Expression of Elongation Factor (EF)-Tu Is Correlated with Prognosis of Gastric Adenocarcinomas
Abstract
:1. Introduction
2. Materials and Methods
2.1. Case Selection
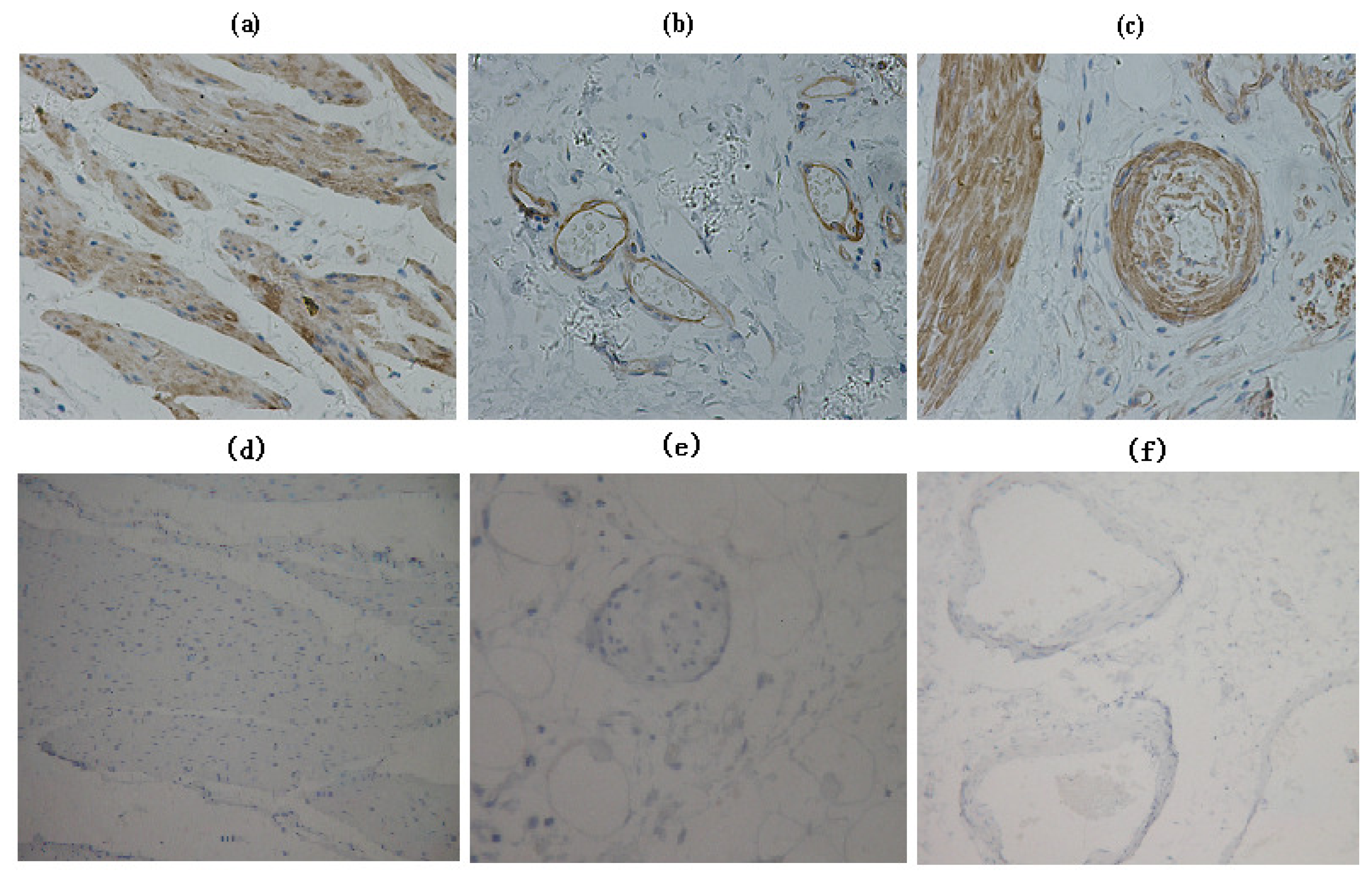
2.2. Immunohistochemistry
2.3. Follow Up
2.4. Statistical Analysis
3. Results
3.1. EF-Tu Expression Correlates with Clinicopathologic Parameters
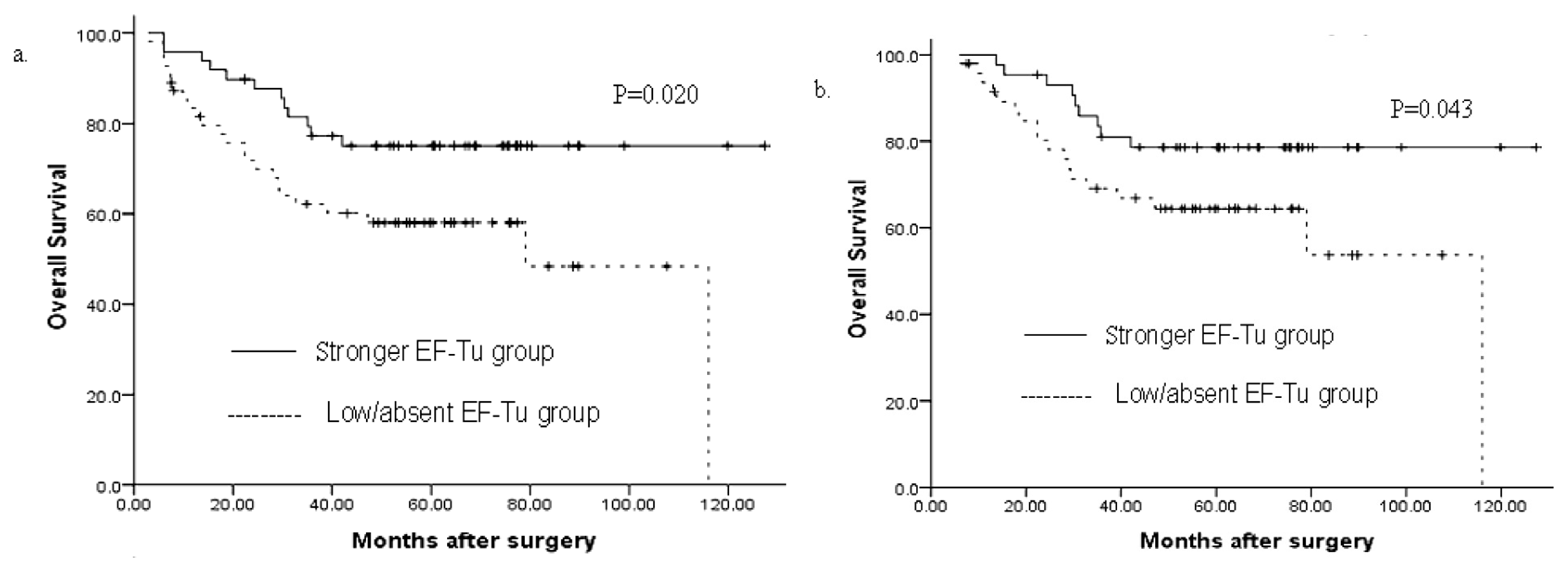
3.2. Univariate Analysis
3.3. Multivariate Analysis
4. Discussion
Acknowledgement
References
- Hohenberger, P; Gretschel, S. Gastric cancer. Lancet 2003, 362, 305–315. [Google Scholar]
- Dicken, BJ; Bigam, DL; Cass, C; Mackey, JR; Joy, AA; Hamilton, SM. Gastric adenocarcinoma: Review and considerations for future directions. Ann. Surg 2005, 241, 27–39. [Google Scholar]
- Zhang, B; Cao, W; Zhang, F; Zhang, L; Niu, R; Niu, Y; Fu, L; Hao, X; Cao, X. Protein interacting with C alpha kinase 1 (PICK1) is involved in promoting tumor growth and correlates with poor prognosis of human breast cancer. Cancer Sci 2010, 101, 1536–1542. [Google Scholar]
- Parmeggiani, A; Nissen, P. Elongation factor Tu-targeted antibiotics: Four different structures, two mechanisms of action. FEBS Lett 2006, 580, 4576–4581. [Google Scholar]
- Suzuki, H; Ueda, T; Taguchi, H; Takeuchi, N. Chaperone properties of mammalian mitochondrial translation elongation factor Tu. J. Biol. Chem 2007, 282, 4076–4084. [Google Scholar]
- Caldas, TD; El Yaagoubi, A; Richarme, G. Chaperone properties of bacterial elongation factor EF-Tu. J. Biol. Chem 1998, 273, 11478–11482. [Google Scholar]
- Archambaud, C; Gouin, E; Pizarro-Cerda, J; Cossart, P; Dussurget, O. Translation elongation factor EF-Tu is a target for Stp, a serine-threonine phosphatase involved in virulence of Listeria monocytogenes. Mol. Microbiol 2005, 56, 383–396. [Google Scholar]
- Sakai, J; Ishikawa, H; Kojima, S; Satoh, H; Yamamoto, S; Kanaoka, M. Proteomic analysis of rat heart in ischemia and ischemia-reperfusion using fluorescence two-dimensional difference gel electrophoresis. Proteomics 2003, 3, 1318–1324. [Google Scholar]
