Effects of Newcastle Disease Virus Strains AF2240 and V4-UPM on Cytolysis and Apoptosis of Leukemia Cell Lines
Abstract
:1. Introduction
2. Results
2.1. Cytotolytic Effects of NDV on Normal Cells and Myelomonocytic Leukemia Cells
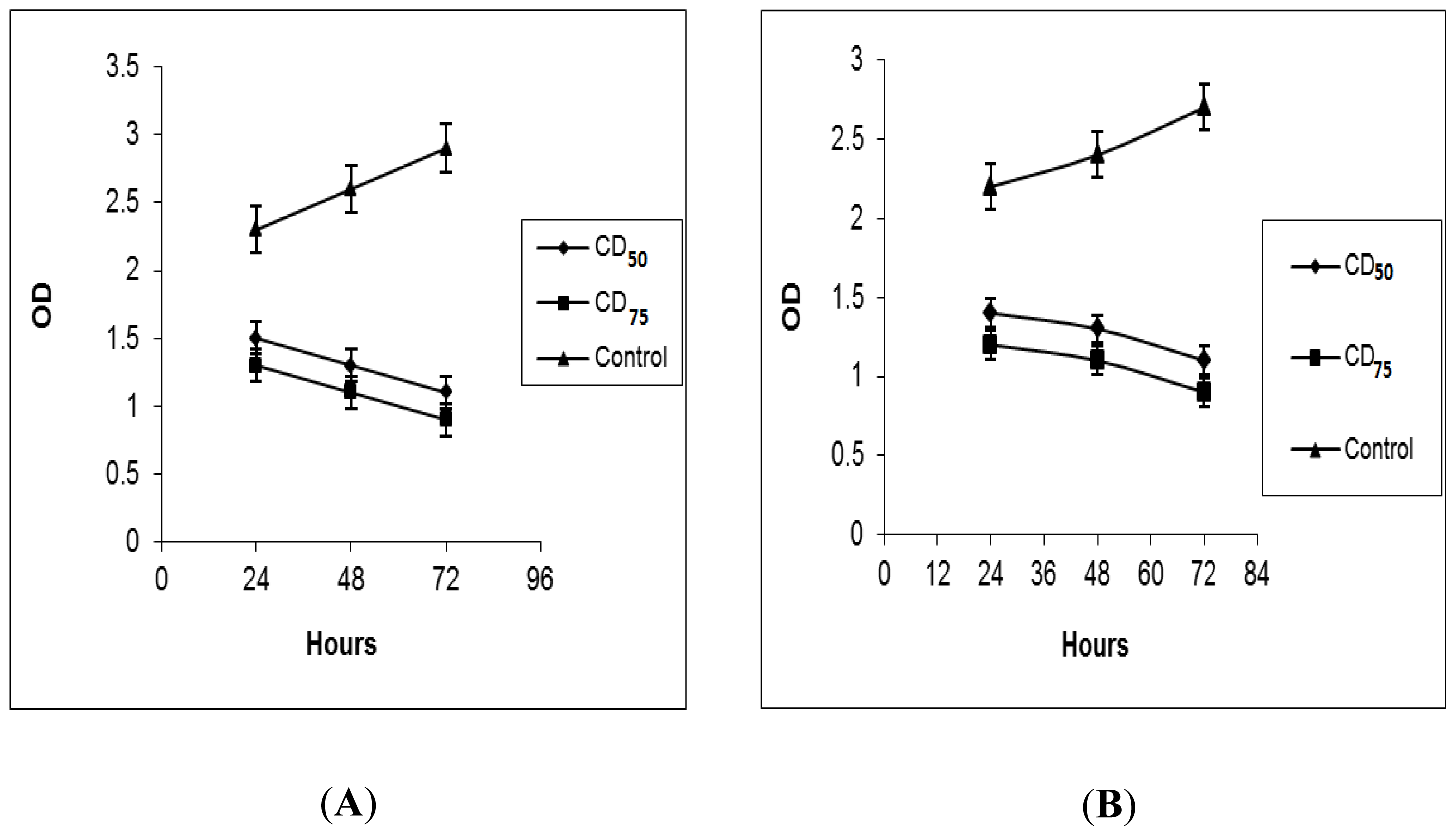
2.2. BrdU Cell Proliferation Assay
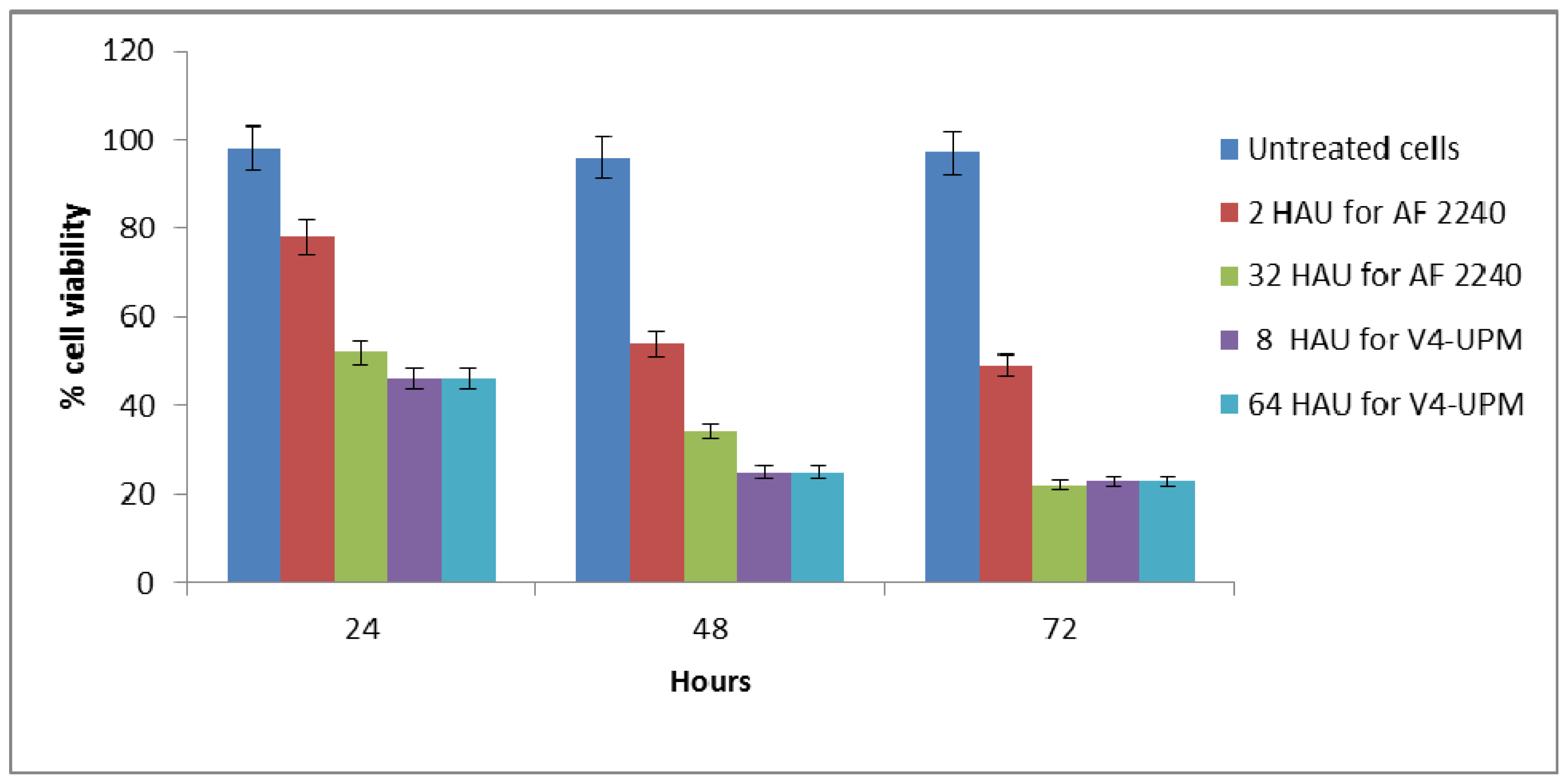
2.3. Trypan Blue Exclusion Assay
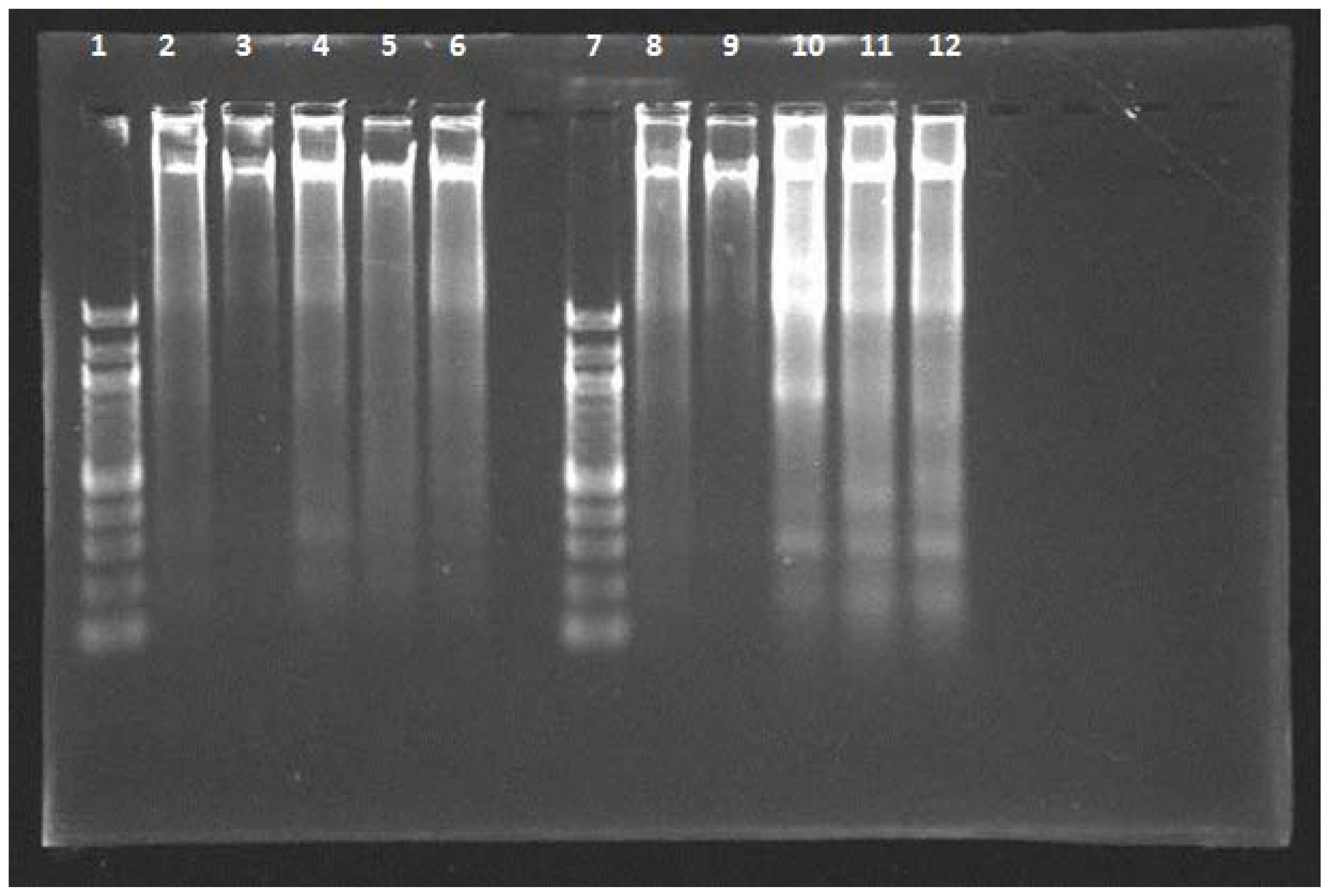
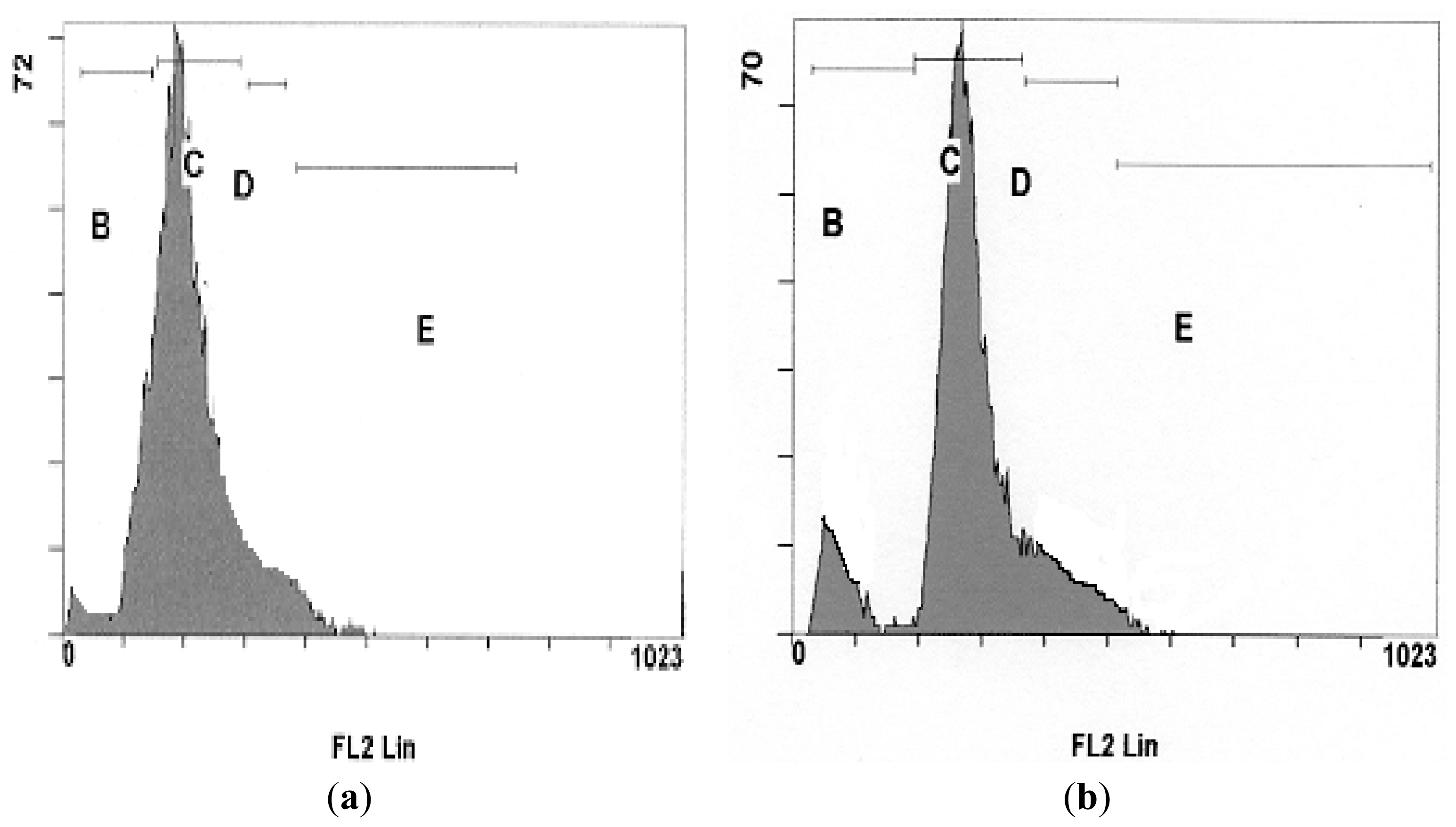
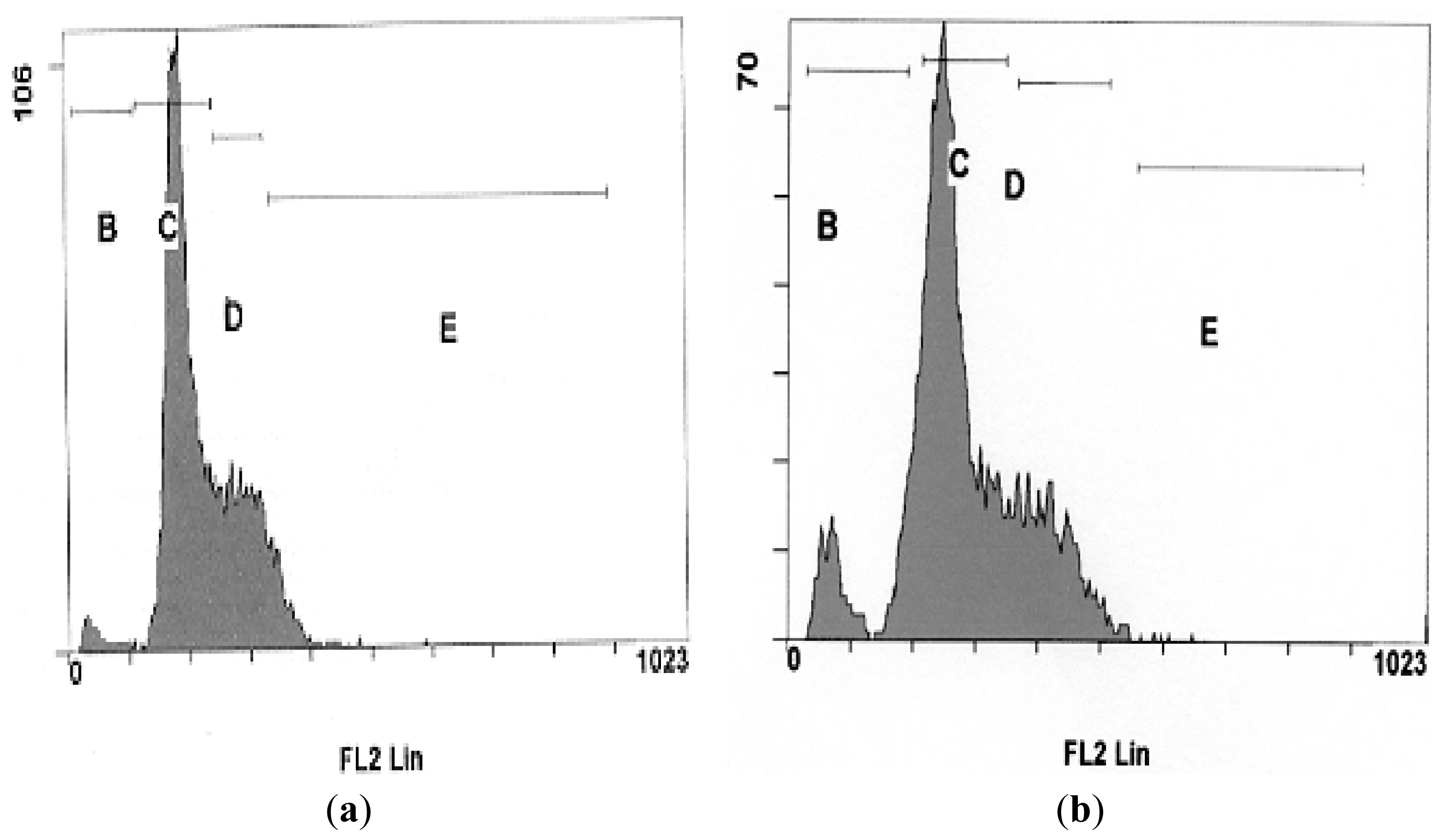
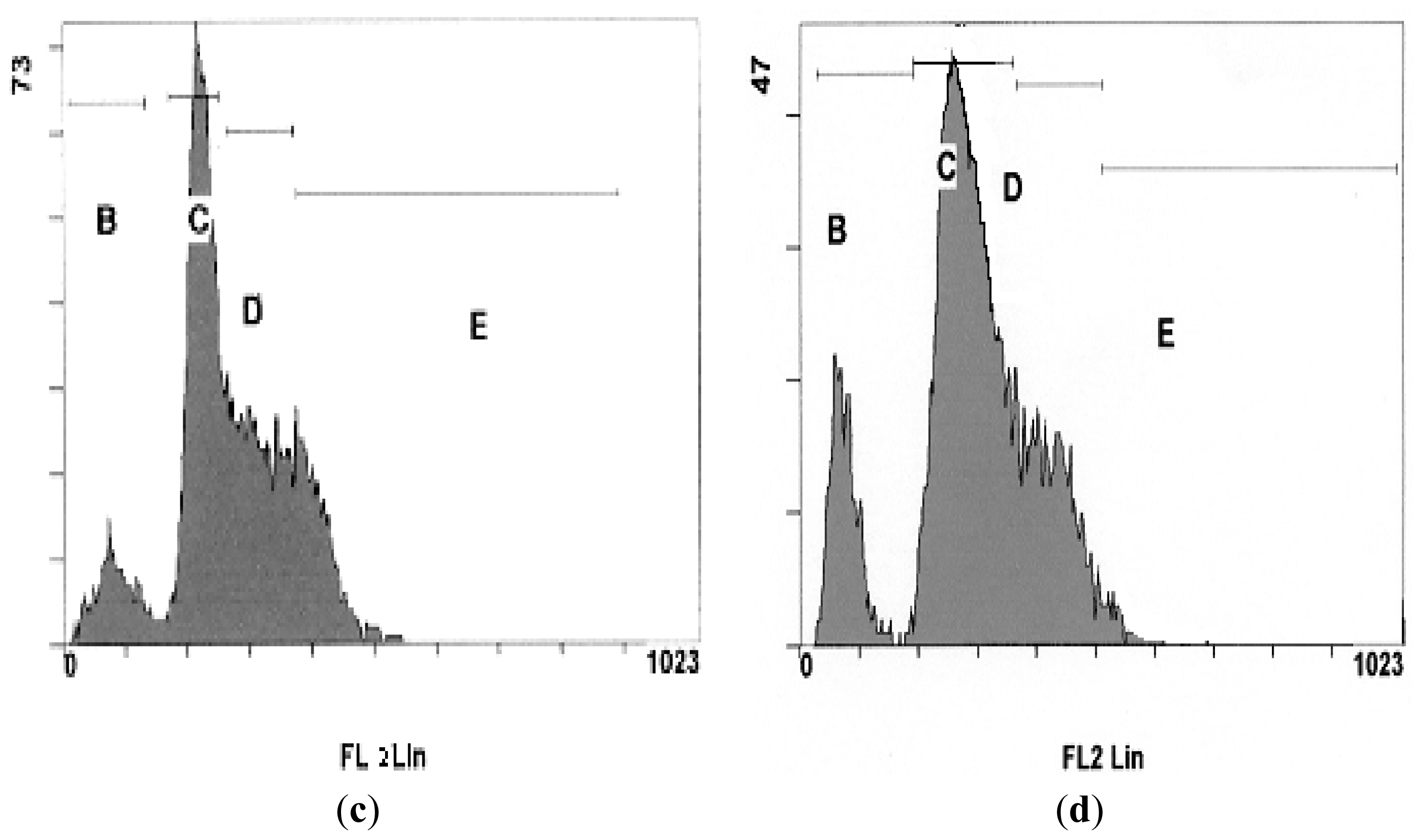
2.4. DNA Fragmentation Assay
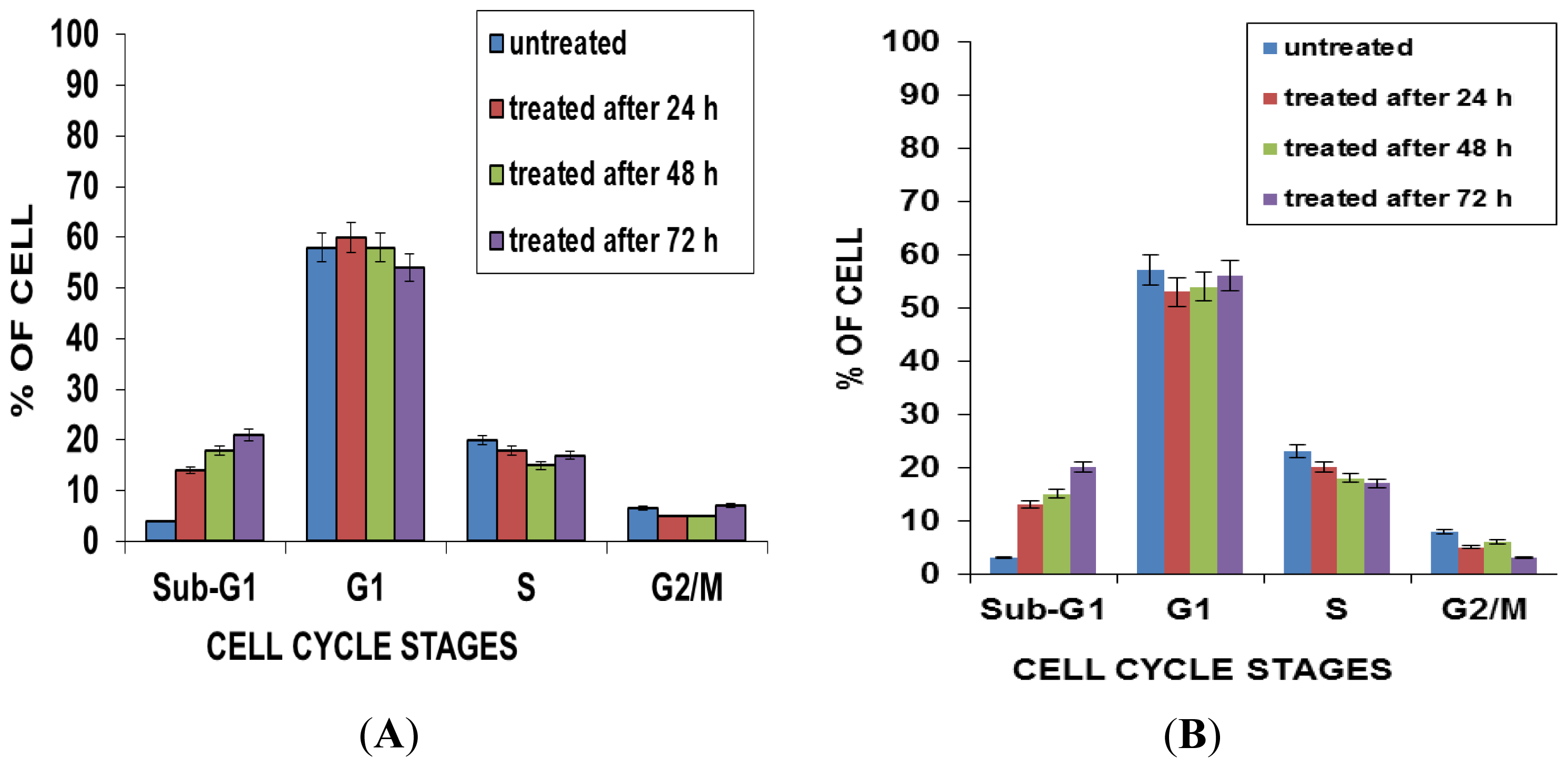
2.5. Effects of NDV on Cell Cycle Distribution
2.6. Activation of Caspase-8, an Initiator Caspase for the Extrinsic Pathway
2.7. Activation of Caspase-9, an Initiator Caspase for the Intrinsic Pathway
2.8. Activation of Effector/Executioner Caspases (Caspase-3/7)
3. Materials and Methods
3.1. Propagation and Purification of NDV Strains AF2240 and V4-UPM
3.2. Cells and Cell Culture
3.3. Cell Viability and Proliferation Assessment
3.3.1. MTT Cytotoxicity Assay
3.3.2. BrdU Proliferation Assay
3.3.3. Trypan Blue Exclusion Assay
3.4. DNA Fragmentation Assay
3.5. Analysis of Cellular DNA Content Using Propidium Iodide
