Antigenotoxic Effect of Chamomilla recutita (L.) Rauschert Essential Oil in Mouse Spermatogonial Cells, and Determination of Its Antioxidant Capacity in Vitro
Abstract
:1. Introduction
2. Material and Methods
2.1. Chemicals and Animals
2.2. SCE Assay
2.3. DPPH Radical Scavenging Activity
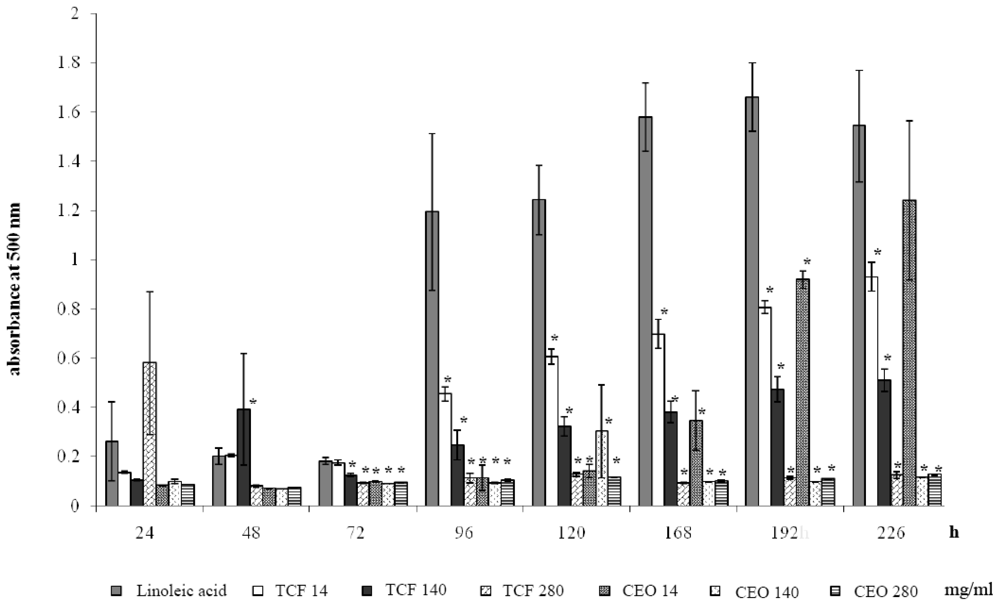
2.4. Ferric Thiocyanate Assay
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusion
References
- Nohmi, T; Kim, SR; Yamada, M. Modulation of oxidative mutagenesis and carcinogenesis by polymorphic forms of human DNA repair enzymes. Mutat. Res 2005, 591, 60–73. [Google Scholar]
- Tesoriere, L; Butera, D; Pintaudi, AM; Allegra, M; Livrea, M. Supplementation with cactus pear (Opuntia ficus-indica) fruit decreases oxidative stress in healthy humans: A comparative study with vitamin C. Am. J. Clin. Nutr 2004, 80, 391–395. [Google Scholar]
- Jiang, T; Glickman, BW; de Boer, JG. Protective effect of green tea against benzo[a]pyrene induced mutations in the liver of Big Blue transgenic mice. Mutat. Res 2001, 480–481, 147–151. [Google Scholar]
- Sharma, N; Trikha, P; Athar, M; Raisuddin, S. Inhibition of benzo[a]pyrene and cyclophosphamide induced mutagenicity by Cinnamomum cassia. Mutat. Res 2001, 480–481, 179–188. [Google Scholar]
- Isaac, O; Schimpke, H. Alte and Erkemtnisse der Kamillenforschung 2. Mitt. Dstch. Pharmaz. Ges 1965, 35, 133–147. [Google Scholar]
- Read, MA. Flavonoids: Naturally occurring anti-inflammatory agents. Am. J. Pathol 1995, 147, 235–237. [Google Scholar]
- Viola, H; Wasowski, C; Levi de Stein, M; Wolfman, C; Silveira, R; Dajas, F; Medina, JH; Paladini, AC. Apigenin, a component of Matricaria recutita flowers is a central benzodiazepine receptor-ligand with anxiolytic effects. Planta Med 1995, 61, 213–216. [Google Scholar]
- Jakolev, V; Issac, O; Flaskamp, E. Pharmacological investigation with compounds of chamazulene and matricine. Planta Med 1983, 49, 67–73. [Google Scholar]
- Matos, F; Machado, M; Alencar, J; Caveiro, A. Constituents of Brazilian chamomile oil. J. Essent. Oil Res 1993, 5, 337–339. [Google Scholar]
- McKay, DL; Blumberg, JB. A review of the bioactivity and potential health benefits of chamomile tea (Matricaria recutita L.). Phytother. Res 2006, 20, 519–530. [Google Scholar]
- Hernández-Ceruelos, A; Madrigal-Bujaidar, E; de la Cruz, C. Inhibitory effect of chamomile essential oil on the sister chromatid exchanges induced by daunorubicin and methyl methanesulfonate in mouse bone marrow. Toxicol. Lett 2002, 135, 103–110. [Google Scholar]
- Hortobágyi, GN. Anthracyclines in the treatment of cancer. An overview. Drugs 1997, 54, 1–7. [Google Scholar]
- Madrigal-Bujaidar, E; Velazquez-Guadarrama, N; Morales-Ramirez, P; Mendiola, M. Effect of disulfiram on the genotoxic potential of acetaldehyde in mouse spermatogonial cells. Teratog. Carcinog. Mutagen 2002, 22, 83–91. [Google Scholar]
