Fractal Hybrid Orbitals Analysis of the Tertiary Structure of Protein Molecules
Abstract
:Introduction
Fractal dimension of proteins

 × mean length,
× mean length,
| Structural class | Protein | No. Resid. | Da | s ratiob | nc |
|---|---|---|---|---|---|
| Only α-helices | calcium binding parvalbumin B | 108 | 1.42 | 0.25 | 3.0 |
| cytochrome C (albacore, reduced) | 103 | 1.42 | 0.25 | 3.0 | |
| hemoglobin (deoxy) | 141 | 1.40 | 0.26 | 2.9 | |
| myoglobin (sperm whale, deoxy) | 153 | 1.42 | 0.25 | 3.1 | |
| myohemerythrin | 118 | 1.40 | 0.26 | 2.9 | |
| virus coat protein | 219 | 1.28 | 0.32 | 2.1 | |
| hemerythrin | 113 | 1.38 | 0.27 | 2.7 | |
| Mean value | 136 | 1.39 | 0.26 | 2.8 | |
| Almost exclusively β-sheet | α-chymotripsin A | 139 | 1.27 | 0.33 | 2.0 |
| concanavalin A | 237 | 1.26 | 0.33 | 2.0 | |
| immunoglobulin | 208 | 1.26 | 0.34 | 2.0 | |
| prealbumin (human, plasma) | 228 | 1.27 | 0.33 | 2.0 | |
| superoxide dismutase | 151 | 1.30 | 0.31 | 2.2 | |
| trypsin (native, pH 8) | 223 | 1.30 | 0.31 | 2.2 | |
| acid protease | 324 | 1.31 | 0.31 | 2.3 | |
| rubredoxin | 53 | 1.38 | 0.27 | 2.7 | |
| streptomyces subtilisin inhibitor | 107 | 1.30 | 0.31 | 2.2 | |
