Enhanced Antiproliferative and Apoptotic Response of HT-29 Adenocarcinoma Cells to Combination of Photoactivated Hypericin and Farnesyltransferase Inhibitor Manumycin A
Abstract
:1. Introduction
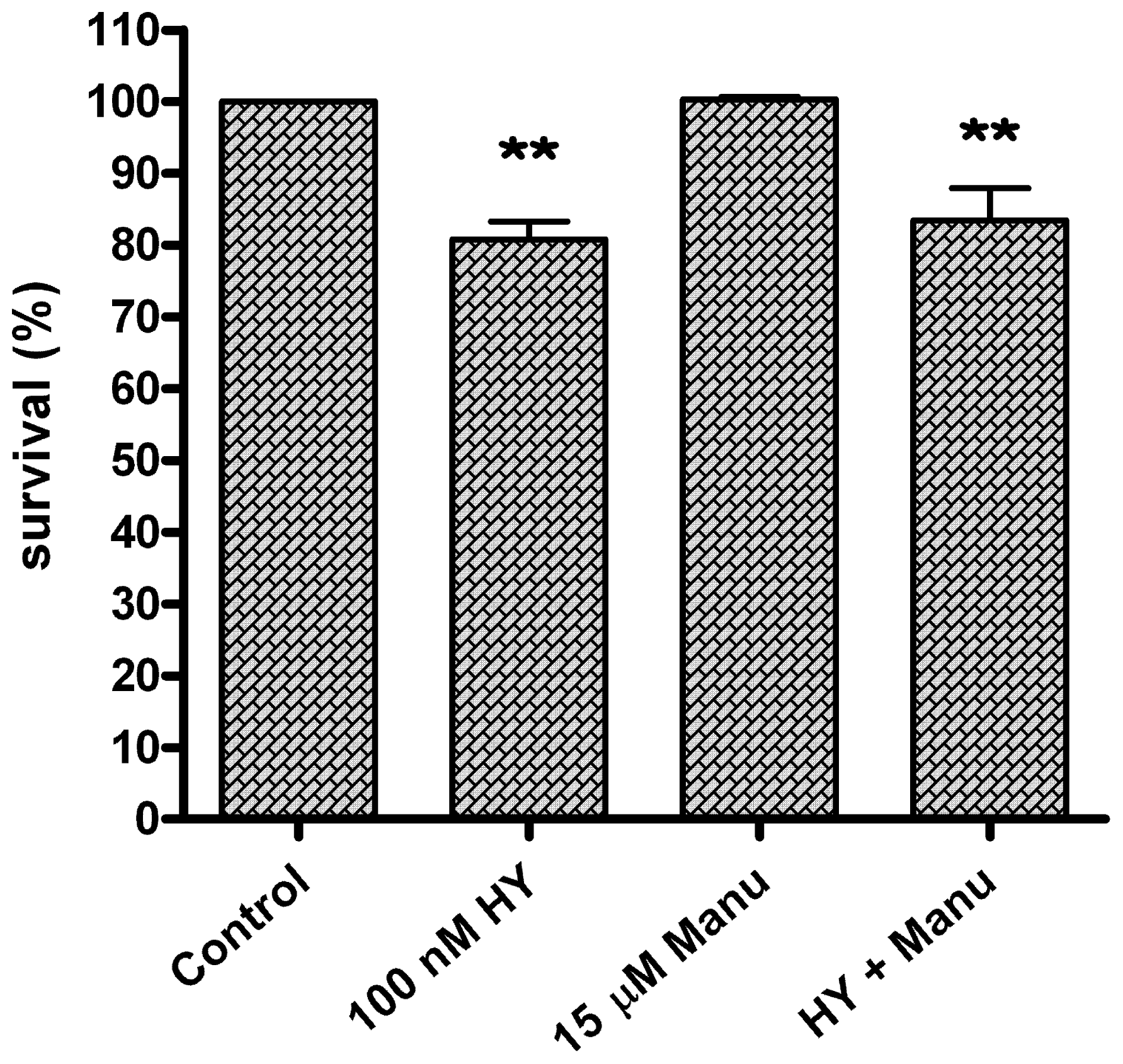
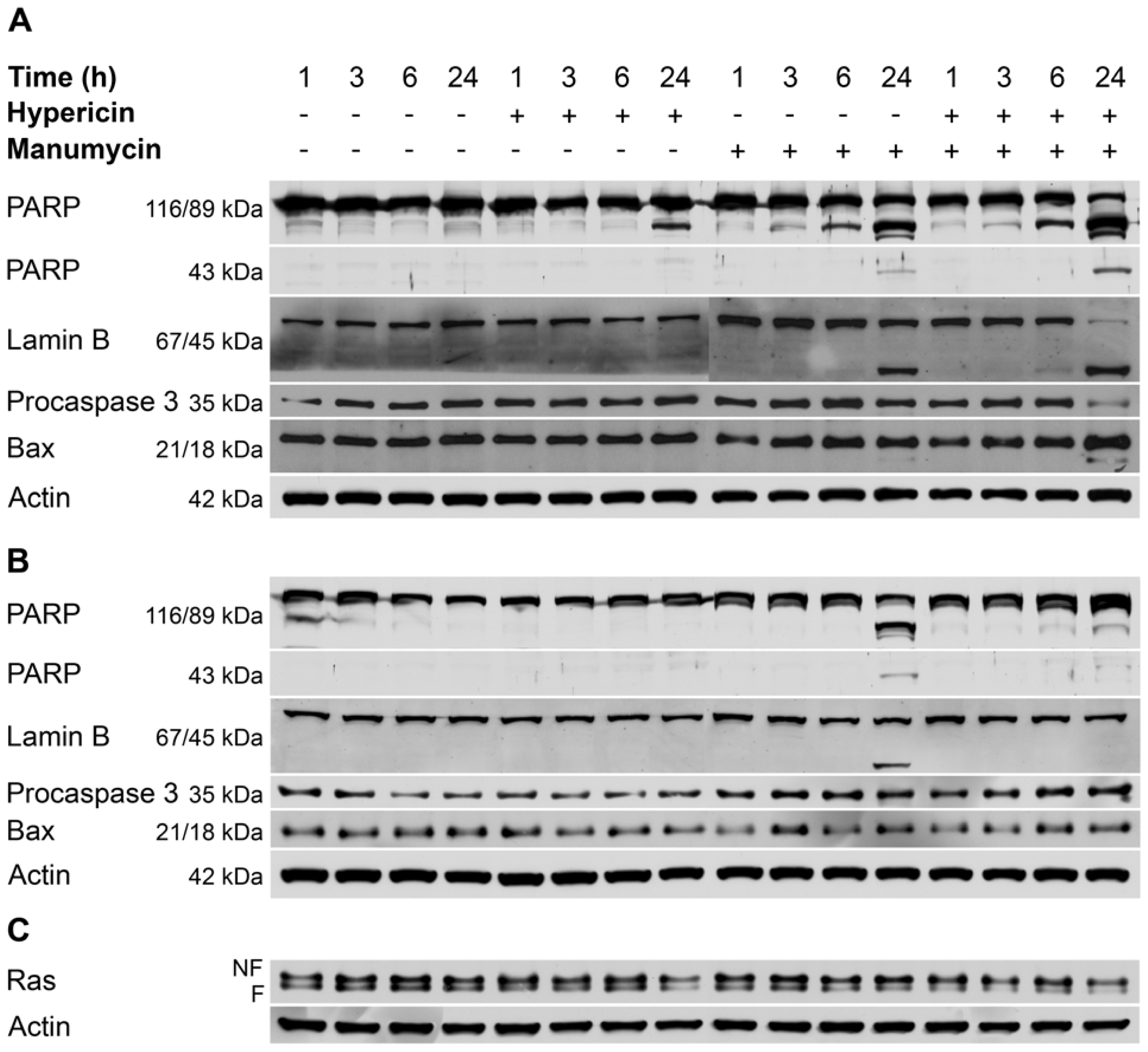
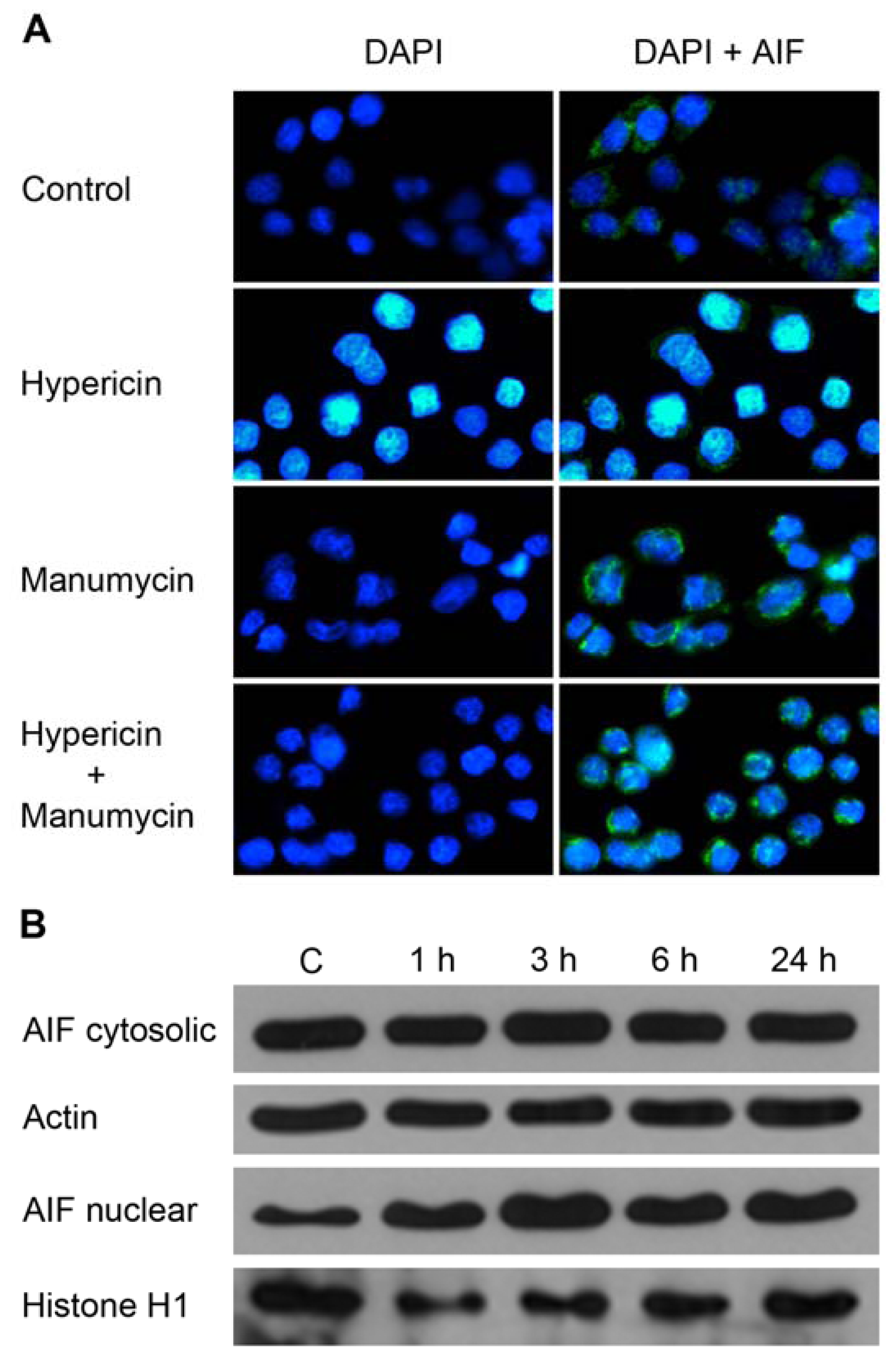
2. Results and Discussion
3. Experimental Section
3.1. Cell Culture
3.2. Reagents
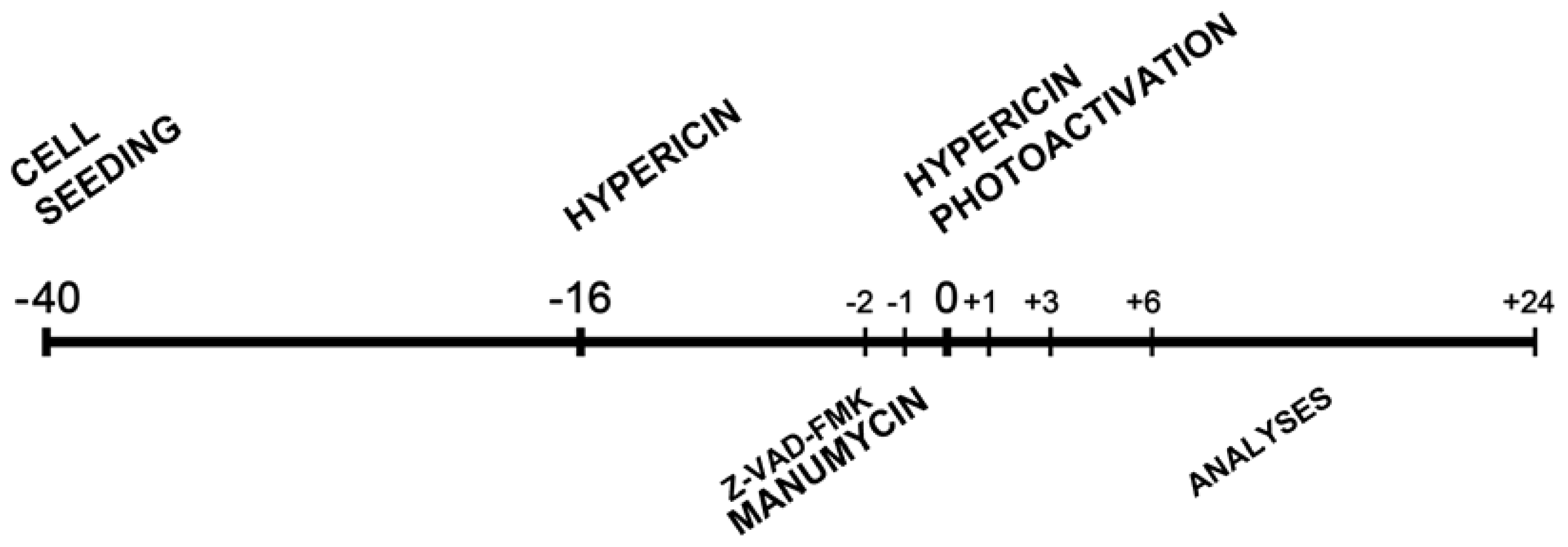
3.3. Experimental Design
3.4. Hypericin Treatment
3.5. Manumycin Treatment
3.6. Pan-Caspase Inhibitor Treatment
3.7. Hypericin Photoactivation
3.8. MTT Assay
3.9. Colony-Forming Assay
3.10. Cell Cycle Distribution Analysis
3.11. Caspase-3/7 Activity Assay
3.12. Western Blot Analysis
3.13. Cytosolic and Nuclear Fractionation
3.14. Antibodies
3.15. Immunofluorescent Staining of AIF
3.16. Statistical Analysis
4. Conclusions
Acknowledgments
- Conflict of interestThe authors declare no conflict of interest.
References
- Brockmann, H.; Haschad, M.N.; Maier, K.; Pohl, F. Hypericin, the photodynamically active pigment from Hypericum perforatum. Naturwissenschaften 1939, 32, 550–555. [Google Scholar]
- Thomas, C.; MacGill, R.S.; Miller, G.C.; Pardini, R.S. Photoactivation of hypericin generates singlet oxygen in mitochondria and inhibits succinoxidase. Photochem. Photobiol 1992, 55, 47–53. [Google Scholar]
- Diwu, Z.; Lown, J.W. Photosensitization with anticancer agents. 17. EPR studies of photodynamic action of hypericin: Formation of semiquinone radical and activated oxygen species on illumination. Free Radic. Biol. Med 1993, 14, 209–215. [Google Scholar]
- Kleban, J.; Szilardiova, B.; Mikes, J.; Horvath, V.; Sackova, V.; Brezani, P.; Hofmanova, J.; Kozubik, A.; Fedorocko, P. Pre-treatment of HT-29 cells with 5-LOX inhibitor (MK-886) induces changes in cell cycle and increases apoptosis after photodynamic therapy with hypericin. J. Photochem. Photobiol. B 2006, 84, 79–88. [Google Scholar]
