Origanum vulgare ssp. vulgare: Chemical Composition and Biological Studies
Abstract
1. Introduction
2. Results and Discussion
2.1. Chromatographic Analysis of Phenolic Compounds
2.2. Polyphenolic Content and Antioxidant Activity of O. vulgare ssp. vulgare Extract
2.3. Determination of Antimicrobial Activity
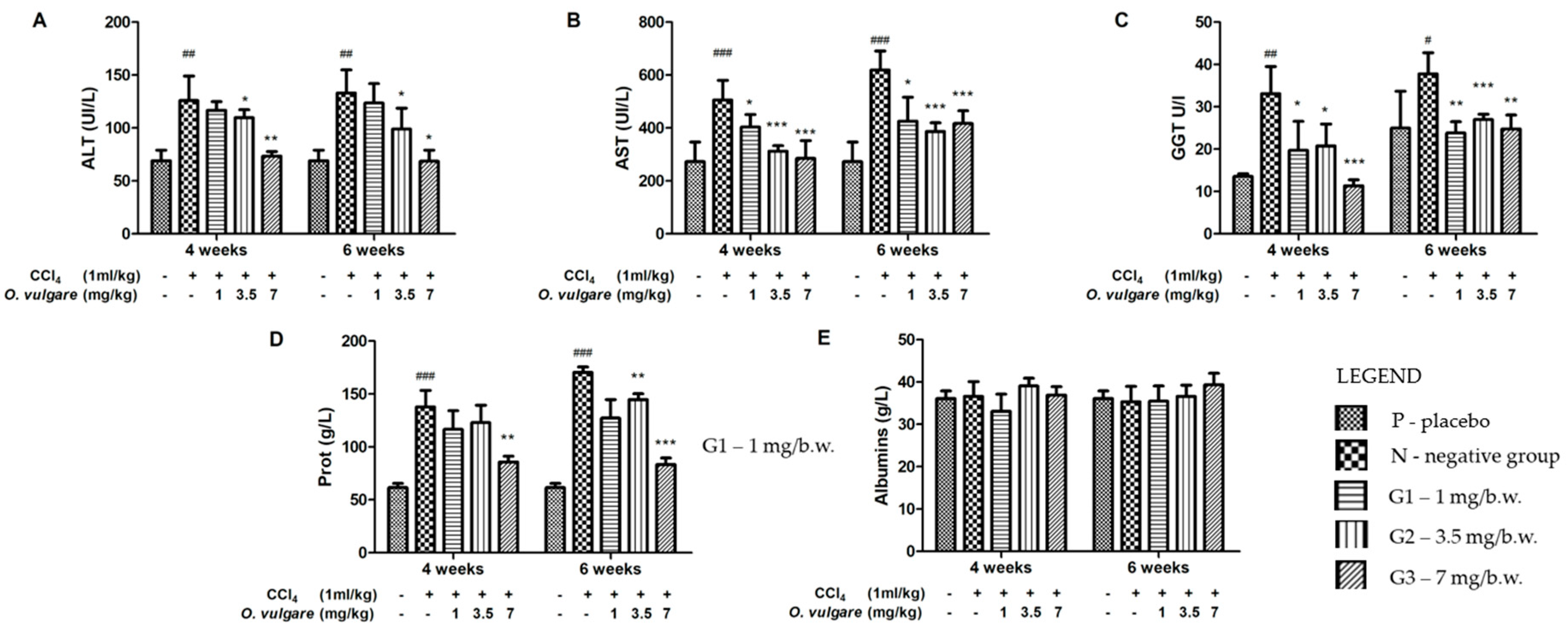
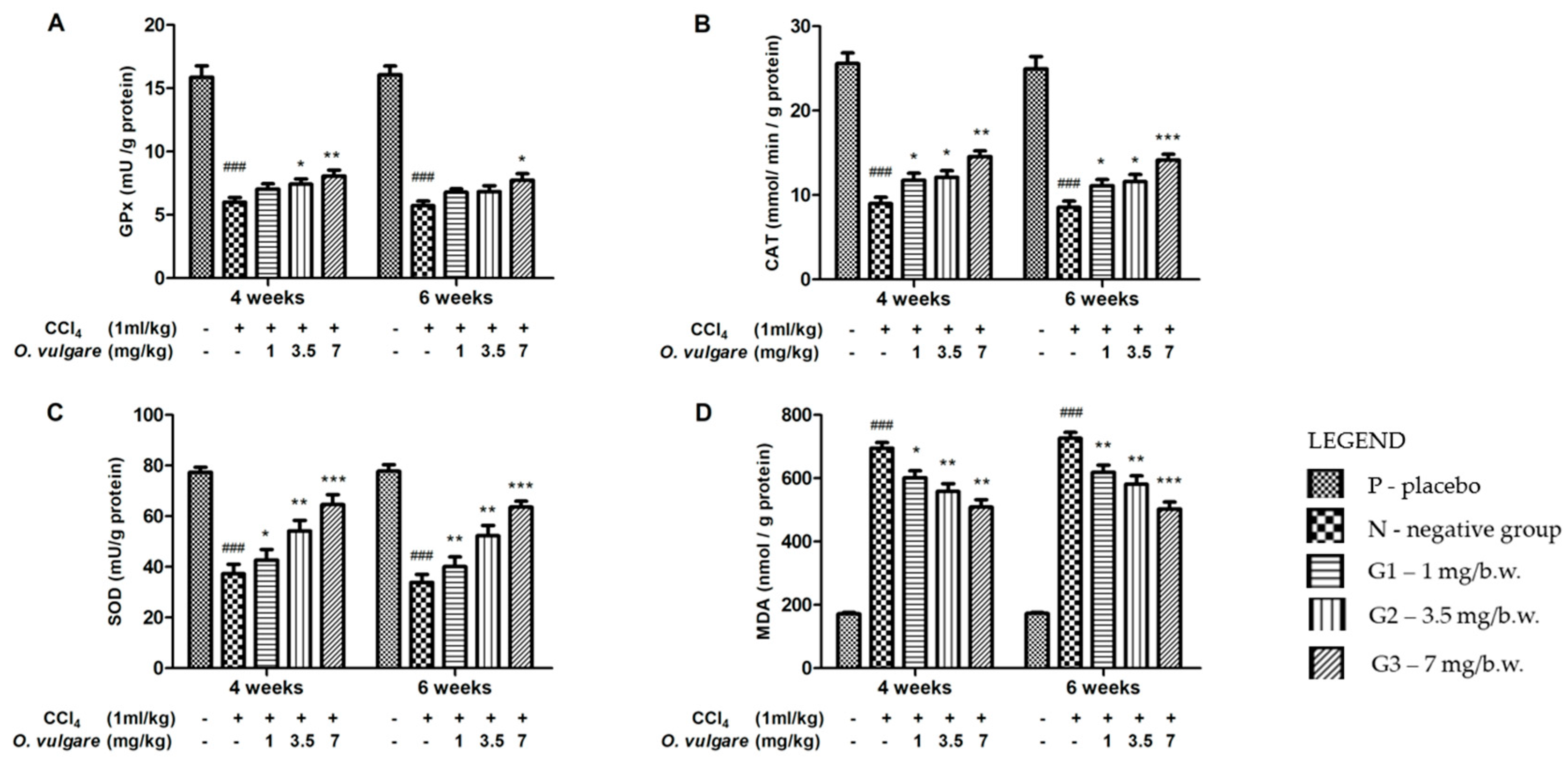
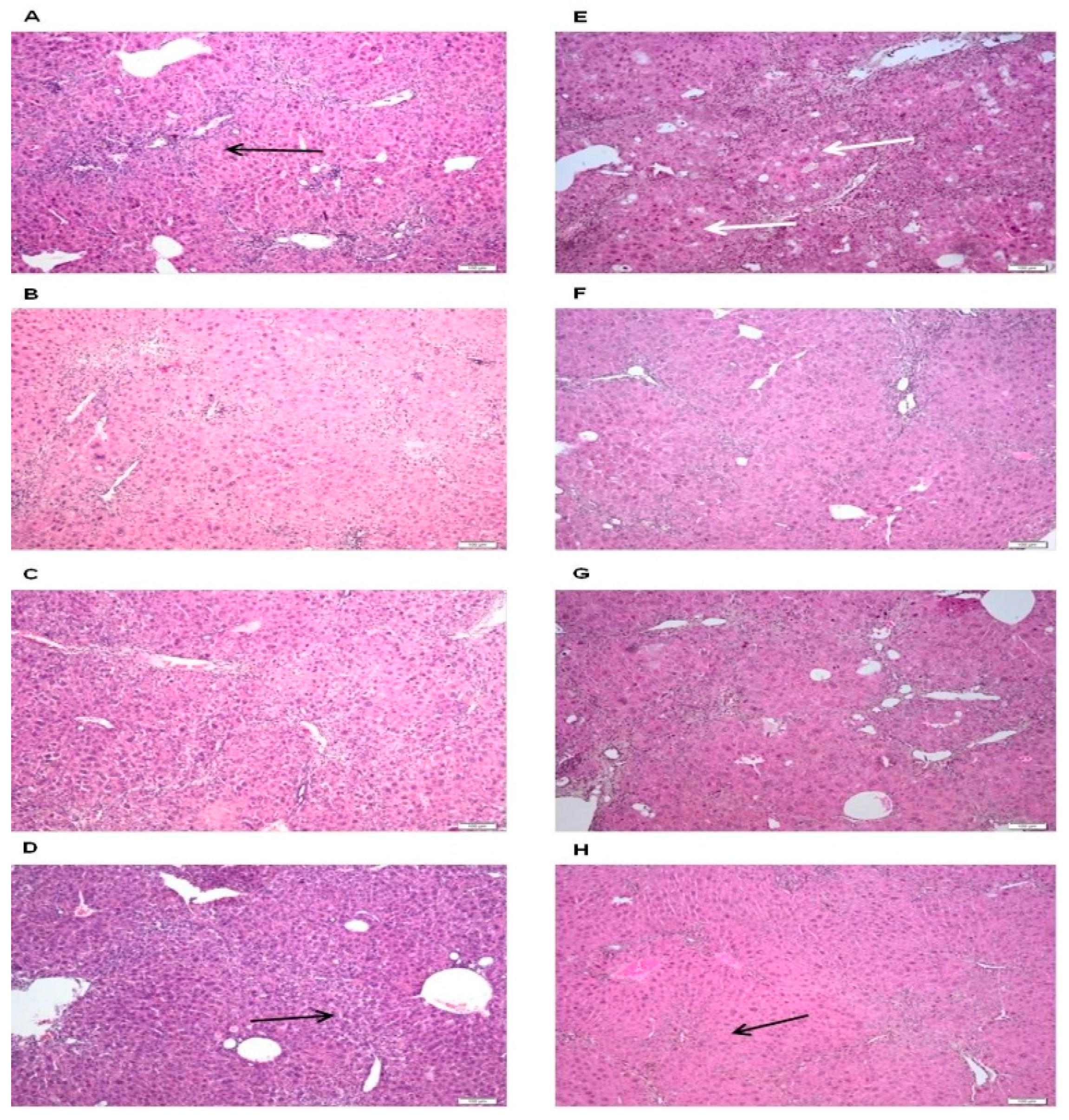
2.4. Hepatoprotective Activity of O. vulgare ssp. vulgare Extract
3. Materials and Methods
3.1. Plant Material
3.2. Chemicals
3.3. Preparation of O. vulgare ssp. vulgare Extract
3.4. HPLC Chromatographic Conditions and Instrumentation
3.5. Determination of Polyphenols Content
3.6. Determination of Antioxidant Properties of O. vulgare Extract
3.6.1. CUPRAC (Cupric Reducing Antioxidant Capacity) Assay
3.6.2. FRAP (Ferric Reducing Antioxidant Power) Assay
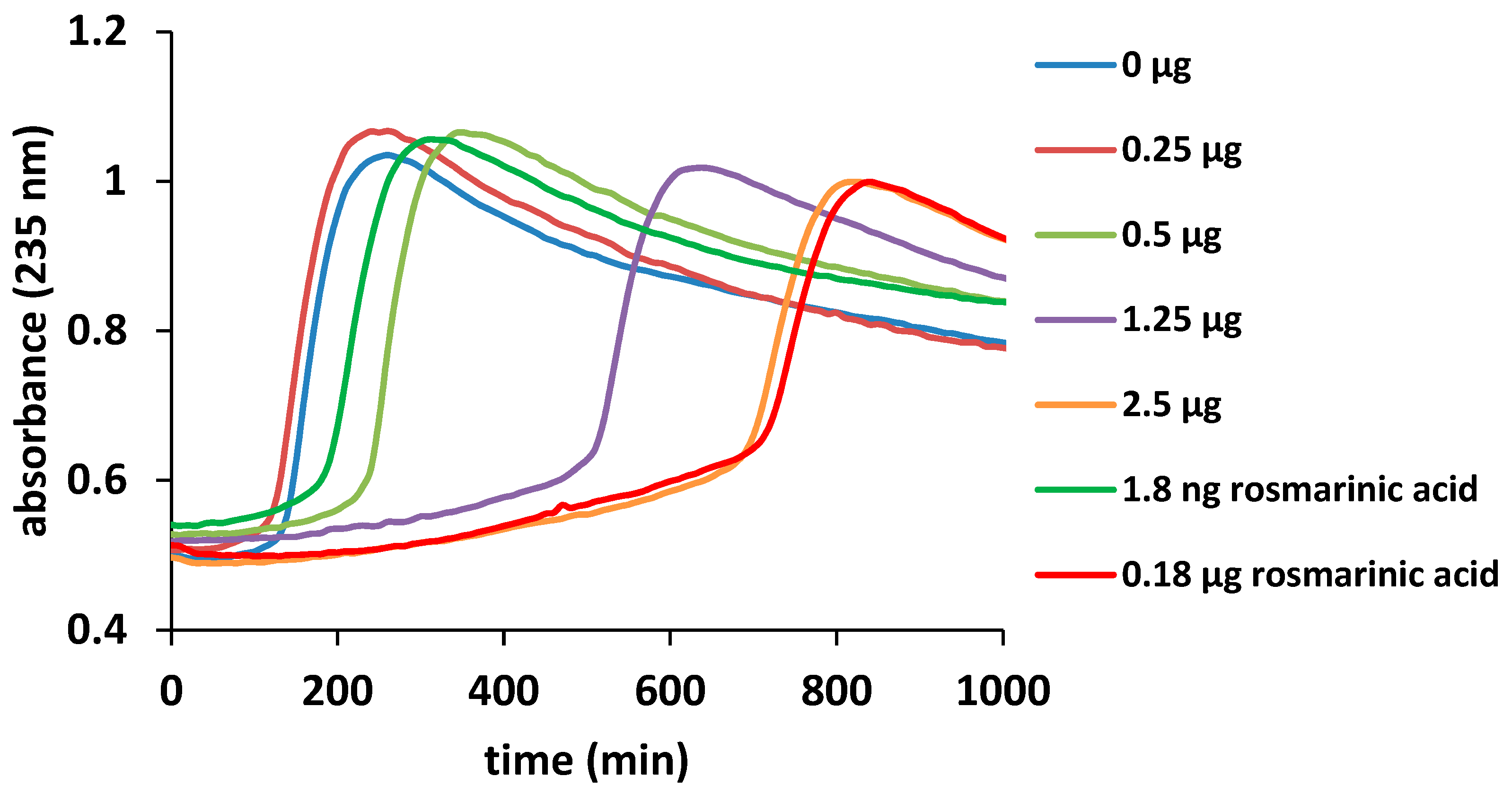
3.6.3. Inhibition of Lipid Peroxidation Catalyzed by Cytochrome c
3.6.4. Superoxide Radical (SO) Scavenging Activity Assay
3.7. Determination of Antimicrobial Activity
3.8. Hepatoprotective Activity Method
Oxidative Stress Markers
3.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sârbu, I.; Ştefan, N.; Oprea, A. Plante Vasculare din România: Determinator Ilustrat de Teren (Vascular Plants of Romania); Victor B Victor: Bucharest, Romania, 2013; p. 640. [Google Scholar]
- Gonceariuc, M.; Balmuș, Z.; Benea, A.; Barsan, V.; Sandu, T. Biochemical diversity of the Origanum vulgare ssp. vulgare L. and Origanum vulgare ssp. hirtum (link) ietswaart genotypes from Moldova. J. ASM Life Sci. 2015, 2, 92–100. [Google Scholar]
