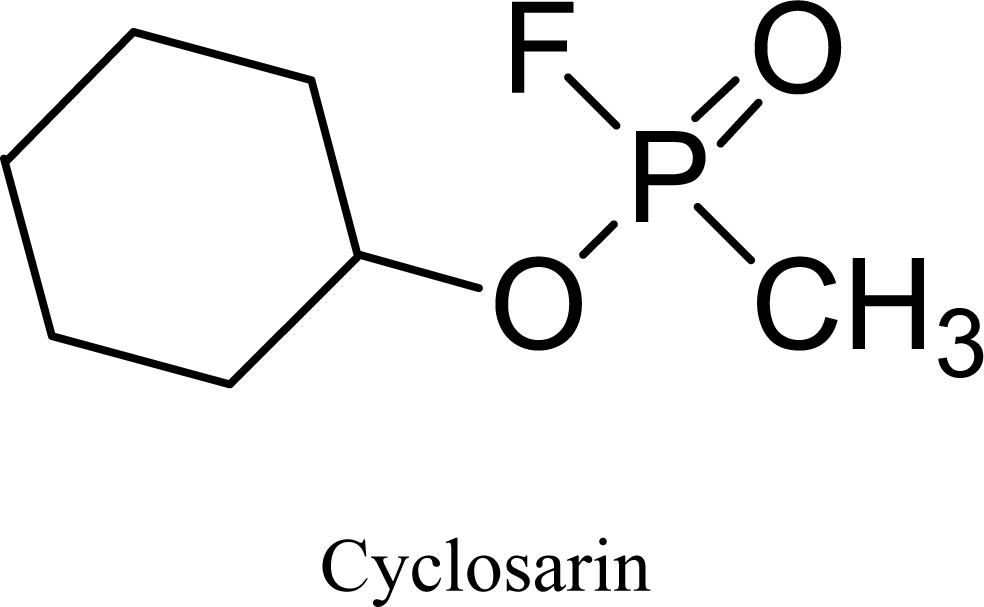
Effect of Seven Newly Synthesized and Currently Available Oxime Cholinesterase Reactivators on Cyclosarin-Intoxicated Rats
Abstract
:1. Introduction
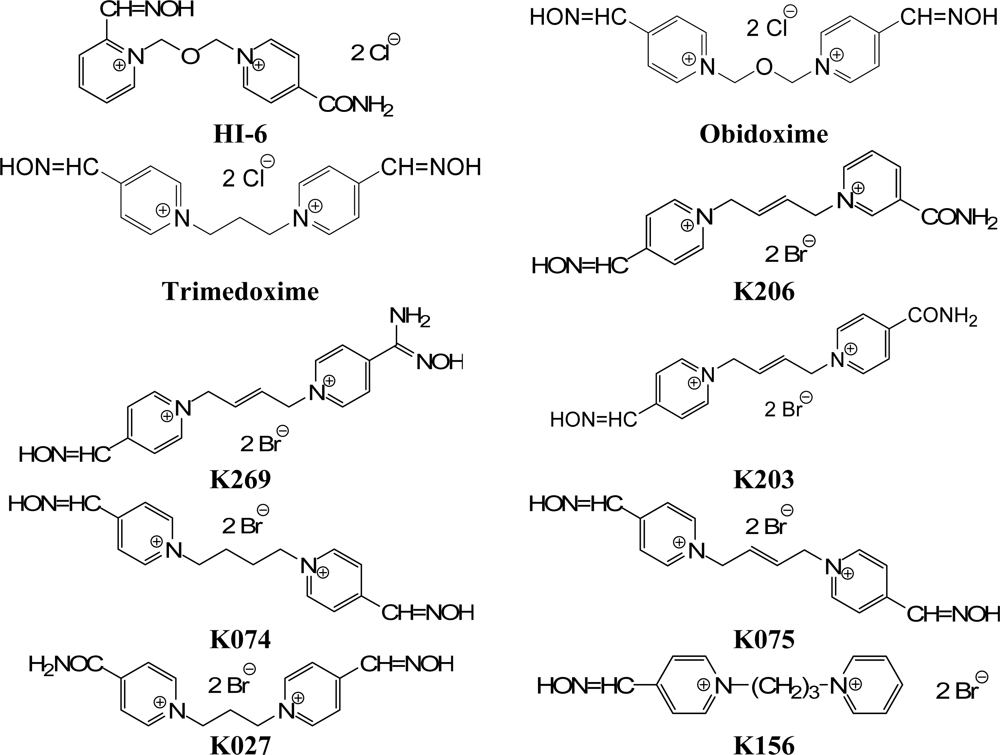
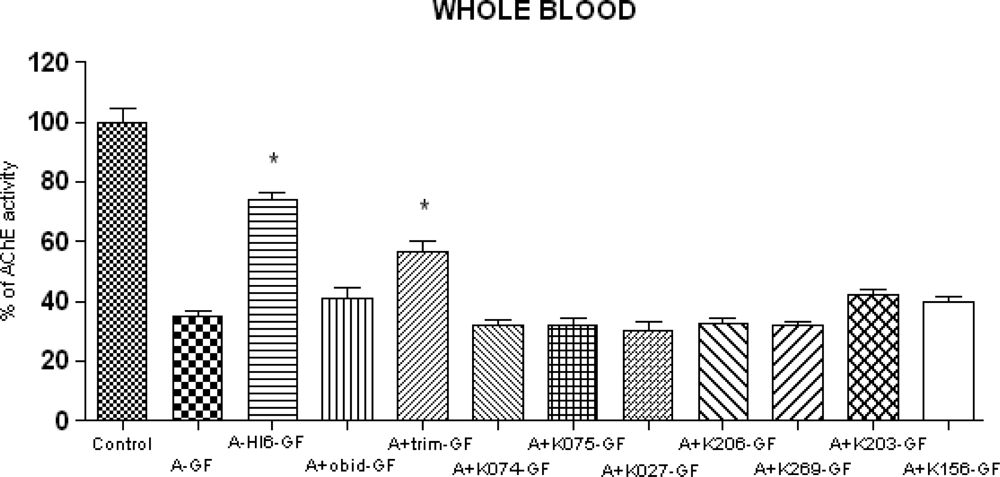
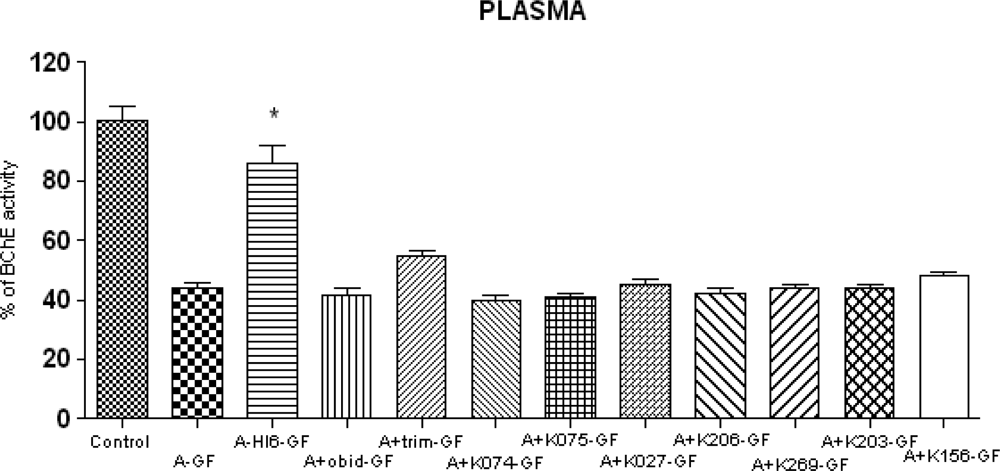
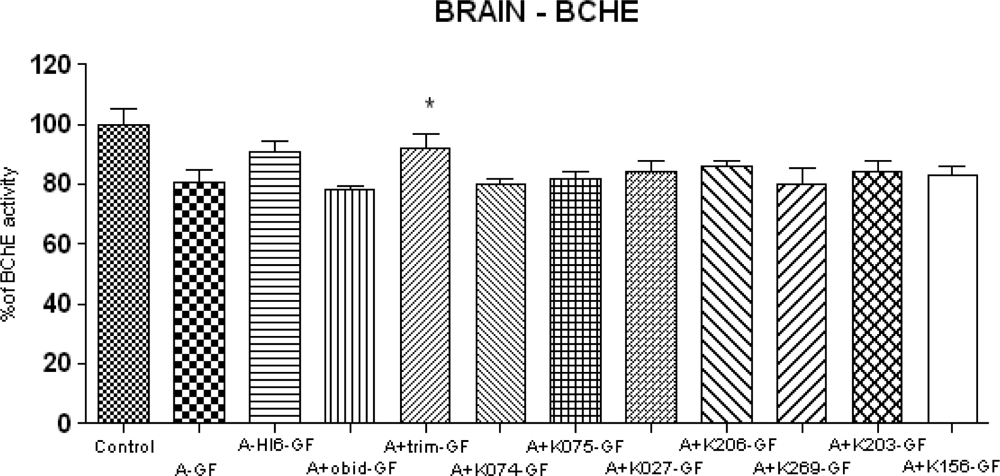
2. Results and Discussion
3. Experimental Section
3.1. Chemicals
3.2. Animals
3.3. Dosing and Sample Collection
3.4. Biochemical Examinations
3.5. Statistical Evaluation
4. Conclusions
Acknowledgments
References and Notes
- Jokanovic, M; Prostran, M. Pyridinium oximes as cholinesterase reactivators. Structure-activity relationship and efficacy in the treatment of poisoning with organophosphorus compounds. Curr. Med. Chem 2009, 16, 2177–2188. [Google Scholar]
- Marrs, TC. Organophosphate poisoning. Pharmacol. Ther 1993, 58, 51–66. [Google Scholar]
- Dawson, RM. Review of oximes available for treatment of nerve agent poisoning. J. Appl. Toxicol 1994, 14, 317–331. [Google Scholar]
- Marrs, TC; Rice, P; Vale, JA. The role of oximes in the treatment of nerve agent poisoning in civilian casualties. Toxicol Rev 2006, 25, 297–323. [Google Scholar]
- Reiter, G; Koller, M; Thiermann, H; Dorandeu, F; Mikler, J; Worek, F. Development and application of procedures for the highly sensitive quantification of cyclosarin enantiomers in hemolysed swine blood samples. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci 2007, 859, 9–15. [Google Scholar]
- Maxwell, DM; Koplovitz, I; Worek, F; Sweeney, RE. A structure-activity analysis of the variation in oxime efficacy against nerve agents. Toxicol Appl Pharmacol 2008, 231, 157–164. [Google Scholar]
- Kovarik, Z; Calic, M; Sinko, G; Bosak, A; Berend, S; Vrdoljak, AL; Radic, B. Oximes: Reactivators of phosphorylated acetylcholinesterase and antidotes in therapy against tabun poisoning. Chem. Biol. Interact 2008, 175, 173–179. [Google Scholar]
