Effect of Mobile Phase Additives on the Resolution of Four Bioactive Compounds by RP-HPLC
Abstract
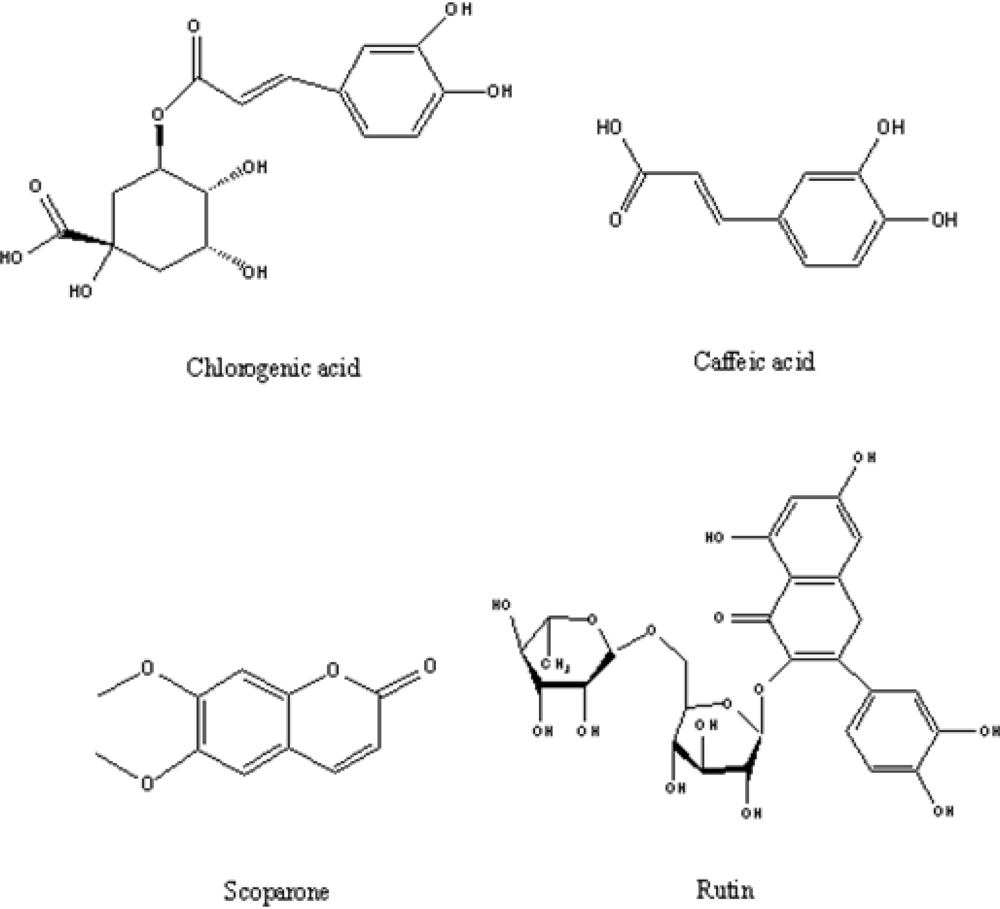
:1. Introduction
2. Results and Discussion
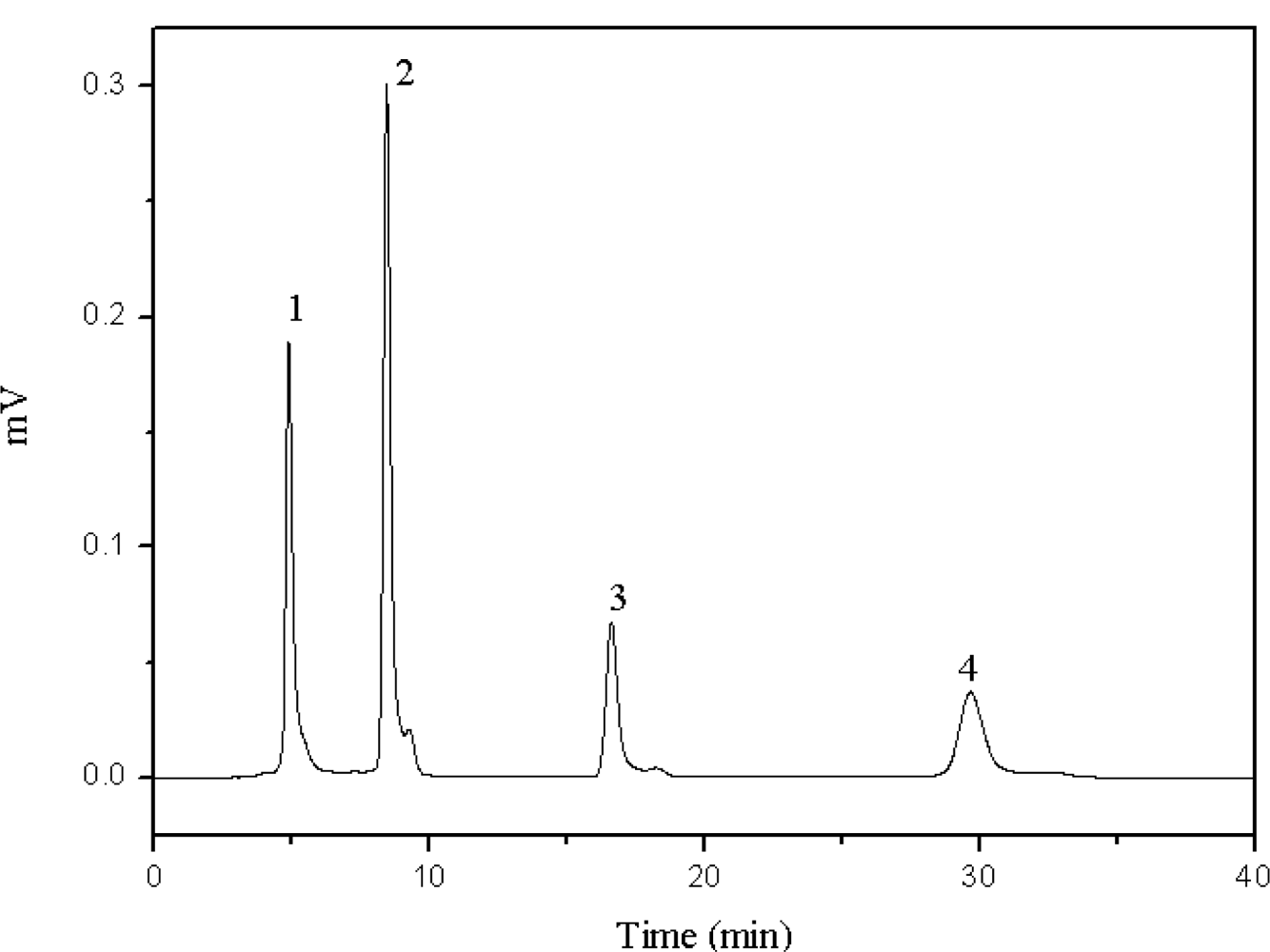
2.1. Retention Factor Estimation
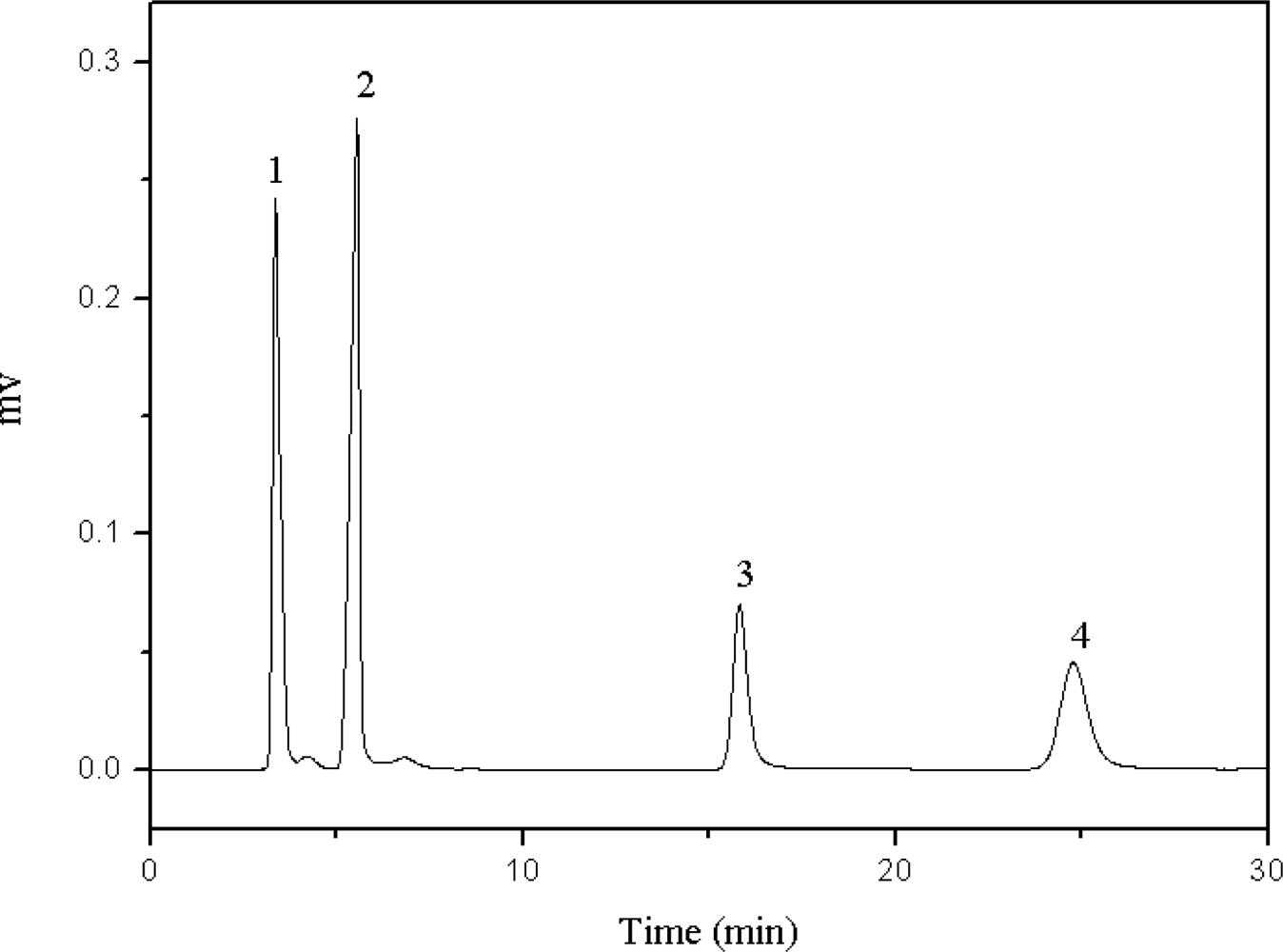
2.2. Effect of Acid and Basic Additives on the Resolution
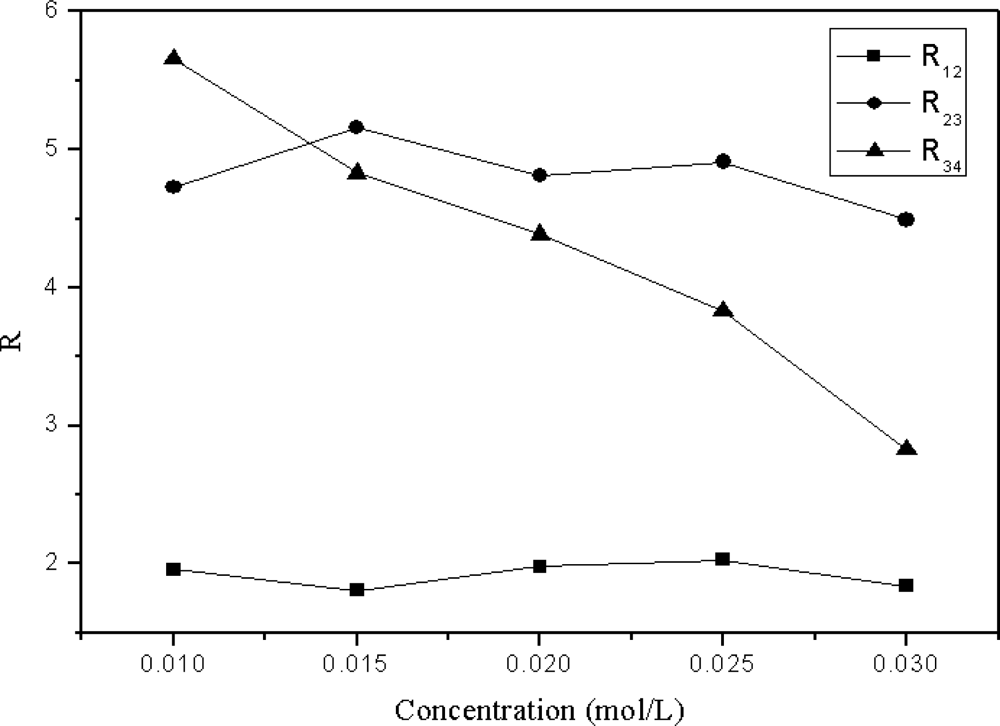
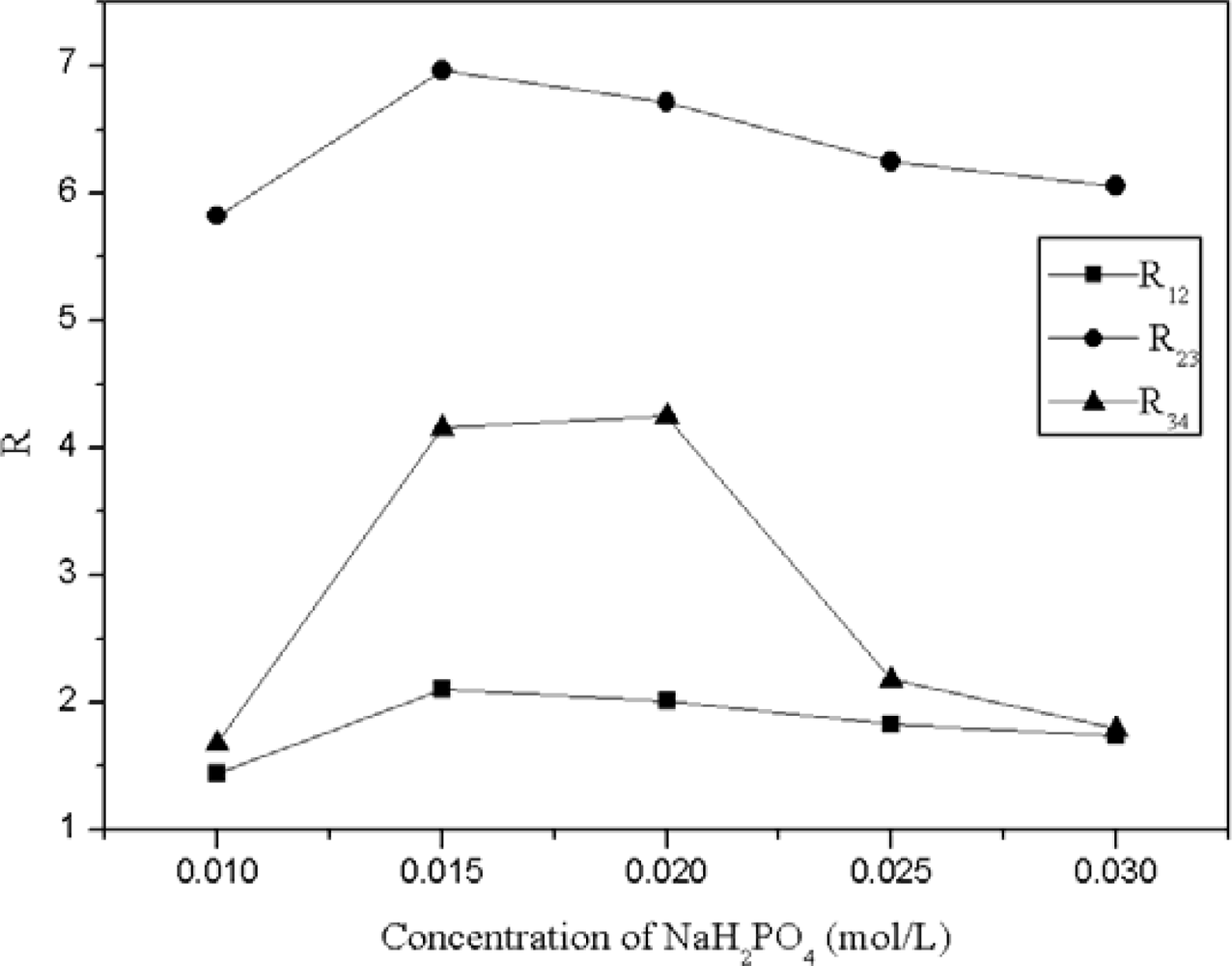
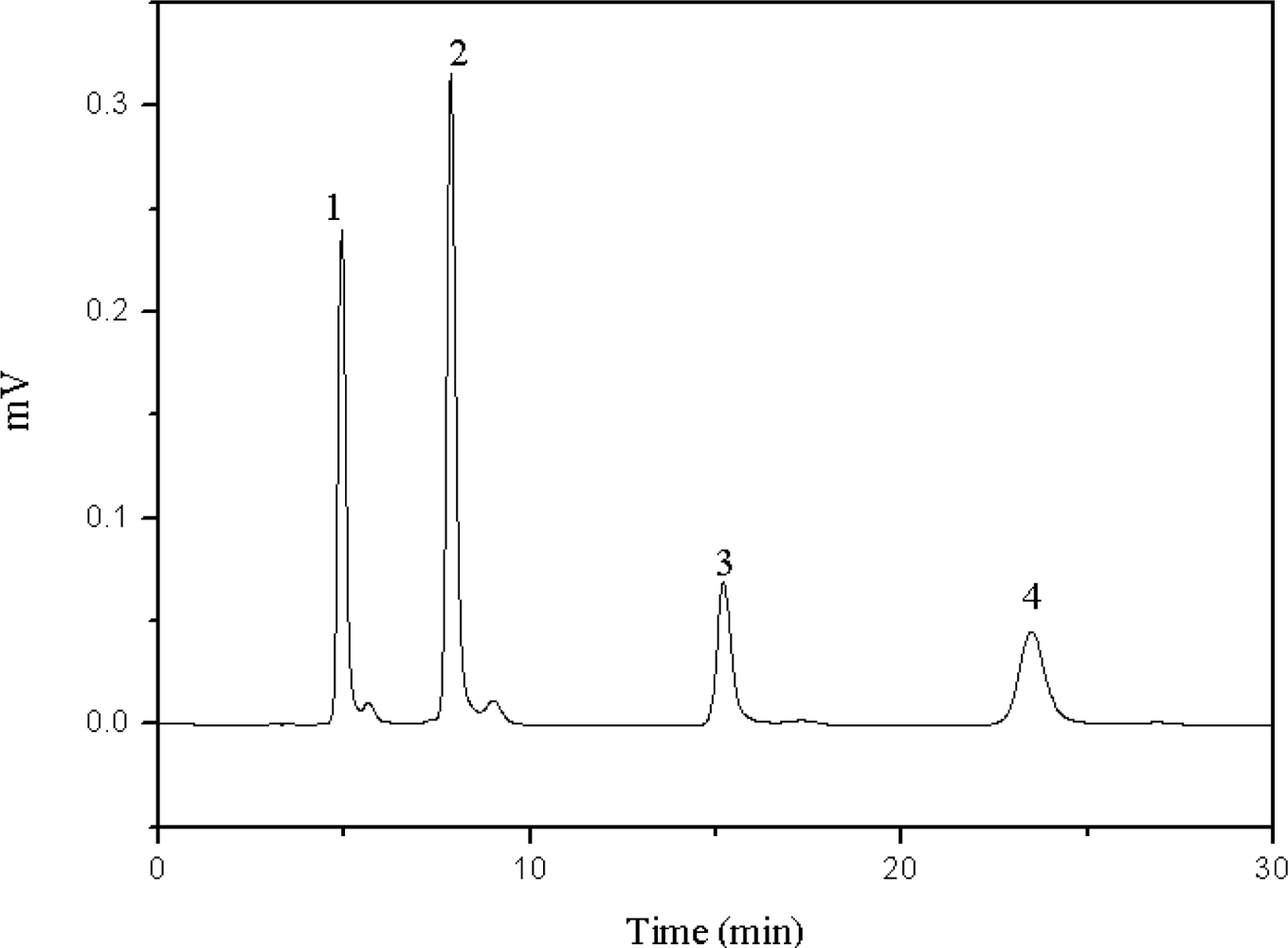
2.3. Effect of the Concentration of Inorganic Salt
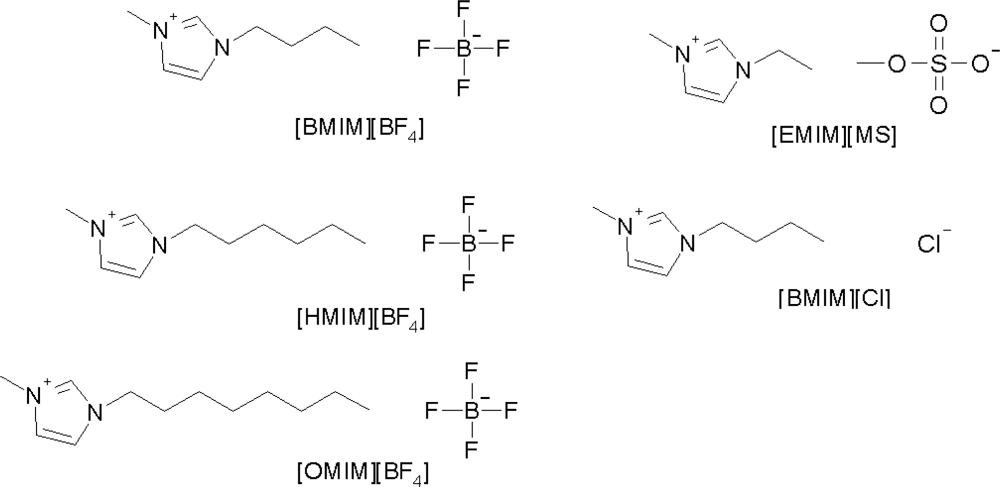
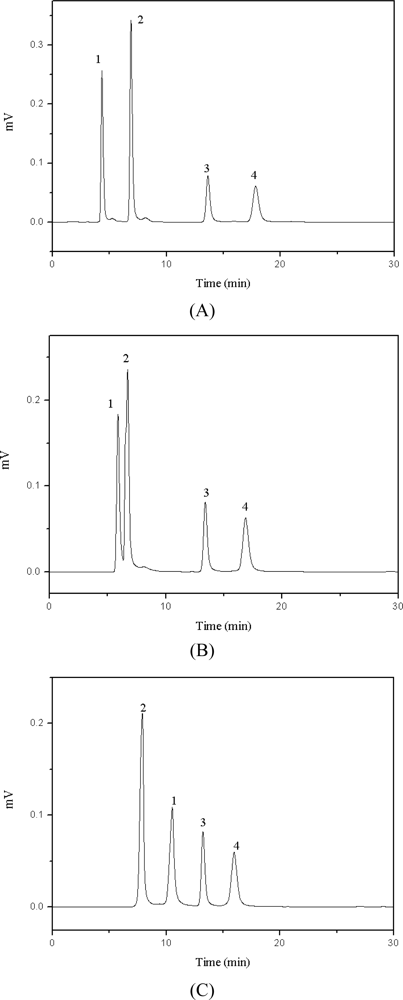
2.4. Effect of Ionic Liquid on the Retention and Resolution of Target Compounds
2.4.1. Different Cations of Ionic Liquid
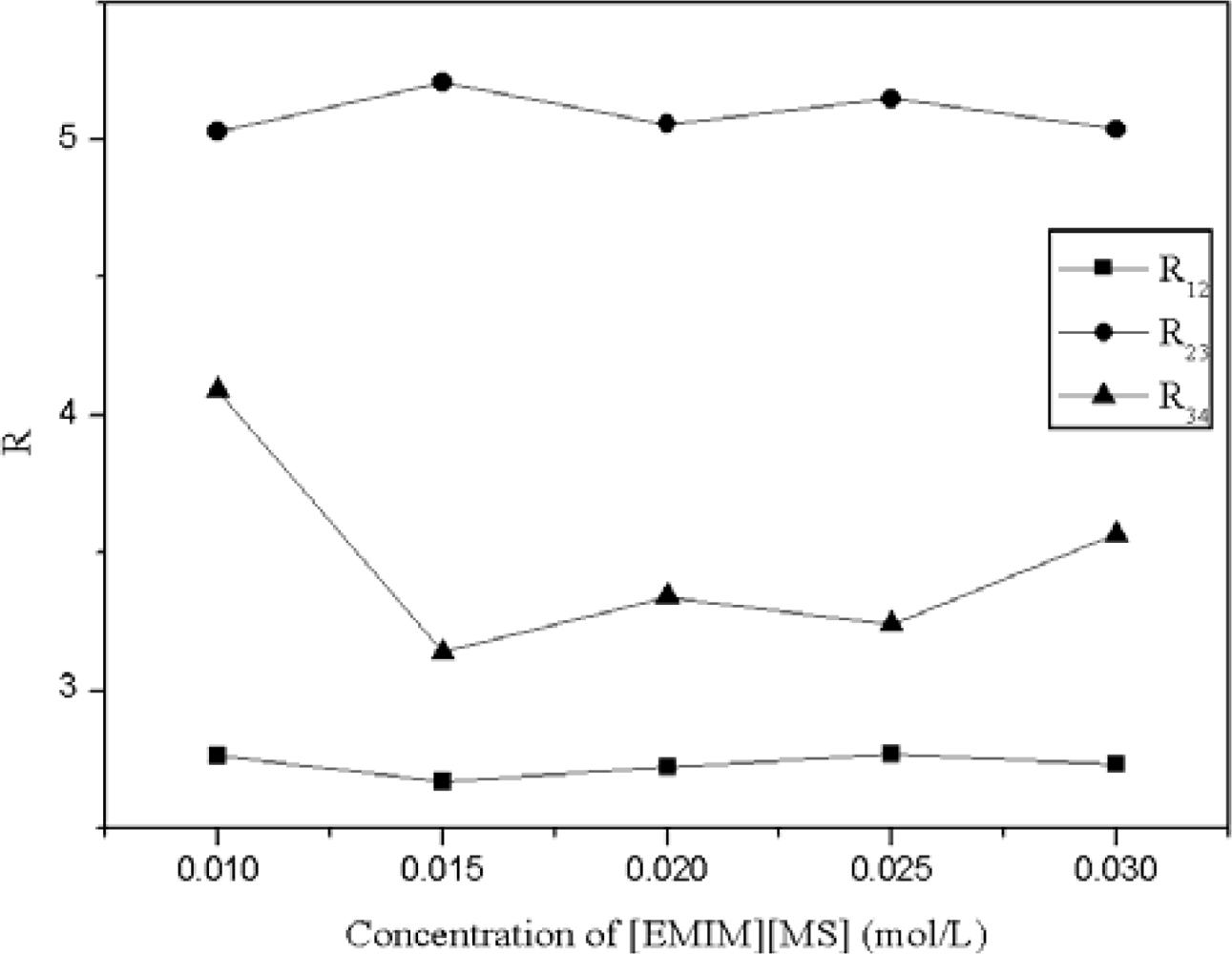
2.4.2. Effect of Anion [MS] of Ionic Liquid
2.4.3. Effect of Anion of Ionic Liquid
3. Experimental Section
3.1. Materials and Reagents
3.2. Preparation of Mobile Phases and Standard Solutions
3.3. Apparatus and Chromatographic Conditions
4. Conclusions
Acknowledgments
References and Notes
- Han, D; Row, KH. Determination of four bioactive compounds in Herba artemisiae scopariae. Asian J. Chem 2010, 22, 2711–2716. [Google Scholar]