- Ciocârlan, V. Illustrated Flora of Romania. Pteridophyta et Spermatophyta; Ceres Publishing House: Bucharest, Romania, 2009; p. 662. [Google Scholar]
- Elshafie, H.S.; Armentano, M.F.; Carmosino, M.; Bufo, S.A.; De Feo, V.; Camele, I. Cytotoxic activity of Origanum vulgare L. on hepatocellular carcinoma cell line HepG2 and evaluation of its biological activity. Molecules 2017, 22, 1435. [Google Scholar] [CrossRef] [PubMed]
- Dragoeva, A.P.; Koleva, V.P.; Nanova, Z.D.; Kaschieva, M.Z. Allelopathy of cold water extracts from Origanum vulgare ssp. vulgare L. J. Agric. Chem. Environ. 2014, 3, 144–150. [Google Scholar] [CrossRef]
- Proestos, C.; Lytoudi, K.; Mavromelanidou, O.K.; Zoumpoulakis, P.; Sinanoglou, V.J. Antioxidant capacity of selected plant extracts and their essential oils. Antioxidants 2013, 2, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Brđjanin, S.; Bogdanović, N.; Kolundžić, M.; Milenković, M.; Golić, N.; Kojić, M.; Kundaković, T. Antimicrobial activity of oregano (Origanum vulgare L.) and basil (Ocimum basilicum L.) extracts. Adv. Technol. 2015, 4, 5–10. [Google Scholar] [CrossRef]
- Hawas, U.W.; El-Desoky, S.K.; Kawashty, S.A.; Sharaf, M. Two new flavonoids from Origanum vulgare. Nat. Prod. Res. 2008, 22, 1540–1543. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Bashir, S.; Khan, S.R.; Gilani, A.H. Antiurolithic activity of Origanum vulgare is mediated through multiple pathways. BMC Complement. Altern. Med. 2011, 11, 96. