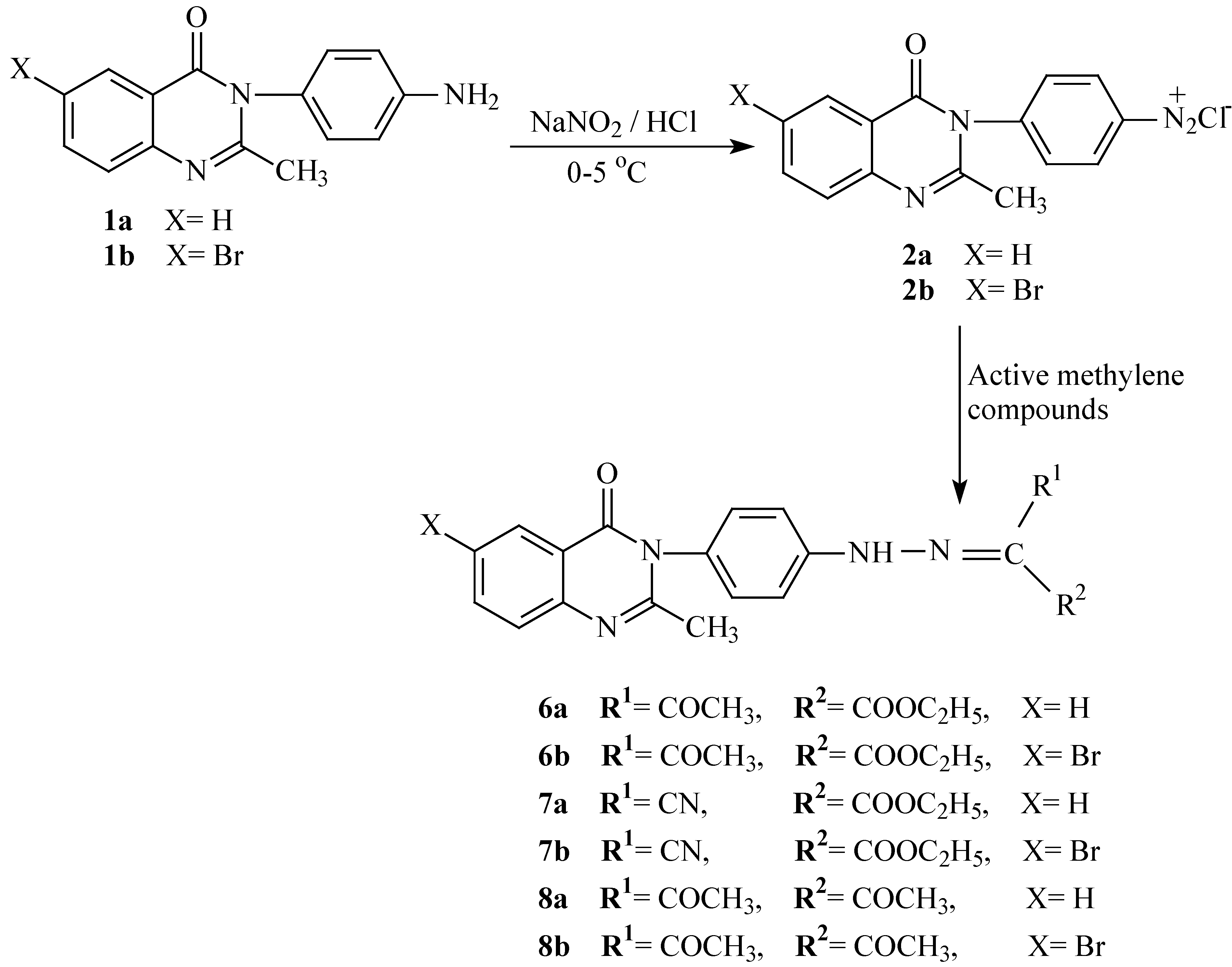
General procedure for the preparation of hydrazono derivatives 6-8
To an ice-cold mixture of the appropriate active methylene compound (ethyl acetoacetate, ethyl cyanoacetate or acetylacetone) (0.01 mole) and sodium acetate (4.10 g; 0.05 mole) in ethanol (50 mL), was added dropwise with stirring a solution of diazonium salt compound 2a or 2b (0.01 mole) over 15 minutes. The stirring was continued for 30 minutes and the reaction mixture then left for 2 hours at room temperature. The solid product was collected and recrystallized from ethanol to give the corresponding hydrazono derivatives 6-8. The following hydrazones were prepared in this manner:
2-{[4-(2-Methyl-4-oxo-4H-quinazolin-3-yl)-phenyl]hydrazono}-3-oxo-butyric acid ethyl ester (6a). Pale yellow crystals; yield 2.04 g (52 %); M.p. 190-192oC; Calculated for C21H20N4O4 (392.41): 64.28 % C, 5.14 % H, 14.28 % N. Found: 64.39 %, 5.32 % H, 13.98 % N; IR, υ cm-1: 3431 (NH), 3089, 2968 (Ar-H), 1773, 1713, 1649 (C=O), 1608, 1603, 1524, 1479 (C=N, C=C ); 1H-NMR δ: 11.60 (1H, s, NH), 7.41-8.10 (8H, m, Ar-H), 4.31 (2H, q, CH2), 2.40 (3H, s, COCH3), 2.15 (3H, s, quinazolinone CH3), 1.28 (3H, s, carboxylic ester CH3); 13C-NMR δ: 167.3 (acetyl C=O), 164.1 (ester C=O), 161.4 (C-4), 154.5 (C-2), 147.3 (C-8a), 142.8 (Ph C-1), 134.5 (C-7), 129.6 (2C, Ph C2,6), 128.5 (Ph C-4), 126.6 (C-5), 126.4 (C-8), 126.3 (2C, Ph C3,5), 126.2 (C-6), 118.9 (C-4a), 61.2 (CH2), 23.9 (quinazolinone CH3), 21.4 (acetyl CH3), 13.9 (ester CH3).