| tosyl-elastase | 240 | 1.29 | 0.32 | 2.1 | |
| Mean value | 191 | 1.29 | 0.32 | 2.2 | |
| α-helix and β-sheet tend to be segregated along the chain | carbonic anhydrase B (human) | 254 | 1.31 | 0.31 | 2.2 |
| oxidized high potential iron protein | 85 | 1.41 | 0.25 | 3.0 | |
| lysozyme (hen egg-white) | 129 | 1.42 | 0.25 | 3.0 | |
| ribonuclease A | 124 | 1.33 | 0.30 | 2.4 | |
| thermolysin | 316 | 1.39 | 0.26 | 2.8 | |
| papain (native) | 212 | 1.32 | 0.30 | 2.3 | |
| trypsin inhibitor (bovine pancreas) | 58 | 1.31 | 0.31 | 2.3 | |
| cytochrome B5 | 85 | 1.35 | 0.29 | 2.5 | |
| bacteriochlorophyl-A protein | 358 | 1.25 | 0.34 | 1.9 | |
| Mean value | 180 | 1.34 | 0.29 | 2.5 | |
| α-helix and β-sheet tend to alternate along the chain | adenylate kinase (porcine muscle) | 194 | 1.36 | 0.28 | 2.6 |
| L-arabinose-binding protein | 306 | 1.34 | 0.29 | 2.5 | |
| carboxypeptidase A (bovine) | 307 | 1.34 | 0.29 | 2.5 | |
| flavodoxin | 138 | 1.31 | 0.30 | 2.3 | |
| glyceraldehyde-3-phosphate dehydrogenase (lobster) | 333 | 1.34 | 0.29 | 2.5 | |
| phosphoglycerate kinase (horse) | 408 | 1.33 | 0.29 | 2.4 | |
| pyruvate kinase (cat) | 432 | 1.34 | 0.29 | 2.5 | |