- Burtis, M; Bucar, F. Antioxidant activity of Nigella sativa essential oil. Phytother. Res 2000, 14, 323–328. [Google Scholar]
- Ono, M; Yamamoto, M; Masuoka, C; Ito, Y; Yamashita, M; Nohara, T. Diterpenes form the fruits of Vitex rotundifolia. J. Nat. Prod 1999, 62, 1532–1537. [Google Scholar]
- Sjöblom, T; West, A; Lähdetie, J. Apoptotic response of spermatogenic cells to the germ cell mutagens etoposide, adriamycin, and diepoxybutane. Environ. Mol. Mutagen 1998, 31, 133–148. [Google Scholar]
- Lahdetie, J; Keiski, A; Suutari, A; Toppari, J. Etoposide (VP-16) is a potent inducer of micronuclei in male rat meiosis: Spermatid micronucleus test and DNA flow cytometry after etoposide treatment. Environ. Mol. Mutagen 1994, 24, 192–202. [Google Scholar]
- Suominen, JS; Linderborg, J; Nikula, H; Hakovirta, H; Parvinen, M; Toppari, J. The effects of mono-2-ethylhexyl phthalate, adriamycin and N-ethyl-N-nitrosourea on stage-specific apoptosis and DNA synthesis in the mouse spermatogenesis. Toxicol. Lett 2003, 143, 163–73. [Google Scholar]
- Rekka, EA; Kourounakis, AP; Kourounakis, PN. Investigation on the effect of chamazulene on lipid peroxidation and free radical processes. Res. Commun. Mol. Pathol. Pharmacol 1996, 92, 361–364. [Google Scholar]
- Safayhi, H; Sabieraj, J; Sailer, ER; Ammon, HP. Chamazulene: An antioxidant-type inhibitor of leukotriene B4 formation. Planta Med 1994, 60, 410–413. [Google Scholar]
- Fujiki, H; Suganuma, M; Okabe, S; Sueoka, E; Sueoka, N; Fujimoto, N; Goto, Y; Matsuyama, S; Imai, K; Nakachi, K. Cancer prevention with green tea and monitoring by a new biomarker, hnRNP B1. Mutat. Res 2001, 480–481, 299–304. [Google Scholar]
- Achterrath-Tuckerman, U; Kunde, R. Investigation on the spasmolytic effect of compounds of chamomile and kamillosan on the isolated guinea pig ileum. Planta Med 1980, 39, 38–50. [Google Scholar]
- Keizer, H; Pinedo, H; Schuurhuis, G; Joenje, H. Doxorubicin (adriamycin): A critical review of free radical-dependent mechanisms of cytotoxicity. Pharmacol. Ther 1990, 47, 219–231. [Google Scholar]
- Lis-Balchin, M; Deans, G; Eaglesham, E. Relationship between bioactivity and chemical composition of commercial essential oils. Flavour Fragrance J 1998, 13, 98–104. [Google Scholar]
- Gülcin, I. Antioxidant activity of caffeic acid (3,4-dihydroxycinnamic acid). Toxicology 2006, 217, 213–220. [Google Scholar]
- Mihaljevic, B; Katusin-Razem, B; Razem, D. The reevaluation of the ferric thiocyanate assay for lipid hydroperoxides with especial considerations of the mechanistic aspects of the response. Free Radic. Biol. Med 1995, 21, 55–63. [Google Scholar]
- Lu, Y; Foo, Y. Polyphenolics of Salvia-a review. Phytochemistry 2002, 59, 117–140. [Google Scholar]