2-{[4-(6-Bromo-2-methyl-4-oxo-4H-quinazolin-3-yl)-phenyl]hydrazono}-3-oxo-butyric acid ethyl ester (6b). Pale yellow crystals; yield 2.73 g (58 %); M.p. 234-236oC; Calculated for C21H19BrN4O4 (471.31): 53.52 % C, 4.06 % H, 11.89 % N. Found: 53.63 %, 3.88 % H, 11.67 % N; IR, υ cm-1: 3442 (NH), 3080, 3048 (Ar-H), 1768, 1718, 1676 (C=O), 1608, 1607, 1548, 1460 (C=N, C=C ); 1H-NMR δ: 11.40 (1H, s, NH), 7.46-8.57 (7H, m, Ar-H), 4.24 (2H, q, CH2), 2.42 (3H, s, COCH3), 2.28 (3H, s, quinazolinone CH3), 1.27 (3H, s, ester CH3); MS, m/z (Ir,%): 472 (M+ for 81Br, 32.43), 470 (M+ for 79Br, 32.62), 316 (52.12), 314 (51.85), 301 (12.32), 299 (13.14), 224 (15.37), 222 (16.00).
Cyano-{[4-(2-methyl-4-oxo-4H-quinazolin-3-yl)-phenyl]hydrazono}acetic acid ethyl ester (7a). Pale yellow crystals; yield 2.44 g (65 %); M.p. 128-130oC; Calculated for C20H17N5O3 (375.38): 63.99 % C, 4.56 % H, 18.66 % N. Found: 63.78 %, 4.77 % H, 18.35 % N; IR, υ cm-1: 3430 (NH), 3115, 3032 (Ar-H), 2366 (CN), 1762, 1674 (C=O), 1605, 1603, 1545, 1458 (C=N, C=C ); 1H-NMR δ: 12.11 (1H, s, NH), 7.30-8.32 (8H, m, Ar-H), 4.30 (2H, q, CH2), 2.23 (3H, s, quinazolinone CH3), 1.22 (3H, s, carboxylic ester CH3); 13C-NMR δ: 163.8 (ester C=O), 160.7 (C-4), 154.9 (C-2), 147.6 (C-8a), 143.3 (C-1 of Ph), 134.7 (C-7), 128.8 (2C, Ph C2,6), 128.5 (Ph C-4), 127.0 (C-5), 126.6 (C-8), 126.4 (2C, Ph C3,5), 126.0 (C-6), 119.2 (C-4a), 117.3 (CN), 61.4 (CH2), 23.4 (quinazolinone CH3), 13.5 (ester CH3).