3.6. Analysis of Caspase-3/7, 8 and 9 Activities
3.7. Statistical Analysis
4. Discussions
Acknowledgments
References
- Cotran, R.S.; Kumar, V.; Robbin, S.L. Neoplasia. In Robbin’s Pathologyical Basis of Diseases, 5th ed.; Saunders: Philiadelphia, PA, USA, 1994; pp. 241–303. [Google Scholar]
- Hughes-Jones, N.C.; Wickramasinghe, S.N. Acute and Chronic Leukemia. In Lecture Notes on Haematology, 6th ed.; Blackwell Science Ltd: Paris, France, 1996; pp. 134–160. [Google Scholar]
- Zuco, V.; Rhigetti, S.C.; Cleris, E.; Marchesi, L.; Passerini, C.G.; Formelli, F. Selective cytotoxicity of betulinic acid on tumor cell lines but not normal cells. Cancer Lett 2002, 175, 17–25. [Google Scholar]
- Parato, K.A.; Senger, D.; Forsyth, P.A.J.; Bell, J.C. Recent progress in the battle between oncolytic viruses and tumours. Nature 2005, 5, 965–976. [Google Scholar]
- Freeman, A.I.; Zakay-Rones, Z.; Gomori, J.M.; Linetsky, E.; Rasooly, L.; Greenbaum, E.; Rozenman-Yair, S.; Panet, A.; Libson, E.; Irving, C.S.; Galun, E.; Siegal, T. Phase I/II trial of intravenous NDV-HUJ oncolytic virus in recurrent glioblastoma multiforme. Mol. Ther 2006, 13, 221–228. [Google Scholar]