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.L.; Guo, Y.S.; Wang, C.H.; Li, G.Q.; Xu, J.J.; Chung, H.Y.; Ye, W.C.; Li, Y.L.; Wang, G.C. Phenolic compounds from Origanum vulgare and their antioxidant and antiviral activities. Food Chem. 2014, 152, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Rao, G.V.; Mukhopadhyay, T.; Annamalai, T.; Radhakrishnan, N.; Sahoo, M.R. Chemical constituents and biological studies of Origanum vulgare linn. Pharmacogn. Res. 2011, 3, 143–145. [Google Scholar] [CrossRef] [PubMed]
- Akrayi, H.; Salih, R.; Hamad, P. In vitro screening of antibacterial properties of rhus coriaria and Origanum vulgare against some pathogenic bacteria. Sci. J. Koya Univ. 2015, 3, 35–41. [Google Scholar] [CrossRef]
- Tusevski, O.; Kostovska, A.; Iloska, A.; Trajkovska, L.; Gadzovska Simic, S. Phenolic production and antioxidant properties of some Macedonian medicinal plants. Cent. Eur. J. Biol. 2014, 9, 888–900. [Google Scholar] [CrossRef]
- Gutiérrez-Grijalva, E.P.; Picos-Salas, M.A.; Leyva-López, N.; Criollo-Mendoza, M.S.; Vazquez-Olivo, G.; Heredia, J.B. Flavonoids and phenolic acids from oregano: occurrence, biological activity and health benefits. Plants 2017, 7, 2. [Google Scholar] [CrossRef] [PubMed]
- Pezzani, R.; Vitalini, S.; Iriti, M. Bioactivities of Origanum vulgare L.: An update. Phytochem. Rev. 2017, 16, 1253–1268. [Google Scholar] [CrossRef]
- Gîrd, C.E.; Duţu, L.E.; Costea, T.; Nencu, I.; Popescu, M.L.; Tudorel, O.O. Preliminary research concerning the obtaining of herbal extracts with potential neuroprotective activity note I. Obtaining and characterization of a selective Origanum vulgare L. dry extract. Farmacia (Bucharest, Rom.) 2016, 64, 680–687. [Google Scholar]
