Expression of an Organic Solvent Stable Lipase from Staphylococcus epidermidis AT2
Abstract
:1. Introduction
2. Results and Discussion
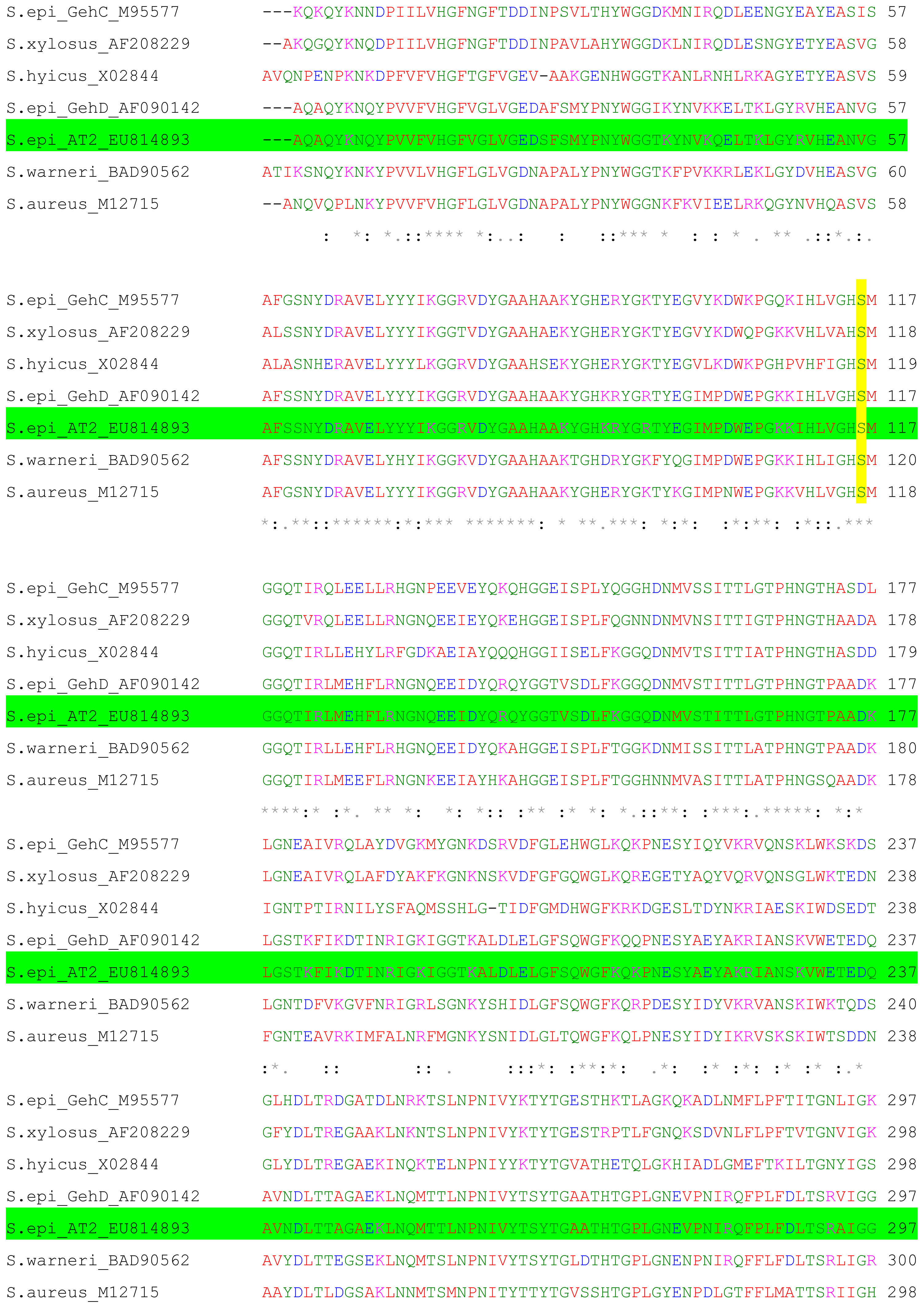
2.1. Cloning and Nucleotide Sequence of the Lipase AT2 Gene
2.2. Expression of AT2 Lipase in Prokaryotic System
2.3. SDS PAGE Analysis
2.4. Stability of Recombinant Lipase in Various Organic Solvents
3. Experimental Section
3.1. Sources of Bacteria
3.2. DNA Manipulation
3.3. Sequencing and Analysis of Lipase Gene
3.4. Cloning of an Organic Solvent-Tolerant AT2 Lipase Gene
FOR: 5′-CAG TGG TCA GCA TGC TCA AGC-3′REV: 5′-GCT AGG TTC ATC ATA CCT ACC TTC-3′
Fm: 5′-CAA TCA ACT TAC TGC GCA AGC-3′Rm: 5′-CAC TAC TTA CGT GTG ATA CCA-3′
3.5. Expression of AT2 Lipase with pTrcHis2 TOPO TA
3.6. Lipase Activity Assay
3.7. Electrophoresis
3.8. Effect of Organic Solvents on Crude Enzyme Stability
4. Conclusions
Acknowledgements
References
- Sakinc, T; Kleine, B; Gatermann, SG. Biochemical characterization of the surface associated lipase of Staphylococcus saprophyticus. FEMS Microbiol. Lett 2007, 274, 335–341. [Google Scholar]
- Sardessai, Y; Bhosle, S. Tolerance of bacteria to organic solvents. Res Microbiol 2002, 153, 263–268. [Google Scholar]
- de Bont, JAM. Solvent-tolerant bacteria in biocatalysis. Trends Biotechnol 1998, 16, 493–499. [Google Scholar]
- Inoue, A; Horikoshi, K. Pseudomonas thrives in high concentrations of toluene. Nature 1989, 338, 264–266. [Google Scholar]
- Gupta, A; Khare, SK. Enzymes from solvent-tolerant microbes: Useful biocatalyst for nonaqueous enzymology. Crit. Rev. Biotechnol 2009, 29, 44–54. [Google Scholar]
- Ogino, H; Miyamoto, K; Ishikiwa, H. Organic solvent stable-tolerant bacterium which secretes an organic solvent-stable lipolytic enzyme. Appl. Environ. Microbiol 1994, 60, 3884–3886. [Google Scholar]
- Yunus, J. Cloning and Expression of an organic solvent tolerant lipase from Staphylococcus epidermidis AT2. Master Thesis, Universiti Putra Malaysia, Serdang, Malaysia, 2009. [Google Scholar]
- Esakkiraj, P; Rajkumarbharathi, M; Palavesam, A; Immanuel, G. Lipase production by Staphylococcus epidermidis CMST-Pi 1 isolated from the gut of shrimp Penaeus indicus. Ann. Microbiol 2010, 60, 37–42. [Google Scholar]
- Longshaw, CM; Farrell, AM; Wright, JD; Holland, KT. Identification of a second lipase gene, gehD, in Staphylococcus epidermidis: comparison of a sequence with those of other staphylococcal lipases. Microbiology 2000, 146, 1419–1427. [Google Scholar]
- Rosenstein, R; Götz, F. Staphylococcal lipases: Biochemical and molecular characterization. J. Biochem 2000, 82, 1005–1014. [Google Scholar]
- Liebl, W; Götz, F. Studies on lipase directed export of E. coli β-lactamse in Staphylococcus carnosus. Mol. Gen. Genet 1986, 204, 166–173. [Google Scholar]