{[4-(6-Bromo-2-methyl-4-oxo-4H-quinazolin-3-yl)-phenyl]hydrazono}cyano-acetic acid ethyl ester (7b). Pale yellow crystals; yield 3.09 g (65 %); M.p. 173-175oC; Calculated for C20H16BrN5O3 (454.28): 52.88 % C, 3.55 % H, 15.42 % N. Found: 52.61 %, 3.83 % H, 15.09 % N; IR, υ cm-1: 3439(NH), 3110, 3000 (Ar-H), 2367 (CN), 1769, 1681 (C=O), 1607, 1602, 1547, 1450 (C=N, C=C ); 1H-NMR δ: 11.74 (1H, s, NH), 7.62-8.74 (7H, m, Ar-H), 4.33 (2H, q, CH2), 2.27 (3H, s, quinazolinone CH3), 1.28(3H, s, ester CH3); 13C-NMR δ: 164.1 (ester C=O), 162.5 (C-4), 158.4 (C-2), 142.8 (C-8a), 140.6(Ph C-1), 135.0 (C-7), 129.3 (2C, Ph C2,6), 128.8 (Ph C-4), 127.6 (C-5), 125.9 (C-8), 125.3 (2C, Ph C3,5), 125.1 (C-6), 120.6 (C-4a), 118.2 (CN), 61.7 (CH2), 23.7 (quinazolinone CH3), 13.8 (ester CH3); MS m/z (Ir,%): 455 (M+ for 81Br, 16.73), 453 (M+ for 79Br, 16.54).
3-{[4-(2-Methyl-4-oxo-4H-quinazolin-3-yl)phenyl]hydrazono}-pentane-2,4-dione (8a). Pale yellow crystals; yield 2.17 g (60 %); M.p. 153-155oC; Calculated for C20H18N4O3 (362.38): 66.29 % C, 4.97 % H, 15.46 % N. Found: 66.42 %, 5.01 % H, 15.62 % N; IR, υ cm-1: 3499 (NH), 3039, 2921 (Ar-H), 1712, 1662 (C=O), 1607, 1605, 1567, 1475 (C=N, C=C); 1H-NMR δ: 13.90 (1H, s, NH), 7.40-8.20 (8H, m, Ar-H), 2.44 (6H, s, 2 x COCH3), 2.25 (3H, s, quinazolinone CH3); 13C-NMR δ: 169.2 (2C, 2 x acetyl C=O), 161.4 (C-4), 155.2 (C-2), 147.2 (C-8a), 142.8 (Ph C-1), 135.0 (C-7), 128.2 (2C, Ph C2,6), 127.8 (Ph C-4), 127.2 (C-5), 126.8 (C-8), 126.6 (2C, Ph C3,5), 126.2 (C-6), 118.5 (C-4a), 23.7 (quinazolinone CH3); MS, m/z (Ir,%): 363 (M+ + 1, 19.38), 362 (M+, 78.74), 319 (20.95), 264 (42.12), 250 (36.46), 236 (31.91), 221 (4.66), 144 (17.06), 143 (100), 115 (4.43), 89 (4.9), 77 (7.53).
3-{[4-(6-Bromo-2-methyl-4-oxo-4H-quinazolin-3-yl)phenyl]hydrazono}-pentane-2,4-dione (8b). Pale yellow crystals; yield 2.91 g (66 %); M.p. 165-167 oC; Calculated for C20H17BrN4O3 (441.28): 54.44 % C, 3.88 % H, 12.70 % N. Found: 54.23 %, 4.02 % H, 12.45 % N; IR, υ cm-1: 3494 (NH), 3040, 2923 (Ar-H), 1718, 1665 (C=O), 1609, 1568, 1477 (C=N, C=C ); 1H-NMR δ: 13.75 (1H, s, NH), 7.43-8.16 (7H, m, Ar-H), 2.45 (6H, s, 2 x COCH3 ), 2.27 (3H, s, quinazolinone CH3); 13C-NMR δ: 168.7 (2C, 2 x acetyl C=O), 161.5 (C-4), 154.6 (C-2), 147.5 (C-8a), 142.2(Ph C-1), 135.4 (C-7), 128.3 (2C, Ph C2,6), 127.7 (Ph C-4), 127.4 (C-5), 127.0 (C-8), 126.5 (2C, Ph C3,5), 126.0 (C-6), 118.8 (C-4a), 23.6 (quinazolinone CH3); MS, m/z (Ir, %): 442 (M+ for 81Br, 32.18), 440 (M+ for 79Br, 32.12).