- Yan, F.; Azizi, A.; Janke, S.; Schwarz, M.; Zeller, S.; Honermeier, B. Antioxidant capacity variation in the oregano (Origanum vulgare L.) collection of the German National Genebank. Ind. Crop. Prod. 2016, 92, 19–25. [Google Scholar] [CrossRef]
- Spiridon, I.; Bodirlau, R.; Teaca, C.A. Total phenolic content and antioxidant activity of plants used in traditional Romanian herbal medicine. Cent. Eur. J. Biol. 2011, 6, 388–396. [Google Scholar] [CrossRef]
- Matei, A.O.; Gatea, F.; Radu, G.L. Analysis of phenolic compounds in some medicinal herbs by LC-MS. J. Chromatogr. Sci. 2015, 53, 1147–1154. [Google Scholar] [CrossRef] [PubMed]
- Shekarchi, M.; Hajimehdipoor, H.; Saeidnia, S.; Gohari, A.R.; Hamedani, M.P. Comparative study of rosmarinic acid content in some plants of Labiatae family. Pharmacogn. Mag. 2012, 8, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Benedec, D.; Hanganu, D.; Oniga, I.; Tiperciuc, B.; Olah, N.; Raita, O.; Bischin, C.; Silaghi, R.; Vlase, L. Assessment of rosmarinic acid content in six Lamiaceae species extracts and their antioxidant and antimicrobial potential. Pak. J. Pharm. Sci. 2015, 28, 2297–2303. [Google Scholar] [PubMed]
- Benedec, D.; Oniga, I.; Kozma-Imre, A.; Hanganu, D.; Țărmure, V.; Bodoki, E. Determination of rosmarinic acid by HPTLC-image analysis in medicinal teas and their biological properties. Farmacia 2017, 65, 605–609. [Google Scholar]
