Rapid Synthesis of Flavor Compound 4-Ethyloctanoic Acid under Microwave Irradiation
Abstract
:1. Introduction
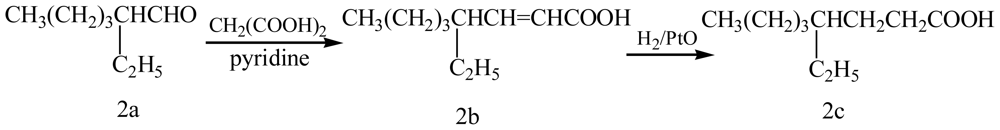
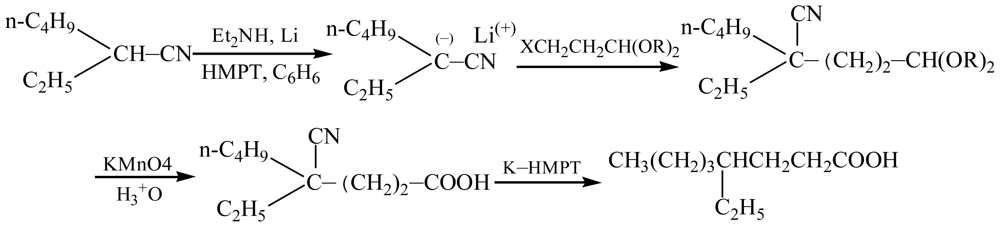
2. Results and Discussion
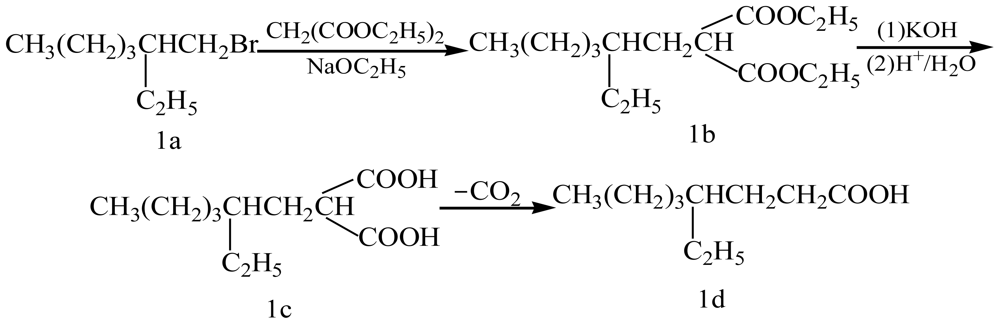
2.1. The Optimization of Synthesis Conditions of Diethyl (2-Ethylhexyl)malonate (1b)
2.2. The Optimization of Reaction Time of Synthesis of (2-Ethylhexyl)propanedioic Acid (1c)
2.3. The Optimization of Reaction Time of Synthesis of 4-Ethyloctanoic Acid (1d)
3. Experimental Section
3.1. General
3.2. Synthesis of Diethyl (2-Ethylhexyl)malonate (1b)
3.3. The Isolation of By-Product (Diethyl Di(2-ethylhexyl)malonate) from the Distill Remainder of 1b
3.4. Synthesis of (2-Ethylhexyl)propanedioic Acid (1c)
3.5. Synthesis of 4-Ethyloctanoic Acid (1d)
4. Conclusions
Acknowledgements
References
- Rijke, DD; Traas, PC; Heide, RT. Acidic components in essential oils of costus root, patchouli and olibanum. Phytochemistry 1978, 17, 1664–1666. [Google Scholar]
- Anderson, RC; Kelly, AG. Identification of a low flavor threshold carboxylic acid constituent, 4-ethyloctanoic acid, in flue-cured Virginia tobacco. J. Agric. Food Chem 1988, 36, 353–354. [Google Scholar]
- Ha, JK; Lindsay, RC. Volatile branched-chain fatty acids and phenolic compounds in aged Italian cheesw flavors. J. Food Sci 1991, 56, 1241–1247. [Google Scholar]
- Ha, JK; Lindsay, RC. Contribution of cow, sheep, and goat milks to characterizing branchedchain fatty acid and phenolic flavors in varietal cheeses. J. Dairy Sci 1991, 74, 3267–3274. [Google Scholar]
- Salles, C; Sommerer, N; Septier, C. Goat cheese flavor: Sensory evaluation of branched-chain fatty acids and small peptides. J. Food Sci 2002, 67, 835–841. [Google Scholar]
- Carunchia-Whetstine, ME; Karagul-Yuceer, Y; Avsar, YK. Identification andquantification of character aroma components in fresh chevre-style goat cheese. J. Food Sci 2003, 68, 2441–2447. [Google Scholar]
- Christlbauer, M; Schieberle, P. Characterization of the key aroma compounds in beef and pork vegetable gravies á la chef by application of the aroma extract dilution analysis. J. Agric. Food Chem 2009, 57, 9114–9122. [Google Scholar]
- Mosciano, G. Organoleptic characteristics of flavor materials. Perfum. Flavor 2007, 32, 16–18. [Google Scholar]
- Oser, BL; Ford, RA. GRAS flavoring Substances 17. Food Tech 1996, 50, 78–80. [Google Scholar]
- Liu, Y; Chen, H; Yin, D; Sun, B. Synthesis and odor evaluation of five new sulfur-containing ester flavor compounds from 4-ethyloctanoic acid. Molecules 2010, 15, 5104–5111. [Google Scholar]
