Abstract
A rapid and simple method for the preparation of 1-ethoxycarbonyl-4-substituted-semicarbazides has been developed. As examples the reaction of six different isocyanates with ethyl carbazate under solvent-free conditions are reported.
Introduction
1-Ethoxycarbonyl-4-substituted-semicarbazides are important organic intermediates for the synthesis of 4-substituted-urazoles, which have usually been prepared by the reaction of the corresponding isocyanates with ethyl carbazate in benzene or toluene solution [1]. The reported methods, however, invariably require long reaction times, low temperatures, and involve large amount of solvents, all of which increase the costs of production. Therefore, the aim of this work was to prepare a wide variety of the semicarbazides under solvent-free conditions.
In recent years, there has been increasing interest in reactions that proceed in the absence of solvents due to reduced pollution, lower costs and simplicity in processing and handling [2]. In continuation of our ongoing program to develop such solvent-free reactions [3], we wish to report here the preparation of semicarbazides from ethyl carbazate and isocyanates under solvent-free conditions. As a model reaction we first decided to investigate the preparation of 1-ethoxycarbonyl-4-phenyl-semicarbazide (2) from ethyl carbazide and phenylisocyanate (1) under solvent-free conditions. The progress of the reaction was monitored by TLC and it was observed that the reaction was complete in a short period of time (ca. 20 min.). A variety of semicarbazides (both aliphatic and aromatic) were then prepared upon simple mixing of ethyl carbazate and the corresponding aliphatic and aromatic isocyanates in a mortar and grinding the mixture with a pestle at room temperature for the time specified in Table 1 (Scheme 1). The reaction yields are high and the reaction times are exceedingly short (20-30 min). The products were isolated by washing the reaction mixtures with small amounts of solvents like n-hexane or toluene followed by filtration. In this method, the optimum molar ratio of ethyl carbazate to isocyanate for complete preparation of semicarbazides was determined to be 1:1.
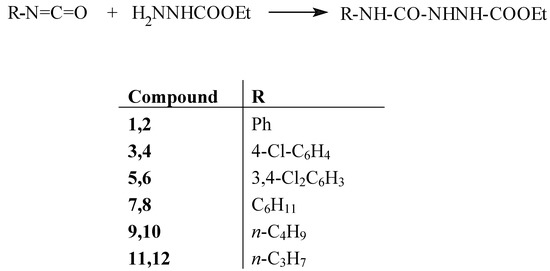
Scheme 1.

Table 1.
Solid-state synthesis of 1-ethoxycarbonyl-4-substituted-semicarbazides
| Substrate | Product | Time (min) | Yield (%)a | mp (oC)/(Lit) |
|---|---|---|---|---|
| 1 | 2 | 20 | 95 | 144-145 ([1a]: 146-147) |
| 3 | 4 | 30 | 87 | 193-195 ([1d]: 202-204) |
| 5 | 6 | 30 | 96 | 206-208 ([1e]: 212-213.5) |
| 7 | 8 | 20 | 95 | 115-117 ([1d]: 116-118) |
| 9 | 10 | 20 | 94 | 96-98 ([1d]: 108-109) |
| 11 | 12 | 20 | 88 | 94-96 ([1d]: 110) |
aYields of isolated pure products after purification.
Conclusions
In this study we have presented a simple method for the preparation of different types of 1-ethoxycarbonyl-4-substituted-semicarbazides from ethyl carbazate and the corresponding isocyanates under solvent-free conditions. This method is superior to previously reported ones in terms of high yields, facile work-up, short reaction times, and the cost of the solvents have been minimized.
Experimental
General
Yields refer to isolated products after purification. All reactions were carried out under solvent-free conditions in a hood with strong ventilation. Ethyl carbazate and isocyanates were purchased from Merck Co. Reactions were followed using silica gel 60 F254 aluminium TLC plates and ethyl acetate/cyclohexane (50:50) as solvent mixture. The products were characterized by comparison of their spectral (IR, UV, 1H-NMR) and physical data with that of authentic samples [1]. All 1H-NMR spectra were recorded at 90 MHz in DMSO-d6 relative using TMS as an internal standard. IR spectra were recorded on Shimadzu 435 IR spectrometer.
Typical Procedure: Preparation of 1-Ethoxycarbonyl-4-phenyl Semicarbazide (2) from Ethyl Carbazate and Phenyl Isocyanate (1)
A mixture of ethyl carbazate (0.104 g, 1 mmol) and phenyl isocyanate (1, 0.119 g, 1 mmol) placed in a mortar was ground with a pestle for the time specified in Table 1. When TLC showed complete disappearance of starting material, the mixture was washed with dry toluene or n-hexane and the solids were filtered off and dried to give 1-ethoxycarbonyl-4-phenyl semicarbazide (2). The yield was 0.212 g (95%) of crystalline white solid, mp 144-145oC (lit. [1a] mp 146-147 oC). IR (KBr): 3360 (s), 3200 (s), 3050 (m), 2980 (m), 1710 (s), 1665 (s), 1600 (s), 1545 (s), 1500 (m), 1475 (m), 1440 (m), 1370 (w), 1310 (s), 1130 (s), 1050 (m), 1010 (m), 890 (m), 750 (m), 630 (m, br) cm-1. 1H-NMR: δ 1.19 (t, 3H, J = 7.0 Hz), 4.07 (q, 2H, J = 7.0 Hz), 7.19 (m, 5H), 8.01 (s, 1H), 8.73 (s, 1H), 8.93 (s, 1H) ppm.
Acknowledgments
We are thankful to the Isfahan University of Technology (IUT) Research Council for the partial support of this work. Further financial support from Center of Excellency in Chemistry Research (IUT) is gratefully acknowledged.
References
- Mallakpour, S.E. J. Chem. Ed. 1992, 69, 238.Mallakpour, S.E.; Butler, G. B.; Aghabozorg, H.; Palenik, G. J. Macromolecules 1985, 18, 342.Mallakpour, S.E.; Zolfigol, M. A. J. Sci. I. R. Iran 1993, 4, 199.Arya, V.P.; Shenoy, S.J. Indian J. Chem. 1976, 14B, 883.Raheno, V.P.; Shenoy, H. M.Sc. Thesis, Isfahan University of Technology, 2002; p. 29.
- Toda, F. Acc. Chem. Res. 1995, 28, 480.Toda, F.; Tanaka, K. Chem. Rev. 2000, 100, 1025.
- Hajipour, A.R.; Mallakpour, S.E.; Khoee, S. Chem. Lett. 2000, 120.Hajipour, A.R.; Mallakpour, S.E.; Imanzadeh, Gh. Indian J. Chem. 2001, 40B, 237.Hajipour, A.R.; Mallakpour, S.E.; Afrousheh, A. Phosphorous, Sulfur and Silicon 2000, 67, 160.Hajipour, A.R.; Mallakpour, S.E.; Khoee, S. Synlett 2000, 740.Hajipour, A.R.; Mallakpour, S.E.; Afrousheh, A. Tetrahedron 1999, 55, 2311.Hajipour, A.R.; Mallakpour, S.E.; Mohammadpour-Baltork, I.; Khoee, S. Synth. Commun. 2001, 31, 1138.Hajipour, A.R.; Mallakpour, S.E.; Adibi, H. Chem. Lett. 2000, 460.
- Sample availability: Samples of compounds 2, 4, 6, 8, 10 and 12 are available from MDPI.
© 2003 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.