- Jendzelovsky, R.; Mikes, J.; Koval, J.; Soucek, K.; Prochazkova, J.; Kello, M.; Sackova, V.; Hofmanova, J.; Kozubik, A.; Fedorocko, P. Drug efflux transporters, MRP1 and BCRP, affect the outcome of hypericin-mediated photodynamic therapy in HT-29 adenocarcinoma cells. Photochem. Photobiol. Sci 2009, 8, 1716–1723. [Google Scholar]
- Ferenc, P.; Solar, P.; Kleban, J.; Mikes, J.; Fedorocko, P. Down-regulation of Bcl-2 and Akt induced by combination of photoactivated hypericin and genistein in human breast cancer cells. J. Photochem. Photobiol. B 2010, 98, 25–34. [Google Scholar]
- Kello, M.; Mikes, J.; Jendzelovsky, R.; Koval, J.; Fedorocko, P. PUFAs enhance oxidative stress and apoptosis in tumour cells exposed to hypericin-mediated PDT. Photochem. Photobiol. Sci 2010, 9, 1244–1251. [Google Scholar]
- Hendrickx, N.; Volanti, C.; Moens, U.; Seternes, O.M.; de Witte, P.; Vandenheede, J.R.; Piette, J.; Agostinis, P. Up-regulation of cyclooxygenase-2 and apoptosis resistance by p38 MAPK in hypericin-mediated photodynamic therapy of human cancer cells. J. Biol. Chem 2003, 278, 52231–52239. [Google Scholar]
- Sarissky, M.; Lavicka, J.; Kocanova, S.; Sulla, I.; Mirossay, A.; Miskovsky, P.; Gajdos, M.; Mojzis, J.; Mirossay, L. Diazepam enhances hypericin-induced photocytotoxicity and apoptosis in human glioblastoma cells. Neoplasma 2005, 52, 352–359. [Google Scholar]
- Zhang, W.; Anker, L.; Law, R.E.; Hinton, D.R.; Gopalakrishna, R.; Pu, Q.; Gundimeda, U.; Weiss, M.H.; Couldwell, W.T. Enhancement of radiosensitivity in human malignant glioma cells by hypericin in vitro. Clin. Cancer Res 1996, 2, 843–846. [Google Scholar]
- Wessels, J.T.; Busse, A.C.; Rave-Frank, M.; Zanker, S.; Hermann, R.; Grabbe, E.; Muller, G.A. Photosensitizing and radiosensitizing effects of hypericin on human renal carcinoma cells in vitro. Photochem. Photobiol 2008, 84, 228–235. [Google Scholar]
- Gupta, V.; Su, Y.S.; Wang, W.; Kardosh, A.; Liebes, L.F.; Hofman, F.M.; Schonthal, A.H.; Chen, T.C. Enhancement of glioblastoma cell killing by combination treatment with temozolomide and tamoxifen or hypericin. Neurosurg. Focus 2006, 20. [Google Scholar] [CrossRef]
