Myricetin Protects Cells against Oxidative Stress-Induced Apoptosis via Regulation of PI3K/Akt and MAPK Signaling Pathways
Abstract
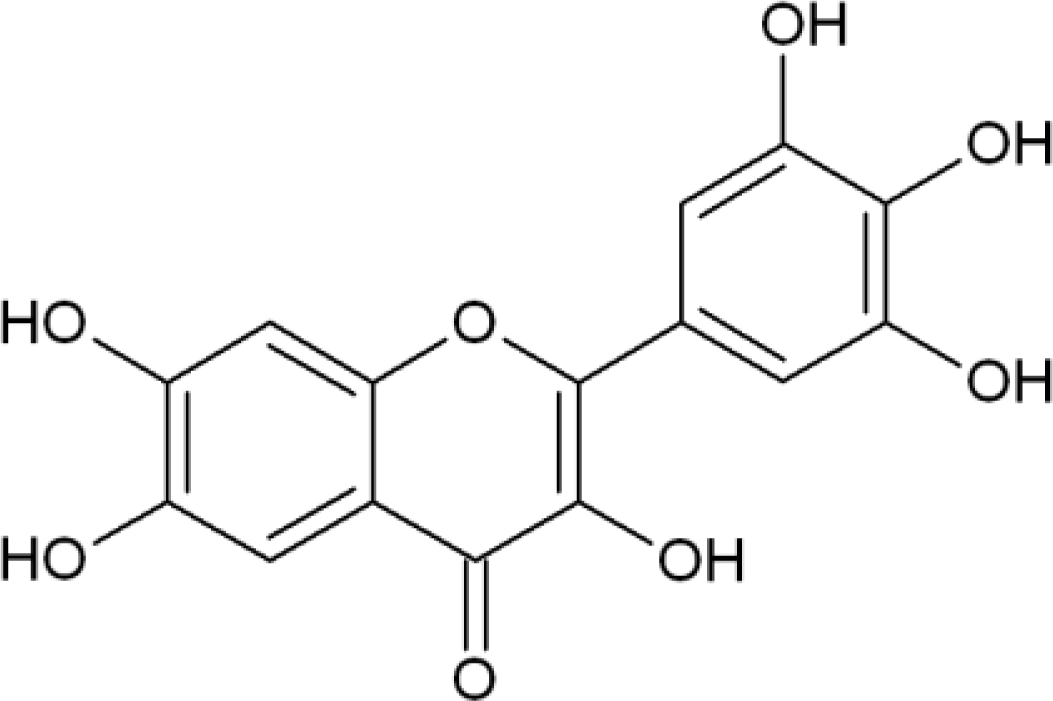
:1. Introduction
2. Results and Discussion
2.1. Myricetin Inhibits H2O2-Induced Cell Death
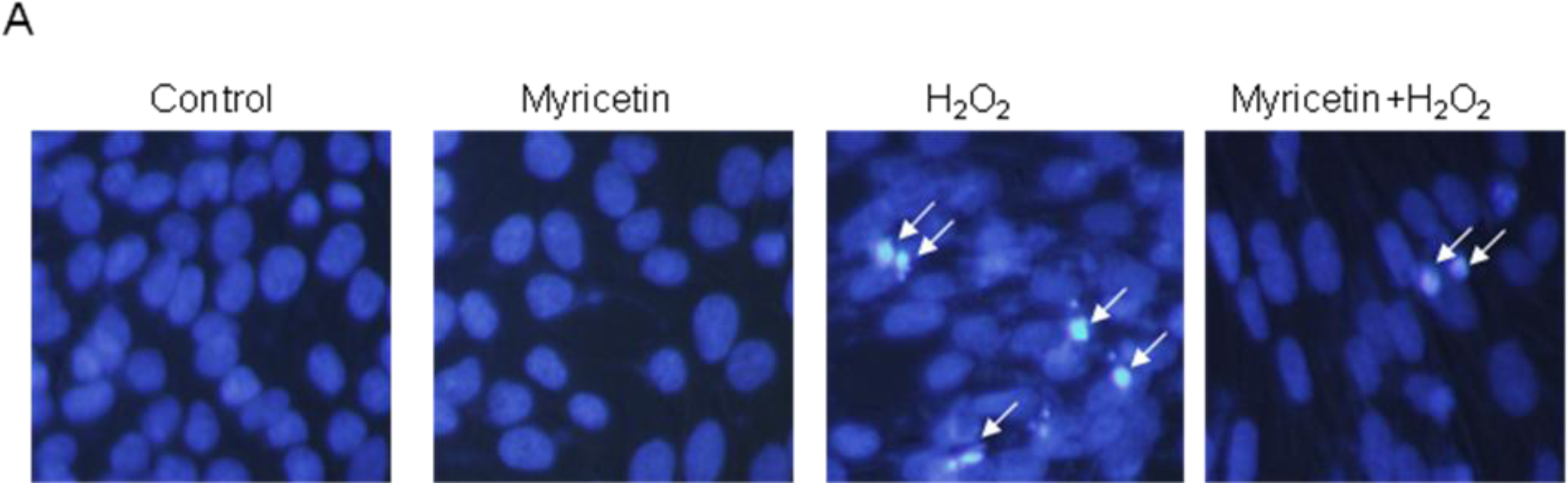
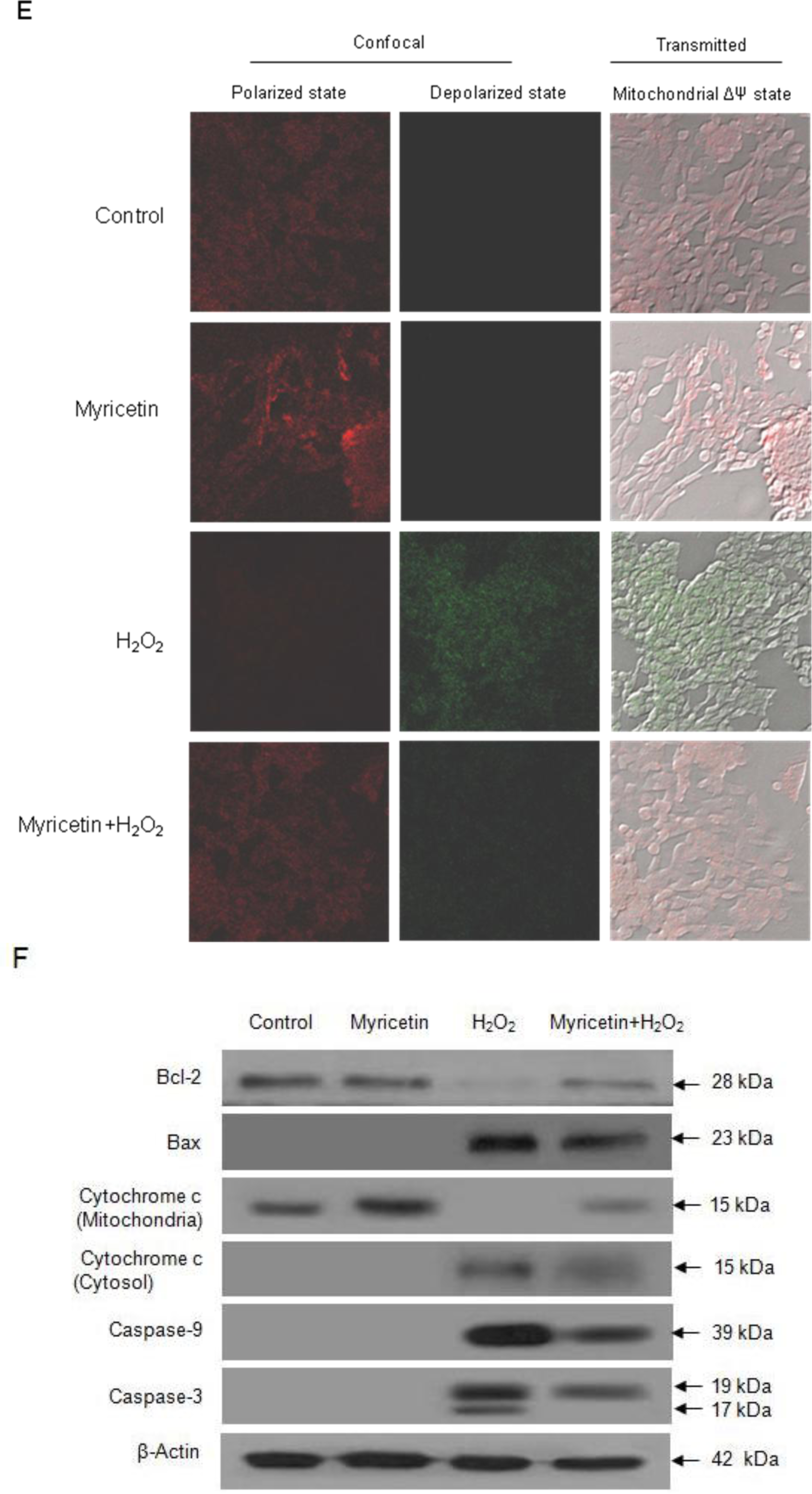
2.2. Protective Effect of Myricetin against Apoptosis Induced by H2O2
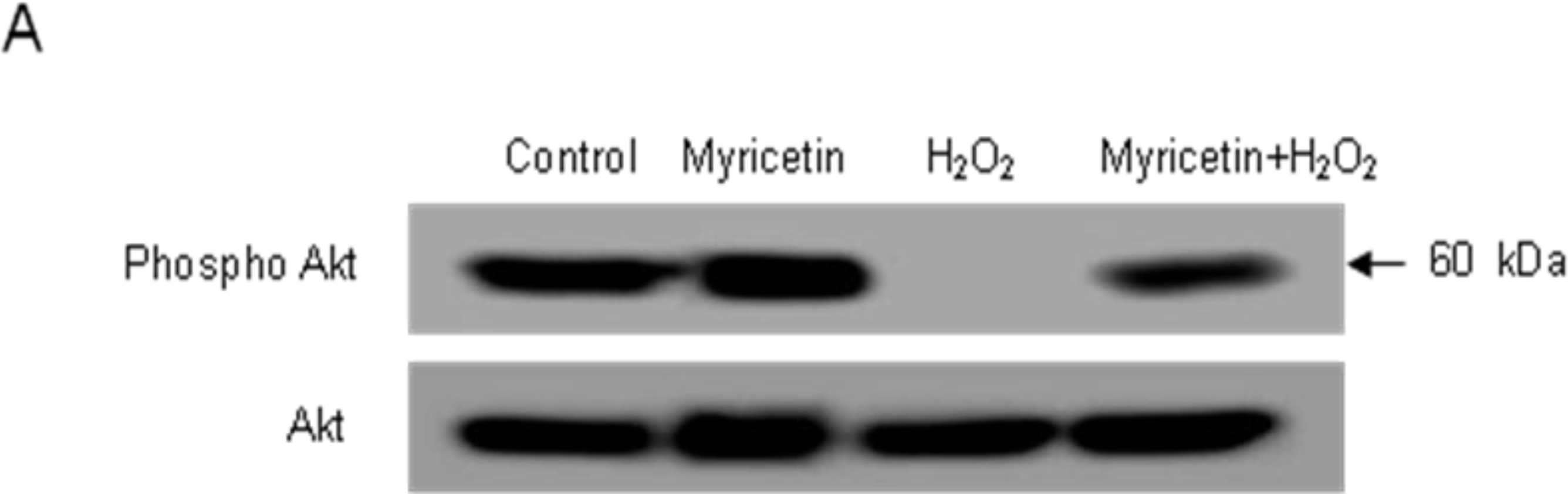
2.3. Involvement of PI3K/Akt and MAPKs in the Anti-apoptotic Effect of Myricetin from H2O2 Treatment
3. Conclusions
4. Experimental Section
4.1. Reagents
4.2. Cell Culture
4.3. Cell Viability
4.4. Nuclear Staining with Hoechst 33342
4.5. DNA Fragmentation
4.6. Flow Cytometry Analysis
4.7. Mitochondrial Membrane Potential (ΔΨ) Analysis
4.8. Western Blot
4.9. Statistical Analysis
Acknowledgments
References
- Halliwell, B; Gutteridge, JM. Free radicals, lipid peroxidation, and cell damage. Lancet 1984, 2, 1095–1100. [Google Scholar]
- Gutteridge, JM; Halliwell, B. Free radicals and antioxidants in the year 2000. A historical look to the future. Ann. N. Y. Acad. Sci 2000, 899, 136–147. [Google Scholar]
- Bergendi, L; Benes, L; Duracková, Z; Ferencik, M. Chemistry, physiology and pathology of free radicals. Life Sci 1999, 65, 1865–1874. [Google Scholar]
- Simon, HU; Haj-Yehia, A; Levi-Schaffer, F. Role of reactive oxygen species (ROS) in apoptosis induction. Apoptosis 2000, 5, 415–418. [Google Scholar]
- Maddika, S; Ande, SR; Panigrahi, S; Paranjothy, T; Weglarczyk, K; Zuse, A; Eshraghi, M; Manda, KD; Wiechec, E; Los, M. Cell survival, cell death and cell cycle pathways are interconnected: Implications for cancer therapy. Drug Resist. Updat 2007, 10, 13–29. [Google Scholar]
