Efficient Dye-Sensitized Solar Cells Using Red Turnip and Purple Wild Sicilian Prickly Pear Fruits
Abstract
:1. Introduction
2. Experimental
2.1. Preparation of Dye-Sensitizer Solutions
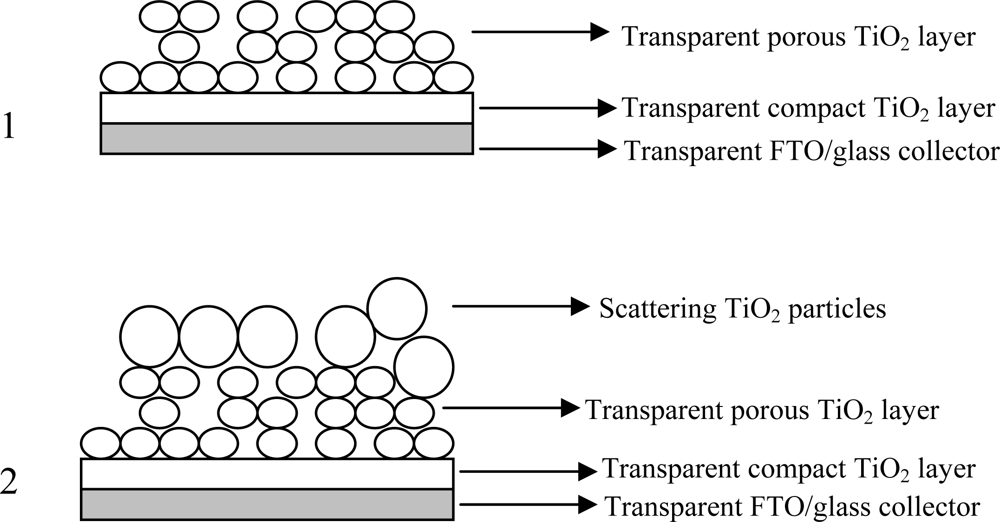
2.2. Preparation of Electrodes
2.3. DSSC Assembling
2.4. Measurements
3. Results and Discussion
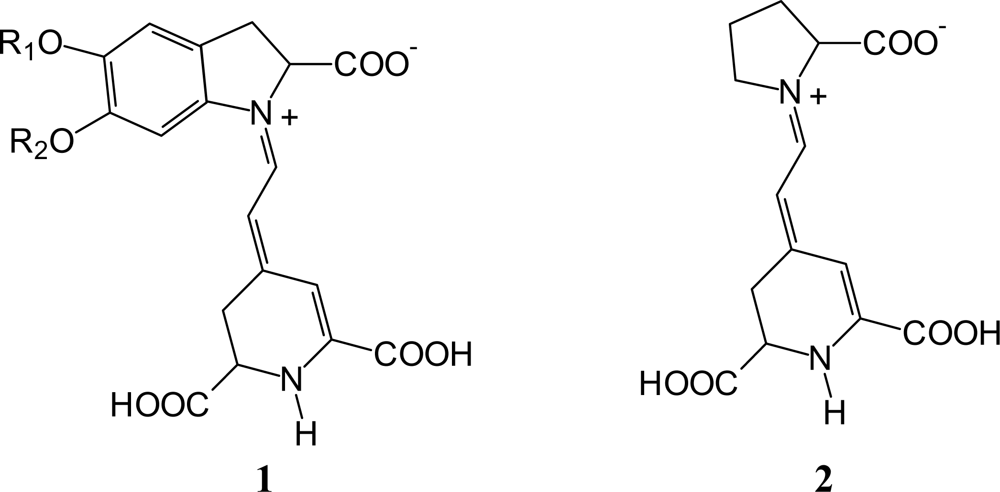
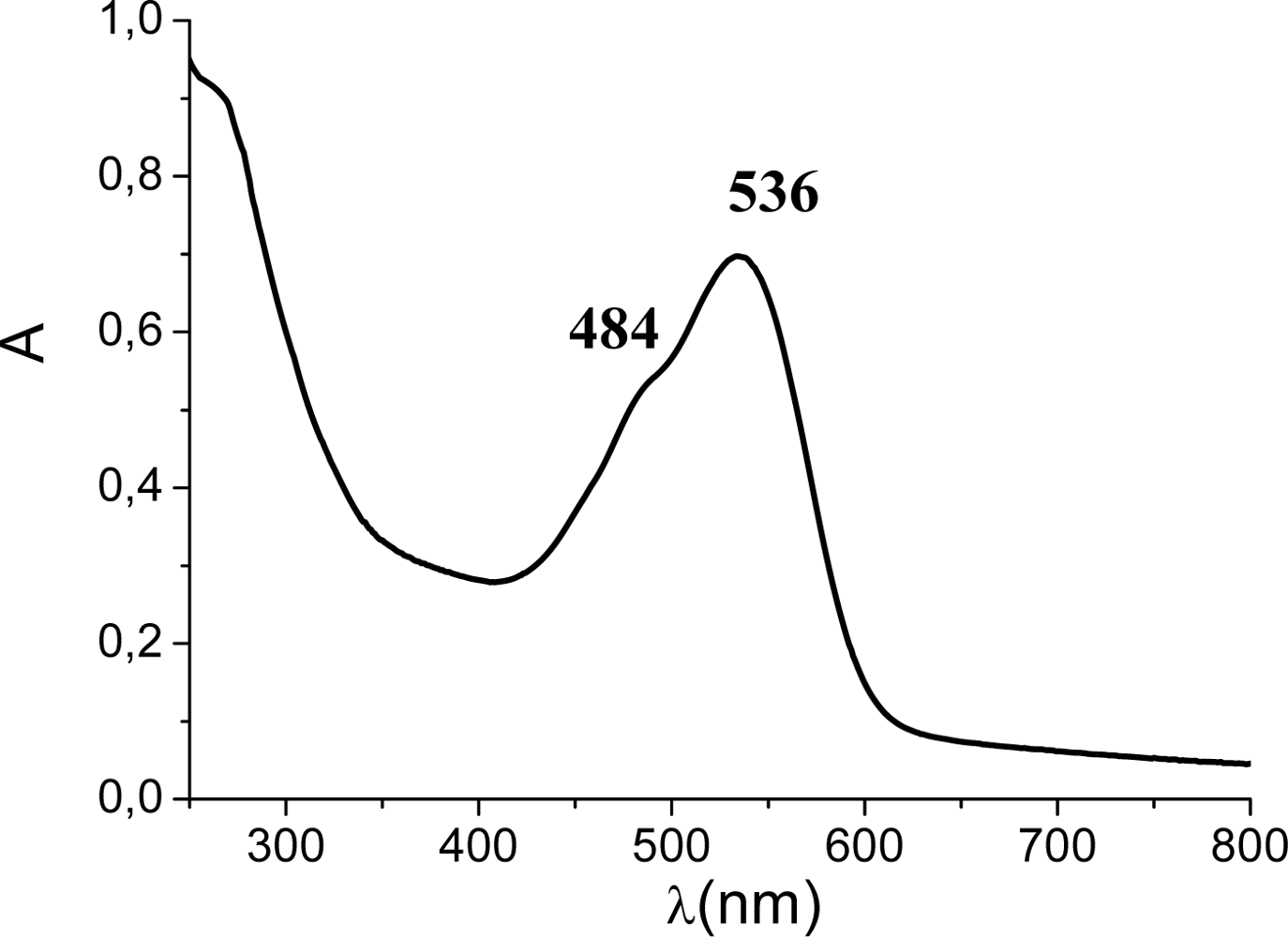
3.1. Absorption Spectra of Raw Natural Dye Extract
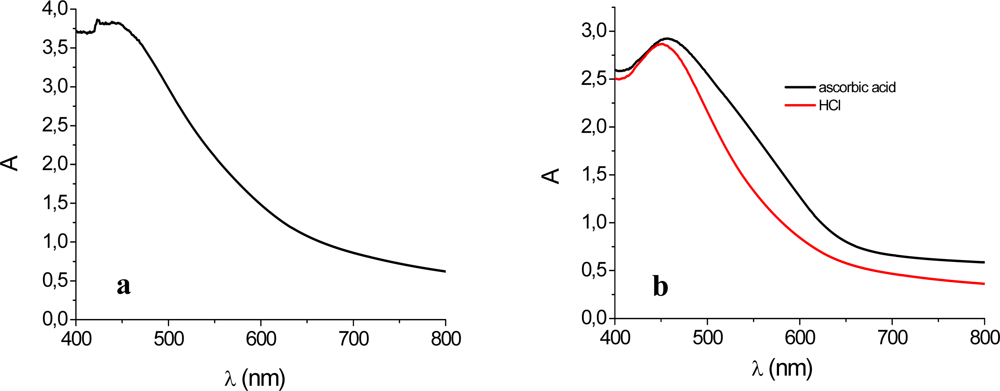
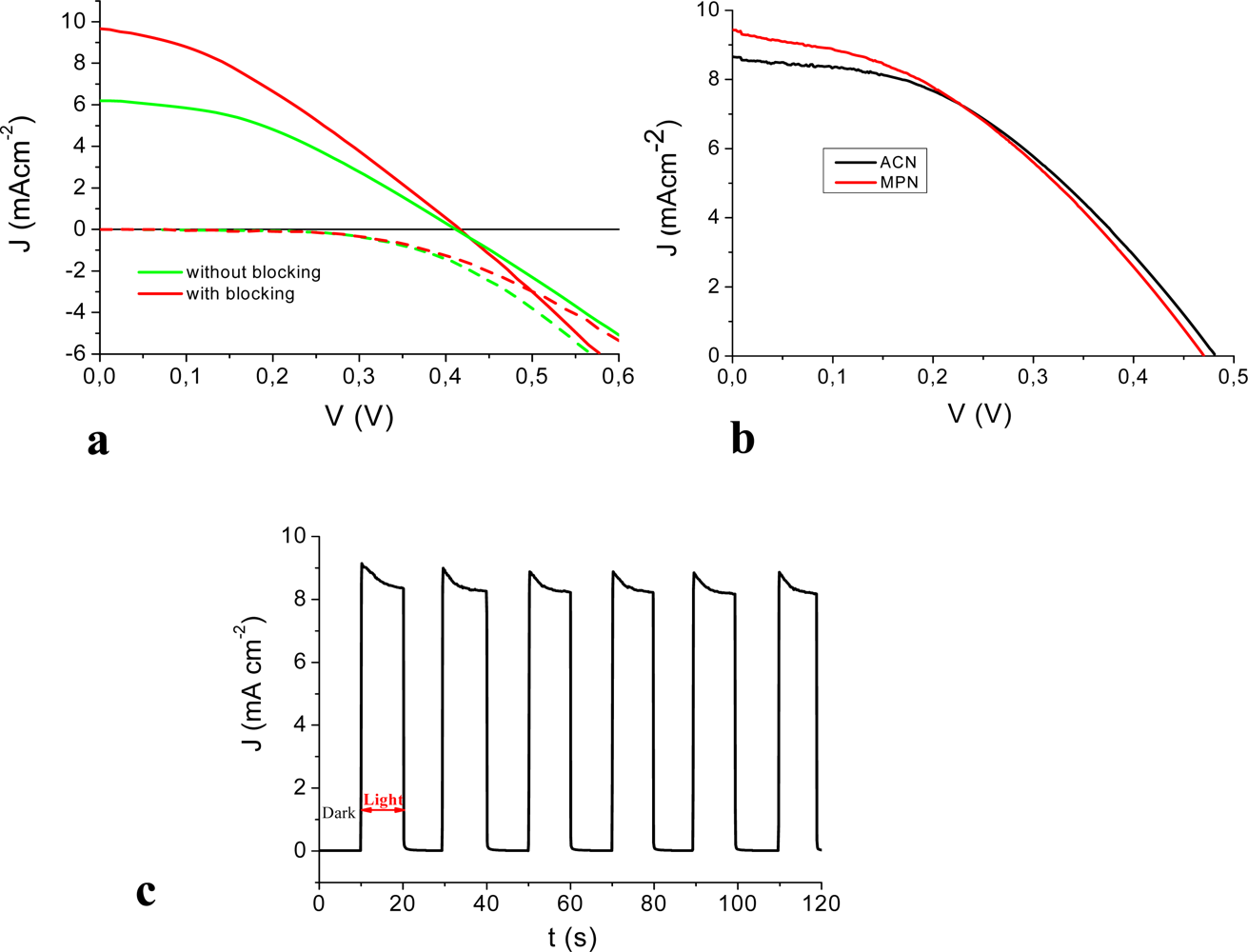
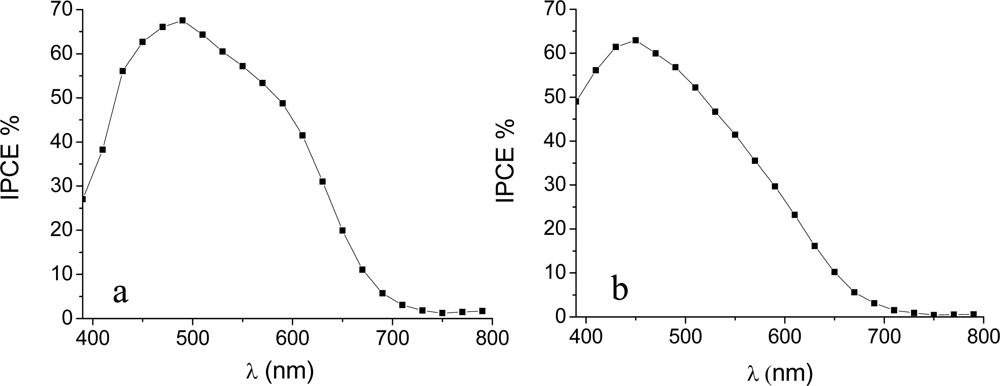
3.2. Photoelectrochemistry
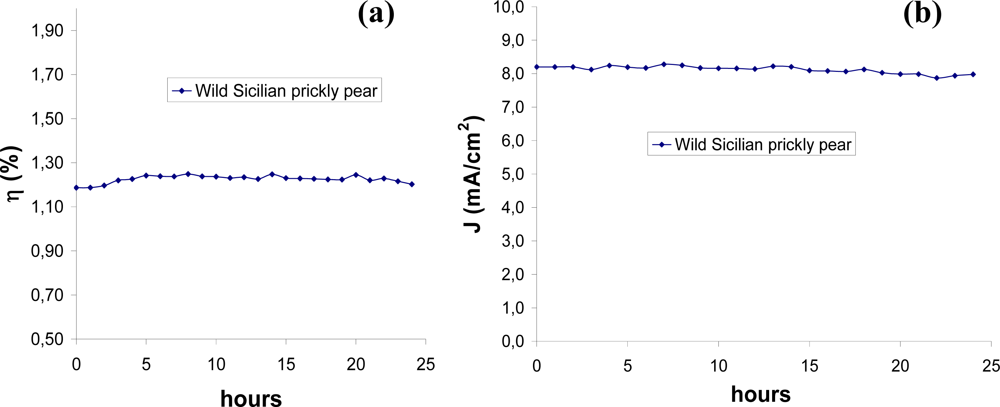
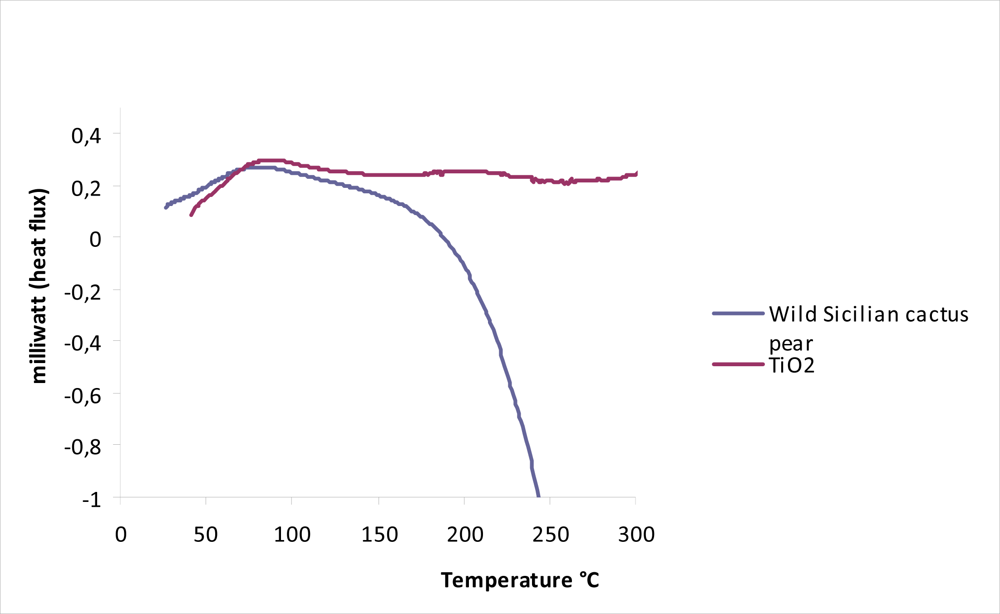
3.3. Stability Test
4. Conclusions
Acknowledgments
References
- Liska, P; Vlachoupoulos, N; Nazeeruddin, MK; Comte, P; Graetzel, M. cis-Diaquabis(2,2′-bipyridyl-4,4′-dicarboxylate)ruthenium(II) sensitizes wide band gap oxide semiconductors very efficiently over a broad spectral range in the visible. J. Am. Chem. Soc 1988, 110, 3686–3687. [Google Scholar]
- O’Regan, B; Graetzel, M. A low-cost, high-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 1991, 353, 737–740. [Google Scholar]
- Graetzel, M. Solar energy conversion by dye sensitized photovoltaic cells. Inorg. Chem 2005, 44, 6841–6851. [Google Scholar]