- Shin, JH; London, J; Le Pecheur, M; Höger, H; Pollak, D; Lubec, G. Aberrant neuronal and mitochondrial proteins in hippocampus of transgenic mice overexpressing human Cu/Zn superoxide dismutase 1. Free Radic. Biol. Med 2004, 37, 643–653. [Google Scholar]
- Wells, J; Henkler, F; Leversha, M; Koshy, R. A mitochondrial elongation factor-like protein is over-expressed in tumours and differentially expressed in normal tissues. FEBS Lett 1995, 358, 119–125. [Google Scholar]
- Grant, AG; Flomen, RM; Tizard, ML; Grant, DA. Differential screening of a human pancreatic adenocarcinoma lambda gt11 expression library has identified increased transcription of elongation factor EF-1 alpha in tumour cells. Int. J. Cancer 1992, 50, 740–745. [Google Scholar]
- Srisomsap, C; Sawangareetrakul, P; Subhasitanont, P; Panichakul, T; Keeratichamroen, S; Lirdprapamongkol, K; Chokchaichamnankit, D; Sirisinha, S; Svasti, J. Proteomic analysis of cholangiocarcinoma cell line. Proteomics 2004, 4, 1135–1144. [Google Scholar]
- Koch, I; Hofschneider, PH; Lottspeich, F; Eckerskorn, C; Koshy, R. Tumour-related expression of a translation-elongation factor-like protein. Oncogene 1990, 5, 839–843. [Google Scholar]
- Kim, HK; Park, WS; Kang, SH; Warda, M; Kim, N; Ko, JH; Prince, Ael-B; Han, J. Mitochondrial alterations in human gastric carcinoma cell line. Am. J. Physiol. Cell Physiol 2007, 293, C761–C771. [Google Scholar]
- Parmeggiani, A; Nissen, P. Elongation factor Tu-targeted antibiotics: Four different structures, two mechanisms of action. FEBS Lett 2006, 580, 4576–4581. [Google Scholar]
- Zuurmond, AM; Martien de Graaf, J; Olsthoorn-Tieleman, LN; van Duyl, BY; Mörhle, VG; Jurnak, F; Mesters, JR; Hilgenfeld, R; Kraal, B. GE2270A-resistant mutations in elongation factor Tu allow productive aminoacyl-tRNA binding to EF-Tu.GTP.GE2270A complexes. J. Mol. Biol 2000, 304, 995–1005. [Google Scholar]