- Chen, B.; Roskams, T.; de Witte, P.A. Enhancing the antitumoral effect of hypericin-mediated photodynamic therapy by hyperthermia. Lasers Surg. Med 2002, 31, 158–163. [Google Scholar]
- Bhuvaneswari, R.; Thong, P.S.; Gan, Y.Y.; Soo, K.C.; Olivo, M. Evaluation of hypericin-mediated photodynamic therapy in combination with angiogenesis inhibitor bevacizumab using in vivo fluorescence confocal endomicroscopy. J. Biomed. Opt 2010, 15. [Google Scholar] [CrossRef]
- Hara, M.; Akasaka, K.; Akinaga, S.; Okabe, M.; Nakano, H.; Gomez, R.; Wood, D.; Uh, M.; Tamanoi, F. Identification of Ras farnesyltransferase inhibitors by microbial screening. Proc. Natl. Acad. Sci. USA 1993, 90, 2281–2285. [Google Scholar]
- Cox, A.D.; Der, C.J. Farnesyltransferase inhibitors and cancer treatment: Targeting simply Ras? Biochim. Biophys. Acta 1997, 1333, F51–F71. [Google Scholar]
- Kainuma, O.; Asano, T.; Hasegawa, M.; Kenmochi, T.; Nakagohri, T.; Tokoro, Y.; Isono, K. Inhibition of growth and invasive activity of human pancreatic cancer cells by a farnesyltransferase inhibitor, manumycin. Pancreas 1997, 15, 379–383. [Google Scholar]
- Yeung, S.C.; Xu, G.; Pan, J.; Christgen, M.; Bamiagis, A. Manumycin enhances the cytotoxic effect of paclitaxel on anaplastic thyroid carcinoma cells. Cancer Res 2000, 60, 650–656. [Google Scholar]
- Pan, J.; Xu, G.; Yeung, S.C. Cytochrome c release is upstream to activation of caspase-9, caspase-8, and caspase-3 in the enhanced apoptosis of anaplastic thyroid cancer cells induced by manumycin and paclitaxel. J. Clin. Endocrinol. Metab 2001, 86, 4731–4740. [Google Scholar]
- Di Paolo, A.; Danesi, R.; Nardini, D.; Bocci, G.; Innocenti, F.; Fogli, S.; Barachini, S.; Marchetti, A.; Bevilacqua, G.; Del Tacca, M. Manumycin inhibits ras signal transduction pathway and induces apoptosis in COLO320-DM human colon tumour cells. Br. J. Cancer 2000, 82, 905–912. [Google Scholar]