- Cha, JD; Jeong, MR; Jeong, SI. Chemical composition and antimicrobial activity of the essential oils of Artemisia scoparia and A. capillaries. Planta Med 2005, 71, 186–190. [Google Scholar]
- Niggeweg, R; Michael, AJ; Martin, C. Increasing chlorogenic acid in tomato, an important antiocxidant for plants and animals. Nat. Biotechnol 2004, 22, 746–754. [Google Scholar]
- Ramezani, M; Fazli-Bazzaz, BS; Saghafi-Khadem, F. Antimicrobial activity of four Artemisia species of Iran. Fitoterapia 2004, 75, 201–203. [Google Scholar]
- Zhang, QW; Zhang, YX; Zhang, Y. Studies on chemical constituents in buds of Artemisia scoparia. J. Chin. Mater. Med 2002, 27, 202–204. [Google Scholar]
- Song, XJ; Zhang, YX; Zhang, Y; Zhang, QW. Methods for qualitative and quantitative determination of chlorogenic acid in commercial Herba Artemisia Scoparia. J. Chin. Mater. Med 2002, 27, 267–269. [Google Scholar]
- Lin, S; Zhang, Q; Zhang, N; Zhang, Y. Determination of flavonoids in buds of Herba Artemisiae Scopariae by HPLC. J. Chin. Mater. Med 2005, 30, 591–594. [Google Scholar]
- Zheng, J; Row, KH. Optimum of mobile phase condition for resolving isoflavones in RP-HPLC. Chin. J. Chem. Eng 2007, 15, 291–295. [Google Scholar]
- Jin, CH; Koo, YM; Choi, DK; Row, KH. Effect of mobile phase additives on resolution of some nucleic compounds in high performance liquid chromatography. Biotechnol. Bioprocess Eng 2007, 12, 525–530. [Google Scholar]