- Yum, JH; Walter, P; Huber, S; Rentsch, D; Geiger, T; Neusch, F; DeAngelis, F; Graetzel, M; Nazeeruddin, MK. Efficient far red sensitization of nanocrystalline TiO2 films by an unsymmetrical squaraine dye. J. Am. Chem. Soc 2007, 129, 10320–10321. [Google Scholar]
- Campbell, WM; Jolley, KW; Wagner, P; Wagner, K; Walsh, PJ; Gordon, KC; Schmidt-Mende, L; Nazeeruddin, MK; Wang, Q; Graetzel, M; Officer, DL. Highly efficient porphyrin sensitizers for dye-sensitized solar cells. J. Phys. Chem. C 2007, 111, 11760–11762. [Google Scholar]
- Nazeeruddin, MK; Kay, A; Rodicio, I; Humphry-Baker, R; Mueller, E; Liska, P; Vlachopoulos, N; Graetzel, M. Conversion of light to electricity by cis-X2bis(2,2′-bipyridyl-4,4′-dicarboxylate)ruthenium(II) charge-transfer sensitizers (X = Cl-, Br-, I-, CN-, and SCN-) on nanocrystalline titanium dioxide electrodes. J. Am. Chem. Soc 1993, 115, 6382–6390. [Google Scholar]
- Nazeeruddin, MK; Pechy, P; Liska, P; Renouard, T; Zakeeruddin, SM; Humphry-Baker, R; Comte, P; Cevey, L; Costa, E; Shklover, V; Spiccia, L; Deacon, GB; Bignozzi, CA; Graetzel, M. Engineering efficient panchromatic sensitizers for nanocrystalline TiO2-based solar cells. J. Am. Chem. Soc 2001, 123, 1613–1624. [Google Scholar]
- Wang, P; Klein, C; Humphry-Baker, R; Zakeeruddin, SH; Graetzel, M. A High Molar extinction coefficient sensitizer for stable dye-sensitized Solar Cells. J. Am. Chem. Soc 2005, 127, 808–809. [Google Scholar]
