The Synthesis of Dicationic Extended Bis-Benzimidazoles
Abstract
:Introduction
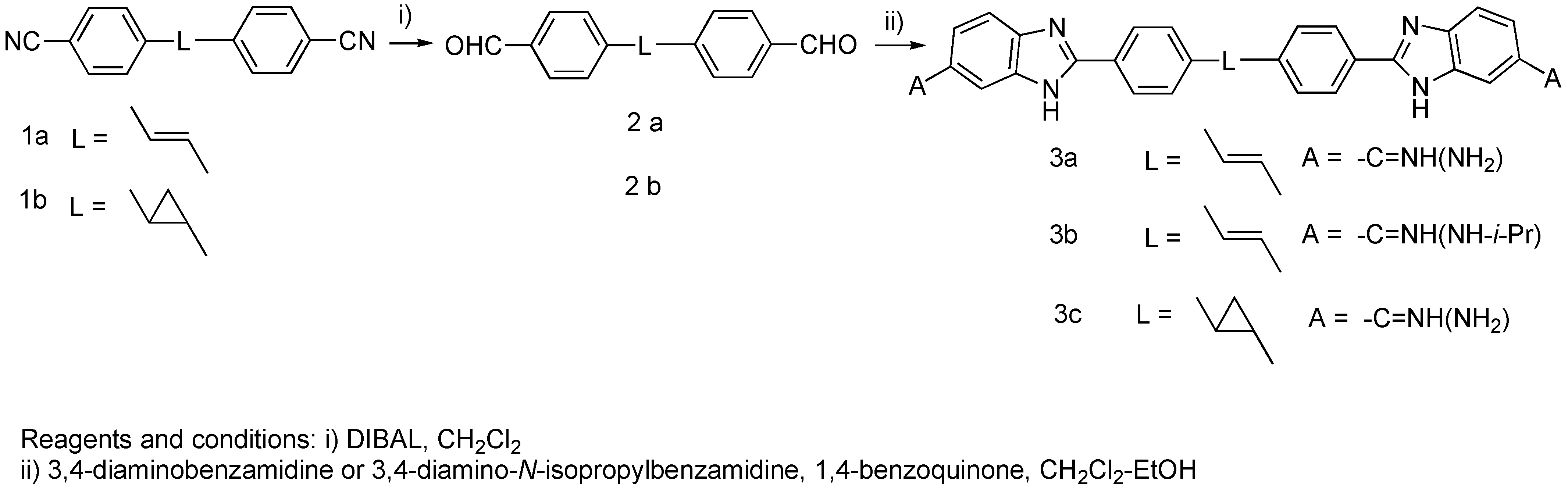
Results and Discussion
| Compound | MIC/MBC B. subtilis N10 | Cell toxicity CV1 IC50 | Therapeutic Index (IC50/MIC) |
| 3a | <0.4/ND | ND | - |
Conclusions
Experimental
General
MIC, MBC and in vitro cell toxicity determinations.