- Han, D; Tian, M; Row, KH. Isolation of four compounds from Herba Artemisiae Scopariae by preparative column HPLC. J. Liq. Chromatogr. Related Tech 2009, 32, 2407–2416. [Google Scholar]
- Wu, Y; Wang, Z; Gu, J; Wang, Y. Determination of acetonitrile volume fraction in mobile phase by HPLC. Chem Res Chin Univ 2008, 24(6), 694–696. [Google Scholar]
- Ma, C; Wang, L; Yan, Y; Che, G; Yin, Y; Wang, R; Li, D. Extraction of tetracycline via ionic liquid two-phase system. Chem Res Chin Univ 2009, 25(6), 832–835. [Google Scholar]
- Zheng, J; Row, KH. Separation of guanine and hypoxanthine with some ionic liquids in RP-HPLC. Am. J. Appl. Sci 2006, 3, 2160–2166. [Google Scholar]
- Tian, M; Liu, J; Row, KH. Effect of ionic liquids as the mobile phase additives on the HPLC resolution of four active compounds from Sophora Flavescens Ait. Mol 2009, 14, 2127–2134. [Google Scholar]
- Wang, Y; Row, KH. Effect of 1-butyl-3-methylimidazolium-based ionic liquid on the resolution of three components in Herba Artemisiae Scopariae by RP-HPLC. J. Indian Chem. Soc 2009, 10, 1082–1086. [Google Scholar]