- Wilson, IB; Sondheimer, F. A specific antidote against lethal alkyl phosphate intoxication. V. Antidotal properties. Arch Biochem Biophys 1957, 69, 468–474. [Google Scholar]
- Kassa, J; Kuca, K; Karasova, J; Musilek, K. The development of new oximes and the evaluation of thein reactivating, therapeutic and neuroprotective efficacy against tabun. Mini Rev. Med. Chem 2008, 8, 1134–1143. [Google Scholar]
- Berend, S; Vrdoljak, AL; Radic, B; Kuca, K. New bispyridinium oximes: in vitro and in vivo evaluation of their biological efficiency in soman and tabun poisoning. Chem. Biol. Interact 2008, 175, 413–416. [Google Scholar]
- Kuca, K; Musilek, K; Paar, M; Jun, D; Stodulka, P; Hrabinova, M; Marek, J. Targeted synthesis of 1-(4-hydroxyiminomethylpyridinium)-3-pyridiniumpropane dibromide-a new nerve agent reactivator. Molecules 2007, 12, 1964–1974. [Google Scholar]
- Musilek, K; Jun, D; Cabal, J; Kassa, J; Gunn-Moore, F; Kuca, K. Design of a potent reactivator of tabun-inhibited acetylcholinesterase--synthesis and evaluation of (E)-1-(4-carbamoylpyridinium)-4-(4-hydroxyiminomethylpyridinium)-but-2-ene dibromide (K203). J Med Chem 2007, 50, 5514–5518. [Google Scholar]
- Musilek, K; Holas, O; Jun, D; Dohnal, V; Gunn-Moore, F; Opletalova, V; Dolezal, M; Kuca, K. Monooxime reactivators of acetylcholinesterase with (E)-but-2-ene linker: preparation and reactivation of tabun- and paraoxon-inhibited acetylcholinesterase. Bioorg. Med. Chem 2007, 15, 6733–6741. [Google Scholar]
- Musilek, K; Holas, O; Kuca, K; Jun, D; Dohnal, V; Opletalova, V; Dolezal, M. Synthesis of monooxime-monocarbamoyl bispyridinium compounds bearing (E)-but-2-ene linker and evaluation of their reactivation activity against tabun- and paraoxon-inhibited acetylcholinesterase. J. Enzyme Inhib. Med. Chem 2008, 23, 70–76. [Google Scholar]
- Kuca, K; Jun, D; Musilek, K. Structural requirements of acetylcholinesterase reactivators. Mini Rev. Med. Chem 2006, 6, 269–277. [Google Scholar]