- Schirrmacher, V.; Ahlert, T.; Pröbstle, T.; Steiner, H.H.; Herold-Mende, C.; Gerhards, R.; Hagmüller, E.; Steiner, H.H. Immunization with virus-modified tumor cells. Semin. Oncol 1998, 25, 677–696. [Google Scholar]
- Omar, A.; Aini, I.; Ali, A.; Othman, F.; Yusoff, K.; Abdullah, J.M.; Wali, H.; Zawawi, M.; Meyyappan, N. An overview on the development of Newcastle disease virus as an anti-cancer therapy. Malays. J. Med. Sci 2003, 10, 4–12. [Google Scholar]
- Alexander, D.J. Newcastle Disease. In Newcastle Disease Virus an Avian Paramyxovirus; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1988; pp. 1–22. [Google Scholar]
- Yusoff, K.; Tan, W.S.; Lau, C.H.; Ng, B.K.; Ibrahim, A.L. Sequence of the haemagglutinin-neuraminadase gene of the Newcastle disease virus oral vaccine strain V4 (UPM). Avian Pathol 1996, 25, 837–844. [Google Scholar]
- Mosmann, T. Rapid Colorimetric Assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar]
- Pujalté, I.; Passagne, I.; Brouillaud, B.; Tréguer, M.; Durand, E.; Ohayon-Courtès, C.; L’Azou1, B. Cytotoxicity and oxidative stress induced by different metallic nanoparticles on human kidney cells. Part. Fibre Toxicol 2011, 8, 10:1–10:16. [Google Scholar]