- Polyakova, Y; Jin, Y; Zheng, J; Row, KH. Effect of concentration of ionic liquid 1-butyl-3-methylimidazolium tetrafuoroborate for retention and separation of some amino and nucleic acids. J. Liq. Chromatogr. Related Tech 2006, 29, 1687–1701. [Google Scholar]
- Zhang, W; He, L; Gu, Y; Liu, X; Jiang, S. Effect of ionic liquids as mobile phase additives on retention of catecholamines in reversed-phase high-performance liquid chromatography. Anal. Lett 2003, 36, 827–838. [Google Scholar]
- Pan, L; LoBrutto, R; Kazakevich, YV; Thompson, R. Influence of inorganic mobile phase additives on the retention, efficiency and peak symmetry of protonated basic compounds in reversed-phase liquid chromatography. J. Chromatogr. A 2004, 1049, 63–73. [Google Scholar]
- Xiao, X; Zhao, L; Liu, X; Jiang, S. Ionic liquids as additives in high performance liquid chromatography: Analysis of amines and the interaction mechanism of ionic liquids. Anal. Chim. Acta 2004, 519, 207–211. [Google Scholar]
- Jiang, Q; Qiu, H; Wang, X; Liu, X; Zhang, S. Effect of ionic liquids as additives on the separation of bases and amino acids in HPLC. J. Liq. Chromatogr. Related Tech 2008, 31, 1448–1457. [Google Scholar]
- Zheng, J; Row, KH. Effects of ionic liquid on the separation of 2-chlorophenol and 2,4,6-trichlorophenol in RP-HPLC. J. Chromatogr. Sci 2009, 47, 392–395. [Google Scholar]
