Phenanthrenes from Juncus Compressus Jacq. with Promising Antiproliferative and Anti-HSV-2 Activities
Abstract
1. Introduction
2. Results and Discussion
2.1. Isolation and Structure Determination of the Compounds
2.2. Antiproliferative Activity of the Isolated Phenanthrenes
2.3. Antiviral Activity of the Phenanthrenes
3. Materials and Methods
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
3.3.1. Compressin A (Compound 1)
3.3.2. Compressin B (Compound 2)
3.4. Antiproliferative Assay
3.5. Anti-HSV-2 Assay
3.5.1. Cultivation and Quantification of Herpes Viruses
3.5.2. MTT Assay for Determination of Non-Toxic Concentration of Compounds in Vero Cells
3.5.3. Assay for Testing Antiviral Activity on Vero Cells
3.5.4. Antiviral Activity Using qPCR
3.5.5. Determination of TCID50 with Virus Yield Reduction Technique
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Li, H.X.; Deng, T.Z.; Chen, Y. Isolation and identification of phenolic constituents from Juncus effusus. Acta Pharm. Sin. 2007, 42, 174–178. [Google Scholar]
- DellaGreca, M.; Fiorentino, A.; Isidori, M.; Previtera, L.; Temussi, F.; Zarrelli, A. Benzocoumarins from the rhizomes of Juncus acutus. Tetrahedron 2003, 59, 4821–4825. [Google Scholar] [CrossRef]
- Shan, C.Y.; Ye, Y.H.; Jiang, H.F.; Zhang, J. Study on chemical constituents isolated from Juncus effusus. J. Chin. Med. Mat. 2008, 31, 374–376. [Google Scholar]
- Tian, X.J.; Li, H.X.; Chen, Y.; Wang, X.Q.; Yang, G.Z. Studies on the chemical constituents of Juncus effusus. Lishizhen Med. Mater. Med. Res. 2007, 18, 2121–2122. [Google Scholar]
- Dong-Zhea, J.; Zhi-Daa, M.; Geroge, C.Y.; Munekazu, I.; Tanaka, T. p-Coumaroyl monoglycerides have been described in the medullae of Juncus effusus. Phytochemistry 1996, 41, 545–547. [Google Scholar] [CrossRef]