| rhodanase | 293 | 1.34 | 0.29 | 2.4 | |
| subtilisin novo | 275 | 1.34 | 0.29 | 2.4 | |
| thioredoxin (E. coli, oxidized) | 108 | 1.36 | 0.28 | 2.6 | |
| triose phosphate isomerase | 247 | 1.33 | 0.29 | 2.4 | |
| hexokinase A | 457 | 1.35 | 0.28 | 2.5 | |
| yeast phosphoglycerate mutase | 218 | 1.34 | 0.29 | 2.4 | |
| dihydrofolate reductase | 148 | 1.30 | 0.31 | 2.2 | |
| d-glucose-6-phosphate isomerase | 514 | 1.35 | 0.28 | 2.6 | |
| Mean value | 292 | 1.34 | 0.29 | 2.5 | |
| No α-helix nor β-sheet | agglutinin (wheat germ) | 164 | 1.48 | 0.22 | 3.6 |
| ferredoxin (Peptococcus aerogenes) | 54 | 1.33 | 0.30 | 2.4 | |
| Mean value | 109 | 1.40 | 0.26 | 3.0 | |
| Mean of the five structural classes | 211 | 1.34 | 0.29 | 2.5 | |
| Gaussian Chain | - | 1.50 | 0.21 | 3.8 | |
Fractals for hybrid orbitals in protein models
 , and the fractal dimension is given by
, and the fractal dimension is given by





Calculation results and discussion
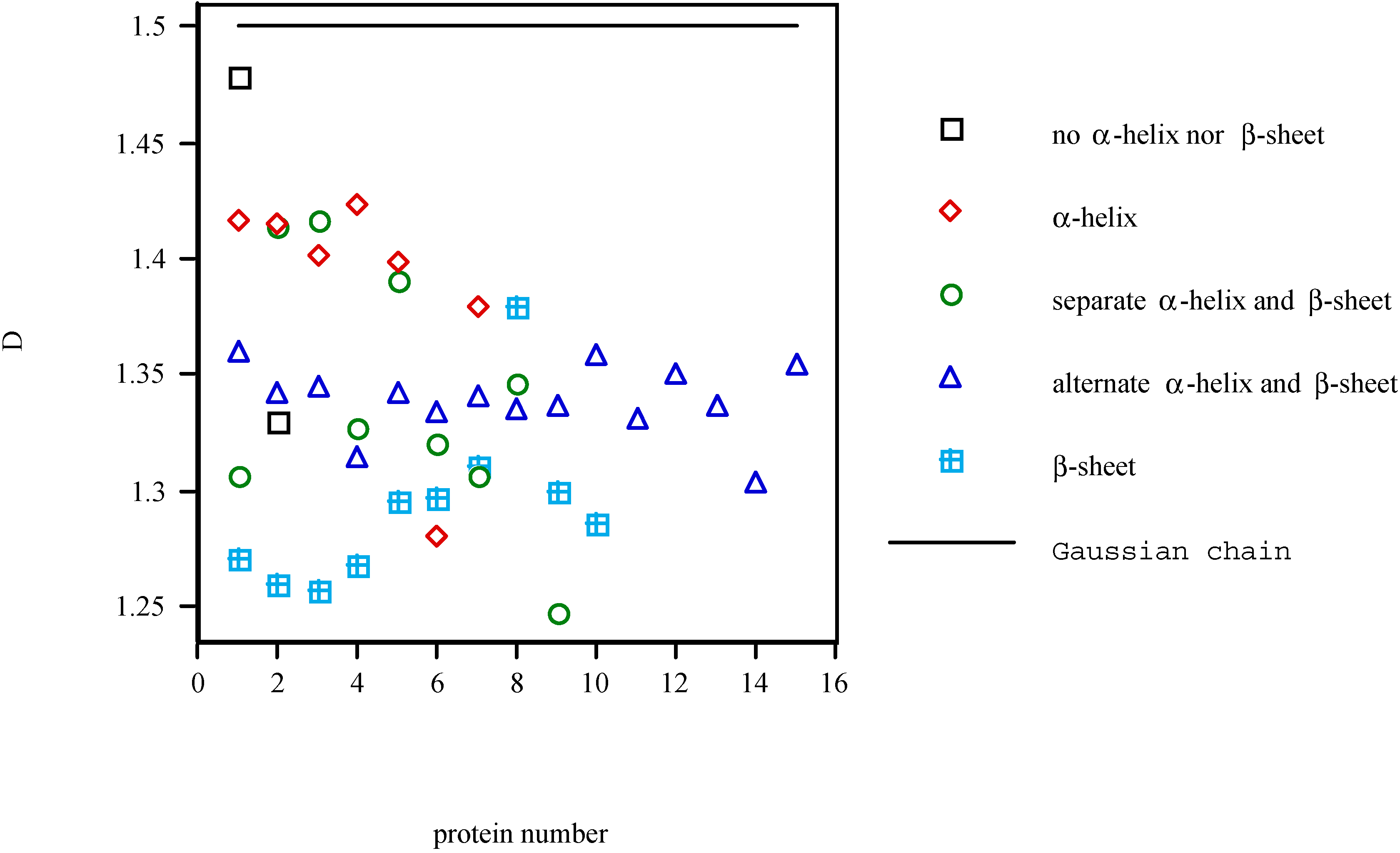
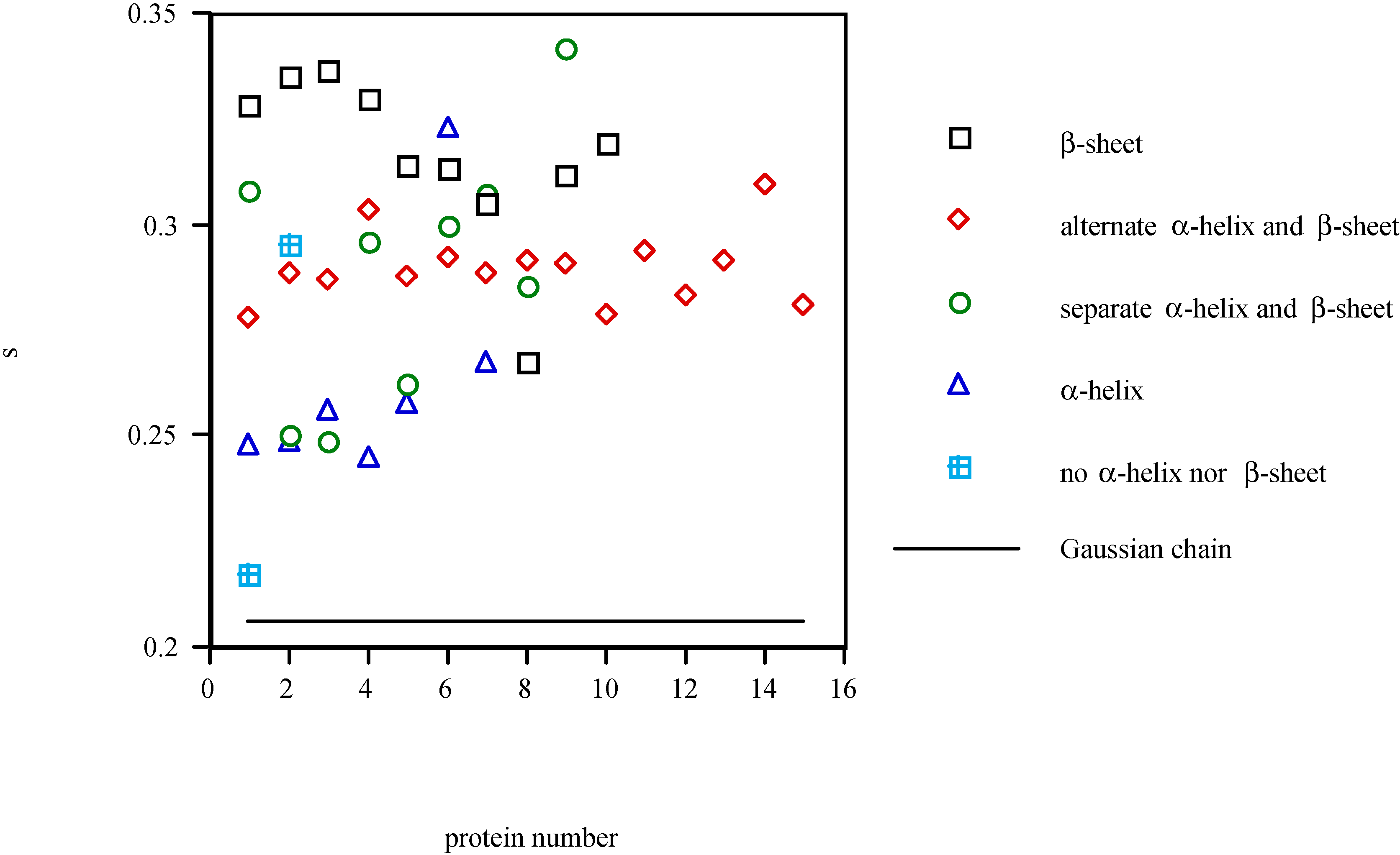
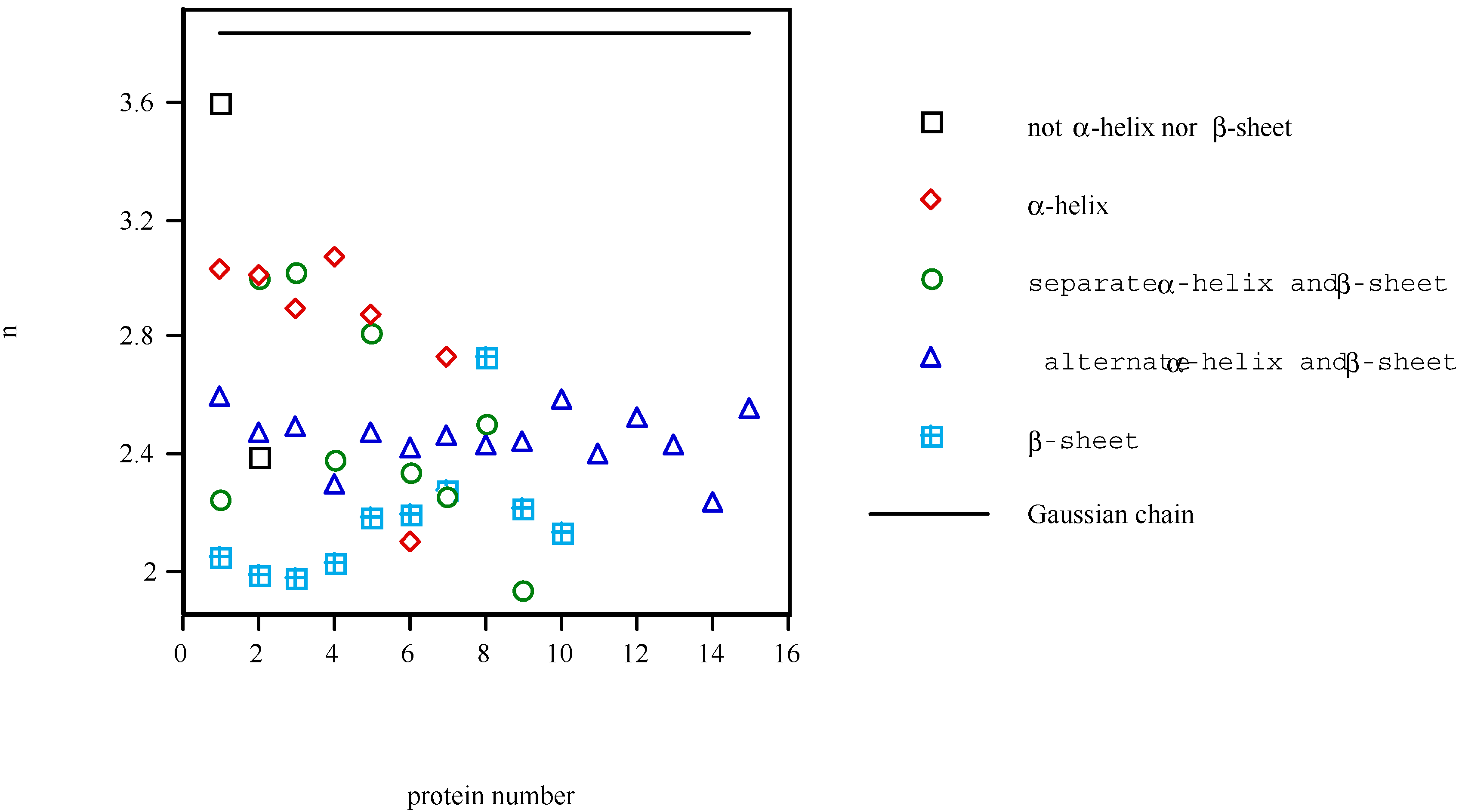
Conclusions
- Defining a measure of the length of protein molecule as a function of coarseness of the scale to see the molecule, the fractal analysis of the tertiary structure of protein has been performed. The fractal dimension D has been calculated for 43 proteins. The fractal dimension D represents the complexity of the local conformation.
- Comparison of this dimension D with that of Gaussian chain revealed that the steric hindrance between atoms is the major factor determining the local conformation. There is nothing new about this conclusion, but there was no quantitative description of the major factors that dictate the formation of local conformation. Namely, the more the steric hindrances dominate the construction of the conformation of protein molecule, the smaller the fractal dimension is. Conversely, the more the attractive forces dominate for it, the greater the fractal dimension is. Thus, the fractal dimension can be used as an indicator of the average strength of the forces acting between the atoms in the protein molecule. The stability of the structure of protein molecule, which has strong connection with the forces maintaining the molecular structure, might possibly be a good object for fractal theory.