- Zhou, J.M.; Zhu, X.F.; Pan, Q.C.; Liao, D.F.; Li, Z.M.; Liu, Z.C. Manumycin induces apoptosis in human hepatocellular carcinoma HepG2 cells. Int. J. Mol. Med 2003, 12, 955–959. [Google Scholar]
- Wang, W.; Macaulay, R.J. Apoptosis of medulloblastoma cells in vitro follows inhibition of farnesylation using manumycin A. Int. J. Cancer 1999, 82, 430–434. [Google Scholar]
- Selleri, C.; Maciejewski, J.P.; Montuori, N.; Ricci, P.; Visconte, V.; Serio, B.; Luciano, L.; Rotoli, B. Involvement of nitric oxide in farnesyltransferase inhibitor-mediated apoptosis in chronic myeloid leukemia cells. Blood 2003, 102, 1490–1498. [Google Scholar]
- She, M.R.; Li, J.G.; Du, X.; Lin, W.; Niu, X.Q.; Guo, K.Y. Involvement of mitochondria apoptotic pathway in the manumycin inducing apoptosis of U937 and HL-60. Zhonghua Xue Ye Xue Za Zhi 2007, 28, 404–406. [Google Scholar]
- Frassanito, M.A.; Cusmai, A.; Piccoli, C.; Dammacco, F. Manumycin inhibits farnesyltransferase and induces apoptosis of drug-resistant interleukin 6-producing myeloma cells. Br. J. Haematol 2002, 118, 157–165. [Google Scholar]
- Sears, K.T.; Daino, H.; Carey, G.B. Reactive oxygen species-dependent destruction of MEK and Akt in Manumycin stimulated death of lymphoid tumor and myeloma cell lines. Int. J. Cancer 2008, 122, 1496–1505. [Google Scholar]
- She, M.; Pan, I.; Sun, L.; Yeung, S.C. Enhancement of manumycin A-induced apoptosis by methoxyamine in myeloid leukemia cells. Leukemia 2005, 19, 595–602. [Google Scholar]
- Hu, W.; Wu, W.; Verschraegen, C.F.; Chen, L.; Mao, L.; Yeung, S.C.; Kudelka, A.P.; Freedman, R.S.; Kavanagh, J.J. Proteomic identification of heat shock protein 70 as a candidate target for enhancing apoptosis induced by farnesyl transferase inhibitor. Proteomics 2003, 3, 1904–1911. [Google Scholar]
- Xu, G.; Pan, J.; Martin, C.; Yeung, S.C. Angiogenesis inhibition in the in vivo antineoplastic effect of manumycin and paclitaxel against anaplastic thyroid carcinoma. J. Clin. Endocrinol. Metab 2001, 86, 1769–1777. [Google Scholar]