General Procedures for the Cyclization Reactions
Method A: With Hydrazine Hydrate
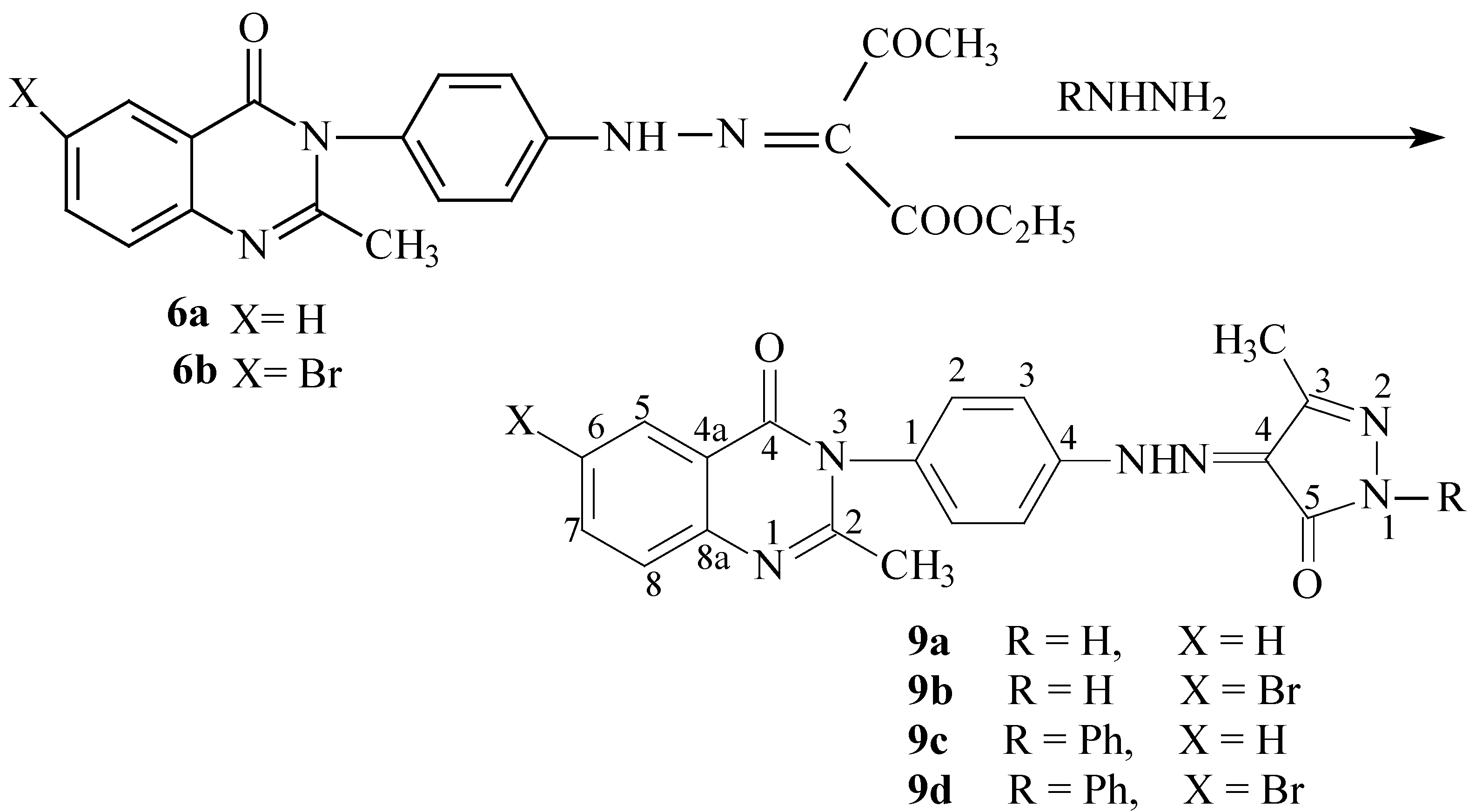
A mixture of the appropriate 6a,b or 7a,b (0.005 mole) and hydrazine hydrate (0.32 mL, 0.01 mole) in ethanol (30 mL) was heated under reflux for 4-6 hours. The solvent was concentrated and the reaction product was allowed to cool. The separated product was filtered off, washed with water, dried and recrystallized from ethanol. The following title compounds were prepared as just described:
2-Methyl-3-{4-[N′-(3-methyl-5-oxo-1,5-dihydropyrazol-4-ylidene)hydrazino] phenyl}-3H-quinazolin-4-one (9a). Orange yellow crystals; yield 1.12 g (62 %); M.p. 261-263oC; Calculated for C19H16N6O2 (360.37): 63.37 % C, 4.48 % H, 23.32 % N. Found: 63.11 %, 4.27 % H, 23.26 % N; IR, υ cm-1: 3325, 3452 (NH), 3044, 2927 (Ar-H), 1680, 1674 (C=O), 1606, 1573, 1475 (C=N, C=C); 1H-NMR δ: 12.36 (1H, s, hydrazino NH), 9.65 (1H, s, pyrazolinone NH), 7.38-8.26 (8H, m, Ar-H), 2.52 (3H, s, pyrazolinone CH3), 2.30 (3H, s, quinazolinone CH3); 13C-NMR δ: 160.8 (C-4), 158.6 (pyrazolinone C-5), 156.5 (pyrazolinone C-4), 154.6 (C-2), 151.3 (pyrazolinone C-3), 148.2 (C-8a), 143.0 (Ph C-1), 136.5 (C-7), 128.7 (2C, Ph C2,6), 128.0(Ph C-4), 127.7 (C-5), 126.8 (C-8), 126.3 (2C, Ph C3,5), 125.7 (C-6), 118.5 (C-4a), 23.4 (quinazolinone CH3), 21.2 (pyrazolinone CH3).