- Argazzi, R; Larramona, G; Contado, C; Bignozzi, CA. Preparation and photoelectrochemical characterization of a red sensitive osmium complex containing 4,4′,4′′-tricarboxy-2,2′:6′,2′′-terpyridine and cyanide ligands. J. Photochem. Photobiol. A: Chemistry 2004, 164, 15–21. [Google Scholar]
- Altobello, S; Argazzi, R; Caramori, S; Contado, C; Da Fre, S; Rubino, P; Chone, C; Larramona, G; Bignozzi, CA. Sensitization of nanocrystalline tio2 with black absorbers based on os and ru polypyridine complexes. J. Am. Chem. Soc 2005, 127, 15342–15343. [Google Scholar]
- Sarto Polo, A; Murakami Iha, NY; Itokazu, MK. Metal complex sensitizers in dye-sensitized solar cells. Coord. Chem. Rev 2004, 248, 1343–1361. [Google Scholar]
- Fernando, JMRC; Sendeera, GKR. Natural anthocyanins as photosensitizers for dye-sensitized solar devices. Res. Comm. Current. Sci 2008, 95, 663–666. [Google Scholar]
- Lai, HW; Su, YH; Teoh, LG; Hon, MH. Commercial and natural dyes as photosensitizers for a water-based dye-sensitized solar cell loaded with gold nanoparticles. J. Photochem. Photobiol. A: Chemistry 2008, 195, 307–313. [Google Scholar]
- Tennakone, K; Kumarasinghe, AR; Kumara, GRRA; Wijayantha, KGU; Sirimanne, PM. Nanoporous TiO2 photoanode sensitized with the flower pigment cyanidin. J. Photochem. Photobiol. A: Chem 1997, 108, 193–195. [Google Scholar]