- Cantley, LC. The phosphoinositide 3-kinase pathway. Science 2000, 296, 1655–1657. [Google Scholar]
- Sussman, M. “AKT”ing lessons for stem cells: Regulation of cardiac myocyte and progenitor cell proliferation. Trends Cardiovasc. Med 2007, 17, 235–240. [Google Scholar]
- Wang, B; Shravah, J; Luo, H; Raedschelders, K; Chen, DD; Ansley, DM. Propofol protects against hydrogen peroxide-induced injury in cardiac H9c2 cells via Akt activation and Bcl-2 up-regulation. Biochem. Biophys. Res. Commun 2009, 389, 105–111. [Google Scholar]
- Kimura, R; Okouchi, M; Fujioka, H; Ichiyanagi, A; Ryuge, F; Mizuno, T; Imaeda, K; Okayama, N; Kamiya, Y; Asai, K; Joh, T. Glucagon-like peptide-1 (GLP-1) protects against methylglyoxal-induced PC12 cell apoptosis through the PI3K/Akt/mTOR/GCLc/redox signaling pathway. Neuroscience 2009, 162, 1212–1219. [Google Scholar]
- Matsuzawa, A; Ichijo, H. Stress-responsive protein kinases in redox-regulated apoptosis signaling. Antioxid. Redox. Signal 2005, 7, 472–481. [Google Scholar]
- Kyosseva, SV. Mitogen-activated protein kinase signaling. Int. Rev. Neurobiol 2004, 59, 201–220. [Google Scholar]
- Benhar, M; Dalyot, I; Engelberg, D; Levitzki, A. Enhanced ROS production in oncogenically transformed cells potentiates c-Jun N-terminal kinase and p38 mitogen-activated protein kinase activation and sensitization to genotoxic stress. Mol. Cell. Biol 2001, 21, 6913–6926. [Google Scholar]
- Ku, BM; Lee, YK; Jeong, JY; Mun, J; Han, JY; Roh, GS; Kim, HJ; Cho, GJ; Choi, WS; Yi, GS; Kang, SS. Ethanol-induced oxidative stress is mediated by p38 MAPK pathway in mouse hippocampal cells. Neurosci. Lett 2007, 419, 64–67. [Google Scholar]