- Mot, A.C.; Bischin, C.; Muresan, B.; Parvu, M.; Damian, G.; Vlase, L.; Silaghi Dumitrescu, R. Antioxidant activity evaluation by physiologically relevant assays based on hemoglobin peroxidase activity and cytochrome c-induced oxidation of liposomes. Nat. Prod. Res. 2015, 30, 1–5. [Google Scholar] [CrossRef]
- Benedec, D.; Oniga, I.; Cuibus, F.; Sevastre, B.; Stiufiuc, G.; Duma, M.; Hanganu, D.; Iacovita, C.; Stiufiuc, R.; Lucaciu, C.M. Origanum vulgare mediated green synthesis of biocompatible gold nanoparticles simultaneously possessing plasmonic, antioxidant and antimicrobial properties. Int. J. Nanomed. 2018, 13, 1041–1058. [Google Scholar] [CrossRef] [PubMed]
- Bunghez, F.; Rotar, M.A.; Pop, R.M.; Romanciuc, F.; Csernatoni, F.; Fetea, F.; Diaconeasa, Z.; Socaciu, C. Comparative phenolic fingerprint and lc-esi+qtof-ms composition of oregano and rosemary hydrophilic extracts in relation to their antibacterial effect. Bull. UASVM Food Sci. Technol. 2015, 72. [Google Scholar] [CrossRef]
- Yotova, I.; Ignatova-Ivanova, T. In vitro study of antifungal activity of oregano (Origanum vulgare). Int. J. Curr. Microbiol. App. Sci. 2015, 4, 321–326. [Google Scholar]
- Sikander, M.; Malik, S.; Parveen, K.; Ahmad, M.; Yadav, D.; Hafeez, Z.B.; Bansal, M. Hepatoprotective effect of Origanum vulgare in wistar rats against carbon tetrachloride-induced hepatotoxicity. Protoplasma 2013, 250, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Benedec, D.; Vlase, L.; Oniga, I.; Mot, A.C.; Damian, G.; Hanganu, D.; Duma, M.; Silaghi-Dumitrescu, R. Polyphenolic composition, antioxidant and antibacterial activities for two Romanian subspecies of achillea distans waldst. et Kit. ex Willd. Molecules 2013, 18, 8725–8739. [Google Scholar] [CrossRef] [PubMed]