- Awaad, A. Phenolic glycosides of Juncus acutus and its anti-eczematic activity. Chem. Nat. Prod. 2006, 42, 152–156. [Google Scholar] [CrossRef]
- DellaGreca, M.; Fiorentino, A.; Mangoni, A.; Molinaro, A.; Monaco, P.; Previtera, L. A bioactive dihydrodibenzoxepin from Juncus effusus. Phytochemistry 1993, 34, 1182–1184. [Google Scholar] [CrossRef]
- Bús, Cs.; Tóth, B.; Stefkó, D.; Hohmann, J.; Vasas, A. Family Juncaceae: Promising source of biologically active natural phenanthrenes. Phytochem. Rev. 2018, 1–19. [Google Scholar] [CrossRef]
- Kovács, A.; Vasas, A.; Hohmann, J. Natural phenanthrenes and their biological activity. Phytochemistry 2008, 69, 1084–1110. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Liu, F.; Ding, Y.Y.; Zhang, Y.; Li, N. Four new phenanthrenoid dimers from Juncus effusus L. with cytotoxic and anti-inflammatory activities. Fitoterapia 2015, 105, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Su, X.H.; Yuan, Z.P.; Li, C.Y.; Zhong, Y.J.; Du, H.J.; Wen, Y.Y.; Li, Y.F.; Liang, B. Phenanthrenes from Juncus effusus. Planta Med. 2013, 79, 1447–1452. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Zhang, Y.Y.; Ding, Y.Y.; Li, N. Cytotoxic and anti-inflammatory activities of phenanthrenes from the medullae of Juncus effusus L. Arch. Pharm. Res. 2016, 39, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Meng, M.; Zhang, B.; Du, L.; Pan, Y.; Yang, P.; Gu, Z.; Zhou, Q.; Cao, Z. Dehydroeffusol effectively inhibits human gastric cell-mediated vasculogenic mimicry with low toxicity. Toxicol. Appl. Pharmacol. 2015, 287, 98–110. