Structural Antitumoral Activity Relationships of Synthetic Chalcones
Abstract
:1. Introduction
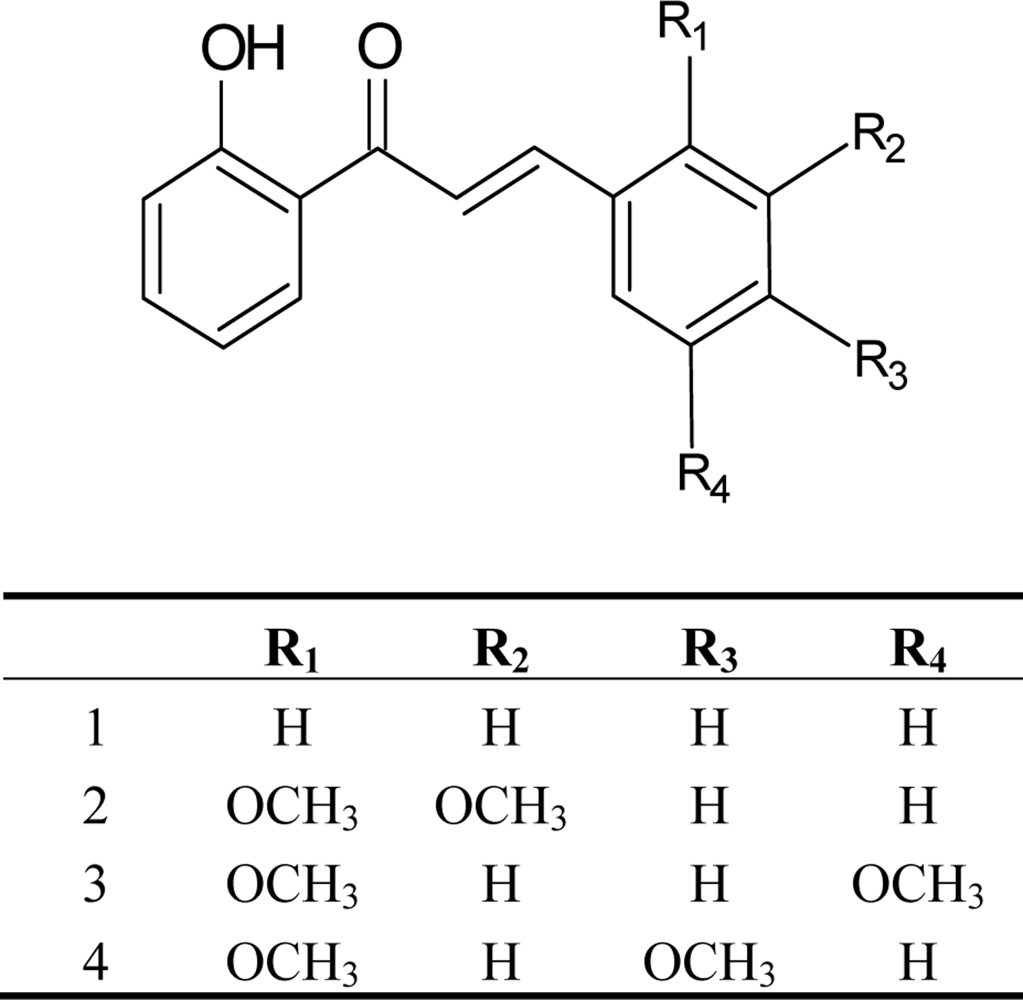
2. Results and Discussion
3. Experimental Section
3.1. Chemicals
3.2. Cell Culture
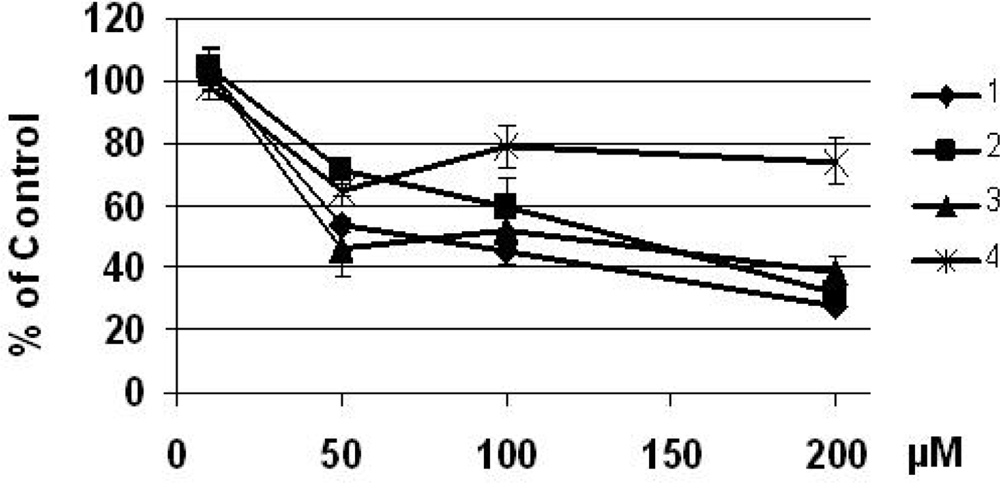