- She, M.; Jim Yeung, S.C. Combining a matrix metalloproteinase inhibitor, a farnesyltransferase inhibitor, and a taxane improves survival in an anaplastic thyroid cancer model. Cancer Lett 2006, 238, 197–201. [Google Scholar]
- Sackova, V.; Fedorocko, P.; Szilardiova, B.; Mikes, J.; Kleban, J. Hypericin-induced photocytotoxicity is connected with G2/M arrest in HT-29 and S-phase arrest in U937 cells. Photochem. Photobiol 2006, 82, 1285–1291. [Google Scholar]
- Mordant, P.; Loriot, Y.; Leteur, C.; Calderaro, J.; Bourhis, J.; Wislez, M.; Soria, J.C.; Deutsch, E. Dependence on phosphoinositide 3-kinase and RAS-RAF pathways drive the activity of RAF265, a novel RAF/VEGFR2 inhibitor, and RAD001 (Everolimus) in combination. Mol. Cancer Ther 2010, 9, 358–368. [Google Scholar]
- Solar, P.; Cavarga, I.; Hofmanova, J.; Cekanova-Figurova, M.; Miskovsky, P.; Brezani, P.; Hrckova, G.; Kozubik, A.; Fedorocko, P. Effect of acetazolamide on hypericin photocytotoxicity. Planta Med 2002, 68, 658–660. [Google Scholar]
- Vantieghem, A.; Xu, Y.; Assefa, Z.; Piette, J.; Vandenheede, J.R.; Merlevede, W.; de Witte, P.A.; Agostinis, P. Phosphorylation of Bcl-2 in G2/M phase-arrested cells following photodynamic therapy with hypericin involves a CDK1-mediated signal and delays the onset of apoptosis. J. Biol. Chem 2002, 277, 37718–37731. [Google Scholar]
- Waldman, T.; Zhang, Y.; Dillehay, L.; Yu, J.; Kinzler, K.; Vogelstein, B.; Williams, J. Cell-cycle arrest versus cell death in cancer therapy. Nat. Med 1997, 3, 1034–1036. [Google Scholar]
- Heiligtag, S.J.; Bredehorst, R.; David, K.A. Key role of mitochondria in cerulenin-mediated apoptosis. Cell Death Differ 2002, 9, 1017–1025. [Google Scholar]
- Altznauer, F.; Conus, S.; Cavalli, A.; Folkers, G.; Simon, H.U. Calpain-1 regulates Bax and subsequent Smac-dependent caspase-3 activation in neutrophil apoptosis. J. Biol. Chem 2004, 279, 5947–5957. [Google Scholar]
- Karlsson, J.; Ora, I.; Porn-Ares, I.; Pahlman, S. Arsenic trioxide-induced death of neuroblastoma cells involves activation of Bax and does not require p53. Clin. Cancer Res 2004, 10, 3179–3188. [Google Scholar]