- Tan, XJ; Li, Q; Chen, XH. Simultaneous determination of 13 bioactive compounds in Herba Artemisiae Scopariae (Yin Chen) from different harvest seasons by HPLC-DAD. J. Pharm. Biomed. Anal 2008, 47, 847–853. [Google Scholar]
- Yao, X; Chen, G. Simultaneous determination of p-hydroxyacetophenone, chlorogenic acid, and caffeic acid in Herba Artemisiae Scopariae by capillary electrophoresis with electrochemical detection. Anal. Bioanal. Chem 2007, 388, 475–481. [Google Scholar]
- Zhang, QW; Zhang, YX; Zhang, Y; Liang, GG. Determination of cirsilineol in Herba Artemisiae scopariae collected in autumn by HPLC. J. Chin. Mater. Med 2002, 27, 23–25. [Google Scholar]
- Zhang, T; Chen, DF. Anticomplementary principles of a Chinese multiherb remedy for the treatment and prevention of SARS. J. Ethnopharmacol 2008, 117, 351–361. [Google Scholar]
- Zheng, J; Polyakova, Y; Row, KH. Effects of ionic liquid as additive and pH of mobile phase on retention factors of amino benzoic acids in reversed-phase high performance liquid chromatography. J. Chromatogr. Sci 2007, 45, 256–262. [Google Scholar]
- Wang, B; Tang, Y; Wen, Z; Wang, H. Dissolution and regeneration of polybenzimidazoles using ionic liquids. Eur. Polym. J 2009, 45, 2962–2965. [Google Scholar]