- Demleitner, G; Götz, F. Evidence for importance of the Staphylococcus hyicus lipase pro-peptide in lipase secretion, stability and activity. FEMS Microbiol. Lett 1994, 121, 189–197. [Google Scholar]
- Rahman, RNZRA; Baharum, SN; Basri, M; Salleh, AB. High-yield purification of an organic solvent-tolerant lipase from Pseudomonas sp. Strain S5. Anal. Biochem 2005, 341, 267–274. [Google Scholar]
- Yusoff, N. Purification and characterization of organic solvent tolerant protease from Pseudomonas aeroginosa strain K. Master Thesis, Universiti Putra Malaysia, Serdang, Malaysia, 2007. [Google Scholar]
- Oh, BC; Kim, HK; Lee, JK; Kang, SC; Oh, TK. Staphylococcus haemolyticus lipase: biochemical properties, substrate specificity and gene cloning. FEMS Microbiol. Lett 1999, 179, 385–392. [Google Scholar]
- Drouault, S; Corthier, G; Ehrlich, SD; Renault, P. Expression of the Staphylococcus hyicus lipase in Lactococcus lactis. Appl. Environ. Microbiol 2000, 66, 588–598. [Google Scholar]
- Laane, C; Boeren, S; Vos, K; Veeger, C. Rules for the optimization of biocatalysis in organic solvents. Biotechnol. Bioeng 1987, 30, 81–87. [Google Scholar]
- Sulong, MR; Rahman, RNZRA; Salleh, AB; Basri, M. A novel organic solvent tolerant lipase from Bacillus sphaericus 205y: Extracellular expression of a novel OST-lipase gene. Protein Express. Pur 2006, 49, 190–195. [Google Scholar]
- Ogino, H; Miyamoto, K; Yasuda, M; Ishimi, K; Ishikawa, H. Growth of organic solventtolerant Pseudomonas aeruginosa LST-03 in the presence of various organic solvents and production of lipolytic enzymes in the presence of cyclohexane. Biochem. Eng. J 1999, 4, 1–6. [Google Scholar]
- Kamini, NR; Fujii, T; Kurosu, T; Iefuji, H. Production, purification and characterization of an extracellular lipase from the yeast, Cryptocococcus sp. S-2. Proc. Biochem 2000, 36, 317–324. [Google Scholar]
- Hun, CJ; Rahman, RNZA; Salleh, AB; Basri, M. A newly isolated organic solvent tolerant Bacillus spahericus 205y producing organic solvent-stable lipase. Biochem. Eng. J 2003, 15, 147–151. [Google Scholar]
- Zhang, A; Gao, R; Diao, N; Xie, G; Gao, G; Cao, S. Cloning, expression and characterization of an organic solvent tolerant from Pseudomonas fluorencens JCM5963. J. Mol. Catal. B: Enzym 2009, 56, 78–84. [Google Scholar]
- Doukyu, N; Ogino, H. Organic solvent-tolerant enzymes. Biochem. Eng. J 2010, 48, 270–282. [Google Scholar]
- Ramos, JL; Duque, E; Gallegos, MT; Godoy, P; Ramos-Gonzalez, MI; Rojas, A; Terán, W; Segura, A. Mechanism of solvent tolerance in Gram negative bacteria. Annu. Rev. Microbiol 2002, 56, 743–768. [Google Scholar]
- Fang, Y; Lu, Z; Lv, F; Bie, X; Liu, S; Ding, Z; Xu, W. A newly isolated organic solvent tolerant Staphylococcus saprophyticus M36 Produced organic solvent stable lipase. Curr. Microbiol 2006, 53, 510–515. [Google Scholar]
- Rúa, ML; Díaz-Mauriño, T; Fernández, VM; Otero, C; Ballesteros, A. Purification and characterization of two distinct lipases from Candida cylindracea. Biochim. Biophys. Acta 1993, 1156, 181–189. [Google Scholar]