- Zipfel, C; Kunze, G; Chinchilla, D; Caniard, A; Jones, JD; Boller, T; Felix, G. Perception of the bacterial PAMP EF-Tu by the receptor EFR restricts Agrobacterium-mediated transformation. Cell 2006, 125, 749–760. [Google Scholar]
- Wells, J; Henkler, F; Leversha, M; Koshy, R. A mitochondrial elongation factor-like protein is over-expressed in tumours and differentially expressed in normal tissues. FEBS Lett 1995, 358, 119–125. [Google Scholar]
- Gross, SR; Kinzy, TG. Translation elongation factor 1A is essential for regulation of the actin cytoskeleton and cell morphology. Nat. Struct. Mol. Biol 2005, 12, 772–778. [Google Scholar]
- Kuramitsu, Y; Baron, B; Yoshino, S; Zhang, X; Tanaka, T; Yashiro, M; Hirakawa, K; Oka, M; Nakamura, K. Proteomic differential display analysis shows up-regulation of 14-3-3 sigma protein in human scirrhous-type gastric carcinoma cells. Anticancer Res 2010, 30, 4459–4465. [Google Scholar]
- Rao, TR; Slobin, LI. The stability of mRNA for eucaryotic elongation factor Tu in Friend erythroleukemia cells varies with growth rate. Mol. Cell. Biol 1988, 8, 1085–1092. [Google Scholar]
- Omori, S; Sato, Y; Hirakawa, S; Isobe, T; Yukawa, M; Murata, K. Two extra chromosomal genomes of Leucocytozoon caulleryi; complete nucleotide sequences of the mitochondrial genome and existence of the apicoplast genome. Parasitol. Res 2008, 103, 953–957. [Google Scholar]
- Krab, IM; Te Biesebeke, R; Bernardi, A; Parmeggiani, A. Elongation factor Ts can act as a steric chaperone by increasing the solubility of nucleotide binding-impaired elongation factor-Tu. Biochemistry 2001, 40, 8531–8535. [Google Scholar]
- Krab, IM; Te Biesebeke, R; Bernardi, A; Parmeggiani, A. Chloroplast protein synthesis elongation factor, EF-Tu, reduces thermal aggregation of rubisco activase. J. Plant Physiol 2007, 164, 1564–1571. [Google Scholar]
- Modica-Napolitano, JS; Kulawiec, M; Singh, KK. Mitochondria and human cancer. Curr. Mol. Med 2007, 7, 121–131. [Google Scholar]
- Modica-Napolitano, JS; Singh, KK. Mitochondrial dysfunction in cancer. Mitochondrion 2004, 4, 755–762. [Google Scholar]