3.3. Cell growth assay
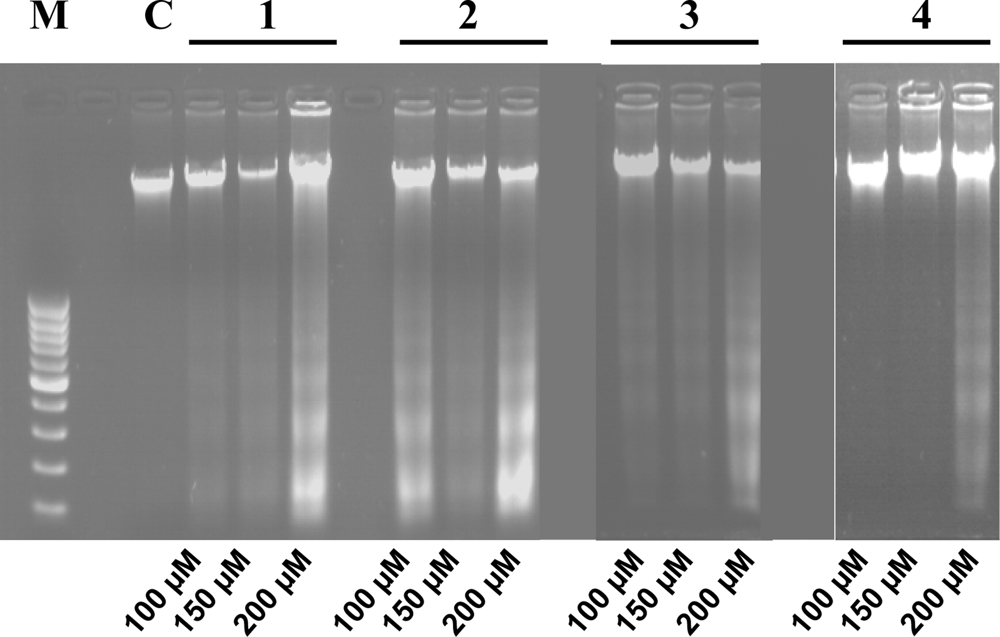
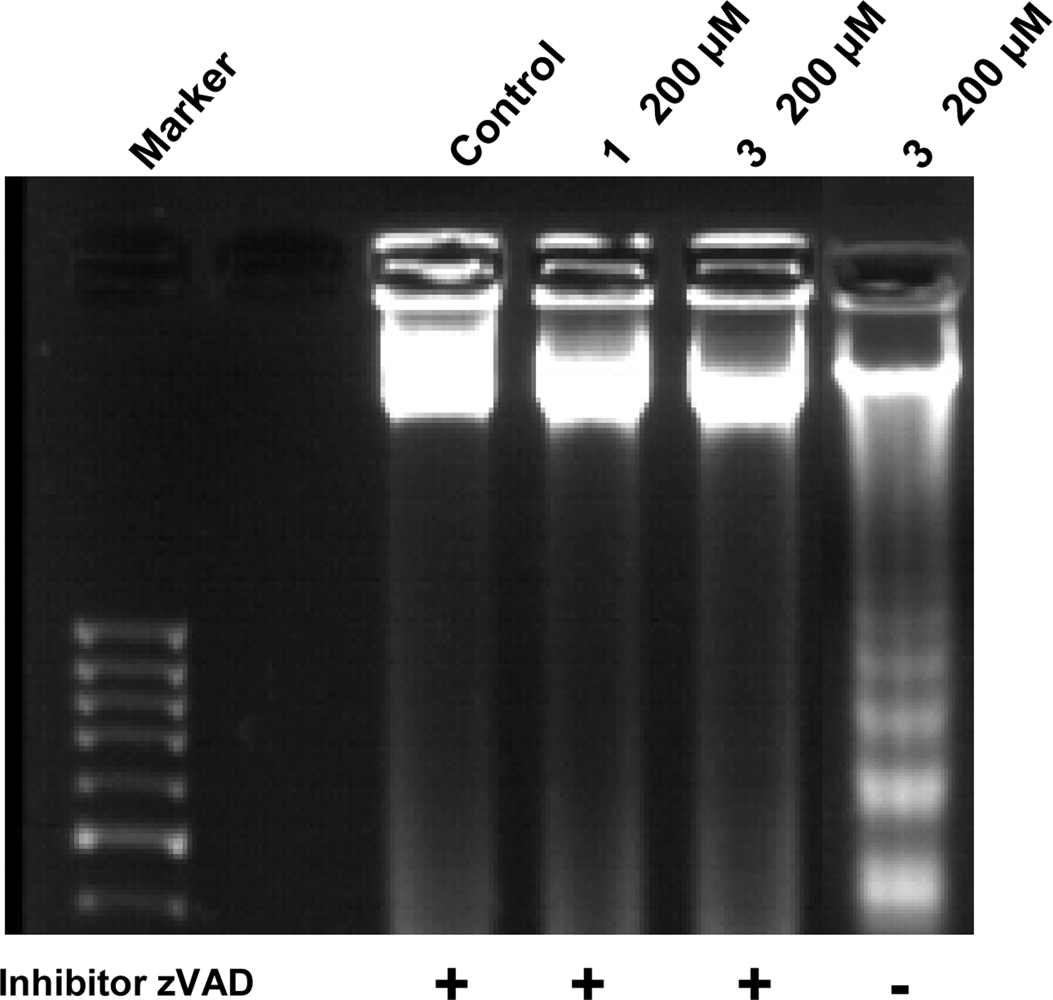
3.4. Internucleosomal DNA fragmentation assay
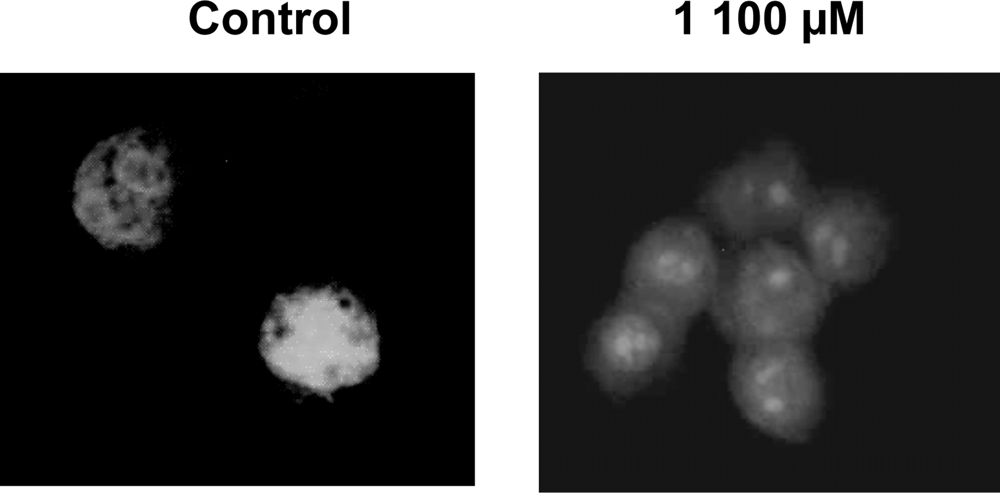
3.5. Nuclear DNA condensation assay
3.6. Activity Caspase-9
3.7. Section model equations
3.8. Computational Details
4. Conclusions
Acknowledgments
References and Notes
- Kuo, S. Dietary flavonoids and cancer prevention: Evidence and potentials mechanisms Oncogenesis 1997, 8, 47–69.
