Phytochemical Study Conyza Sophiaefolia. Antiinflammatory Activity
Abstract
:Introduction
Experimental
Plant Material
Isolation Procedure
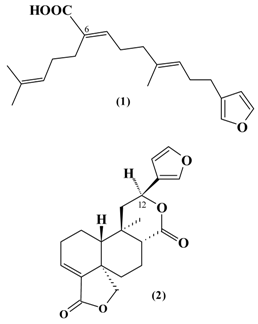
Result and Discussion
References and Notes
- Bohlmann, F.; Grenz, M.; Wegner, P.; Jakupovic. Liebigs Ann. Chem. 1983, 2008.
- Jolad, D. S.; Timmermann, B. N.; Hoffmann, J. J.; Bates, R. B.; Camou, F. A. Phytochemistry 1988, 27, 1211.
- Wagner, H.; Seitz, R.; Lotter, H.; Herz, W. J. Org. Chem. 1978, 43, 3339.
- Favier, L.S.; Tonn, C.E.; Guerreiro, E.; Rotelli, A.; Peltzer, L. Planta Medica 1998, 64, 587.
| H/C | δH (Compound 1) | δC |
| 1 | 1.68 br s | 25.7 q |
| 2 | 132.1 s | |
| 3 | 5.19 br t (6.0) | 124.1 d |
| 4 | 2.10 br t (4.0) | 28.0 t |
| 5 | 2.25 m | 28.7 t |
| 6 | 131.7 s | |
| 7 | 6.72 t (7.3) | 145.6 d |
| 8 | 2.35 m | 27.0 t |
| 9 | 2.30 m | 38.5 t |
| 10 | 134.4 s | |
| 11 | 5.20 br t (6.8) | 125.1 d |
| 12 | 2.23 m | 27.4 t |
| 13 | 2.45 br q (7.5) | 25.1 t |
| 14 | 128.2 s | |
| 15 | 6.28 br s | 111.2 d |
| 16 | 7.31 br s | 142.8 d |
| 17 | 7.20 br s | 139.2 d |
| 18 | 1.60 br s | 15.8 q |
| 19 | 174.3 s | |
| 20 | 1.60 br s | 17.6 q |
| Product | Acute inflammation inhibit % | Dunnet’s Test | |||
|---|---|---|---|---|---|
| Acetonic extract | 1H | 3Hs. | 5Hs. | 7Hs. | (a) p<0.02 |
| - | 37 | 45(b) | 49(a) | ||
| Chloroformic extract A | 14 | 22 | 45(b) | 35 | (b) p<0.04 |
| n-hexane extract B | - | 12 | 36 | 26 | (c) p<0.002 |
| Phenylbutazone | 55 | 65(d) | 65(c) | 52(a) | (d) p<0.0003 |
Share and Cite
Simirgiotis, M.J.; Favier, L.S.; Rossomando, P.C.; Tonn, C.E.; Juarez, A.; Giordano, O.S. Phytochemical Study Conyza Sophiaefolia. Antiinflammatory Activity. Molecules 2000, 5, 605-607. https://doi.org/10.3390/50300605
Simirgiotis MJ, Favier LS, Rossomando PC, Tonn CE, Juarez A, Giordano OS. Phytochemical Study Conyza Sophiaefolia. Antiinflammatory Activity. Molecules. 2000; 5(3):605-607. https://doi.org/10.3390/50300605
Chicago/Turabian StyleSimirgiotis, M. J., L. S. Favier, P. C. Rossomando, C. E. Tonn, A. Juarez, and O. S. Giordano. 2000. "Phytochemical Study Conyza Sophiaefolia. Antiinflammatory Activity" Molecules 5, no. 3: 605-607. https://doi.org/10.3390/50300605
APA StyleSimirgiotis, M. J., Favier, L. S., Rossomando, P. C., Tonn, C. E., Juarez, A., & Giordano, O. S. (2000). Phytochemical Study Conyza Sophiaefolia. Antiinflammatory Activity. Molecules, 5(3), 605-607. https://doi.org/10.3390/50300605