| Concentration (mol/L) | Compound | k (error, %) | |||
|---|---|---|---|---|---|
| Acetic acid | Triethylamine | NaH2PO4 | Na2HPO4 | ||
| 0.01 | CGA | 1.61 (0.62) | 0.12 (0.05) | 0.66 (0.60) | 0.51 (0.06) |
| CA | 3.51 (0.52) | 1.51 (0.57) | |||
| SC | 7.88 (0.63) | 5.83 (0.42) | 6.32 (0.6) | 7.49 (0.73) | |
| Rutin | 14.86 (1.82) | 0.12 (0.05) | 7.99 (0.98) | 0.51 (0.06) | |
| 0.015 | CGA | 1.57 (0.60) | 0.12 (0.05) | 0.80 (0.67) | 0.53 (0.06) |
| CA | 3.34 (0.81) | 1.96 (0.49) | |||
| SC | 7.53 (0.65) | 5.53 (0.43) | 7.45 (0.86) | 6.88 (0.68) | |
| Rutin | 13.24 (1.74) | 0.12 (0.05) | 12.24 (1.36) | 0.53 (0.06) | |
| 0.02 | CGA | 1.55 (0.62) | 0.12 (0.05) | 0.84 (0.67) | 0.56 (0.06) |
| CA | 3.25 (0.55) | 2.09 (0.58) | |||
| SC | 7.35 (0.62) | 4.85 (0.42) | 7.59 (0.90) | 6.63 (0.70) | |
| Rutin | 12.39 (1.25) | 0.12 (0.05) | 12.63 (0.99) | 0.56 (0.06) | |
| 0.025 | CGA | 1.51 (0.61) | 0.12 (0.05) | 0.81 (0.60) | 0.58 (0.063) |
| CA | 3.12 (0.56) | 1.86 (0.54) | |||
| SC | 7.11 (0.66) | 4.59 (0.40) | 6.81 (0.58) | 6.59 (0.66) | |
| Rutin | 11.44 (1.12) | 0.12 (0.05) | 9.64 (0.87) | 0.58 (0.06) | |
| 0.03 | CGA | 1.49 (0.43) | 0.12 (0.05) | 0.82 (0.60) | 0.60 (0.07) |
| CA | 2.99 (0.47) | 1.81 (0.56) | |||
| SC | 6.85 (0.58) | 4.30 (0.40) | 6.58 (0.74) | 7.93 (0.82) | |