- Cherepy, NJ; Smestad, GP; Graetzel, M; Zhang, GJ. Ultrafast electron injection: implications for a photoelectrochemical cell utilizing an anthocyanin dye-sensitized TiO2 nanocrystalline electrode. J. Phys. Chem. B 1997, 101, 9342–9351. [Google Scholar]
- Dai, Q; Rabani, J. Photosensitization of nanocrystalline TiO2 films by anthocyanin dyes. J. Photochem. Photobiol. A: Chemistry 2002, 148, 17–24. [Google Scholar]
- Sarto Polo, A; Murakami Iha, NY. Blue sensitizers for solar cells: Natural dyes from Calafate and Jaboticaba. Sol. Energ. Mat. Sol. Cel 2006, 90, 1936–1944. [Google Scholar]
- Calogero, G; Di Marco, G. Red Sicilian orange and purple eggplant fruits as natural sensitizers for dye-sensitized solar cells. Sol. Energ. Mat. Sol. Cel 2008, 92, 1341–1346. [Google Scholar]
- Calogero, G; Di Marco, G. Photoelectrochemical solar cell comprising sensitizing anthocyanin and betalain dyes of vegetal or synthetic origin, or mixtures thereof. International patent n°PCT/IT2009/0004682008. [Google Scholar]
- Gao, FG; Bard, AJ; Kispert, LD. Photocurrent generated on a carotenoid-sensitized TiO2 nanocrystalline mesoporous electrode. J. Photochem. Photobiol. A: Chemistry 2000, 130, 49–56. [Google Scholar]
- Zhang, D; Lanier, SM; Downing, JA; Avent, JL; Lum, J; McHale, JL. Betalain pigments for dye-sensitized solar cells. J. Photochem. Photobiol. A: Chem 2008, 195, 72–80. [Google Scholar]
