The Stimulation of IGF-1R Expression by Lewis(y) Antigen Provides a Powerful Development Mechanism of Epithelial Ovarian Carcinoma
Abstract
:Objective
Methods
Results
Conclusion
1. Introduction
2. Results
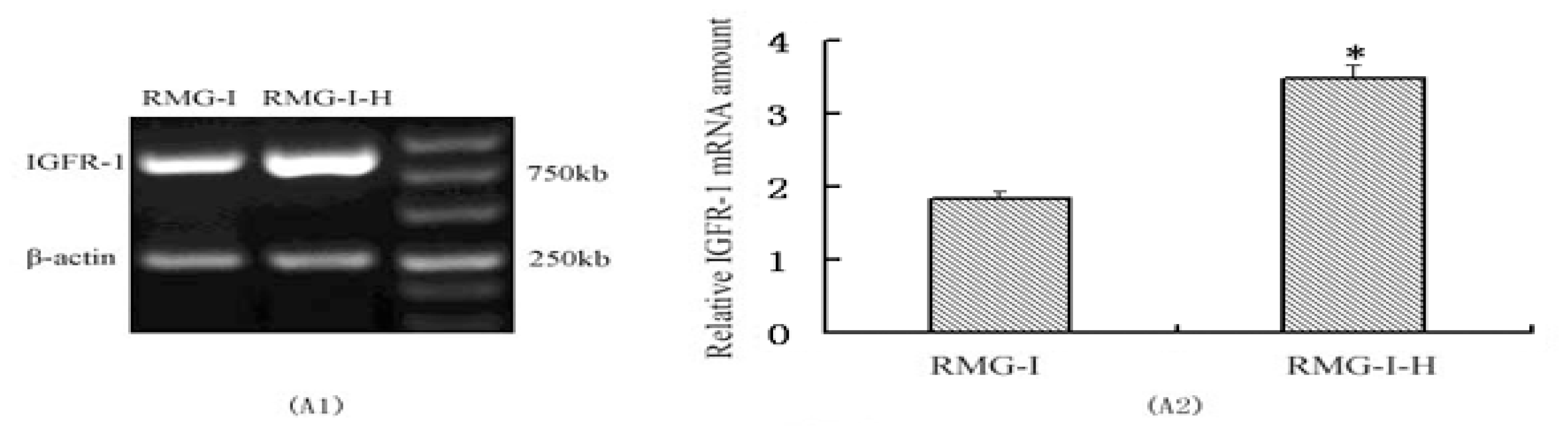
2.1. IGF-1R mRNA and Protein Expression in RMG-I and RMG-I-H Cells before and after Antibody Blocking
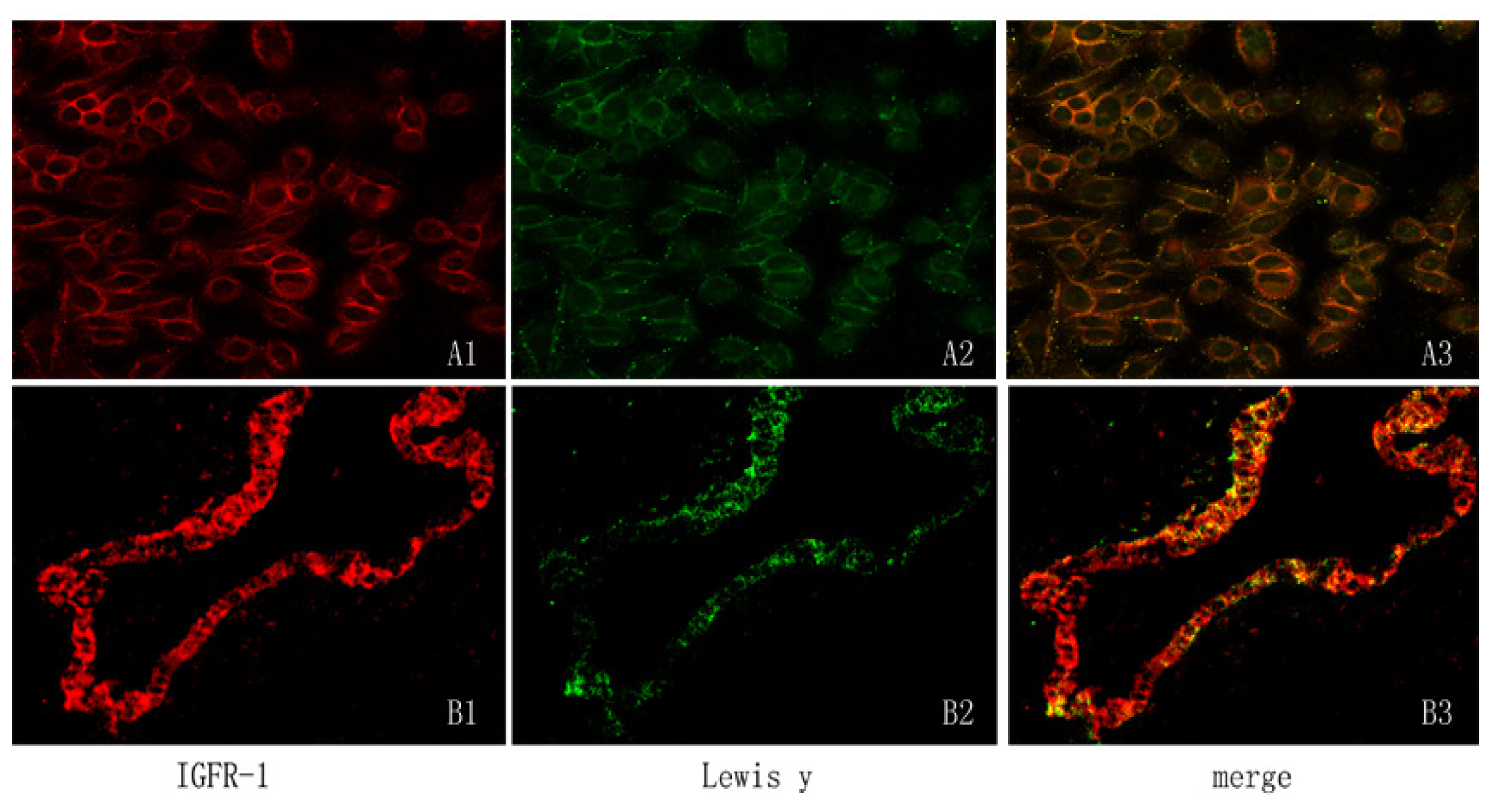
2.2. Expression of IGF-1R Protein and Lewis(y) on the Surface of RMG-I and RMG-I-H Cells and in Epithelial Ovarian Cancer Tissues
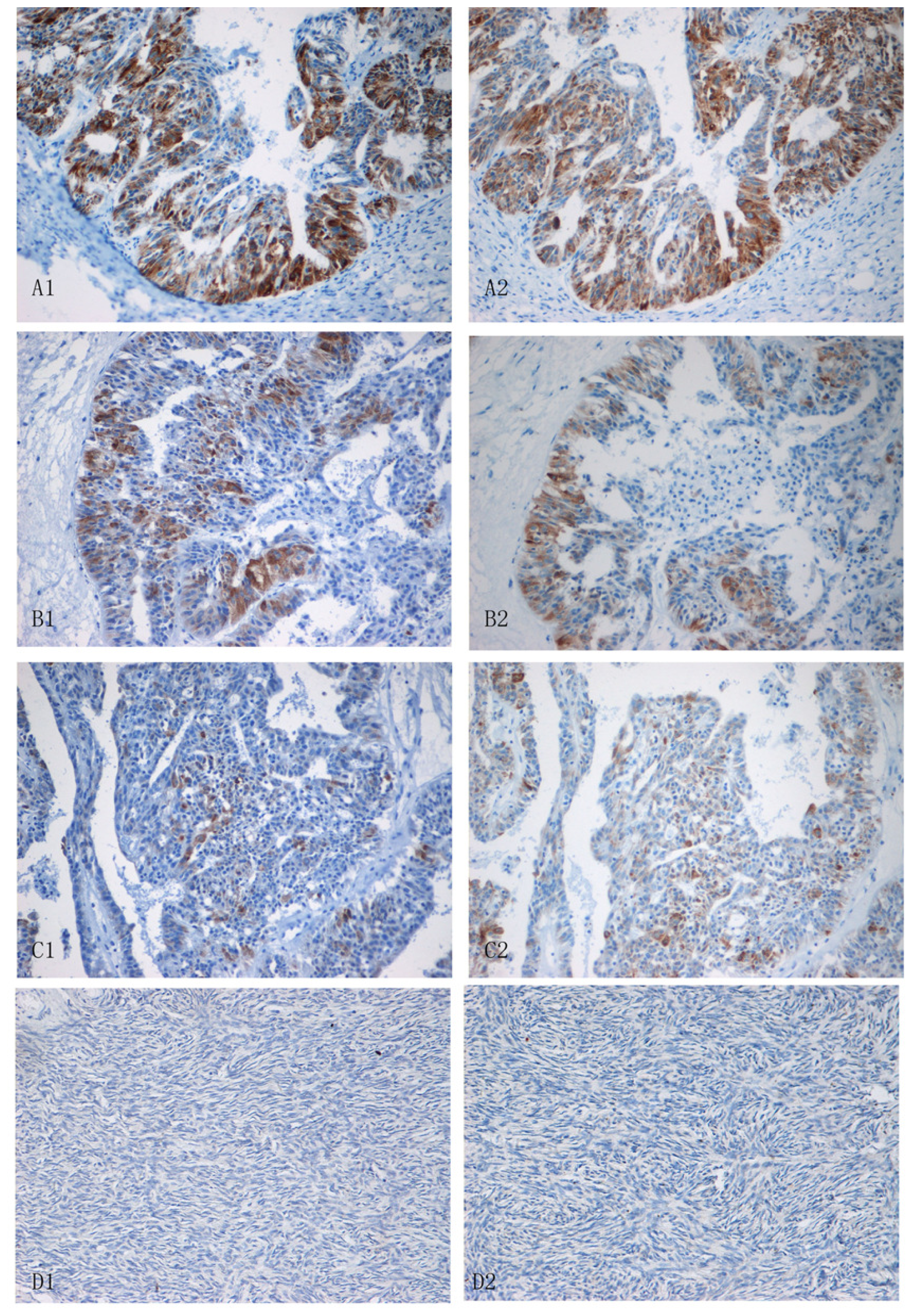
2.3. Expression of Lewis(y) and IGF-1R in Various Ovarian Tissues
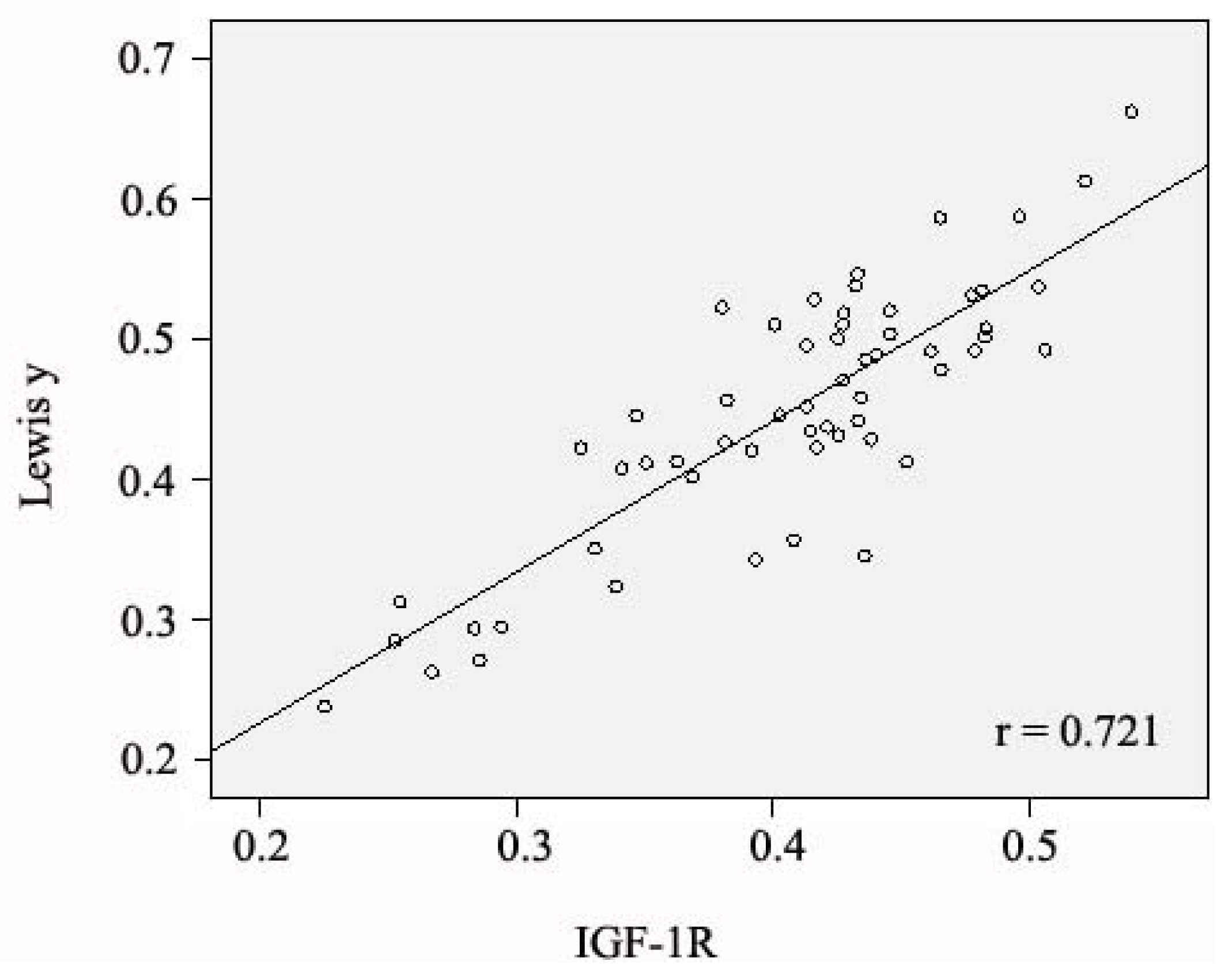
2.4. Relationship between Expression of IGF-1R and Antigen Lewis(y), and Clinical Features of Ovarian Cancer
2.5. Staining Intensity of Lewis(y) and IGF-1R with Relation to Clinicopathological Parameters of Ovarian Cancer
3. Discussion
4. Material and Method
4.1. Reagents and Cells
4.2. Patients and Tissue Samples
4.3. Cell Culture
4.4. Determination of the Expression of IGF-1R mRNAs with Semi-Quantitative RT-PCR
4.5. Analysis of the Proteins of IGF-1R with Western Blotting
4.6. Analysis of Lewis(y) Expression on IGF-1R Using Immunoprecipitation and Western Blotting
4.7. Detection of Lewis(y) and IGF-1R Location on Cells and Tumor Tissue by Double-Labeling Immunofluorescence
4.8. Immunohistochemistry
4.9. Assessment Standard and Statistical Analysis
5. Conclusions
Acknowledgements
References