| Rutin | 10.20 (1.10) | 0.12 (0.05) | 8.49 (0.86) | 1.03 (0.83) | |
| Concentration (mol/L) | Compound | k (error, %) | ||||
|---|---|---|---|---|---|---|
| [BMIM][BF4] | [HMIM][BF4] | [OMIM][BF4] | [EMIM][MS] | [BMIM][Cl] | ||
| 0.01 | CGA | 1.20 (0.75) | 2.12 (0.80) | 4.63 (1.00) | 1.63 (0.80) | 0.94 (1.50) |
| CA | 2.67 (0.73) | 2.54 (0.69) | 3.22 (0.83) | 3.22 (0.83) | ||
| SC | 6.51 (0.72) | 6.07 (0.66) | 6.08 (0.82) | 7.16 (0.81) | 6.13 (0.91) | |
| Rutin | 9.41 (1.20) | 7.74 (1.18) | 7.54 (1.18) | 11.66 (1.09) | 7.03 (0.96) | |
| 0.015 | CGA | 1.29 (0.75) | 2.18 (0.80) | 4.47 (0.95) | 1.57 (0.80) | 0.98 (1.50) |
| CA | 2.86 (0.73) | 2.95 (0.69) | 3.28 (0.83) | 3.01 (0.83) | ||
| SC | 6.76 (0.74) | 6.71 (0.69) | 6.38 (0.82) | 6.95 (0.79) | 6.60 (0.77) | |
| Rutin | 10.22 (1.23) | 9.72 (1.16) | 8.00 (1.21) | 10.28 (1.10) | 8.06 (0.99) | |
| 0.02 | CGA | 1.30 (0.75) | 2.28 (0.79) | 4.20 (0.86) | 1.55 (0.80) | 0.93 (1.40) |
| CA | 2.91 (0.72) | 2.90 (0.72) | 3.29 (0.88) | 2.98 (0.70) | ||
| SC | 6.74 (0.70) | 6.73 (0.70) | 6.50 (0.67) | 6.72 (0.72) | 4.65 (0.58) | |
| Rutin | 10.17 (1.18) | 9.63 (1.17) | 8.15 (1.09) | 10.13 (1.05) | 6.12 (0.87) | |
| 0.025 | CGA | 1.21 (0.72) | 2.29 (0.79) | 5.09 (0.92) | 1.52 (0.80) | 0.93 (1.40) |
