol. Toxicol 1981, 21, 83–111. [Google Scholar]
- Hinton, J.M.; Marley, E. The effects of meprobamate and pentobarbitone sodium on sleep and motility during sleep: A controlled trial with psychiatric patients. J. Neurol. Neurosurg. Psychiatry 1959, 22, 137–140. [Google Scholar]
- Rakotonirina, V.S.; Bum, E.N.; Rakotonirina, A.; Bopelet, M. Sedative properties of the decoction of the rhizome of Cyperus articulatus. Fitoterapia 2001, 72, 22–29. [Google Scholar]
- Rocha, F.F.; Lapa, A.J.; DeLima, T.C. Evaluation of the anxiolytic-like effects of Cecropia glazioui Sneth in mice. Pharmacol. Biochem. Behav 2002, 71, 183–190. [Google Scholar]
- Di Rosa, M.; Giroud, P.P.; Willoughby, D.A. Studies on the mediators of the acute inflammatory response induced in rats in different sites by carrageenan and turpentine. J. Pathol 1971, 104, 15–29. [Google Scholar]
- Seibert, K.; Zhang, Y.; Leahy, K.; Hauser, S.; Masferrer, J.; Perkins, W.; Lee, L.; Isakson, P. Pharmacological and biochemical demonstration of the role of cyclooxygenase 2 in inflammation and pain. Proc. Natl. Acad. Sci. USA 1994, 54, 7–10. [Google Scholar]
- Nantel, F.; Denis, D.; Gordon, R.; Northey, A.; Cirino, M.; Metters, K.M.; Chan, C.C. Distribution and regulation of cyclooxygenase-2 in carrageenan-induced inflammation. Br. J. Pharmacol 1999, 128, 853–859. [Google Scholar]
- Stochla, K.; Maslinski, S. Carrageenan-induced oedema in the rat paw-histamine participation. Agents Actions 1982, 12, 201–202. [Google Scholar]
- Hwang, S.B.; Lam, M.H.; Li, C.L.; Shen, T.Y. Release of platelet activation factor and its involvement in the first phase of carrageenin-induced rat foot edema. Eur. J. Pharmacol 1986, 120, 33–41. [Google Scholar]
- De Campos, R.O.; Alves, R.V.; Kyle, D.J.; Chakravarty, S.; Mavunkel, B.J.; Calixto, J.B. Antioedematogenic and antinociceptive actions of NPC 18521, a novel bradykinin B2 receptor antagonist. Eur. J. Pharmacol 1996, 316, 277–286. [Google Scholar]
- Gilligan, J.P.; Lovato, S.J.; Erion, M.D.; Jeng, A.Y. Modulation of carrageenan-induced hind paw edema by substance P. Inflammation 1994, 18, 285–292. [Google Scholar]
- Vinegar, R.; Truax, J.F.; Selph, J.L. Some quantitative temporal characteristics of carrageenin-induced pleurisy in the rat. Proc. Soc. Exp. Biol. Med 1973, 143, 711–714. [Google Scholar]
- Ammendola, G.; Di Rosa, G.M.; Sorrentino, L. Leucocyte migration and lysosomal enzymes release in rat carrageenin pleurisy. Agents Actions 1975, 5, 250–255. [Google Scholar]
- Compasso, F.; Dunn, C.J.; Yamamoto, S.; Willougbby, D.A.; Giroud, J.P. Further studies on carrageenan-induced pleurisy in rats. J. Pathol 1975, 116, 117–124. [Google Scholar]
- Miyasaka, K.; Mikami, T. Comparison of the anti-inflammatory effects of dexamethasone, indomethacin and BW755C on carrageenin-induced pleurisy in rats. Eur. J. Pharmacol 1982, 77, 229–236. [Google Scholar]
- Matos, F.J.A. Introdução à Fitoquímica Experimental, 2nd ed; Edições UFC: Fortaleza, Brazil, 1997; pp. 41–75. [Google Scholar]
- Dietrich, L. A new approach to practical acute toxicity testing. Arch. Toxicol 1983, 54, 275–287. [Google Scholar]
- Litchfield, J.T.; Wilcoxon, F. A simplified method of evaluating dose-effect experiments. J. Pharmacol. Exp. Therap 1949, 96, 99–113. [Google Scholar]
- Hunskaar, S.; Hole, K. The formalin test in mice: Dissociation between inflammatory and non-inflammatory pain. Pain 1987, 30, 103–114. [Google Scholar]