- Yu, R; Lei, W; Mandlekar, S; Weber, MJ; Der, CJ; Wu, J; Kong, AN. Role of a mitogen-activated protein kinase pathway in the induction of phase II detoxifying enzymes by chemicals. J. Biol. Chem 1999, 274, 27545–27552. [Google Scholar]
- Yu, R; Mandlekar, S; Lei, W; Fahl, WE; Tan, TH; Kong, AN. p38 mitogen-activated protein kinase negatively regulates the induction of phase II drug-metabolizing enzymes that detoxify carcinogens. J. Biol. Chem 2000, 275, 2322–2327. [Google Scholar]
- Wang, ZH; Kang, KA; Zhang, R; Piao, MJ; Jo, SH; Kim, JS; Sang, SS; Lee, JS; Park, DH; Hyun, JW. Myricetin suppresses oxidative stress-induced cell damage via both direct and indirect antioxidant action. Environ. Toxicol. Pharmacol 2010, 29, 12–18. [Google Scholar]
- Cohen, GM; d’Arcy Doherty, M. Free radical mediated cell toxicity by redox cycling chemicals. Br. J. Cancer Suppl 1987, 8, 46–52. [Google Scholar]
- Powis, G. Free radical formation by antitumor quinones. Free Radic. Biol. Med 1989, 6, 63–101. [Google Scholar]
- Lin, YC; Huang, YC; Chen, SC; Liaw, CC; Kuo, SC; Huang, LJ; Gean, PW. Neuroprotective effects of ugonin K on hydrogen peroxide-induced cell death in human neuroblastoma SH-SY5Y cells. Neurochem. Res 2009, 34, 923–930. [Google Scholar]
- Tetich, M; Kutner, A; Leskiewicz, M; Budziszewska, B; Lasoń, W. Neuroprotective effects of (24R)-1,24-dihydroxycholecalciferol in human neuroblastoma SH-SY5Y cell line. J Steroid Biochem Mol Biol 2004, 89–90, 365–370. [Google Scholar]
- Kim, SS; Park, RY; Jeon, HJ; Kwon, YS; Chun, W. Neuroprotective effects of 3,5-dicaffeoylquinic acid on hydrogen peroxide-induced cell death in SH-SY5Y cells. Phytother. Res 2005, 19, 243–245. [Google Scholar]