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Han, H.; Fu, S.; Yang, P.; Gu, Z.; Zhou, Q.; Cao, Z. Dehydroeffusol inhibits gastric cancer cell growth and tumorigenicity by selectively inducing tumor-suppressive endoplasmic reticulum stress and a moderate apoptosis. Biochem. Pharmacol. 2016, 104, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Chapatwala, K.D.; de la Cruz, A.A.; Miles, D.H. Antimicrobial activity of juncusol, a novel 9,10-dihydrophenanthrene from the marsh plant Juncus roemerianus. Life Sci. 1981, 29, 1997–2001. [Google Scholar] [CrossRef]
- Hanawa, F.; Okamoto, M.; Towers, G.H.N. Antimicrobial DNA-binding photosensitizers from the common rush, Juncus effusus. Photochem. Photobiol. 2002, 76, 51–56. [Google Scholar] [CrossRef]
- Sahly, R.; Rivière, C.; Siah, A.; Smaoui, A.; Samaillie, J.; Hennebelle, T.; Roumy, V.; Ksouri, R.; Halama, P. Biocontrol activity of effusol from the extremophile plant, Juncus maritimus, against the wheat pathogen Zymoseptoria tritici. Environ. Sci. Pollut. Res. 2017, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Tóth, B.; Liktor-Busa, E.; Kúsz, N.; Szappanos, Á.; Mándi, A.; Kurtán, T.; Urbán, E.; Hohmann, J.; Chang, F.R.; Vasas, A. Phenanthrenes from Juncus inflexus with antimicrobial activity against methicillin-resistant Staphylococcus aureus. J. Nat. Prod. 2016, 79, 2814–2823. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Duffhues, G.; Calzado, M.A.; de Vinuesa, A.G.; Caballero, F.J.; Ech-Chahad, A.; Appendino, G.; Krohn, K.; Fiebich, B.L.; Muñoz, E. Denbinobin, a naturally occurring 1,4-phenanthrenequinone, inhibits HIV-1 replication through an NF-κB-dependent pathway. Biochem. Pharmacol. 2008, 76, 1240–1250. [Google Scholar] [CrossRef] [PubMed]
- Staikuniene, J.; Staneviciute, J. Long-term valacyclovir treatment and immune modulation for Herpes-associated erythema multiforme. Cent. Eur. J. Immunol. 2015, 3, 387–390. [Google Scholar] [CrossRef] [PubMed]