- Eddy, N.B.; Leimbach, D. Synthetic analgesics. II. Dithienylbutenyl and dithienylbutilamines. J. Pharmacol. Exp. Ther 1953, 107, 385–393. [Google Scholar]
- Fujimori, H. Potentiation of barbital hypnosis as an evaluation method for central nervous system depressants. Psychopharmacologia 1965, 7, 374–378. [Google Scholar]
- Winter, C.A.; Risley, E.A.; Nuss, G.W. Carrageenin-induced edema in hind paw of the rat as an assay for anti-inflammatory drugs. Proc. Soc. Exp. Biol. Med 1962, 111, 544–547. [Google Scholar]


| Group | Dose (mg/kg) | Time after drug administration (seconds)
| |||
|---|---|---|---|---|---|
| 0 min | 30 min | 60 min | 90 min | ||
| Control | Saline | 5.01 ± 0.31 | 5.56 ± 0.83 | 5.55 ± 0.47 | 5.94 ± 0.64 |
| 100 | 5.03 ± 0.46 | 4.88 ± 0.84 | 5.71 ± 0.40 | 5.94 ± 0.87 | |
| Ethanol extract | 200 | 5.46 ± 0.54 | 6.01 ± 0.38 | 6.85 ± 0.45 | 7.50 ± 0.77 |
| 400 | 5.50 ± 0.67 | 7.64 ± 0.99 | 10.13 ± 0.39*** | 11.14 ± 1.33*** | |
| Morphine | 1 | 6.24 ± 0.77 | 9.77 ± 0.44*** | 11.82 ± 0.39*** | 14.51 ± 0.94*** |
| Naloxone + Morphine | 1 + 1 | 5.57 ± 0.68 | 9.99 ± 0.96*** | 8.24 ± 0.56** | 7.01 ± 0.59 |
| Naloxone + Extract | 1 + 400 | 5.54 ± 0.74 | 8.39 ± 0.67* | 12.16 ± 0.74*** | 12.03 ± 1.05*** |
| Group | Dose (mg/kg) | Volume of hind paw (mL)
| |||
|---|---|---|---|---|---|
| 1 h | 2 h | 3 h | 4 h | ||
| Control | Saline | 0.65 ± 0.09 | 0.85 ± 0.09 | 0.93 ± 0.06 | 1.10 ± 0.08 |
| 100 | 0.42 ± 0.02 | 0.72 ± 0.03 | 0.73 ± 0.07 * | 0.85 ± 0.05 * | |
| Ethanol extract | 200 | 0.46 ± 0.04 | 0.51 ± 0.06 * | 0.72 ± 0.03 * | 0.70 ± 0.09 ** |
| 400 | 0.45 ± 0.03 | 0.59 ± 0.07 * | 0.63 ± 0.04 ** | 0.74 ± 0.06 ** | |
| Indomethacin | 10 | 0.41 ± 0.09 | 0.43 ± 0.08 ** | 0.51 ± 0.08 *** | 0.55 ± 0.06 *** |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Da Silva, J.B.; Temponi, V.d.S.; Fernandes, F.V.; Alves, G.d.A.D.; Matos, D.M.d.; Gasparetto, C.M.; Ribeiro, A.; De Pinho, J.d.J.R.G.; Alves, M.S.; De Sousa, O.V. New Approaches to Clarify Antinociceptive and Anti-Inflammatory Effects of the Ethanol Extract from Vernonia condensata Leaves. Int. J. Mol. Sci. 2011, 12, 8993-9008. https://doi.org/10.3390/ijms12128993
Da Silva JB, Temponi VdS, Fernandes FV, Alves GdAD, Matos DMd, Gasparetto CM, Ribeiro A, De Pinho JdJRG, Alves MS, De Sousa OV. New Approaches to Clarify Antinociceptive and Anti-Inflammatory Effects of the Ethanol Extract from Vernonia condensata Leaves. International Journal of Molecular Sciences. 2011; 12(12):8993-9008. https://doi.org/10.3390/ijms12128993
Chicago/Turabian StyleDa Silva, Jucélia Barbosa, Vanessa dos Santos Temponi, Felipe Valente Fernandes, Geórgia de Assis Dias Alves, Dalyara Mendonça de Matos, Carolina Miranda Gasparetto, Antônia Ribeiro, José de Jesus R. G. De Pinho, Maria Silvana Alves, and Orlando Vieira De Sousa. 2011. "New Approaches to Clarify Antinociceptive and Anti-Inflammatory Effects of the Ethanol Extract from Vernonia condensata Leaves" International Journal of Molecular Sciences 12, no. 12: 8993-9008. https://doi.org/10.3390/ijms12128993
APA StyleDa Silva, J. B., Temponi, V. d. S., Fernandes, F. V., Alves, G. d. A. D., Matos, D. M. d., Gasparetto, C. M., Ribeiro, A., De Pinho, J. d. J. R. G., Alves, M. S., & De Sousa, O. V. (2011). New Approaches to Clarify Antinociceptive and Anti-Inflammatory Effects of the Ethanol Extract from Vernonia condensata Leaves. International Journal of Molecular Sciences, 12(12), 8993-9008. https://doi.org/10.3390/ijms12128993