- Kroemer, G; Zamzami, N; Susin, SA. Mitochondrial control of apoptosis. Immunol. Today 1997, 18, 44–51. [Google Scholar]
- Green, DR; Reed, JC. Mitochondria and apoptosis. Science 1998, 281, 1309–1312. [Google Scholar]
- Zamzami, N; Marchetti, P; Castedo, M; Zanin, C; Vayssiere, JL; Petit, PX; Kroemer, G. Reduction in mitochondrial potential constitutes an early irreversible step of programmed lymphocyte death in vivo. J. Exp. Med 1995, 181, 1661–1672. [Google Scholar]
- Lim, JH; Kim, SS; Boo, DH; No, H; Kang, BY; Kim, EM; Hwang, O; Choi, HJ. Protective effect of bromocriptine against BH4-induced Catha cell death involving up-regulation of antioxidant enzymes. Neurosci. Lett 2009, 451, 185–189. [Google Scholar]
- Gu, Q; Wang, D; Wang, X; Peng, R; Liu, J; Jiang, T; Wang, Z; Wang, S; Deng, H. Basic fibroblast growth factor inhibits radiation-induced apoptosis of HUVECs. I. The PI3K/AKT pathway and induction of phosphorylation of BAD. Radiat. Res 2004, 161, 692–702. [Google Scholar]
- Povsic, TJ; Kohout, TA; Lefkowitz, RJ. Beta-arrestin1 mediates insulin-like growth factor 1 (IGF-1) activation of phosphatidylinositol 3-kinase (PI3K) and anti-apoptosis. J. Biol. Chem 2003, 278, 51334–51339. [Google Scholar]
- Marte, BM; Downward, J. PKB/Akt: Connecting phosphoinositide 3-kinase to cell survival and beyond. Trends Biochem. Sci 1997, 22, 355–358. [Google Scholar]
- Downward, J. PI 3-kinase, Akt and cell survival. Semin. Cell Dev. Biol 2004, 15, 177–182. [Google Scholar]
- Carmichael, J; DeGraff, WG; Gazdar, AF; Minna, JD; Mitchell, JB. Evaluation of a tetrazolium-based semiautomated colorimetric assay: assessment of chemosensitivity testing. Cancer Res 1987, 47, 936–941. [Google Scholar]