- Diao, B; Huang, J; Lin, B; Zhou, D; Hao, YY; Zhang, SL. The expression and significance of Le-y antigen in ovarian epithelial carcinoma. J. Mod. Oncol 2008, 3, 335–338. [Google Scholar]
- Dettke, M; Palfi, G; Loibner, H. Activation-dependent expression of the blood group-related Lewis(y) antigen on peripheral blood granulocytes. J. Leukoc. Biol 2000, 68, 511–514. [Google Scholar]
- Hellstrom, I; Garrigues, HJ; Garrigues, U; Hellstrom, KE. Highly tumor-reactive, internalizing, mouse monoclonal antibodies to Le(y)-related cell surface antigens. Cancer Res 1990, 50, 2183–2190. [Google Scholar]
- Madjd, Z; Parsons, T; Watson, NF; Spendlove, I; Ellis, I; Durrant, LG. High expression of Lewis(y)/b antigens is associated with decreased survival in lymph node negative breast carcinomas. Breast Cancer Res 2005, 7, R780–R787. [Google Scholar]
- Lin, B; Hao, YY; Wang, DD; Zhu, LC; Zhang, SL; Saito, M; Iwamori, M. Transfection of α1,2-Fucosyltransferase gene increases the antigenic expression of Lewis(y) in ovarian cancer cell line RMG-I. Acta Acad. Med. Sin 2008, 30, 284–289. [Google Scholar]
- Zhao, Y; Lin, B; Hao, YY; Yan, LM; Liu, JJ; Zhu, LC; Zhang, SL. The effects of Lewis(y) antigen content on drug resistance to carboplatin in ovarian cancer line RMG-I. Prog. Biochem. Biophys 2008, 35, 1175–1182. [Google Scholar]
- Hao, Y; Lin, B; Zhao, Y; Zhang, YH; Li, FF; Diao, B; Ou, YL; Zhang, SL. α1,2- Fucosyltransferase gene translection in fluences on biological behavior of ovarian carcinomaderived RMG-1 cells. Fen Zi Xi Bao Sheng Wu Xue Bao 2008, 41, 435–442. [Google Scholar]