- Powell, SG; Baldwin, MM. The condensation of 2-butanone with aldehydes of the type RR1CHCHO. J. Am. Chem. Soc 1936, 58, 1871–1872. [Google Scholar]
- Keil, W. The fat from fatty acids with odd numbers of carbon atoms. III. Z. Physiol. Chem 1942, 274, 175–185. [Google Scholar]
- Petrov, AD; Nikishin, GI; Ogibin, YN. The synthesis and surface-active properties of branched chain acids of the series, CnH2n+1COOH, with a C chain, C10–C20. Vses. Nauchn.-Issled. Inst. Zhirov 1960, 20, 278–292. [Google Scholar]
- Dziomko, VM; Dunaevskaya, KA. Synthesis of some aliphatic ketones with branched chains and different distance of branches from oxo group. Tr. Vses. Nauch. Issled. Inst 1966, 28, 184–192. [Google Scholar]
- Hedenstrom, E; Nguyen, BV; Silks, LA. Do enzymes recognize remotely located stereocentres? Highly enantioselective Candida rugosa lipase-catalysed esterification of the 2- to 8-methyldecanoic acids. Tetrahedron Asymmetry 2002, 13, 835–844. [Google Scholar]
- Neth, NV. Naarden International. Branched-chain carboxylic acids as perfume components. GB Patent 1,503,241, 8 March 1978. [Google Scholar]
- Larcheveque, M; Cuvigny, T. A new preparation of β and γ-alkyl aldehydes or acids by using activating cyano group. Tetrahedron Lett 1975, 44, 3851–3854. [Google Scholar]
- Sugiyama, T; Sasada, H; Masaki, J; Yamashita, K. Unusual fatty acids with specific odor from mature male goat. Agric. Boil. Chem 1981, 45, 2655–2658. [Google Scholar]
- Srivastava, KP; Mishra, PK. Green syntheses of 4-substitued-3-phenyl sydnones under microwave irradiation. Int. J. Pure Appl. Chem 2008, 3, 171–174. [Google Scholar]
- Reyes, L; Corona, S; Arroyo, G. Eco-contribution for the production of N-arylnitrones: Solventfree and assisted by microwaves. Int. J. Mol. Sci 2010, 11, 2576–2583. [Google Scholar]
- Sinha, AK; Sharma, A; Joshi, BP. One-pot two-step synthesis of 4-vinylphenols from 4-hydroxy substituted benzaldehydes under microwave irradiation: A new perspective on the classical Knoevenagel-Doebner reaction. Tetrahedron 2007, 63, 960–965. [Google Scholar]
- Nomura, E; Hosoda, A; Mori, H; Taniguchi, H. Rapid base-catalyzed decarboxylation and amide-forming reaction of substituted cinnamic acids via microwave heating. Green Chem 2005, 7, 863–866. [Google Scholar]
- Sharma, A; Kumar, R; Sharma, N; Sinha, AK. Unique versatility of ionic liquids as clean decarboxylation catalyst cum solvent: A metal- and quinoline-free paradigm towards synthesis of indoles, styrenes, stilbenes and arene derivatives under microwave irradiation in aqueous conditions. Adv. Synth. Catal 2008, 350, 2910–2920. [Google Scholar]