References
- Tidwell, R. R.; Boykin, D. W. Dicationic DNA Minor Groove Binders as Antimicrobial Agents, in Small Molecule DNA and RNA Binders: From Synthesis to Nucleic Acid Complexes. Demeunynck, M., Bailly, C., Wilson, W. D., Eds.; Wiley-VCH: New York, 2003; Vol 2, pp. 414–460. [Google Scholar]
- Tidwell, R. R.; Jones, S. K.; Naiman, N. A.; Berger, I. C.; Brake, W. R.; Dykstra, C. C.; Hall, J. E. Activity of dicationically substituted bis-benzimidazoles against experimental Pneumocystis carinii pneumonia. Antimicrob. Agents Chemother. 1993, 3, 1713–1716. [Google Scholar]
- Hopkins, K.; Wilson, W. D.; Bender, B.; McCurdy, D.; Hall, J.E; Tidwell, R. R.; Kumar, A.; Bajic, M.; Boykin, D.W. Extended Aromatic Furan Amidino Derivatives as Anti-Pneumocystis carinii Agents. J. Med. Chem. 1998, 41, 3872–3878. [Google Scholar]
- Del Poeta, M.; Schell, W. A.; Dykstra, C. C.; Jones, S.; Tidwell, R. R.; Czarny, A.; Bajic, M.; Kumar, A.; Boykin, D. W.; Perfect, J. R. Structure-In Vitro Activity Relationships of Pentamidine Analogues and Dication-Substituted Bis-Benzimidazoles as New Antifungal Agents. Antimicrob. Agents Chemother. 1998, 42, 2495–2502. [Google Scholar]
- Del Poeta, M.; Schell, W.A.; Dykstra, C.C.; Jones, S.; Tidwell, R.R.; Kumar, A.; Boykin, D.W.; Perfect, J.R. In Vitro Antifungal Activities of a Series of Dicationic-Substituted Carbazoles, Furans, and Benzimidazoles. Antimicrob. Agents Chemother. 1998, 42, 2503–2509. [Google Scholar]
- Ismail, M.A.; Brun, R.; Tanious, F .A.; Wilson, W. D.; Boykin, D. W. Synthesis and Anti-Protozoal Activity of Aza-Analogs of Furamidine. J. Med. Chem. 2003, 46, 4761–4769. [Google Scholar]
- Givens, M. D.; Dykstra, C. C.; Brock, K. V.; Stringfellow, D.A.; Kumar, A.; Stephens, C. E.; Goker, H.; Boykin, D. W. In Vitro Inhibition of Replication of Bovine Viral Diarrhea Virus by Aromatic Cationic Molecules. Antimicrob. Agents Chemother. 2003, 47, 2223–2230. [Google Scholar]
- Bance, S.; Barber, H. J.; Woolman, A. M. Bromination of diphenylalkanes and the preparation of some stilbene derivatives. J. Chem. Soc. 1943, 1–4. [Google Scholar]
- Dann, O.; Hieke, E.; Hahn, H.; Miserre, H.H.; Luerding, G.; Roessler, R. Polynuclear thiophenes VII. Trypanocidal diamidines of 2-phenylthionaphththene. Ann. Chem. 1970, 734, 23–45. [Google Scholar]
- Fairly, T. A.; Tidwell, R. R.; Donkor, I.; Naiman, N.A.; Ohemeng, K. A.; Lombardy, R. J.; Bentley, J.; Cory, M. Struture, DNA minor groove binding and base pair specificity of alkyl- and aryl linked bis(amidinobenzimidazoles) and bis(amidinoindoles). J. Med. Chem. 1993, 36, 1746–1753. [Google Scholar]
- Patrick, D. G.; Boykin, D. W.; Spychala, J.; Wilson, W. D.; Bender, B.K.; Hall, J. E.; Tidwell, R. R. Anti-pneumocystis carinii pneumonia activity of dicationic carazoles. Eur. J. Med. Chem. 1997, 32, 781–793. [Google Scholar]
- Hamada, M. The preparation and properties of 1,2-diphenyl cyclopropanes. Ann. Chem. 1951, 22–28. [Google Scholar]
- Sambrook, J.; Fritchsch, E.F.; Maniatis, T. Molecular Cloning: A Laboratory Manual, 2nd ed; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, N.Y, 1989. [Google Scholar]
- Sample Availability: Not Available
© 2004 by MDPI (http:www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Kang, Z.; Dykstra, C.C.; Boykin, D.W. The Synthesis of Dicationic Extended Bis-Benzimidazoles. Molecules 2004, 9, 158-163. https://doi.org/10.3390/90300158
Kang Z, Dykstra CC, Boykin DW. The Synthesis of Dicationic Extended Bis-Benzimidazoles. Molecules. 2004; 9(3):158-163. https://doi.org/10.3390/90300158
Chicago/Turabian StyleKang, Zhijan, Christine C. Dykstra, and David W. Boykin. 2004. "The Synthesis of Dicationic Extended Bis-Benzimidazoles" Molecules 9, no. 3: 158-163. https://doi.org/10.3390/90300158