- Szeberenyi, J.; Fabian, Z.; Torocsik, B.; Kiss, K.; Csatary, L.K. Newcastle disease virus-induced apoptosis in PC12 pheochromocytoma cells. Am. J. Ther 2003, 10, 282–288. [Google Scholar]
- Reichard, K.W.; Lorence, R.M.; Cascino, C.J.; Peeples, M.E.; Walter, R.J.; Fernando, M.B.; Reyes, H.M.; Greager, J.A. Newcastle disease virus selectively kills human tumor cells. J. Surg. Res 1992, 52, 448–453. [Google Scholar]
- Adams, J. Proteasome inhibition in cancer: Development of PS-341. Semin. Oncol 2001, 28, 613–619. [Google Scholar]
- Zulkifli, M.M.; Ibrahim, R.; Ali, A.M.; Aini, I.; Jaafar, H.; Hilda, S.S.; Alitheen, N.B.; Abdullah, J.M. Newcastle diseases virus strain V4UPM displayed oncolytic ability against experimental human malignant glioma. Neurol. Res 2009, 13, 3–10. [Google Scholar]
- Othman, F.; Ideris, A.; Motalleb, G.; Eshak, Z.B.; Rahmat, A. Oncolytic effect of newcastle disease virus AF2240 strain on the MCF-7 breast cancer cell line. Yakhteh Med. J 2010, 12, 17–24. [Google Scholar]
- Ali, R.; Alabsi, A.M.; Ali, A.M.; Ainideris Omar, A.R.; Yousoff, K. Apoptosis induction and cytolytic effects of newcastle disease virus strain AF2240 on DBTRG.05 mg brain tumor cell line. Int. J. Cancer Res. 2011, 7, 25–35. [Google Scholar]