- Sinha, AK; Joshi, BP; Sharma, A. Microwave induced process for the preparation of substituted 4-vinylphenols. US Patent 6,989,467, 24 January 2006. [Google Scholar]

| Run | n (sodium) | n(diethyl malonate) | n(2-ethylhexyl bromide) | Yield of 1b (%) |
|---|---|---|---|---|
| 1 | 0.105 | 0.105 | 0.1 | 78.1 |
| 2 | 0.110 | 0.105 | 0.1 | 70.6 |
| 3 | 0.110 | 0.110 | 0.1 | 77.3 |
| 4 | 0.115 | 0.110 | 0.1 | 73.4 |
| 5 | 0.1 | 0.105 | 0.105 | 76.1 |
| 6 | 0.1 | 0.105 | 0.110 | 76.6 |
| 7 | 0.1 | 0.110 | 0.110 | 78.7 |
| 8 | 0.1 | 0.110 | 0.115 | 78.8 |
| Reaction temperature (°C) | Maximum concentration of 1b in reaction mixture (%) | Reaction time (h) | Yield of 1b (%) |
|---|---|---|---|
| 75 | 70.92 | 3.0 | 73.3 |
| 80 | 76.37 | 2.5 | 79.1 |
| 85 | 74.22 | 2.0 | 78.2 |
| 90 | 70.53 | 1.5 | 72.8 |
| Reaction time (h) | 6 | 12 | 18 | 24 |
|---|---|---|---|---|
| Yield of 1b (%) | 71.0 | 75.5 | 77.2 | 78.9 |
| In oil bath | Under microwave irradiation | ||
|---|---|---|---|
| Reaction time (min) | Yield of 1c (%) | Reaction time (min) | Yield of 1c (%) |
| 60 | 93.0 | 5 | 94.3 |
| 90 | 95.5 | 10 | 96.1 |
| 120 | 95.9 | 20 | 97.4 |
| 150 | 96.1 | 30 | 97.7 |
| In oil bath | Under microwave irradiation | ||
|---|---|---|---|
| Reaction time (min) | The reducing amount of reaction mixture (g) | Reaction time min) | The reducing amount of reaction mixture (g) |
| 5 | 4.186 | 4 | 4.679 |
| 15 | 4.445 | 8 | 4.719 |
| 30 | 4.536 | 12 | 4.746 |
| 60 | 4.575 | 16 | 4.759 |
| Separated yield of 1d(%) | 87.0 | Separated yield of 1d(%) | 90.6 |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liu, Y.-P.; Yin, D.-C.; Chen, H.-T.; Sun, B.-G. Rapid Synthesis of Flavor Compound 4-Ethyloctanoic Acid under Microwave Irradiation. Int. J. Mol. Sci. 2010, 11, 4165-4174. https://doi.org/10.3390/ijms11104165
Liu Y-P, Yin D-C, Chen H-T, Sun B-G. Rapid Synthesis of Flavor Compound 4-Ethyloctanoic Acid under Microwave Irradiation. International Journal of Molecular Sciences. 2010; 11(10):4165-4174. https://doi.org/10.3390/ijms11104165
Chicago/Turabian StyleLiu, Yu-Ping, De-Cai Yin, Hai-Tao Chen, and Bao-Guo Sun. 2010. "Rapid Synthesis of Flavor Compound 4-Ethyloctanoic Acid under Microwave Irradiation" International Journal of Molecular Sciences 11, no. 10: 4165-4174. https://doi.org/10.3390/ijms11104165
APA StyleLiu, Y.-P., Yin, D.-C., Chen, H.-T., & Sun, B.-G. (2010). Rapid Synthesis of Flavor Compound 4-Ethyloctanoic Acid under Microwave Irradiation. International Journal of Molecular Sciences, 11(10), 4165-4174. https://doi.org/10.3390/ijms11104165




