A Novel Synthesis of 1,2,4-Triazolopteridines
Abstract
:Introduction
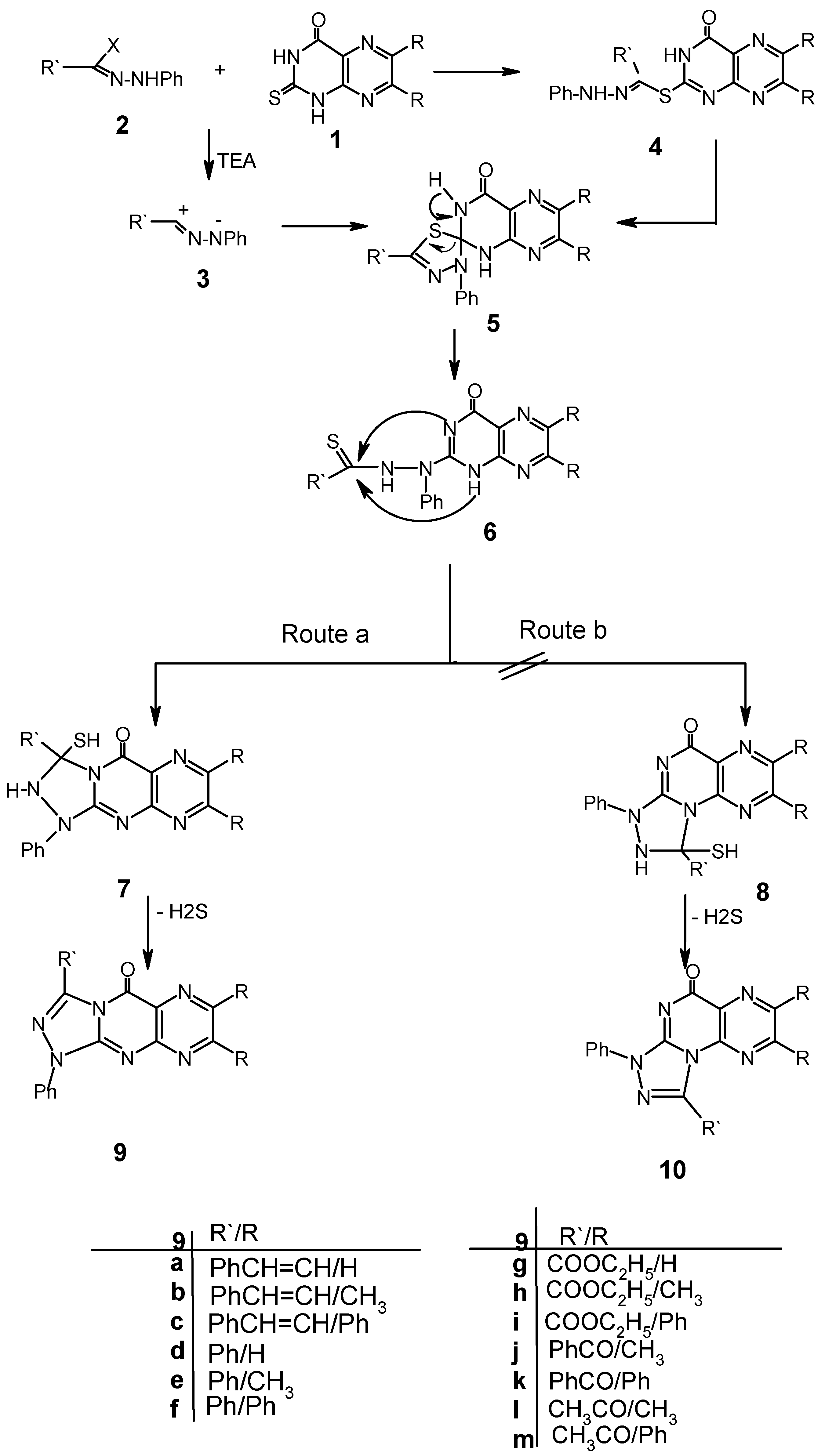
Results and Discussion


Experimental
General
| Compound | m.p. (°C) (solvent) | Yield (%) | IR (ν) | MS(70 ev) m/e (M+) | Molecular Formula | Analysis Calcd Found | 1H NMR | ||
|---|---|---|---|---|---|---|---|---|---|
| 9a | 253 (DMF) | 80 | 1709 | 366 | C21H14N6O (366) | C H N | 68.8 3.8 22.9 | 68.8 3.8 22.7 | 7.2-7.6 (m, 8H); 7.9 (d, J=5 Hz, 1H); 8.1 (d, J=5 Hz, 1H); 8.3 (s, 1H); 8.4 (s, 1H); 8.8 (d, J=14 Hz, 1H) and 8.9 (d, J=14 Hz, 1H). |
| 9b | 277 (DMF) | 75 | 1705 | 394 | C23H18N6O (394) | C | 70.0 | 69.8 | 2.7 (s, 3H); 2.8 (s, 3H) and 7.2-8.4 (m, 12H). |
| H | 4.6 | 4.7 | |||||||
| N | 21.3 | 21.4 | |||||||
| 9c | 302 (DMF) | 82 | 1714 | 518 | C33H22N6O (518) | C | 76.4 | 76.4 | 7.3-8.5 (m, Ar-H). |
| H | 4.2 | 4.3 | |||||||
| N | 16.2 | 16.0 | |||||||
| 9d | 314 (DMF) | 78 | 1727 | 340 | C19H12N6O (340) | C | 67.1 | 67.3 | 7.2-7.9 (m, 10H); 8.7 (d, J=14 Hz, 1H) and 8.8 (d, J=14 Hz, 1H). |
| H | 3.5 | 3.3 | |||||||
| N | 24.7 | 24.4 | |||||||
| 9e | 287 (DMF) | 75 | 1708 | 368 | C21H16N6O (368) | C | 68.5 | 68.7 | 2.7 (s, 3H); 2.7 (s, 3H) and 7.3- 8.4 (m, 10H). |
| H | 4.4 | 4.4 | |||||||
| N | 22.8 | 22.6 | |||||||
| 9f | 308 (DMF) | 75 | 1725 | 492 | C31H20N6O (492) | C | 75.6 | 75.6 | 7.3-8.5(m,Ar-H) |
| H | 4.1 | 4.0 | |||||||
| N | 17.1 | 16.9 | |||||||