- Ali, R.; Alabsi, A.M.; Ali, A.M.; Ideris, A.; Omar, A.R.; Yusoff, K.; Saif-Ali, R. Cytolytic effects and apoptosis induction of newcastle disease virus strain AF2240 on anaplastic astrocytoma brain tumor cell line. Neurochem. Res 2011, 36, 2051–2062. [Google Scholar]
- Ruben, R.L.; Neubuer, R.H. Semiautomated colorimeteric assay for in vitro screening of anticancer compounds. Cancer Treat. Rep 1987, 71, 1141–1149. [Google Scholar]
- Strober, W. Trypan Blue Exclusion Test of Cell Viability. In Current Protocols in Immunology; Coligan, J.E., Kruisbeek, A.M., Margulies, D.H., Shevach, W.M., Strober, W., Eds.; John Wiley and Sons, Inc: Hoboken, NJ, USA, 2003. [Google Scholar]
- Muneesh, T.; Vishva, M.D. Fas- and tumor necrosis factor-induced apoptosis is inhibited by the poxvirus crmA gene product. Am. Soc. Biochem. Mol. Biol 1995, 270, 3255–3260. [Google Scholar]
- Cohen, G.M.; Sun, X.M.; Snowden, R.T.; Dinsdale, D.; Skilleter, D.N. Key morphological features of apoptosis may occur in the absence of internucleosomal DNA fragmentation. Biochem. J 1992, 286, 331–334. [Google Scholar]