- Nicoletti, I; Migliorati, G; Pagliacci, MC; Grignani, F; Riccardi, C. A rapid and simple method for measuring thymocyte apoptosis by propidium iodide staining and flow cytometry. J. Immunol. Methods 1991, 139, 271–279. [Google Scholar]
- Troiano, L; Ferraresi, R; Lugli, E; Nemes, E; Roat, E; Nasi, M; Pinti, M; Cossarizza, A. Multiparametric analysis of cells with different mitochondrial membrane potential during apoptosis by polychromatic flow cytometry. Nat. Protoc 2007, 2, 2719–2727. [Google Scholar]



© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kang, K.A.; Wang, Z.H.; Zhang, R.; Piao, M.J.; Kim, K.C.; Kang, S.S.; Kim, Y.W.; Lee, J.; Park, D.; Hyun, J.W. Myricetin Protects Cells against Oxidative Stress-Induced Apoptosis via Regulation of PI3K/Akt and MAPK Signaling Pathways. Int. J. Mol. Sci. 2010, 11, 4348-4360. https://doi.org/10.3390/ijms11114348
Kang KA, Wang ZH, Zhang R, Piao MJ, Kim KC, Kang SS, Kim YW, Lee J, Park D, Hyun JW. Myricetin Protects Cells against Oxidative Stress-Induced Apoptosis via Regulation of PI3K/Akt and MAPK Signaling Pathways. International Journal of Molecular Sciences. 2010; 11(11):4348-4360. https://doi.org/10.3390/ijms11114348
Chicago/Turabian StyleKang, Kyoung Ah, Zhi Hong Wang, Rui Zhang, Mei Jing Piao, Ki Cheon Kim, Sam Sik Kang, Young Woo Kim, Jongsung Lee, Deokhoon Park, and Jin Won Hyun. 2010. "Myricetin Protects Cells against Oxidative Stress-Induced Apoptosis via Regulation of PI3K/Akt and MAPK Signaling Pathways" International Journal of Molecular Sciences 11, no. 11: 4348-4360. https://doi.org/10.3390/ijms11114348