- Erlich, K.S.; Mills, J.; Chatis, P.; Mertz, G.J.; Busch, D.F.; Follansbee, S.E.; Grant, R.M.; Crumpacker, C.S. Acyclovir-resistant Herpes simplex virus infections in patients with the Acquired Immunodeficiency Syndrome. N. Engl. J. Med. 1989, 320, 293–296. [Google Scholar] [CrossRef] [PubMed]
- DellaGreca, M.; Fiorentino, A.; Nangoni, L.; Molinaro, A.; Monaco, P.; Previtera, L. 9,10-Dihydrophenanthrene metabolites from Juncus effusus L. Tetrahedron Lett. 1992, 33, 5257–5260. [Google Scholar]
- DellaGreca, M.; Isidori, M.; Lavorgna, M.; Monaco, P.; Previtera, L.; Zarrelli, A. Bioactivity of phenanthrenes from Juncus acutus on Selenastrum capricornutum. J. Chem. Ecol. 2004, 30, 867–879. [Google Scholar] [CrossRef] [PubMed]
- Ishiuchi, K.; Kosuge, Y.; Hamagami, H.; Ozaki, M.; Ishige, K.; Ito, Y.; Kitanaka, S. Chemical constituents isolated from Juncus effusus induce cytotoxicity in HT22 cells. J. Nat. Med. 2015, 69, 421–426. [Google Scholar] [CrossRef] [PubMed]
- DellaGreca, M.; Monaco, P.; Previtera, L.; Zarrelli, A. Minor bioactive dihydrophenanthrenes from Juncus effusus. J. Nat. Prod. 1997, 60, 1265–1268. [Google Scholar] [CrossRef]
- Shima, K.; Toyota, M.; Asakawa, Y. Phenanthrene derivatives from the medullae of Juncus effusus. Phytochemistry 1991, 30, 3149–3151. [Google Scholar] [CrossRef]
- Shen, C.C.; Chang, Y.S.; Ho, L.K. Nuclear magnetic resonance studies of 5,7-dihydroxyflavonoids. Phytochemistry 1993, 34, 843–845. [Google Scholar] [CrossRef]