- Oberhammer, F.; Wilson, J.W.; Dive, C.; Morris, I.D.; Hickman, J.A.; Wakeling, A.E.; Walker, P.R.; Sikorska, M. Apoptotic death in epithelial cells: Cleavage of DNA to 300 and/or 50 kb fragments prior to or in the absence of internucleosomal fragmentation. EMBO J 1993, 12, 3679–3684. [Google Scholar]
- Gooch, J.L.; Yee, D. Strain-specific differences in formation of apoptotic DNA ladders in MCF-7 breast cancer cells. Cancer Lett 1999, 144, 31–37. [Google Scholar]
- Telford, W.G.; King, L.E.; Fraker, P.J. Rapid quantitation of apoptosis in pure and heterogeneous cell populations using flow cytometry. J. Immunol. Methods 1994, 172, 1–16. [Google Scholar]
- Fried, J.; Perez, A.G.; Clarkson, B.D. Flow cytofluorometric analysis of cell cycle distributions using propidium iodide, Properties of the method and mathematical analysis of the data. J. Cell Biol 1976, 71, 172–181. [Google Scholar]
- Nagata, S. Apoptosis by death factor. Cell 1997, 88, 355–365. [Google Scholar]
- Ashkenazi, A.; Dixit, V.M. Death receptors: Signaling and modulation. Science 1998, 281, 1305–1308. [Google Scholar]
- Locksley, R.M.; Killeen, N.; Lenardo, M.J. The TNF and TNF receptor superfamilies: Integrating mammalian biology. Cell 2001, 104, 487–501. [Google Scholar]