6-Bromo-2-methyl-3-{4-[N′-(3-methyl-5-oxo-1,5-dihydropyrazol-4-ylidene)hydrazino]phenyl}-3H-quinazolin-4-one (9b). Orange yellow crystals; yield 1.50 g (68 %); M.p. 308-310oC; Calculated for C19H15BrN6O2 (439.27): 51.95 % C, 3.44 % H, 19.13 % N. Found: 51.69 %, 3.65 % H, 19.10 % N; IR, υ cm-1: 3331, 3446 (NH), 3047, 2927 (Ar-H), 1683, 1672 (C=O), 1604, 1576, 1468 (C=N, C=C ); 1H-NMR δ: 12.24 (1H, s, hydrazino NH), 9.56 (1H, s, pyrazolinone NH), 7.32-8.42 (7 H, m, Ar-H), 2.68 (3H, s, pyrazolinone CH3), 2.34 (3H, s, quinazolinone CH3); MS, m/z (Ir, %): 441 (M+ for 81Br, 35.00), 439 (M+ for 79Br, 34.84), 331(C15H1281BrN3O+, 13.24), 329 (C15H1279BrN3O+, 13.42), 316 (C15H1181BrN2O+, 100), 314 (C15H1179BrN2O+, 100), 275 (23.95), 273 (23.94), 198 (10.23), 196 (10.20), 110 (C4H4N3O+, 42.36), 96 (C3H2N3O+ , 7.40).
3-{4-N′-(3-Amino-5-oxo-1,5-dihydropyrazol--4-ylidene)hydrazino]phenyl}-2-methyl-3H-quinazolin-4-one (10a). Orange yellow crystals; yield 0.87 g (48 %); M.p. 180-182oC; Calculated for C18H15N7O2 (361.36): 59.83 % C, 4.18 % H, 27.13 % N. Found: 59.52 %, 4.29 % H, 27.04 % N; IR, υ cm-1: 3426, 3450 (NH), 3312 (NH2), 3186, 2923 (Ar-H), 1680, 1674 (C=O), 1603, 1601, 1508, 1464 (C=N, C=C); 1H-NMR δ: 12.10 (1H, s, hydrazino NH), 9.72 (1H, s, pyrazolinone NH), 7.30-8.05 (8H, m, Ar-H), 5.05 (2H, s, NH2), 2.27 (3H, s, quinazolinone CH3); MS, m/z (Ir, %): 362 (M+ +1, 4.32), 361 (M+, 27.53), 251(C15H13N3O+, 17.10), 236 (C15H12N2O+, 100), 195 (24.14), 118 (2.75), 111 (C3H3N4O+, 54.26), 77 (38.38).