- Bieger, D; Wasserman, O. Ionization constants of cholinesterase-reactivating bispyridinium aldoximes. J. Pharm. Pharmacol 1967, 19, 844–847. [Google Scholar]
- Kuca, K; Patocka, J. Reactivation of the cyclosarin-inhibited rat brain acetylcholinesterase by pyridinium-oximes. J. Enzyme Inhib. Med. Chem 2004, 19, 39–43. [Google Scholar]
- Zdarova Karasova, J; Kassa, J; Jung, YS; Musilek, K; Pohanka, M; Kuca, K. Effect of several new and currently available oxime cholinesterase reactivators on tabun-inhibited rats. Int J Mol Sci 2008, 9, 2243–2252. [Google Scholar]
- Kassa, J; Jun, D; Karasova, J; Bajgar, J; Kuca, K. A comparison of reactivating efficacy of newly developed oximes (K 074, K 075) and currently available oximes (obidoxime, HI-6) in soman, cyclosarin and tabun-poisoned rats. Chem. Biol. Interact 2008, 175, 425–427. [Google Scholar]
- Kassa, J; Cabal, J. A comparison of the efficacy of a new asymmetric bispyridium oxime BI-6 with currently available oximes and H oximes against soman by in vitro and in vivo methods. Toxicology 1999, 132, 111–118. [Google Scholar]
- Patocka, J; Cabal, J; Kuca, K; Jun, D. Oxime reactivation of acetylcholinesterase inhibited by toxic organophosphorus ester: In vitro kinetics and thermodynamics. J. Appl. Biomed 2005, 3, 91–99. [Google Scholar]
- Kuca, K; Bielavsky, J; Cabal, J; Bielavska, M. Synthesis of a potential reactivator of acetylcholinesterase 1-(4-hydroxyiminomethylpyridinium)-3-(carbamoyl-pyridinium)-propane dibromide. Tetrahedron Lett 2003, 44, 3123–3125. [Google Scholar]
- Bajgar, J. Biological monitoring of exposure to nerve agents. Br. J. Ind. Med 1992, 49, 648–653. [Google Scholar]
- Bartosova, L; Kuca, K; Kunesova, G; Jun, D. The acute toxicity of the acetylcholinesterase reactivators in mice in relation to their structure. Neurotox. Res 2006, 9, 291–296. [Google Scholar]