- Hiden, U; Glitzner, E; Hartmann, M; Desoye, G. Insulin and the IGF system in the human placenta of normal and diabetic pregnancies. J. Anat 2009, 215, 60–68. [Google Scholar]
- Dufourny, B; Alblas, J; van Teeffelen, HA; van Schaik, FM; van der Burg, B; Steenbergh, PH; Sussenbach, JS. Mitogenic signaling of insulin-like growth factor-I in MCF-7 human breast cancer cells requires phosphatidylinositol 3-kinase and is independent of mitogen-activated protein kinase. J. Biol. Chem 1997, 272, 31163–31171. [Google Scholar]
- Khandwala, HM; McCutcheon, IE; Flyvbjerg, A; Friend, KE. The effects of insulin-like growth factors on tumorigenes is and neoplastic growth. Endocr. Rev 2000, 21, 215–244. [Google Scholar]
- Baserga, R; Hongo, A; Rubini, M; Baserga, R; Hongo, A; Rubini, M; Prisco, M; Valentinis, B. The IGF-I receptor in cell growth, transformation and apoptosis. Biochim. Biophys. Acta 1997, 1332, F105–F126. [Google Scholar]
- Blakesley, VA; Stannard, BS; Kalebic, T; Helman, LJ; LeRoith, D. Role of the IGF-I receptor in mutagensis and tumor promotion. J. Endocrinol 1997, 152, 339–344. [Google Scholar]
- Lau, KS; Partridge, EA; Grigorian, A; Silvescu, CI; Reinhold, VN; Demetriou, M; Dennis, JW. Complex N-glycan number and degree of branching cooperate to regulate cell proliferation and differentiation. Cell 2007, 129, 123–134. [Google Scholar]