- The fractal dimension D of various proteins was examined separately for the five structural classes of proteins. The α and β type classes have shown the smallest and largest fractal dimension D, respectively, reflecting the extended character of β-structure and the closely packed character of α-helix, respectively. The β type class has been shown to be significantly different from other classes in terms of the fractal dimension D, but Isogai et al. failed to discriminate the differences between the pairs of other classes with the fractal dimension D, showing the availability and the limitation of the fractal dimension D.
- The results of an attempt to apply fractal theory to the analysis of the tertiary structure of protein have been reported in this article. The analysis has been carried out focusing on the relationship between the fractal dimension and the structural classes, which was widely accepted. Availability and limitations of the theory have been made clear. Detecting the minute difference of protein conformations is not within its scope, but grasping the general character of global conformation is possible. Phenomena deeply connected with global conformation might possibly be good objects of study for fractal theory.
- The structure and shape of the polypeptide chain of proteins are determined by the hybridized states of atomic orbitals in the molecular chain. The calculated s ratio in the spn hybrid orbitals is computed from the fractal dimension. The proteins in the β type structural class are distinguished quantitatively from other classes with this representation.
 ), C–H bond energy, C–H bond length, bond angle and pKa.
), C–H bond energy, C–H bond length, bond angle and pKa.Acknowledgements
References
- Schulz, G. E.; Schirmer, R. H. Principles of Protein Structure; (Springer, Berlin, 1979); p. 66.
- Jou, D.; Llebot, J. E. Introduction to the Thermodynamics of Biological Processes; Prentice Hall: Englewood Cliffs, 1990. [Google Scholar]
- Cohen, C.; Holmes, K. C. X−ray diffraction evidence for α−helical coiled-coils in native muscle. J. Mol. Biol. 1963, 6, 423–432. [Google Scholar]
- Richardson, J. S. Handedness of crossover connections in β sheets. Proc. Natl. Acad. Sci. USA 1976, 73, 2619–2623. [Google Scholar]
- Richardson, J. S. β−Sheet topology and the relatedness of proteins. Nature (London) 1977, 268, 495–500. [Google Scholar]
- Wetlaufer, D. B. Nucleation, rapid folding, and globular intrachain regions in proteins. Proc. Natl. Acad. Sci. USA 1973, 70, 697–701. [Google Scholar]
- Rackovsky, S.; Scheraga, H. A. Differential geometry and polymer conformation. 1. Comparison of protein conformations. Macromolecules 1978, 11, 1168–1174. [Google Scholar]
- Mandelbrot, B. B. Fractals: Form, Chance, and Dimension; Freeman: San Francisco, 1977. [Google Scholar]
- Mandelbrot, B. B. The Fractal Geometry of Nature; Freeman: San Francisco, 1982. [Google Scholar]
- Mandelbrot, B. B. Multifractals and 1/F Noise; Springer: Berlin, 1998. [Google Scholar]
- Gardner, M. White and brown music, fractal curves and one-over−f fluctuations. Sci. Am. 1978, 238, 16–32. [Google Scholar]
- McDermott, J. Smithsonian 1983, 14, 110–117.