- Slee, E.A.; Adrain, C.; Martin, S.J. Serial killers: Ordering caspase activation events in apoptosis. Cell Death Differ 1999, 6, 1067–1074. [Google Scholar]
- Srinivasula, S.M.; Ahmad, M.; Fernandes-Alnemri, T.; Alnemri, E.S. Autoactivation of procaspase-9 by Apaf-1-mediated oligomerization. Mol. Cell 1998, 1, 949–957. [Google Scholar]
- Slee, E.A.; Adrian, C.; Martin, S.J. Executioner caspase-3, -6, and -7 perform distinct, non-redundant roles during the demolition phase of apoptosis. J. Biol. Chem 2001, 280, 4663–4673. [Google Scholar]
- Kuida, K.; Zheng, T.S.; Na, S.Q.; Kuan, C.Y.; Yang, D.; Karasuyama, H.; Rakic, P.; Flavell, R.A. Decreased apoptosis in the brain and premature lethality in CPP32-deficient mice. Nature 1996, 384, 368–372. [Google Scholar]
- Woo, M.; Hakem, R.; Soengas, M.S.; Duncan, G.S.; Shahinian, A.; Kägi, D.; Hakem, A.; McCurrach, M.; Khoo, W.; Kaufman, S.A.; et al. Essential contribution of caspase 3/CPP32 to apoptosis and its associated nuclear changes. Genes Dev 1998, 12, 806–819. [Google Scholar]



| Cell Lines | AF 2240 | V4-UPM | Doxorubicin | Arabinocytocine (Ara-C) |
|---|---|---|---|---|
| WEHI-3B | 2 ± 0.2 | 8 ± 0.2 | 0.8 ± 0.6 | 1.0 ± 0.13 |
| HL-60 | 25 ± 0.3 | 110 ± 0.8 | 1.8 ± 0.1 | 0.3 ± 0.05 |