- Rudd, PM; Dwek, RA. Glycosylation: Heterogeneity and the 3D structure of proteins. Crit. Rev. Biochem. Mol. Biol 1997, 32, 1–100. [Google Scholar]
- Mann, M; Jensen, ON. Proteomic analysis of post-translational modifications. Nat. Biotechnol 2003, 21, 255–261. [Google Scholar]
- Rebbaa, A; Yamamoto, H; Saito, T; Meuillet, E; Kim, P; Kersey, DS; Bremer, EG; Taniguchi, N; Moskal, JR. Gene transfection-mediated overexpression of β1,4-N- acetylglucosamine- bisecting oligosaccharides in glioma cell line U373 MG inhibits epidermal growth factor receptor function. J. Biol. Chem 1997, 272, 9275–9279. [Google Scholar]
- Wang, QY; Zhang, Y; Chen, HJ; Shen, ZH; Chen, HL. Alpha 1,3-fucosyltransferase-VII regulates the signaling molecules of the insulin receptor pathway. FEBS J 2007, 274, 526–538. [Google Scholar]
- Kitamura, K; Stockert, E; Garin-Ghesa, P; Welt, S; Lloyd, KO; Armour, KL; Wallace, TP; Harris, WJ; Carr, FJ; Old, LJ. Specificity anaiysis of blood group Lewis-y (Le(y)) antibodies generated against synthetic and natural Le(y) determinants. Proc. Natl. Acad. Sci. USA 1994, 91, 12957–12961. [Google Scholar]
- Yin, BW; Finstad, CL; Kitamura, K. Serological and immunochemical analysis of Lewis(y) (Ley) blood group antigen expression in epithelial ovarian cancer. Int. J. Cancer 2006, 65, 406–412. [Google Scholar]
- Baldus, SE; Hanisch, FG; Pütz, C. Immunoreactivity of Lewis blood group and mucin peptide core antigens: correlations with grade of dysplasia and malignant transformation in the colorectal adenoma-carcinoma sequence. Histol. Histopathol 2002, 17, 191–198. [Google Scholar]
