A Simple and Rapid Method for DNA Isolation from Xylophagous Insects
Abstract
:1. Introduction
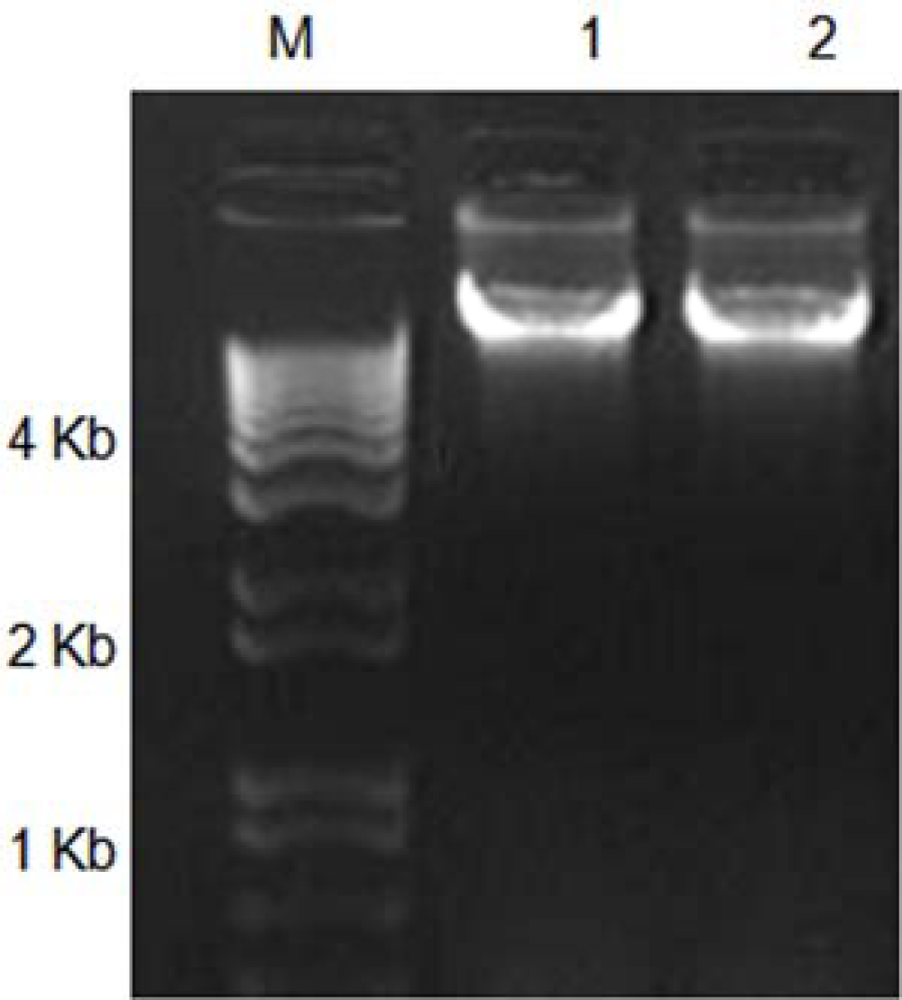
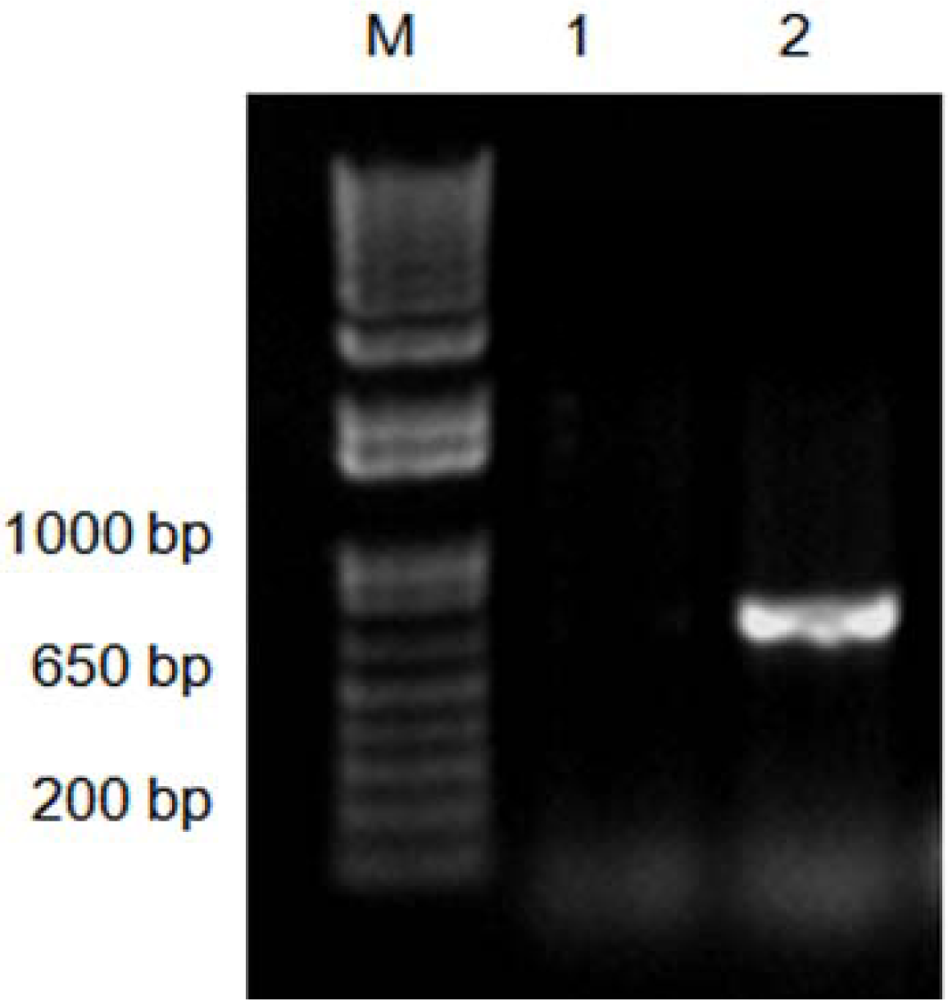
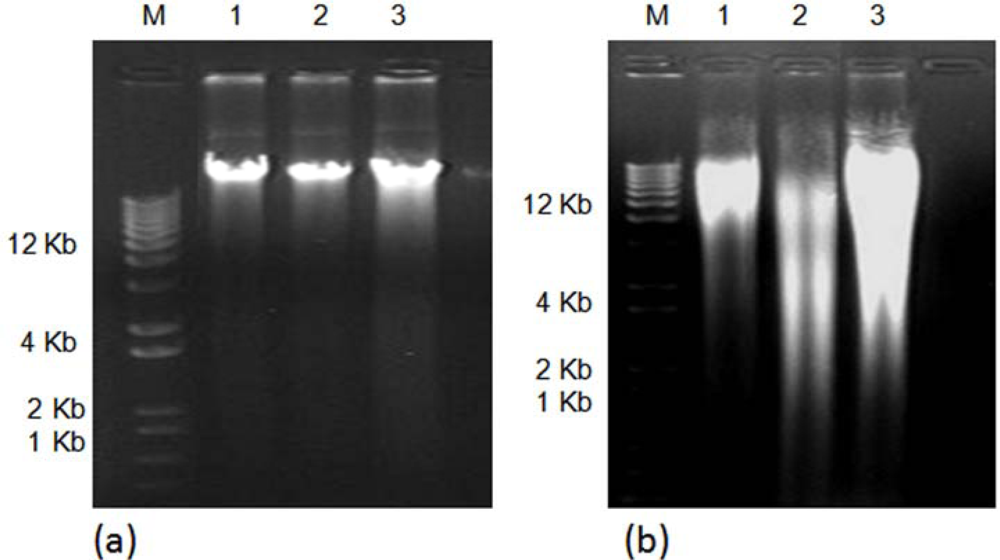
2. Results and Discussion
3. Experimental Section
3.1. DNA Isolation
3.2. Comparison of the Efficacy for the DNA Isolation Methods
3.3. Evaluation of the CTAB-PVP Modified Method in Other Xylophagous Species
4. Conclusions
Acknowledgments
References
- Amman, GD. The role of the mountain pine beetle in lodgepole pine ecosystems: impact on sucession. In The Role of Arthropods in Forest Ecosystems; Mattson, WJ, Ed.; Springer-Verlag: New York, NY, USA, 1976. [Google Scholar]
- Schowalter, TD. Insect herbivore relationship to the state of the host plant: biotic regulation of the ecosystem nutrient cycling through ecological succession. Oikos 1981, 37, 126–130. [Google Scholar]
- Feller, IC. The role of herbivory by wood-boring insects in mangrove ecosystems in Belize. Oikos 2002, 97, 167–176. [Google Scholar]
- Allison, JD; Borden, JH; Seybold, ST. A review of the chemical ecology of the Cerambycidae (Coleoptera). Chemoecology 2004, 14, 123–150. [Google Scholar]
- Knizek, M; Beave, R. Taxonomy and systematic of bark and Ambrosia beetles. In Bark and Wood-Boring Insects in Living Trees in Europe, a Synthesis; Lieutier, F, Day, KR, Battisti, A, Grégoire, JC, Evans, H, Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2004. [Google Scholar]
- Caterino, MS; Cho, S; Sperling, FAH. The current state of insect molecular systematics: a thriving tower of Babel. Annu. Rev. Entomol 2000, 45, 1–54. [Google Scholar]
- Brockerhoff, EG; Liebhold, AM; Jactel, H. The ecology of forest insect invasions and advances in their management. Can. J. Forest Res 2006, 36, 263–268. [Google Scholar]