- Adlercreutz, P. Koskinen, AMP, Klibanov, AM, Eds.; Modes of using enzymes in organic media. In Enzymatic Reactions in Organic Media; Chapman & Hall: London, UK, 1996; pp. 9–37. [Google Scholar]
- Geok, LP; Razak, CNA; Rahman, RNZA; Basri, M; Salleh, AB. Isolation and screening of an extracellular organic solvent tolerant protease producer. Biochem. Eng. J 2003, 13, 73–77. [Google Scholar]
- Ghatorae, AS; Bell, G; Halling, PJ. Inactivation of enzymes by organic solvents: new technique with well-defined interfacial area. Biotechnol. Bioeng 1994, 43, 331–336. [Google Scholar]
- Sambrook, J; Fritsch, EF; Maniatis, T. Molecular Cloning: A Laboratory Manual, 2nd ed; Cold Spring Harbor Laboratory: New York, NY, USA, 1989. [Google Scholar]
- Kwon, DY; Rhee, JS. A simple and rapid colorimetric method for determination of free fatty acid for lipase assay. J. Am. Oil Chem. Soc 1986, 63, 82–89. [Google Scholar]
- Laemmli, UK. Most commonly used discontinuous buffer system for SDS electrophoresis. Nature 1970, 227, 680–686. [Google Scholar]
- Sellek, GA; Chaudhuri, JB. Biocatalysis in organic media using enzymes from extremophiles. Enzyme Microb. Technol 1999, 25, 471–482. [Google Scholar]






| Solvent | Log P* | Relative activity (%) |
|---|---|---|
| Control | - | 100 |
| Dimethylsulfoxide | −1.3 | 112 |
| Methanol | −0.76 | 1.2 |
| Acetonitrile | −0.33 | 0 |
| Ethanol | −0.24 | 0 |
| Acetone | −0.23 | 0 |
| Diethylether | 0.68 | 0 |
| Ethylacetate | 0.68 | 15 |
| Chloroform | 2.0 | 14 |
| Benzene | 2.0 | 56 |
| Toluene | 2.5 | 230 |
| Octanol | 2.9 | 295 |
| Ethylbenzene | 3.1 | 58 |
| p-xylene | 3.1 | 190 |
| n-hexane | 3.5 | 154 |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Rahman, R.N.Z.R.A.; Kamarudin, N.H.A.; Yunus, J.; Salleh, A.B.; Basri, M. Expression of an Organic Solvent Stable Lipase from Staphylococcus epidermidis AT2. Int. J. Mol. Sci. 2010, 11, 3195-3208. https://doi.org/10.3390/ijms11093195
Rahman RNZRA, Kamarudin NHA, Yunus J, Salleh AB, Basri M. Expression of an Organic Solvent Stable Lipase from Staphylococcus epidermidis AT2. International Journal of Molecular Sciences. 2010; 11(9):3195-3208. https://doi.org/10.3390/ijms11093195
Chicago/Turabian StyleRahman, Raja Noor Zaliha Raja Abd., Nor Hafizah Ahmad Kamarudin, Jalimah Yunus, Abu Bakar Salleh, and Mahiran Basri. 2010. "Expression of an Organic Solvent Stable Lipase from Staphylococcus epidermidis AT2" International Journal of Molecular Sciences 11, no. 9: 3195-3208. https://doi.org/10.3390/ijms11093195
APA StyleRahman, R. N. Z. R. A., Kamarudin, N. H. A., Yunus, J., Salleh, A. B., & Basri, M. (2010). Expression of an Organic Solvent Stable Lipase from Staphylococcus epidermidis AT2. International Journal of Molecular Sciences, 11(9), 3195-3208. https://doi.org/10.3390/ijms11093195