- An, Y; Cai, L; Wang, Y; Zhu, D; Guan, Y; Zheng, J. Local expression of insulin-like growth factor-I, insulin-like growth factor-I receptor, and estrogen receptor alpha in ovarian cancer. Onkologie 2009, 32, 638–644. [Google Scholar]
- Basu, A; Murthy, U; Rodeck, U; Herlyn, M; Mattes, L; Das, M. Presence of tumor associated antigens in epidermal growth factor receptors from different human carcinomas. Cancer Res 1987, 47, 2531–2536. [Google Scholar]
- Liu, JJ; Lin, B; Hao, YY; Li, FF; Liu, DW; Qi, Y; Zhu, LC; Zhang, SL; Iwamori, M. Lewis(y) antigen stimulates the growth of ovarian cancer cells via regulation of the epidermal growth factor receptor pathway. Oncol. Rep 2010, 23, 833–841. [Google Scholar]
- Gao, LL; Yan, LM; Lin, B; Gao, J; Liang, XY; Wang, YY; Liu, JJ; Zhang, SL; Iwamori, M. Enhancive effects of Lewis(y) antigen on CD44-mediated adhesion and spreading of human ovarian cancer cell line RMG-I. J. Exp. Clin. Cancer Res 2011, 30, 15. [Google Scholar]
- Gregory, CW; DeGeorges, A; Sikes, RA. The IGF axis in the development and progression of prostate cancer. Recent Res. Dev. Cancer 2001, 3, 437–462. [Google Scholar]
- Cong, JP; Lin, B; Liu, JJ; Liu, Q; Li, FF; Liu, SC; Gao, S; Zhang, SL. The effect of α1, 2-Fucosyl Transferase Gene Transfection on p38MAPK signaling pathway-mediated apoptosis of ovarian carcinoma RMG-I cells. Prog. Biochem. Biophys 2010, 37, 175–183. [Google Scholar]