- Strauss, SY; Zangerl, AR. Plant-insect interactions in terrestrial ecosystems. In Plant-Animal Interactions: An Evolutionary Approach; Herrera, CM, Pellmyr, O, Eds.; Blackwell Publishing: Oxford, UK, 2002. [Google Scholar]
- Chown, SL; Nicolson, SW. Insect Physiological Ecology; Oxford University Press: Oxford, UK, 2004. [Google Scholar]
- Henry, JM; Raina, AK; Ridgway, RL. Isolation of High Molecular-weight DNA from insects. Anal. Res 1990, 185, 147–150. [Google Scholar]
- Aljanabi, SM; Martinez, I. Universal and rapid salt-extraction of high quality genomic DNA for PCR-based techniques. Nucleic Acids Res 1997, 25, 4692–4693. [Google Scholar]
- Reineke, A; Karlovsky, P; Zebitz, PW. Preparation and purification of DNA from insects for AFLP analysis. Insect Mol. Biol 1998, 7, 95–99. [Google Scholar]
- Hill, CA; Gutierrez, JA. A method for extraction and analysis of high quality genomic DNA from ixodid ticks. Med. Vet. Entomol 2003, 17, 224–227. [Google Scholar]
- Juen, A; Traugott, M. Amplification facilitators and multiplex PCR: tools to overcome PCR-inhibition in DNA-gut-content analysis of soil-living invertebrates. Soil Biol. Biochem 2006, 38, 1872–1879. [Google Scholar]
- Stone, GN; Challis, RJ; Atkinson, RJ; Csóka, G; Hayward, A; Melika, G; Mutun, S; Preuss, S; Rokas, A; Sadeghi, E; Schönrogge, K. The phylogeographical clade trade: tracing the impact of human-mediated dispersal on the colonization of northern Europe by the oak gallwasp Andricus kollari. Mol. Ecol 2007, 16, 2768–2781. [Google Scholar]
- Ball, SL; Armstrong, KF. Rapid, one step DNA extraction for insect pest identification using barcodes. J. Econ. Entomol 2008, 101, 523–532. [Google Scholar]
- Serrano, MG; Nunes, LR; Campaner, M; Buck, GA; Camargo, EP. Trypanosomatidae: Phytomonas detection in plants and phytophagous insects by PCR amplification of a genus-specific sequence of the spliced leader gene. Exp. Parasitol 1999, 91, 268–279. [Google Scholar]