- Mucsi, I.; Molnár, J.; Motohashi, N. Combination of benzo[a]phenothiazines with acyclovir against Herpes simplex virus. Int. J. Antimicrob. Agents 2001, 18, 67–72. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Elshafie, H.S.; Armentano, M.F.; Carmosino, M.; Bufo, S.A.; De Feo, V.; Camele, I. Cytotoxic activity of Origanum vulgare L. on hepatocellular carcinoma cell line HepG2 and evaluation of its biological activity. Molecules 2017, 22, 1435. [Google Scholar] [CrossRef] [PubMed]
- Matos, A.M.; Reis, M.; Duarte, N.; Spengler, G.; Molnár, J.; Ferreira, M.J. Epoxylathyrol derivatives: Modulation of ABCB1-mediated multidrug resistance in human colon adenocarcinoma and mouse T-lymphoma cells. J. Nat. Prod. 2015, 78, 2215–2228. [Google Scholar] [CrossRef] [PubMed]
- Virók, D.P.; Eszik, I.; Mosolygó, T.; Önder, K.; Endrész, V.; Burián, K. A direct quantitative PCR-based measurement of Herpes simplex virus susceptibility to antiviral drugs and neutralizing antibodies. J. Virol. Methods 2017, 242, 46–52. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 1–11 are available from the authors. |
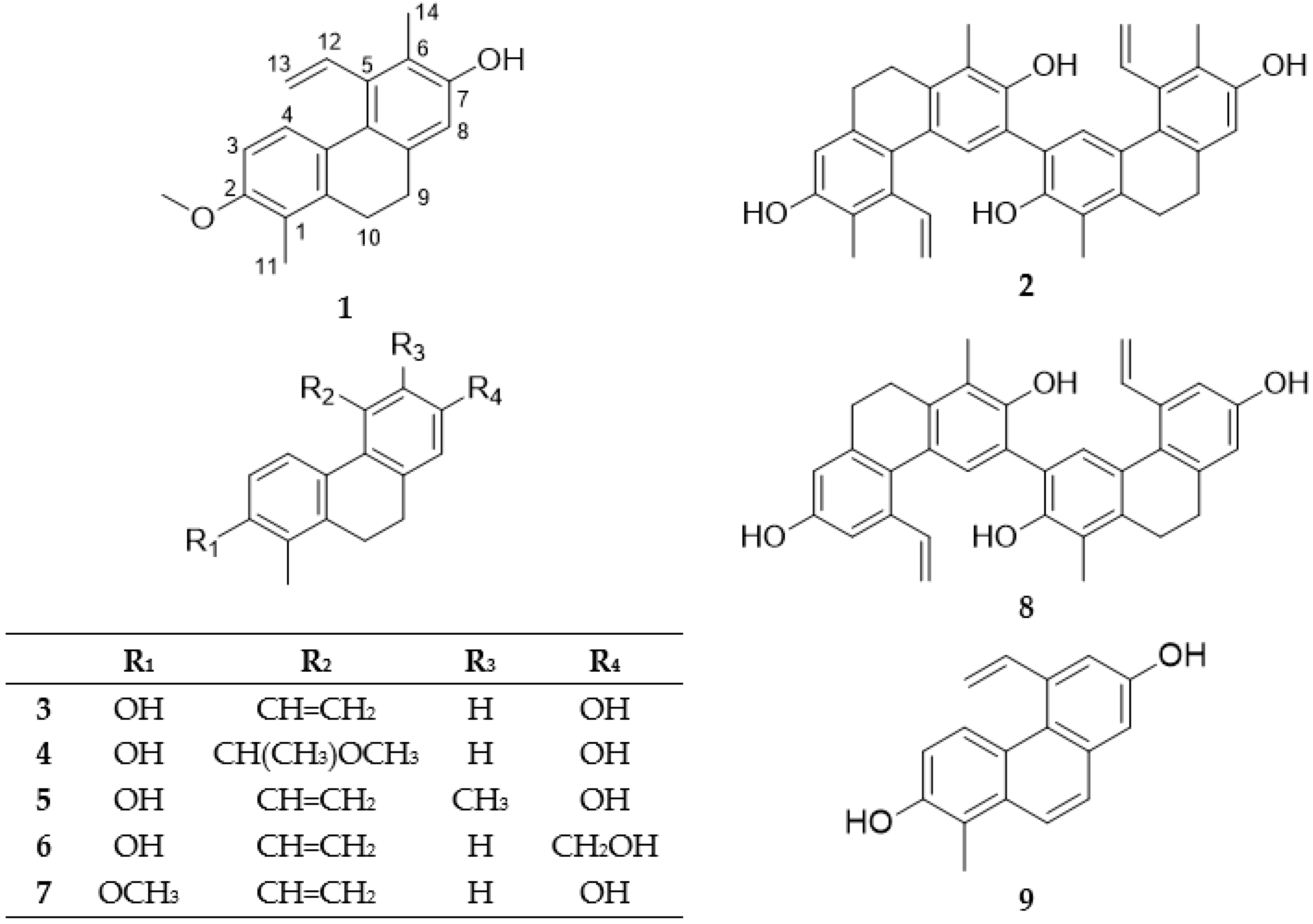

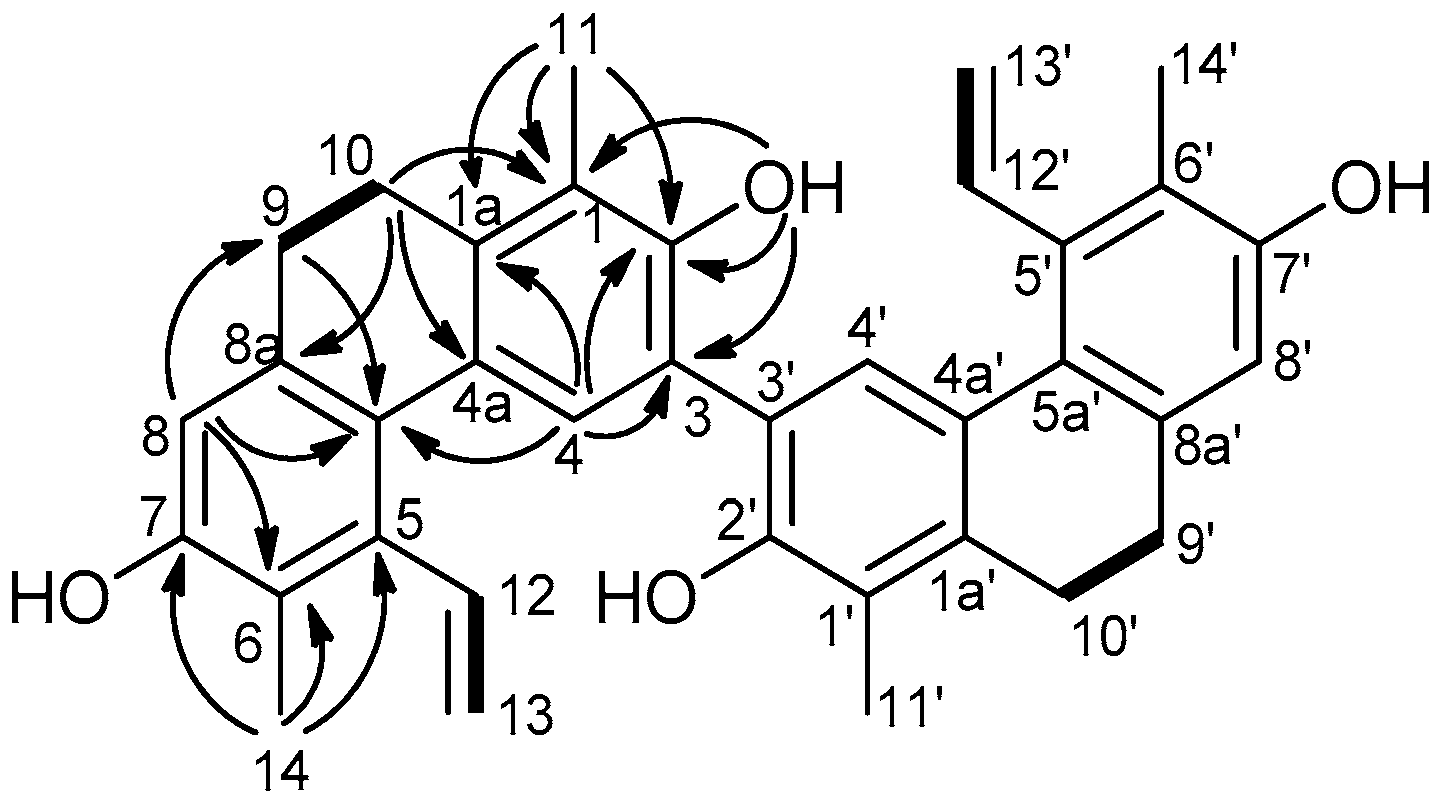
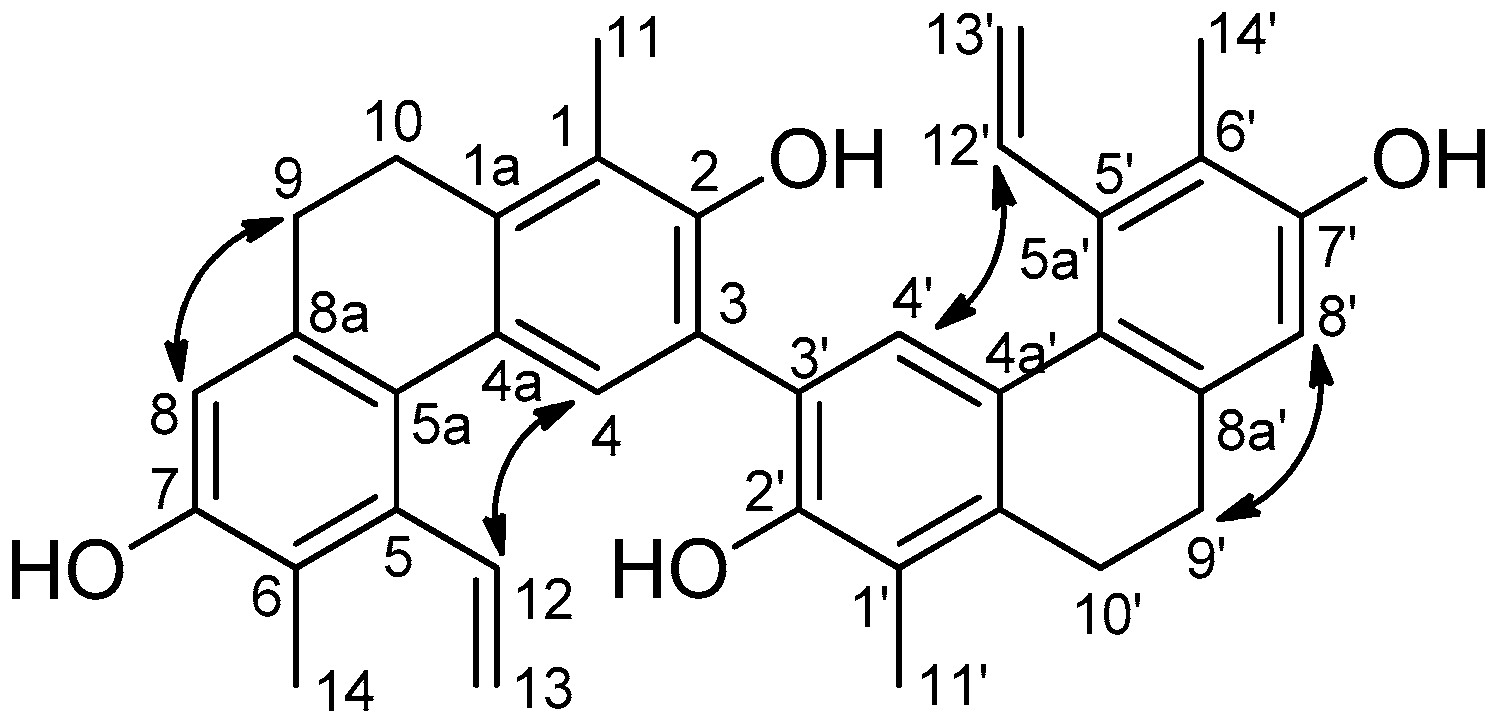
| Position | δH (J in Hz) | δC, Type | HMBC (H→C) |
|---|---|---|---|
| 1 | 122.7, C | ||
| 1a | 139.5, C | ||
| 2 | 156.2, C | ||
| 3 | 6.71, d (8.6) | 106.9, CH | 1, 2, 4a |
| 4 | 7.62, d (8.6) | 128.3, CH | 1a, 2, 5a |
| 4a | 127.5, C | ||
| 5 | 137.1, C | ||
| 5a | 127.6, C | ||
| 6 | 120.6, C | ||
| 7 | 152.5, C | ||
| 8 | 6.67, s | 113.2, CH | 5a, 6, 7, 9 |
| 8a | 137.9, C | ||
| 9 | 2.64, m (2H) | 30.5, CH2 | 1a, 5a, 8, 8a |
| 10 | 2.70, m (2H) | 25.9, CH2 | 1, 1a, 4a, 8a |
| 11 | 2.22, s | 11.9, CH3 | 1, 1a, 2 |
| 12 | 6.78, dd (17.9, 11.4) | 137.8, CH | 5, 5a, 6 |
| 13 | 5.52, d (11.4) 5.25, d (18.0) | 119.8, CH2 | |
| 14 | 2.29, s | 13.4, CH3 | 5, 6, 7 |
| OCH3 | 3.85, s | 55.7, CH3 | 2 |