3-{4-N′-(3-Amino-5-oxo-1,5-dihydropyrazol--4-ylidene)hydrazino]phenyl}-6-bromo-2-methyl-3H-quinazolin-4-one (10b). Orange yellow crystals; yield 1.17 g (53 %); M.p. 244-246oC; Calculated for C18H14BrN7O2 (440.25): 49.11 % C, 3.21 % H, 22.27 % N. Found: 49.37 %, 3.51 % H, 22.10 % N; IR, υ cm-1: 3428, 3451 (NH), 3318 (NH2), 3176, 2927 (Ar-H), 1681, 1675 (C=O), 1606, 1603, 1511, 1473 (C=N, C=C); 1H-NMR δ: 12.46 (1H, s, hydrazino NH), 9.82 (1H, s, pyrazolinone NH), 7.42-8.38 (7H, m, Ar-H), 5.12 (2H, s, NH2), 2.31 (3H, s, quinazolinone CH3).
Method B: With Phenyl Hydrazine
To a solution of the appropriate 6a,b or 8a,b (0.01 mole) in glacial acetic acid (30 mL) was added phenyl hydrazine (1.30 g, 0.012 mole) and anhydrous sodium acetate (0.82 g, 0.01 mole). The reaction mixture was heated under reflux for 4 hours. The mixture was poured into ice-cold water and stored in a refrigerator. The crude product, which separated, was washed with water, dried and recrystallized from methanol. The following title compounds were prepared as just described:
2-Methyl-3-{4-[N′-(3-methyl-5-oxo-1-phenyl-1,5-dihydropyrazol-4-ylidene)hydrazino]phenyl}-3H-quinazolin-4-one (9c). Orange yellow crystals; yield 2.00 g (46 %); M.p. 302-304oC; Calculated for C25H20N6O2 (436.47): 68.80 % C, 4.62 % H, 19.25 % N. Found: 69.13 %, 4.88 % H, 19.54 % N; IR, υ cm-1: 3458 (NH), 3189, 2928 (Ar-H), 1682, 1672 (C=O), 1604, 1602, 1512, 1468 (C=N, C=C ); 1H-NMR δ: 13.35 (1H, s, hydrazino NH), 7.51-8.38 (13H, m, Ar-H), 2.54 (3H, s, pyrazolinone CH3), 2.27 (3H, s, quinazolinone CH3); MS, m/z (Ir, %): 437 (M+ +1, 7.32), 436 (M+, 41.50).
6-Bromo-2-methyl-3-{4-[N′-(3-methyl-5-oxo-1-phenyl-1,5-dihydropyrazol-4-ylidene)hydrazino]-phenyl}-3H-quinazolin-4-one (9d). Orange yellow crystals; yield 2.16 g (42 %); M.p. 259-261oC; Calculated for C25H19BrN6O2 (515.36): 58.26 % C, 3.72 % H, 16.31 % N. Found: 58.34 %, 3.92 % H, 16.18 % N; IR, υ cm-1: 3455 (NH), 3185, 2922 (Ar-H), 1680, 1673 (C=O), 1606, 1603, 1517, 1470 (C=N, C=C ); 1H-NMR δ: 13.40 (1H, s, hydrazino NH), 7.43-8.28 (12H, m, Ar-H), 2.60 (3H, s, pyrazolinone CH3), 2.25 (3H, s, quinazolinone CH3); MS, m/z (Ir, %): 517 (M+ +1 for 81Br, 10.25), 515 (M+ +1 for 79Br, 10.25), 516 (M+ for 81Br, 43.50), 514 (M+ for 79Br, 43.50).
3-[4-(3,5-Dimethyl-1-phenyl-1H-pyrazol-4-ylazo)phenyl]-2-methyl-3H-quinazolin-4-one(11a). Orange crystals; yield 2.60 g (60 %); M.p. 205-207oC; Calculated for C26H22N6O (434.50): 71.87 % C, 5.10 % H, 19.34 % N. Found: 72.03 %, 4.09 % H, 19.15 % N; IR, υ cm-1: 3051, 2923, 2852 (Ar-H), 1678 (C=O quinazolinone), 1596 (C=N), 1450 (N=N); 1H-NMR δ: 7.50-8.18 (13H, m, Ar-H), 2.66, 2.51 (6H, 2 s, 2 x pyrazole CH3), 2.23 (3H, s, quinazolinone CH3); MS, m/z (Ir, %): 435 (M+ +1, 5.24), 434 (M+, 16.42), 264 (C15H12N4O+, 37.36), 263 (C15H11N4O+, 28.80), 171 (C11H11N2+, 52.33), 95 (C5H7N2+, 100).