- Liu, JJ; Lin, B; Hao, YY; Qi, Y; Zhu, LC; Li, FF; Liu, DW; Cong, JP; Zhang, SL; Iwamori, M. Lewis(y) antigen promotes the proliferation of ovarian carcinoma-derived RMG-I cell through the PI3K/Akt signaling pathway. J. Exp. Clin. Cancer Res 2009, 28, 154. [Google Scholar]

| Groups | Cases | Lewis(y) | Positive Cases | Rate (%) | IGF-1R | Positive Cases | Rate (%) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| − | + | ++ | +++ | − | + | ++ | +++ | ||||||
| Malignant group | 60 | 7 | 15 | 20 | 18 | 53 | 88.33 * | 4 | 10 | 23 | 23 | 56 | 93.33 * |
| Borderline group | 30 | 12 | 6 | 11 | 1 | 18 | 60.00† | 11 | 9 | 7 | 3 | 19 | 63.33‡ |
| Benign group | 30 | 20 | 6 | 4 | 0 | 10 | 33.33 | 14 | 9 | 6 | 1 | 16 | 53.33 |
| Normal group | 20 | 20 | 0 | 0 | 0 | 0 | 00.00 | 12 | 6 | 2 | 0 | 8 | 40.00 |
| Features | Case | Lewis(y) | IGF-1R | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Positive Cases | Rate (%) | P | MOD | P | Positive Cases | Rate (%) | P | MOD | P | |||
| Pathological type | Mucous | 30 | 26 | 86.67 | >0.05 | 0.463 ± 0.068 | >0.05 | 24 | 80.00 | >0.05 | 0.418 ± 0.044 | >0.05 |
| Serous | 30 | 27 | 90.00 | 0.477 ± 0.016 | 27 | 90.00 | 0.437 ± 0.051 | |||||
| FIGO stage | I–II | 39 | 33 | 84.62 | >0.05 | 0.455 ± 0.065 | <0.05 | 33 | 84.62 | >0.05 | 0.413 ± 0.047 | <0.05 |
| III–IV | 21 | 20 | 95.24 | 0.505 ± 0.072 | 18 | 85.71 | 0.444 ± 0.051 | |||||