| Position | δH (J in Hz) | δC, Type | HMBC (H→C) |
|---|---|---|---|
| 1, 1′ | 122.0, C | ||
| 1a, 1a′ | 138.6, C | ||
| 2, 2′ | 147.6, C | ||
| 3, 3′ | 116.4, C | ||
| 4, 4′ | 7.66, s | 127.3, CH | 1a, 1a′, 2, 2′, 3, 3′, 5a, 5a′ |
| 4a, 4a′ | 128.1, C | ||
| 5, 5′ | 137.8, C | ||
| 5a, 5a′ | 126.3, C | ||
| 6, 6′ | 120.7, C | ||
| 7, 7′ | 152.3, C | ||
| 8, 8′ | 6.66, s | 113.2, CH | 5a, 5a′, 6, 6′, 7, 7′, 9, 9′ |
| 8a, 8a′ | 137.2, C | ||
| 9, 9′ | 2.62, m | 30.2, CH2 | 1a, 1a′, 5a 5a′, 8, 8′, 8a, 8a′, 10, 10′ |
| 10, 10′ | 2.67, m | 25.6, CH2 | 1, 1′, 1a, 1a′, 4a, 4a′, 8a, 8a′, 9, 9′ |
| 11, 11′ | 2.28, s | 12.6, CH3 | 1, 1′, 1a, 1a′, 2, 2′ |
| 12, 12′ | 6.77, dd (18.0, 11.4) | 137.1, CH | 13, 13′ |
| 13, 13′ | 5.57, dd (11.4, 1.7) 5.25, dd (18.0, 1.7) | 120.5, CH2 | 12, 12′ |
| 14, 14′ | 2.27, s | 13.3, CH3 | 5, 5′, 6, 6′, 7, 7′ |
| 2-OH, 2′-OH | 5.56, s | 1, 1′, 2, 2′, 3, 3′ | |
| 7-OH, 7-OH’ | 4.73, s |
| Compound | Concentration (μM) | Growth Inhibition (%) ± SEM (Calculated IC50 Value (μM)) | ||
|---|---|---|---|---|
| HeLa | SiHa | A2780 | ||
| 1 | 10 | 41.67 ± 1.10 | – * | 47.64 ± 1.58 |
| 30 | 93.73 ± 0.36 | 38.56 ± 0.78 | 73.00 ± 0.47 | |
| 11.27 | 13.19 | |||
| 2 | 10 | 92.03 ± 0.44 | – | – |
| 30 | 92.38 ± 0.15 | 32.82 ± 0.88 | 64.85 ± 1.95 | |
| 1.86 | ||||
| 3 | 10 | 96.37 ± 0.28 | – | – |
| 30 | 97.77 ± 0.27 | – | 50.53 ± 1.59 | |
| 3.68 | ||||
| 4 | 10 | – | – | 18.33 ± 0.42 |
| 30 | 30.45 ± 1.91 | 14.23 ± 2.75 | 72.05 ± 1.06 | |
| 5 | 10 | 97.71 ± 0.37 | 17.05 ± 2.37 | 18.06 ± 1.26 |
| 30 | 97.76 ± 0.23 | 29.26 ± 2.00 | 63.43 ± 0.98 | |
| 1.31 | ||||
| 6 | 10 | 96.80 ± 0.28 | – | – |
| 30 | 98.77 ± 0.24 | 10.25 ± 1.47 | – | |
| 4.17 | ||||
| 7 | 10 | 45.38 ± 1.57 | 14.23 ± 0.98 | – |