- Lodhi, MA; Ye, GN; Weeden, NF; Reisch, BI. A simple and efficient method for DNA extraction from gravepine cultivars, Vitis species and Ampelopsis. Plant Mol. Biol. Rep 1994, 12, 6–13. [Google Scholar]
- Zidani, S; Ferchichi, A; Chaieb, M. Genomic DNA extraction method from pearl millet (Pennisetum glaucum) Leaves. Afr. J. Biotechnol 2005, 4, 862–866. [Google Scholar]
- Couch, JA; Fritz, PJ. Isolation of DNA from plants high in polyphenolics. Plant Mol. Biol. Rep 1990, 8, 8–12. [Google Scholar]
- Kim, CS; Lee, CH; Shin, JS; Chung, YS; Hyung, NI. A simple and rapid method for isolation of high quality genomic DNA from fruit trees and conifers using PVP. Nucleic Acids Res 1997, 25, 1085–1086. [Google Scholar]
- Horne, EC; Kumpatla, SP; Patterson, MG; Thompson, SA. Improved high-throughput sunflower and cotton genomic DNA extraction and PCR fidelity. Plant Mol. Biol. Rep 2004, 22, 83a–83i. [Google Scholar]
- Friar, EA. Isolation of DNA from plants with large amounts of secondary metabolites. Method Enzymol 2005, 395, 3–14. [Google Scholar]
- Padmalatha, K; Prasad, MNV. Optimization of DNA isolation and PCR protocol for RAPD analysis of selected medicinal and aromatic plants of conservation concern from Peninsular India. Afr. J. Biotechnol 2006, 5, 230–234. [Google Scholar]
- Arif, IA; Bakir, MA; Khan, HA; Ahamed, A; Al Farhan, AH; Al Homaidan, AA; Al Sadoon, M; Bahkali, AH; Shobrak, M. A simple method for DNA extraction from mature date palm leaves: impact of sand grinding and composition of lysis buffer. Int. J. Mol. Sci 2010, 11, 3149–3157. [Google Scholar]
- Chen, DH; Ronald, PC. A rapid DNA minipreparation method suitable for AFLP and other PCR applications. Plant Mol. Biol. Rep 1999, 17, 53–57. [Google Scholar]