- Wood, D.E.; Thomas, A.; Devi, L.A.; Berman, Y.; Beavis, R.C.; Reed, J.C.; Newcomb, E.W. Bax cleavage is mediated by calpain during drug-induced apoptosis. Oncogene 1998, 17, 1069–1078. [Google Scholar]
- Yeo, J.K.; Cha, S.D.; Cho, C.H.; Kim, S.P.; Cho, J.W.; Baek, W.K.; Suh, M.H.; Kwon, T.K.; Park, J.W.; Suh, S.I. Se-methylselenocysteine induces apoptosis through caspase activation and Bax cleavage mediated by calpain in SKOV-3 ovarian cancer cells. Cancer Lett 2002, 182, 83–92. [Google Scholar]
- Sanchez-Gomez, M.V.; Alberdi, E.; Perez-Navarro, E.; Alberch, J.; Matute, C. Bax and calpain mediate excitotoxic oligodendrocyte death induced by activation of both AMPA and kainate receptors. J. Neurosci 2011, 31, 2996–3006. [Google Scholar]
- Rokudai, S.; Fujita, N.; Kitahara, O.; Nakamura, Y.; Tsuruo, T. Involvement of FKHR-dependent TRADD expression in chemotherapeutic drug-induced apoptosis. Mol. Cell Biol 2002, 22, 8695–8708. [Google Scholar]
- Cao, X.; Deng, X.; May, W.S. Cleavage of Bax to p18 Bax accelerates stress-induced apoptosis, and a cathepsin-like protease may rapidly degrade p18 Bax. Blood 2003, 102, 2605–2614. [Google Scholar]
- Yang, Y.; Zhao, S.; Song, J. Caspase-dependent apoptosis and -independent poly(ADP-ribose) polymerase cleavage induced by transforming growth factor beta1. Int. J. Biochem. Cell Biol 2004, 36, 223–234. [Google Scholar]
- Ghosh, A.K.; Steele, R.; Ryerse, J.; Ray, R.B. Tumor-suppressive effects of MBP-1 in non-small cell lung cancer cells. Cancer Res 2006, 66, 11907–11912. [Google Scholar]
- Bhaskara, V.K.; Challa, S.; Panigrahi, M.; Babu, P.P. Differential PARP cleavage: An indication for existence of multiple forms of cell death in human gliomas. Neurol. India 2009, 57, 264–268. [Google Scholar]
- Chaitanya, G.V.; Steven, A.J.; Babu, P.P. PARP-1 cleavage fragments: Signatures of cell-death proteases in neurodegeneration. Cell Commun. Signal 2010, 8. [Google Scholar] [CrossRef]