| CA | 2.43 (0.66) | 2.84 (0.71) | 3.50 (0.89) | 2.94 (0.70) | ||
| SC | 5.88 (0.58) | 6.61 (0.68) | 6.70 (0.68) | 6.63 (0.67) | 5.47 (0.92) | |
| Rutin | 7.21 (0.99) | 9.11 (0.98) | 9.50 (1.18) | 9.90 (1.13) | 6.43 (0.83) | |
| 0.03 | CGA | 1.33 (0.75) | 2.32 (0.79) | 4.43 (0.86) | 1.56 (0.81) | 0.93 (1.30) |
| CA | 2.68 (0.66) | 2.94 (0.71) | 3.32 (0.88) | 3.04 (0.72) | ||
| SC | 6.30 (0.67) | 6.64 (0.68) | 6.48 (0.66) | 6.80 (0.65) | 3.93 (0.47) | |
| Rutin | 8.54 (1.08) | 8.63 (0.96) | 7.36 (0.91) | 10.57 (1.05) | 6.07 (0.78) | |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, S.; Tian, M.; Row, K.H. Effect of Mobile Phase Additives on the Resolution of Four Bioactive Compounds by RP-HPLC. Int. J. Mol. Sci. 2010, 11, 2229-2240. https://doi.org/10.3390/ijms11052229
Li S, Tian M, Row KH. Effect of Mobile Phase Additives on the Resolution of Four Bioactive Compounds by RP-HPLC. International Journal of Molecular Sciences. 2010; 11(5):2229-2240. https://doi.org/10.3390/ijms11052229
Chicago/Turabian StyleLi, Shengnan, Minglei Tian, and Kyung Ho Row. 2010. "Effect of Mobile Phase Additives on the Resolution of Four Bioactive Compounds by RP-HPLC" International Journal of Molecular Sciences 11, no. 5: 2229-2240. https://doi.org/10.3390/ijms11052229