| 9g | 199 (DMF) | 77 | 17451707 | 336 | C16H12N6O3 (336) | C | 57.1 | 57.1 | 1.5 (t, J=7 Hz, 3H); 4.7 (q, J=7 Hz, 2H); 7.3-7.6 (m, 5H); 8.3 (d, J=14 Hz, 1H) and 8.8 (d, J=14Hz, 1H). |
| H | 3.6 | 3.6 | |||||||
| N | 25.0 | 24.9 | |||||||
| 9h* | 211 (DMF) | 77 | 17141675 | C18H16N6O3 | C | 59.3 | 59.4 | 1.4 (t, J=7 Hz, 3H); 2.7 (s, 3H); 2.8 (s, 3H); 4.6 (q, J=7Hz, 2H) and 7.3-8.3 (m, 5H). | |
| H | 4.4 | 4.2 | |||||||
| N | 23.1 | 22.9 | |||||||
| 9i | 206 (DMF) | 84 | 17301640 | 488 | C28H20N6O3 (488) | C | 68.9 | 68.7 | 1.5 (t, J=7Hz, 3H); 4.6 (q, J=7 Hz, 2H) and 7.2-8.3 (m, 15H). |
| H | 4.1 | 3.9 | |||||||
| N | 17.2 | 16.9 | |||||||
| 9j | 298 (DMF) | 78 | 17231683 | 396 | C22H16N6O2 (396) | C | 66.7 | 66.5 | 2.7 (s, 3H); 2.8 (s, 3H) and 7.3-8.3 (m, 10H). |
| H | 4.0 | 3.9 | |||||||
| N | 21.2 | 21.2 | |||||||
| 9k | 183 (DMF) | 72 | 17731637 | 520 | C32H20N6O2 (520) | C | 73.9 | 73.9 | 7.2-8.3 (m, Ar-H) |
| H | 3.9 | 3.9 | |||||||
| N | 16.2 | 16.1 | |||||||
| 9l | 197 (DMF) | 66 | 17101680 | 334 | C17H14N6O2 (334) | C | 61.1 | 61.1 | 2.6 (s, 3H), 2.7 (s, 6H) and 7.3-8.2 (m, 5H). |
| H | 4.2 | 4.3 | |||||||
| N | 25.1 | 25.0 | |||||||
| 9m | 196 (DMF) | 70 | 17621640 | 458 | C27H18N6O2 (458) | C | 70.7 | 70.8 | 2.6 (s,3H) and 7.2-7.8 (m, 15H). |
| H | 3.9 | 3.6 | |||||||
| N | 18.3 | 18.2 | |||||||
References
- Fusco, R; Romani, R. Gazz. Chim. Ital. 1946, 76, 419.