| 30 | 92.36 ± 0.23 | 29.48 ± 3.36 | 63.24 ± 2.54 | |
| 10.66 | ||||
| 8 | 10 | – | – | – |
| 30 | 10.45 ± 0.47 | 14.42 ± 1.48 | 72.73 ± 1.82 | |
| 9 | 10 | 75.15 ± 2.48 | – | – |
| 30 | 96.50 ± 0.32 | 28.69 ± 2.01 | 57.27 ± 2.58 | |
| 7.75 | ||||
| Cisplatin | 10 | 42.61 ± 2.33 | 88.64 ± 0.50 | 83.57 ± 1.21 |
| 30 | 99.93 ± 0.26 | 90.18 ± 7.78 | 95.02 ± 0.28 | |
| 12.43 | 7.84 | 1.30 | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bús, C.; Kúsz, N.; Jakab, G.; Senobar Tahaei, S.A.; Zupkó, I.; Endrész, V.; Bogdanov, A.; Burián, K.; Csupor-Löffler, B.; Hohmann, J.; et al. Phenanthrenes from Juncus Compressus Jacq. with Promising Antiproliferative and Anti-HSV-2 Activities. Molecules 2018, 23, 2085. https://doi.org/10.3390/molecules23082085
Bús C, Kúsz N, Jakab G, Senobar Tahaei SA, Zupkó I, Endrész V, Bogdanov A, Burián K, Csupor-Löffler B, Hohmann J, et al. Phenanthrenes from Juncus Compressus Jacq. with Promising Antiproliferative and Anti-HSV-2 Activities. Molecules. 2018; 23(8):2085. https://doi.org/10.3390/molecules23082085
Chicago/Turabian StyleBús, Csaba, Norbert Kúsz, Gusztáv Jakab, Seyyed Ashkan Senobar Tahaei, István Zupkó, Valéria Endrész, Anita Bogdanov, Katalin Burián, Boglárka Csupor-Löffler, Judit Hohmann, and et al. 2018. "Phenanthrenes from Juncus Compressus Jacq. with Promising Antiproliferative and Anti-HSV-2 Activities" Molecules 23, no. 8: 2085. https://doi.org/10.3390/molecules23082085
APA StyleBús, C., Kúsz, N., Jakab, G., Senobar Tahaei, S. A., Zupkó, I., Endrész, V., Bogdanov, A., Burián, K., Csupor-Löffler, B., Hohmann, J., & Vasas, A. (2018). Phenanthrenes from Juncus Compressus Jacq. with Promising Antiproliferative and Anti-HSV-2 Activities. Molecules, 23(8), 2085. https://doi.org/10.3390/molecules23082085