| CEM-SS | 16 ± 0.6 | 64 ± 0.5 | 3.5 ± 0.1 | 3.8 ± 0.30 |
| 3T3 | NO CD50 | NO CD50 | 7.0 ± 0.4 | 2.8 ± 0.20 |
| PBMC | NO CD50 | NO CD50 | 11.0 ± 0.8 | 7.0 ± 0.80 |
| Mouse spleen lymphocyte | NO CD50 | NO CD50 | 6.5 ± 0.2 | 4.7 ± 0.6 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Alabsi, A.M.; Bakar, S.A.A.; Ali, R.; Omar, A.R.; Bejo, M.H.; Ideris, A.; Ali, A.M. Effects of Newcastle Disease Virus Strains AF2240 and V4-UPM on Cytolysis and Apoptosis of Leukemia Cell Lines. Int. J. Mol. Sci. 2011, 12, 8645-8660. https://doi.org/10.3390/ijms12128645
Alabsi AM, Bakar SAA, Ali R, Omar AR, Bejo MH, Ideris A, Ali AM. Effects of Newcastle Disease Virus Strains AF2240 and V4-UPM on Cytolysis and Apoptosis of Leukemia Cell Lines. International Journal of Molecular Sciences. 2011; 12(12):8645-8660. https://doi.org/10.3390/ijms12128645
Chicago/Turabian StyleAlabsi, Aied M., Siti Aishah Abu Bakar, Rola Ali, Abdul Rahman Omar, Mohd Hair Bejo, Aini Ideris, and Abdul Manaf Ali. 2011. "Effects of Newcastle Disease Virus Strains AF2240 and V4-UPM on Cytolysis and Apoptosis of Leukemia Cell Lines" International Journal of Molecular Sciences 12, no. 12: 8645-8660. https://doi.org/10.3390/ijms12128645