- Rao, L.; Perez, D.; White, E. Lamin proteolysis facilitates nuclear events during apoptosis. J. Cell. Biol 1996, 135, 1441–1455. [Google Scholar]
- Kaufmann, S.H. Induction of endonucleolytic DNA cleavage in human acute myelogenous leukemia cells by etoposide, camptothecin, and other cytotoxic anticancer drugs: A cautionary note. Cancer Res 1989, 49, 5870–5878. [Google Scholar]
- Neamati, N.; Fernandez, A.; Wright, S.; Kiefer, J.; McConkey, D.J. Degradation of lamin B1 precedes oligonucleosomal DNA fragmentation in apoptotic thymocytes and isolated thymocyte nuclei. J. Immunol 1995, 154, 3788–3795. [Google Scholar]
- Shimizu, T.; Cao, C.X.; Shao, R.G.; Pommier, Y. Lamin B phosphorylation by protein kinase calpha and proteolysis during apoptosis in human leukemia HL60 cells. J. Biol. Chem 1998, 273, 8669–8674. [Google Scholar]
- Dynlacht, J.R.; Earles, M.; Henthorn, J.; Seno, J.D. Different patterns of DNA fragmentation and degradation of nuclear matrix proteins during apoptosis induced by radiation, hyperthermia or etoposide. Radiat Res 2000, 154, 515–530. [Google Scholar]
- Di Matola, T.; D’Ascoli, F.; Luongo, C.; Bifulco, M.; Rossi, G.; Fenzi, G.; Vitale, M. Lovastatin-induced apoptosis in thyroid cells: Involvement of cytochrome c and lamin B. Eur. J. Endocrinol 2001, 145, 645–650. [Google Scholar]
- Zhivotovsky, B.; Gahm, A.; Orrenius, S. Two different proteases are involved in the proteolysis of lamin during apoptosis. Biochem. Biophys. Res. Commun 1997, 233, 96–101. [Google Scholar]
- Granville, D.J.; Cassidy, B.A.; Ruehlmann, D.O.; Choy, J.C.; Brenner, C.; Kroemer, G.; van Breemen, C.; Margaron, P.; Hunt, D.W.; McManus, B.M. Mitochondrial release of apoptosis-inducing factor and cytochrome c during smooth muscle cell apoptosis. Am. J. Pathol 2001, 159, 305–311. [Google Scholar]
- Furre, I.E.; Moller, M.T.; Shahzidi, S.; Nesland, J.M.; Peng, Q. Involvement of both caspase-dependent and -independent pathways in apoptotic induction by hexaminolevulinate-mediated photodynamic therapy in human lymphoma cells. Apoptosis 2006, 11, 2031–2042. [Google Scholar]