- Gogvadze, V; Orrenius, S; Zhivotovsky, B. Mitochondria in cancer cells: What is so special about them? Trends Cell Biol 2008, 18, 165–173. [Google Scholar]
- Calabrese, C; Pisi, A; Di Febo, G; Liguori, G; Filippini, G; Cervellera, M; Righi, V; Lucchi, P; Mucci, A; Schenetti, L; et al. Biochemical alterations from normal mucosa to gastric cancer by ex vivo magnetic resonance spectroscopy. Cancer Epidemiol. Biomarkers Prev 2008, 17, 1386–1395. [Google Scholar]
- Zhang, L; Li, L; Liu, H; Prabhakaran, K; Zhang, X; Borowitz, JL; Isom, GE. HIF-1alpha activation by a redox-sensitive pathway mediates cyanide-induced BNIP3 upregulation and mitochondrial-dependent cell death. Free Radic. Biol. Med 2007, 43, 117–127. [Google Scholar]
- Gillies, RJ; Robey, I; Gatenby, RA. Causes and consequences of increased glucose metabolism of cancers. J. Nucl. Med 2008, 49, 24S–42S. [Google Scholar]
- Kondoh, H. Cellular life span and the Warburg effect. Exp. Cell Res 2008, 314, 1923–1928. [Google Scholar]
- Kim, JW; Tchernyshyov, I; Semenza, GL; Dang, CV. HIF-1-mediated expression of pyruvate dehydrogenase kinase: A metabolic switch required for cellular adaptation to hypoxia. Cell Metab 2006, 3, 177–185. [Google Scholar]
- Papandreou, I; Cairns, RA; Fontana, L; Lim, AL; Denko, NC. HIF-1 mediates adaptation to hypoxia by actively down regulating mitochondrial oxygen consumption. Cell Metab 2006, 3, 187–197. [Google Scholar]

| Negative/low (n = 55) | High (n = 49) | P value | |
|---|---|---|---|
| Age | 54.9 ± 11.3 | 57.2 ± 12.5 | 0.329 |
| Gender | 0.104 | ||
| Male | 40 (61.5%) | 28 (51.7) | |
| Female | 15 (38.5%) | 21 (48.3) | |
| ECOG | 0.544 | ||
| 0 | 18 (32.7) | 17 (35.4) | |
| 1 | 37 (67.3) | 31 (62.5) | |
| 2 | 0 | 1 (2.1) | |
| Type of operation | 1.000 | ||
| Total gastrectomy | 6 (10.9) | 5 (10.2) | |
| Subtotal gastrectomy | 49 (89.1) | 44 (89.8) |
| Negative/low (n = 55) | High (n = 49) | P value | |
|---|---|---|---|
| Location | 0.753 | ||