- Council of Europe. European Pharmacopoeia, 5th ed.; Council of Europe: Strasbourg, France, 2005; p. 221. [Google Scholar]
- Romanian Pharmacopoeia Commission National Medicines Agency. Romanian Pharmacopoeia; Medical Publishing House: Bucharest, Romania, 1993; p. 335. [Google Scholar]
- Özyürek, M.; Güçlü, K.; Tütem, E.; Sözgen Başkan, K.; Erçağ, E.; Çelik, S.E.; Baki, S.; Yıldız, L.; Karaman, Ş.; Apak, R. A comprehensive review of cuprac methodology. Anal. Methods 2011, 3, 2439–2453. [Google Scholar] [CrossRef]
- Olah, N.K.; Osser, G.; Câmpean, R.F.; Furtuna, F.R.; Benedec, D.; Filip, L.; Raita, O.; Hanganu, D. The study of polyphenolic compounds profile of some Rosmarinus officinalis L. extracts. Pak. J. Pharm. Sci. 2016, 29, 2355–2361. [Google Scholar] [PubMed]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of plasma (frap) as a measure of “antioxidant power”: The frap assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.N.; Bristi, N.J.; Rafiquzzaman, M. Review on in vivo and in vitro methods evaluation of antioxidant activity. Saudi Pharm. J. 2013, 21, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Reeves, D.S.; White, L.O. Principles of methods of assaying antibiotics. In Pharmaceutical Microbiology, 3rd ed.; Hugo, W.B., Russel, A.D., Eds.; Blackwell Scientific Publication: Oxford, UK, 1983; pp. 140–162. [Google Scholar]
- Pasca, C.; Mărghitas, L.; Dezmirean, D.; Bobiș, O.; Bonta, V.; Chirilă, F.; Matei, I.; Fiț, N. Medicinal plants based products tested on pathogens isolated from mastitis milk. Molecules 2017, 22, 1473. [Google Scholar] [CrossRef] [PubMed]