- Tallarida, R; Murray, R. Manual of pharmacological calculation with computer programs, 2nd ed; Springer-Verlag: New York, NY, USA, 1987; p. 145. [Google Scholar]
- Zdarova Karasova, J; Kuca, K; Jun, D; Bajgar, J. Using of Ellman method for In vivo testing of cholinesterases' activities (In Czech). Chem Listy 2009, in press.. [Google Scholar]
- Ellman, GL; Coutney, DK; Andres, V; Fear-Stone, RM. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 1961, 7, 88–95. [Google Scholar]
- Pohanka, M; Jun, D; Kuca, K. Improvement of acetylcholinesterase-based assay for organophosphates in way of identification by reactivators. Talanta 2008, 77, 451–454. [Google Scholar]

| OXIME REACTIVATOR | DOSE CORRESPONING TO 5 % OF LD50 |
|---|---|
| HI-6 | 39.0 mg/kg |
| Obidoxime | 10.5 mg/kg |
| Trimedoxime | 7.5 mg/kg |
| K206 | 19.3 mg/kg |
| K269 | 5.7 mg/kg |
| K203 | 16.3 mg/kg |
| K074 | 23.0 mg/kg |
| K075 | 22.9 mg/kg |
| K027 | 22.3 mg/kg |
| K156 | 6.4 mg/kg |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Karasova, J.Z.; Kassa, J.; Musilek, K.; Pohanka, M.; Novotny, L.; Kuca, K. Effect of Seven Newly Synthesized and Currently Available Oxime Cholinesterase Reactivators on Cyclosarin-Intoxicated Rats. Int. J. Mol. Sci. 2009, 10, 3065-3075. https://doi.org/10.3390/ijms10073065
Karasova JZ, Kassa J, Musilek K, Pohanka M, Novotny L, Kuca K. Effect of Seven Newly Synthesized and Currently Available Oxime Cholinesterase Reactivators on Cyclosarin-Intoxicated Rats. International Journal of Molecular Sciences. 2009; 10(7):3065-3075. https://doi.org/10.3390/ijms10073065
Chicago/Turabian StyleKarasova, Jana Zdarova, Jiri Kassa, Kamil Musilek, Miroslav Pohanka, Ladislav Novotny, and Kamil Kuca. 2009. "Effect of Seven Newly Synthesized and Currently Available Oxime Cholinesterase Reactivators on Cyclosarin-Intoxicated Rats" International Journal of Molecular Sciences 10, no. 7: 3065-3075. https://doi.org/10.3390/ijms10073065