- Singh, G; Marrimuthu, P; de Heluani, CS; Catalan, CA. Antioxidant biocidal activities of Carum nigrum (seed) essential oil, oleoresin and their selected components. J. Agric. Food Chem 2006, 54, 174–181. [Google Scholar]

| Compound | RTa | CAS no. | Area (%) |
|---|---|---|---|
| (E)-β-Farnecene | 38.46 | 28973-97-9 | 28.17 |
| Germacrene-d | 39.23 | 23986-74-5 | 2.19 |
| Unidentified sesquiterpene | 40.07 | 1.40 | |
| Unidentified sesquiterpene | 41.17 | 0.78 | |
| (Z,E)-α-Farnecene | 41.35 | 26560-14-5 | 1.59 |
| Unidentified sesquiterpene | 48.52 | 0.71 | |
| α-Bisabolol oxide A | 54.46 | 22567-36-8 | 41.77 |
| α-Bisabolol oxide B | 49.28 | 26184-88-3 | 4.31 |
| α-Bisabolol oxide | 50.65 | 22567-38-0 | 5.30 |
| α-Bisabolol | 51.18 | 515-69-5 | 2.31 |
| Chamazulene | 52.80 | 529-05-5 | 2.39 |
| 1,6-Dioxaspiro[4,4]non-3-ene,2-(2,4hexadyn-1-ylidene) | 60.73 | 2.19 | |
| Hexatriacontane | 67.49 | 630-06-8 | 0.50 |
| Agent | Dose (mg/kg) | SCE ± S.D. | Inhibition (%) |
|---|---|---|---|
| Controla | 0 | * 1.90 ± 0.40 | |
| CEO | 500 | * 2.36 ± 0.44 | |
| DAU | 10 | 11.21 ± 1.38 | |
| CEO+DAU | 5+10 | * 6.78 ± 1.05 | 47.5 |
| CEO+DAU | 50+10 | * 5.44 ± 0.33 | 61.9 |
| CEO+DAU | 500+10 | * 2.50 ± 0.31 | 93.5 |
| Agent | Concentration(mg/mL) | Absorbance OD (517 nm) | Absorbance Inhibition (%) |
|---|---|---|---|
| DPPH | 0.04 | 1.059 ± 0.001 | |
| α-cariophylene | 100 | 1.048 ± 0.009 | 1.0 |
| α-tocopherol | 1 | 0.362 ± 0.015* | 65.8 |
| CEO | 5 | 1.057 ± 0.003 | 0.001 |
| CEO | 10 | 1.013 ± 0.004* | 4.3 |
| CEO | 50 | 0.872 ± 0.014* | 17.6 |
| CEO | 100 | 0.621 ± 0.022* | 41.3 |
| CEO | 500 | 0.208 ± 0.013* | 80.4 |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hernández-Ceruelos, A.; Madrigal-Santillán, E.; Morales-González, J.A.; Chamorro-Cevallos, G.; Cassani-Galindo, M.; Madrigal-Bujaidar, E. Antigenotoxic Effect of Chamomilla recutita (L.) Rauschert Essential Oil in Mouse Spermatogonial Cells, and Determination of Its Antioxidant Capacity in Vitro. Int. J. Mol. Sci. 2010, 11, 3793-3802. https://doi.org/10.3390/ijms11103793
Hernández-Ceruelos A, Madrigal-Santillán E, Morales-González JA, Chamorro-Cevallos G, Cassani-Galindo M, Madrigal-Bujaidar E. Antigenotoxic Effect of Chamomilla recutita (L.) Rauschert Essential Oil in Mouse Spermatogonial Cells, and Determination of Its Antioxidant Capacity in Vitro. International Journal of Molecular Sciences. 2010; 11(10):3793-3802. https://doi.org/10.3390/ijms11103793
Chicago/Turabian StyleHernández-Ceruelos, Alejandra, Eduardo Madrigal-Santillán, José Antonio Morales-González, Germán Chamorro-Cevallos, Martha Cassani-Galindo, and Eduardo Madrigal-Bujaidar. 2010. "Antigenotoxic Effect of Chamomilla recutita (L.) Rauschert Essential Oil in Mouse Spermatogonial Cells, and Determination of Its Antioxidant Capacity in Vitro" International Journal of Molecular Sciences 11, no. 10: 3793-3802. https://doi.org/10.3390/ijms11103793