- Tesoriere, L; Allegra, M; Butera, D; Livrea, MA. Absorption, excretion, and distribution of dietary antioxidant betalains in LDLs: potential health effects of betalains in humans. Am. J. Clin. Nutr 2004, 80, 941–945. [Google Scholar]
- Nazeeruddin, MK; Zakeeruddin, SM; Humphry-Baker, R; Jirousek, M; Liska, P; Vlachoupoulos, N; Shklover, V; Fischer, C-H; Graetzel, M. Acid–Base equilibria of (2,2‘-Bipyridyl-4,4‘-dicarboxylic acid)ruthenium(II) complexes and the effect of protonation on charge-transfer sensitization of nanocrystalline titania. Inorg. Chem 1999, 38, 6298–6305. [Google Scholar]
- Castellar, R; Obon, JM; Alacid, M; Fernandez-lopez, JA. Color properties and stability of betacyanins from opuntia fruits. J. Agric. Food Chem 2003, 51, 2772–2776. [Google Scholar]
- Zabri, H; Gillaizeau, I; Bignozzi, CA; Caramori, S; Charlot, M-F; Cano-Boquera, J; Odobel, F. Synthesis and comprehensive characterizations of new cis-RuL2X2 (X = Cl, CN, and NCS) sensitizers for nanocrystalline tio2 solar cell using bis-phosphonated bipyridine ligands (L). Inorg. Chem 2003, 42, 6655–6666. [Google Scholar]
- Quin, C; Clark, AE. DFT characterization of the optical and redox properties of natural pigments relevant to dye-sensitized solar cells. Chem. Phys. Lett 2007, 438, 26–30. [Google Scholar]