| Upper body or whole | 13 (23.6) | 9 (18.5) | |
| Lower or middle body | 42 (76.4) | 40 (81.5) | |
| Tumor differentiated | 0.061 | ||
| Well | 7 (12.7) | 2 (4.1) | |
| Moderate | 14 (25.5) | 22 (40.0) | |
| Poor | 34 (61.8) | 25 (55.9) | |
| Tumor size | 5.3 ± 2.3 | 4.3 ± 2.0 | 0.021 |
| Serosal invasion | 0.042 | ||
| No | 14 (25.5) | 22 (44.9) | |
| Yes | 41 (74.5) | 27 (55.1) | |
| Node involveme | 0.005 | ||
| No | 13 (23.6) | 25 (51.0) | |
| Yes | 42 (76.4) | 24 (49.0) | |
| Mean no. of tLNs | 21 | 19 | 0.489 |
| Distant metastasis | 1.000 | ||
| No | 48 (87.3) | 43 (23.6) | |
| Yes | 7 (12.7) | 6 (23.6) | |
| Normal specimens EF-Tu expression | 0.015 | ||
| Negative/low | 27 (49.1) | 12 (24.5) | |
| High | 28 (50.9) | 37 (75.5) |
| Variable | HR | 95% CI | P * value |
|---|---|---|---|
| EF-Tu | 0.460 | 0.220–0.960 | 0.038 |
| Distant metastasis | 4.323 | 1.442–12.963 | 0.009 |
| Serosal invasion (yes vs. no) | 6.242 | 1.808–21.548 | 0.004 |
| mLNs | 1.082 | 1.001–1.169 | 0.047 |
| tLNs | 0.053 | ||
| Type of operation | 0.096 | ||
| Age (years) (>55 vs ≤55) | 0.187 | ||
| Tumor size (≦4 cm vs >4 cm) | 0.547 | ||
| Histological type (undifferentiated vs differentiated) | 0.812 | ||
| Gender | 0.810 | ||
| Tumor location | 0.856 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xu, C.; Wang, J.; Li, J.; Fang, R. Expression of Elongation Factor (EF)-Tu Is Correlated with Prognosis of Gastric Adenocarcinomas. Int. J. Mol. Sci. 2011, 12, 6645-6655. https://doi.org/10.3390/ijms12106645
Xu C, Wang J, Li J, Fang R. Expression of Elongation Factor (EF)-Tu Is Correlated with Prognosis of Gastric Adenocarcinomas. International Journal of Molecular Sciences. 2011; 12(10):6645-6655. https://doi.org/10.3390/ijms12106645
Chicago/Turabian StyleXu, Chaoyang, Jianjun Wang, Jiajia Li, and Rengui Fang. 2011. "Expression of Elongation Factor (EF)-Tu Is Correlated with Prognosis of Gastric Adenocarcinomas" International Journal of Molecular Sciences 12, no. 10: 6645-6655. https://doi.org/10.3390/ijms12106645