| Differentiation level | High | 21 | 17 | 80.95 | >0.05 | 0.448 ± 0.017 | <0.05 * | 16 | 76.19 | >0.05 | 0.412 ± 0.052 | <0.05 * |
| Middle | 21 | 18 | 85.71 | 0.461 ± 0.054 | >0.05 † | 18 | 85.71 | 0.413 ± 0.038 | <0.05 † | |||
| Low | 18 | 18 | 100 | 0.498 ± 0.084 | >0.05 ‡ | 17 | 94.44 | 0.455 ± 0.049 | >0.05 ‡ | |||
| Lymphatic metastasis | No | 48 | 41 | 85.42 | >0.05 | 0.459 ± 0.078 | >0.05 | 40 | 83.33 | >0.05 | 0.420 ± 0.054 | >0.05 |
| Yes | 12 | 12 | 100 | 0.476 ± 0.057 | 11 | 91.67 | 0.433 ± 0.060 | |||||
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liu, D.; Liu, J.; Wang, C.; Lin, B.; Liu, Q.; Hao, Y.; Zhang, S.; Iwamori, M. The Stimulation of IGF-1R Expression by Lewis(y) Antigen Provides a Powerful Development Mechanism of Epithelial Ovarian Carcinoma. Int. J. Mol. Sci. 2011, 12, 6781-6795. https://doi.org/10.3390/ijms12106781
Liu D, Liu J, Wang C, Lin B, Liu Q, Hao Y, Zhang S, Iwamori M. The Stimulation of IGF-1R Expression by Lewis(y) Antigen Provides a Powerful Development Mechanism of Epithelial Ovarian Carcinoma. International Journal of Molecular Sciences. 2011; 12(10):6781-6795. https://doi.org/10.3390/ijms12106781
Chicago/Turabian StyleLiu, Dawo, Juanjuan Liu, Changzhi Wang, Bei Lin, Qing Liu, Yingying Hao, Shulan Zhang, and Masao Iwamori. 2011. "The Stimulation of IGF-1R Expression by Lewis(y) Antigen Provides a Powerful Development Mechanism of Epithelial Ovarian Carcinoma" International Journal of Molecular Sciences 12, no. 10: 6781-6795. https://doi.org/10.3390/ijms12106781