- Xagao, AP; Bernard, MC; Hugot-La Goffe, A; Spyellis, N; Loizos, Z; Falaras, P. Surface modification and photosensitisation of TiO2 nanocrystalline films with ascorbic acid. J. Photochem. Photobiol 2000, 132, 115–120. [Google Scholar]
- Burke, A; Ito, S; Snaith, H; Bach, U; Kwiatkowski, J; Graetzel, M. The function of a TiO2 compact layer in dye-sensitized solar cells incorporating “planar” organic dyes. Nano Lett 2008, 977–981. [Google Scholar]
- Pedreno, MA; Escribano, J. Correlation between antiradical activity and stability of betanine from Beta vulgaris L roots under different pH, temperature and light conditions. J. Sci. Food. Agric 2001, 81, 627–631. [Google Scholar]
- Calogero, G; Di Marco, G; Cazzanti, S; Caramori, S; Argazzi, R; Bignozzi, CA. Natural dye sensitizers for photoelectrochemical cells. Energ. Environ. Sci 2009, 2, 1162–1172. [Google Scholar]
| Dye Source | Jsc (mA/cm) | Voc (mV) | FF | η (%) | Anode Type (active area 0.5 cm2) |
|---|---|---|---|---|---|
| N719 | 17.73 | 530 | 0.35 | 3.3 | Method B |
| Red Turnip* (Beta vulgaris rubra) | 9.5 | 425 | 0.37 | 1.7 | Method B |
| Wild Sicilian Prickly pear* (Opuntia engelmannii) | 9.4 | 350 | 0.38 | 1.26 | Scattering layer |
| Wild Sicilian Prickly pear* (Opuntia engelmannii) | 8.20 | 375 | 0.38 | 1.19 | Method B |
| Wild Sicilian Prickly pear* (Opuntia engelmannii) | 7.32 | 400 | 0.41 | 1.21 | Method A |
| Sicilian Indian Fig* (Opuntia ficus indica) | 2.7 | 375 | 0.54 | 0.50 | Method A |