- Karmakar, S.; Banik, N.L.; Patel, S.J.; Ray, S.K. 5-Aminolevulinic acid-based photodynamic therapy suppressed survival factors and activated proteases for apoptosis in human glioblastoma U87MG cells. Neurosci. Lett 2007, 415, 242–247. [Google Scholar]
- Vittar, N.B.; Awruch, J.; Azizuddin, K.; Rivarola, V. Caspase-independent apoptosis, in human MCF-7c3 breast cancer cells, following photodynamic therapy, with a novel water-soluble phthalocyanine. Int. J. Biochem. Cell Biol 2010, 42, 1123–1131. [Google Scholar]
- Di Paolo, A.; Danesi, R.; Caputo, S.; Macchia, M.; Lastella, M.; Boggi, U.; Mosca, F.; Marchetti, A.; Del Tacca, M. Inhibition of protein farnesylation enhances the chemotherapeutic efficacy of the novel geranylgeranyltransferase inhibitor BAL9611 in human colon cancer cells. Br. J. Cancer 2001, 84, 1535–1543. [Google Scholar]



© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sačková, V.; Kuliková, L.; Kello, M.; Uhrinová, I.; Fedoročko, P. Enhanced Antiproliferative and Apoptotic Response of HT-29 Adenocarcinoma Cells to Combination of Photoactivated Hypericin and Farnesyltransferase Inhibitor Manumycin A. Int. J. Mol. Sci. 2011, 12, 8388-8405. https://doi.org/10.3390/ijms12128388
Sačková V, Kuliková L, Kello M, Uhrinová I, Fedoročko P. Enhanced Antiproliferative and Apoptotic Response of HT-29 Adenocarcinoma Cells to Combination of Photoactivated Hypericin and Farnesyltransferase Inhibitor Manumycin A. International Journal of Molecular Sciences. 2011; 12(12):8388-8405. https://doi.org/10.3390/ijms12128388
Chicago/Turabian StyleSačková, Veronika, Lucia Kuliková, Martin Kello, Ivana Uhrinová, and Peter Fedoročko. 2011. "Enhanced Antiproliferative and Apoptotic Response of HT-29 Adenocarcinoma Cells to Combination of Photoactivated Hypericin and Farnesyltransferase Inhibitor Manumycin A" International Journal of Molecular Sciences 12, no. 12: 8388-8405. https://doi.org/10.3390/ijms12128388