- Mark, C; Nagarathnam, D. Cytotoxicities of some flavonoid analogues. J. Nat. Prod 1991, 54, 1656–1660. [Google Scholar]
- Dharn, DN. The Chemistry of Chalcones and Related Compounds; Wiley: New York, 1981; p. 213. [Google Scholar]
- Torigoo, T; Arisawa, M; Iloch, S; Fujiu, M; Mayuyama, HB. Anti-mutagenic chalcones: Antagonizing the mutagenicity of benzo(a)pyrene in Salmonella typhymurium. Biophys. Res. Commun 1983, 112, 833–842. [Google Scholar]
- Sukumaran, K; Kuttan, R. Screening of 11 ferns of cytotoxic and antitumor potential with special reference to Pityrogramma calomelanos. J. Ethnopharmacol 1991, 36, 93–96. [Google Scholar]
- Calliste, C; Le Bail, J; Trouilas, P; Pouget, C; Habrious, G; Chulea, A; Duroux, JL. Chalcones: structural requirements for antioxidant, estrigenic and antiproliferative activities. J. Anticancer Res 2001, 21, 3949–3956. [Google Scholar]
- Won, S; Liu, C; Tsao, L; Weng, J; Ko, H; Wang, J; Lin, C. Synthetics chalcones as potential anti-inflammatory and cancer chemopreventive agents. Europ. J. Med. Chem 2005, 40, 103–112. [Google Scholar]
- De Vincenazo, R; Ferlini, C; Distefano, M; Gaggini, C; Riva, A; Bombardelli, E; Morazzoni, P; Valenti, P; Belluti, F; Ranelletti, FO; Mancuso, S; Scambia, G. In vitro evaluation of newly developed chalcone analogues in human cancer cells. Cancer Chemother. Pharmacol 2000, 46, 305–312. [Google Scholar]
- Saydam, G; Aydin, H; Sahin, F; Kucukogin, O; Erciyas, E; Terzioglu, E; Buyukkececi, F; Omay, S. Cytotoxic and inhibitory effects os 4,4’-dihydrochalcone RVC-588 on proliferation of human leukemic HL-60 cells. Leuk. Res 2003, 27, 57–64. [Google Scholar]
- Sabzevari, O; Galati, G; Moridani, M; Siraki, A; Obrien, P. Molecular cytotoxic mechanisms of anticancer hydrochalcones. Chem. Biol. Interact 2004, 148, 57–67. [Google Scholar]
- Edwards, M; Stemerick, D; Sunkara, P. Chalcones: A new class of antimitotic agents. J. Med. Chem 1990, 33, 1948–1954. [Google Scholar]
- Tanigawa, S; Fujii, M; Hou, D. Stabilization of p53 is involved in Quercetin-induced Cell Cycle Arrest and apoptosis in HepG2 cells. Biosci. Biotechnol. Biochem 2008, 72, 797–804. [Google Scholar]
- Allen, P; Newland, A. Electrophoresis DNA analysis for the detection of apoptosis. Mol. Biotechnol 1998, 9, 247–251. [Google Scholar]
- Kohler, C; Orrenius, S. Evaluation of caspase activity in apoptotic cells. J. Immunol. Meth 2002, 265, 97–110. [Google Scholar]
- Fernandes, R; McGowan, A; Cotter, T. Mutant Ha-Ras overexpression inhibits drug and U.V. induced apoptosis. Anticancer Res 1996, 16, 1691–1706. [Google Scholar]
- Parr, R; Pearsons, R. Absolute hardness: Companion parameter to absolute electronegativity. J. Am. Chem. Soc 1983, 105, 7512–7516. [Google Scholar]
- Parr, R; Szentpaly, L; Liu, S. Electrophilicity Index. J. Am. Chem. Soc 1999, 121, 1922–1924. [Google Scholar]
- Amsterdam Density Functional (ADF) code, release 2007; Vrije Universiteit: Amsterdam, The Netherlands.