6-Bromo-3-[4-(3,5-dimethyl-1-phenyl-1H-pyrazol-4-ylazo)phenyl]-2-methyl-3H-quinazolin-4-one (11b). Orange crystals; yield 2.29 g (64 %); M.p. 238-240oC; Calculated for C26H21BrN6O (513.39): 60.83 % C, 4.12 % H, 16.37 % N. Found: 60.98 %, 4.93 % H, 16.53 % N; IR, υ cm-1: 3052, 2925, 2850 (Ar-H), 1674 (C=O quinazolinone), 1602, 1594, 1547, 1450, 1421 ( C=N, C=C, N=N); 1H-NMR δ: 7.53-8.26 (12H, m, Ar-H), 2.68, 252 (6H, 2 s, 2 x pyrazole CH3), 2.21 (3H, s, quinazolinone CH3).
Method C: With Urea
A mixture of hydrazono derivatives 8a,b (0.005 mole) and urea (0.6 g, 0.01 mole) in ethanol (40 mL) was heated under reflux for 5 hours. After cooling to room temperature, crushed ice was added and the mixture was stirred for 1 hour. The separated product was collected by filtration and recrystallized from aqueous ethanol. The following title compounds were prepared as just described:
3-{4-[N′(4,6-Dimethyl-2-oxo-2H-pyrimidin-5-ylidene)hydrazino]phenyl}-2-methyl-3H-quinazolin-4-one (12a). Orange yellow crystals; yield 0.83 g (43 %); M.p. 285-286oC; Calculated for C21H18N6O2 (386.41): 65.27 % C, 4.70 % H, 21.75 % N. Found: 64.97 %, 4.88 % H, 21.82 % N; IR, υ cm-1: 3424 (NH), 3052 (Ar-H), 1688, 1676 (C=O), 1592, 1565, 1481 (C=N, C=C ), 1284 (N-N=C); 1H-NMR δ: 13.28 (1H, s, hydrazino NH), 7.42-8.26 (8H, m, Ar-H), 2.58 (6H, s, 2 x pyrimidin-5-one CH3), 2.27(3H, s, quinazolinone CH3); 13C-NMR δ: 166.7 (pyrimidin-5-one C-6), 165.7 (pyrimidin-5-one C-4), 160.6 (quinazolinone C-4), 157.3 (pyrimidin-5-one C-2), 152.3 (quinazolinone C-2), 149.7 (pyrimidin-5-one C-5), 145.4 (C-8a), 140.6 (Ph C-1), 133.8 (C-7), 128.0 (2C, Ph C2,6), 127.6 (Ph C-4), 126.6 (C-8), 126.1 (C-5), 126.0 (2C, Ph C3,5), 125.3 (C-6), 120.5 (C-4a), 23.7 (quinazolinone CH3), 20.8 (2C, 2 x pyrimidin-5-one CH3).
6-Bromo-3-{4-[N′(4,6-dimethyl-2-oxo-2H-pyrimidin-5-ylidene)hydrazino]phenyl}-2-methyl-3H-quinazolin-4-one (12b). Orange yellow crystals; yield 1.09 g (47 %); M.p. 210-212oC; Calculated for C21H17BrN6O2 (465.21): 54.21 % C, 3.68 % H, 18.06 % N. Found: 53.89 %, 3.97 % H, 18.31 % N; IR, υ cm-1: 3427 (NH), 3063, 2928 (Ar-H), 1692, 1679 (C=O), 1579, 1507, 1429 (C=N, C=C), 1282 (N-N=C); 1H-NMR δ: 13.68 (1H, s, hydrazino NH), 7.36-8.35 (7H, m, Ar-H), 2.55 (6H, s, 2 x pyrimidin-5-one CH3), 2.25 (3H, s, quinazolinone CH3); MS, m/z (Ir, %): 467 (M+ +1 for 81Br, 4.45), 465 (M+ +1 for 79Br, 4.45), 466 (M+ for 81Br, 27.75), 464 (M+ for 79Br, 27.75).