- Li, JT; Yang, J; Chen, DC; Zhang, XL; Tang, ZS. An optimized mini-preparation method to obtain high-quality genomic DNA from mature leaves of sunflower. Gen. Mol. Res 2007, 6, 1064–1071. [Google Scholar]
- Feeley, CJ; Hart, ER; Thompson, JR; Harrington, TC. Ocurrence, associated symptoms, and potential insect vectors of the ash yellows phytoplasma in Iowa, U.S. J. Arboric 2001, 27, 331–340. [Google Scholar]
- Doyle, JJ; Doyle, JL. Isolation of plant DNA from fresh tissue. Focus 1990, 12, 13–15. [Google Scholar]
- Puchooa, D; Venkatasamy, K. A protocol for the isolation of DNA from Trochetia boutoniana. Int. J. Agric. Biol 2005, 7, 82–85. [Google Scholar]
- Michiels, A; van den Ende, W; Tucker, M; van Riet, L; van Laere, A. Extraction of high-quality genomic DNA from latex-containing plants. Anal. Biochem 2003, 315, 85–89. [Google Scholar]
- Calderón, CN. Center for Ecosystems Research (CIEco), National Autonomous University of Mexico (UNAM), Morelia, Mich, Mexico. Unpublished work. 2010.


© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Calderón-Cortés, N.; Quesada, M.; Cano-Camacho, H.; Zavala-Páramo, G. A Simple and Rapid Method for DNA Isolation from Xylophagous Insects. Int. J. Mol. Sci. 2010, 11, 5056-5064. https://doi.org/10.3390/ijms11125056
Calderón-Cortés N, Quesada M, Cano-Camacho H, Zavala-Páramo G. A Simple and Rapid Method for DNA Isolation from Xylophagous Insects. International Journal of Molecular Sciences. 2010; 11(12):5056-5064. https://doi.org/10.3390/ijms11125056
Chicago/Turabian StyleCalderón-Cortés, Nancy, Mauricio Quesada, Horacio Cano-Camacho, and Guadalupe Zavala-Páramo. 2010. "A Simple and Rapid Method for DNA Isolation from Xylophagous Insects" International Journal of Molecular Sciences 11, no. 12: 5056-5064. https://doi.org/10.3390/ijms11125056