- Te Velde, G; Bickelhaupt, FM; Van Gisberger, SJA; Fonseca Guerra, C; Baerends, EJ; Snijders, JG; Ziegler, TJ. Chemistry with ADF. Comput. Chem 2001, 22, 931–967. [Google Scholar]
- Verluis, L; Ziegler, T. The determination of molecular structures by density functional theory. The evaluation of analytical energy gradients by numerical integration. J. Chem. Phys 1988, 88, 322–328. [Google Scholar]
- Vosko, SH; Wilk, L; Nusair, M. Accurate spin-dependent electron liquid correlation energies for local spin density calculations: A critical analysis. Can. J. Phys 1980, 58, 1200–1211. [Google Scholar]
- Klamt, A. Conductor-like screening model for real solvents: A new approach to the quantitative calculation of salvation phenomena. J. Chem. Phys 1995, 99, 2224–2235. [Google Scholar]
- Klamt, A; Jonas, V. Treatment of the outlying charge in continuum salvation models. J. Chem. Phys 1996, 105, 9972–9981. [Google Scholar]
- Quintana, P; Yáñez, C; Escobar, CA; Sicker, D; Araya-Maturana, R; Squella, JA. Electrochemical approach to the radical anion formation from 2’-hydroxy chalcone derivatives. Electroanalysis 2006, 18, 521–525. [Google Scholar]
- Thornberry, N; Lazebnik, Y. Caspases: Enemies within. Science 1998, 281, 1312–1316. [Google Scholar]
- Kobori, M; Iwashita, K; Shinmoto, H; Tsushida, T. Phloretin-induced apoptosis in B16 melanoma 4A5 cells and HL60 human leukemia cells. Biosci. Biotechnol. Biochem 1999, 63, 719–725. [Google Scholar]
- Mancini, M; Nicholson, D; Roy, S. The caspase-3 precursor has a cytosolic and mitochondrial distribution: Implication for apoptotic signaling. J. Cell Biol 1998, 140, 1485–1495. [Google Scholar]
- Perez, P; Toro-Labbe, A; Aizman, A; Contreras, R. Comparison between experimental and theoretical scales of electrophilicity in benzhydryl cations. J. Org. Chem 2002, 67, 4747–4752. [Google Scholar]
- Li, X; Cai, ZI; Sevilla, MD. DFT Calculations of the electron affinities of nucleic acid bases: dealing with negative electron affinities. J. Phys. Chem. A 2002, 106, 1596–1603. [Google Scholar]
- Javitt, N. HepG2 cells as a resource for metabolic studies: lipoprotein, cholesterol, and bile acids. FASEB J 1990, 4, 161–168. [Google Scholar]
- Sakahira, H; Enari, M; Ohsawa, Y; Uchiyama, Y; Nagata, S. Apoptotic nuclear morphological change without DNA fragmentation. Curr. Biol 1999, 20, 543–546. [Google Scholar]

| Compound Nº | η | μ | ω | AEA |
|---|---|---|---|---|
| 1 | 84.63 | −42.20 | 142.28 | 72.52 |
| 2 | 83.71 | −41.74 | 137.21 | 73.40 |
| 3 | 78.41 | −39.20 | 113.69 | 73.96 |
| 4 | 75.87 | −37.81 | 102.39 | 69.64 |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/). This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Echeverria, C.; Santibañez, J.F.; Donoso-Tauda, O.; Escobar, C.A.; Ramirez-Tagle, R. Structural Antitumoral Activity Relationships of Synthetic Chalcones. Int. J. Mol. Sci. 2009, 10, 221-231. https://doi.org/10.3390/ijms10010221
Echeverria C, Santibañez JF, Donoso-Tauda O, Escobar CA, Ramirez-Tagle R. Structural Antitumoral Activity Relationships of Synthetic Chalcones. International Journal of Molecular Sciences. 2009; 10(1):221-231. https://doi.org/10.3390/ijms10010221
Chicago/Turabian StyleEcheverria, Cesar, Juan Francisco Santibañez, Oscar Donoso-Tauda, Carlos A. Escobar, and Rodrigo Ramirez-Tagle. 2009. "Structural Antitumoral Activity Relationships of Synthetic Chalcones" International Journal of Molecular Sciences 10, no. 1: 221-231. https://doi.org/10.3390/ijms10010221
APA StyleEcheverria, C., Santibañez, J. F., Donoso-Tauda, O., Escobar, C. A., & Ramirez-Tagle, R. (2009). Structural Antitumoral Activity Relationships of Synthetic Chalcones. International Journal of Molecular Sciences, 10(1), 221-231. https://doi.org/10.3390/ijms10010221