| Bougainvillea* | 2.1 | 300 | 0.57 | 0.36 | Method A |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Calogero, G.; Di Marco, G.; Cazzanti, S.; Caramori, S.; Argazzi, R.; Di Carlo, A.; Bignozzi, C.A. Efficient Dye-Sensitized Solar Cells Using Red Turnip and Purple Wild Sicilian Prickly Pear Fruits. Int. J. Mol. Sci. 2010, 11, 254-267. https://doi.org/10.3390/ijms11010254
Calogero G, Di Marco G, Cazzanti S, Caramori S, Argazzi R, Di Carlo A, Bignozzi CA. Efficient Dye-Sensitized Solar Cells Using Red Turnip and Purple Wild Sicilian Prickly Pear Fruits. International Journal of Molecular Sciences. 2010; 11(1):254-267. https://doi.org/10.3390/ijms11010254
Chicago/Turabian StyleCalogero, Giuseppe, Gaetano Di Marco, Silvia Cazzanti, Stefano Caramori, Roberto Argazzi, Aldo Di Carlo, and Carlo Alberto Bignozzi. 2010. "Efficient Dye-Sensitized Solar Cells Using Red Turnip and Purple Wild Sicilian Prickly Pear Fruits" International Journal of Molecular Sciences 11, no. 1: 254-267. https://doi.org/10.3390/ijms11010254
APA StyleCalogero, G., Di Marco, G., Cazzanti, S., Caramori, S., Argazzi, R., Di Carlo, A., & Bignozzi, C. A. (2010). Efficient Dye-Sensitized Solar Cells Using Red Turnip and Purple Wild Sicilian Prickly Pear Fruits. International Journal of Molecular Sciences, 11(1), 254-267. https://doi.org/10.3390/ijms11010254