- Fusco, R.; Musante, C. Gazz. Chim. Ital. 1938, 68, 147.
- Bacchetti, T. Gazz. Chim. Ital. 1961, 91, 86.
- Eweiss, N.F; Osman, A. J. Heterocycl. Chem. 1980, 17, 1713.
- Eweiss, N.F; Osman, A. Tetrahedron Letts. 1979, 1169.
- Shawali, A.S; Abdelhamid, A.O. Bull. Chem. Soc. Jpn. 1976, 49, 321.
- Gibson, M.S. Can. J. Chem. 1975, 53, 3211.
- Sasaki, T.; Ito, E. J. Heterocycl. Chem. 1981, 18, 1553.
- Dawnis, J.; Lopez, H.; Murry, G. J. Org. Chem. 1977, 42, 1018.
- Abdelhadi, H.A.; Abdallah, T.A.; Hassaneen, H.M. Heterocycles 1995, 41, 1999.
- Mansour, A.K.; Elwan, N.M.; Abdelhadi, H.A.; Abdallah, T.A.; Hassaneen, H.M. Sulfur Letts. 1994, 18, 105.
- Hassaneen, H.M.; Mousa, H.A.H.; Abed, N.M. Heterocycles 1988, 3, 27.
- Shawali, A.S.; Hassaneen, H.M.; Sherif, M.S. J. Heterocycl. Chem. 1980, 17, 1745.
- Hassaneen, H.M.; Abdelhamid, A.O.; Fahmi, A.A.; Shawali, A.S. J. Heterocycl. Chem. 1985, 22, 395.
- Skaric, V.; Skaric, F.; Cizmek, A. J. Chem. Soc. Perkin Tran.1. 1984, 2221.
- Talukdar, P.B.; Sengupta, S.K.; Datta, A.K. Ind. J. Chem. 1986, 25B, 275.
- Mizutani, M.; Sanamitsu, Y.; Tamaru, Y.; Yoshida, Z. J. Org. Chem. 1985, 50, 764.
- Mizutani, M.; Sanamitsu, Y; Tamaru, Y.; Yoshida, Z. J. Org. Chem. 1983, 48, 4585.
- Hübsch, W; Pfleiderer, W. Helv. Chim. Acta 1988, 71, 1379.
- Hassaneen, H.M; Hilal, R.H; Elwan, N.M; Harhash, A; Shawali, A.S. J.Heterocycl. Chem. 1984, 21, 1013.
- Wolkoff, P. Can. J. Chem. 1975, 53, 1333.
- Shawali, A.S; Eweiss, N.F; Hassaneen, H.M; Sami, M. Bull. Chem. Soc. Jpn. 1975, 48, 365.
- North, A.C.T; Philip, D.C; Mathews, F.S. Acta Crystallogr. Sect. A. 1994, 24, 351.
- Sample Availability: Samples of entries 1, 5, 7 and 11 are available from MDPI
© 2002 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Abdallah, T.A.; Darwish, M.A.; Hassaneen, H.M. A Novel Synthesis of 1,2,4-Triazolopteridines. Molecules 2002, 7, 494-500. https://doi.org/10.3390/70600494
Abdallah TA, Darwish MA, Hassaneen HM. A Novel Synthesis of 1,2,4-Triazolopteridines. Molecules. 2002; 7(6):494-500. https://doi.org/10.3390/70600494
Chicago/Turabian StyleAbdallah, Tayseer A., Manal A. Darwish, and Hamdi M. Hassaneen. 2002. "A Novel Synthesis of 1,2,4-Triazolopteridines" Molecules 7, no. 6: 494-500. https://doi.org/10.3390/70600494