- Havlin, S.; Ben-Avraham, D. Fractal dimensionality of polymer chains. J. Phys. A 1982, 15, L311–L316. [Google Scholar]
- Havlin, S.; Ben-Avraham, D. New method of analysing self−avoiding walks in four dimensions. J. Phys. A 1982, 15, L317–L320. [Google Scholar]
- Havlin, S.; Ben-Avraham, D. New approach to self−avoiding walks as a critical phenomenon. J. Phys. A 1982, 15, L321–L328. [Google Scholar]
- Torrens, F.; Sánchez-Marín, J.; Nebot-Gil, I. New dimension indices for the characterization of the solvent-accessible surface. J. Comput. Chem. 2001, 22, 477–487. [Google Scholar]
- Tanford, C. Physical Chemistry of Macromolecules; Wiley: New York, 1961; pp. 152–154. [Google Scholar]
- Bernstein, F. C.; Koetzle, T. F.; Williams, G. J. B.; Meyer Jr., E. F.; Brice, M. D.; Rodgers, J. D.; Kennard, O.; Shimanouchi, T.; Tasumi, M. The Protein Data Bank: A computer-based archival file for macromolecular structures. J. Mol. Biol. 1977, 112, 535–542. [Google Scholar]
- Levitt, M.; Chothia, C. Structural patterns in globular proteins. Nature (London) 1976, 261, 552–558. [Google Scholar]
- Schulz, G. E. Structural rules for globular proteins. Angew. Chem., Int. Ed. Engl. 1977, 16, 23–32. [Google Scholar]
- Walters, P.; Stahl, M. Program BABEL. University of Arizona: Tucson, 1994. [Google Scholar]
- Torrens, F.; Ortí, E.; Sánchez-Marín, J. Vectorized TOPO program for the theoretical simulation of molecular shape. J. Chim. Phys. Phys.-Chim. Biol. 1991, 88, 2435–2441. [Google Scholar]
- Vicsek, T. Fractal Growth Phenomena; World Scientific: Singapore, 1989. [Google Scholar]
- Li, H. Q.; Li, Y.; Zhao, H. M. personal communication.
- Stapleton, H. J.; Allen, J. P.; Flynn, C.P.; Stinson, D. G.; Kurtz, S. R. Fractal form of proteins. Phys. Rev. Lett. 1980, 45, 1456–1459. [Google Scholar]
- Allen, J. P.; Colvin, J. T.; Stinson, D. G.; Flynn, C. P.; Stapleton, H. J. Protein conformation from electron spin relaxation data. Biophys. J. 1982, 38, 299–310. [Google Scholar]
- Wagner, G. C.; Colvin, J. T.; Allen, J. P.; Stapleton, H. J. Fractal models of protein structure, dynamics, and magnetic relaxation. J. Am. Chem. Soc. 1985, 107, 5589–5594. [Google Scholar]
- Colvin, J. T.; Stapleton, H. J. Fractal and spectral dimensions of biopolymer chains: Solvent studies of electron spin relaxation rates in myoglobin azide. J. Chem. Phys. 1985, 82, 4699–4706. [Google Scholar]
- Li, H. Q.; Zhao, H. M. Nature J. (Shanghai) 1989, 12, 894–901.
- Mosher, M. D.; Ojha, S. Hybridization and structural properties. J. Chem. Educ. 1998, 75, 888–890. [Google Scholar]
- Friebolin, H. Basic One- and Two-Dimensional NMR Spectroscopy; VCH: Weinheim, 1993; p. 95. [Google Scholar]
- Laszlo, P.; Stang, P. Organic Spectroscopy; Harper and Row: New York, 1971. [Google Scholar]
- Williams, D. H.; Fleming, I. Spectoscopic Methods in Organic Chemistry; McGraw-Hill: London, 1980; pp. 125–127. [Google Scholar]
- Torrens, F. Fractal hybrid orbitals in biopolymer chains. Russ. J. Phys. Chem. (Engl. Transl.) 2000, 74, 115–120. [Google Scholar]
- Torrens, F. Fractals for hybrid orbitals in protein models. Complexity Int. in press.
- Pascual-Ahuir, J. L.; Silla, E.; Tomasi, J.; Bonaccorsi, R. Electrostatic interaction of a solute with a continuum. Improved description of the cavity and of the surface cavity bound charge distribution. J. Comput. Chem. 1987, 8, 778–787. [Google Scholar]
- Isogai, Y.; Itoh, T. Fractal analysis of tertiary structure of protein molecule. J. Phys. Soc. Jpn. 1984, 53, 2162–2171. [Google Scholar]
- Sample availability: Not applicable.
© 2002 by MDPI (http://www.mdpi.org). Reproduction is permitted for non commercial purposes.
Share and Cite
Torrens, F. Fractal Hybrid Orbitals Analysis of the Tertiary Structure of Protein Molecules. Molecules 2002, 7, 26-37. https://doi.org/10.3390/70100026
Torrens F. Fractal Hybrid Orbitals Analysis of the Tertiary Structure of Protein Molecules. Molecules. 2002; 7(1):26-37. https://doi.org/10.3390/70100026
Chicago/Turabian StyleTorrens, Francisco. 2002. "Fractal Hybrid Orbitals Analysis of the Tertiary Structure of Protein Molecules" Molecules 7, no. 1: 26-37. https://doi.org/10.3390/70100026