- Organization for Economic Co-operation and Development (OECD/OCDE). Test No. 425: Acute Oral Toxicity: Up-and-Down Procedure. In OECD Guidelines for the Testing of Chemicals; OECD: Paris, France, 2008; pp. 1–27. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |
| Compounds | [M − H]−, m/z | Retention Time (tR), min | UV Detection | MS Detection | Concentration (mg/g) |
|---|---|---|---|---|---|
| Gentisic acid | 153 | 3.69 ± 0.04 | NO | YES | <0.02 |
| Chlorogenic acid | 353 | 6.43 ± 0.05 | YES | YES | 2.10 ± 0.14 |
| p-Coumaric acid | 163 | 9.48 ± 0.08 | NO | YES | <0.02 |
| Hyperoside | 463 | 18.60 ± 0.12 | YES | YES | 1.05 ± 0.03 |
| Isoquercitrin | 463 | 20.29 ± 0.10 | YES | YES | 0.71 ± 0.19 |
| Rutin | 609 | 20.76 ± 0.15 | YES | YES | 0.64 ± 0.15 |
| Rosmarinic acid | 360 | 21.80 ± 0.10 | YES | YES | 12.83 ± 2.19 |
| Quercitrin | 447 | 23.64 ± 0.13 | YES | YES | 0.50 ± 0.08 |
| Quercetin | 301 | 27.55 ± 0.15 | NO | YES | <0.02 |
| Luteolin | 285 | 29.64 ± 0.19 | YES | YES | 0.10 ± 0.04 |
| Sample | TPC (mg GAE/g) | Flavonoid (mg RE/g) | Caffeic Acids (mg CAE/g) | CUPRAC (µM TE/g) | FRAP (µM TE/g) | SO Scavenging (µM TE/g) |
|---|---|---|---|---|---|---|
| O. vulgare | 94.69 ± 4.03 | 38.46 ± 3.54 | 29.92 ± 1.08 | 1284 ± 66 | 794.40 ± 25.80 | 44.00 ± 0.56 |
| Sample | Diameter of Inhibition Zone (mm) | ||||
|---|---|---|---|---|---|
| S. enteritidis | E. coli | L. monocytogenes | S. aureus | A. niger | |
| O. vulgare ssp. vulgare | 18.0 ± 0.00 | 16.0 ± 0.00 | 17.0 ± 1.00 | 16.0 ± 1.00 | 19.0 ± 0.00 |
| Gentamicin | 19.0 ± 1.00 | 18.0 ± 1.06 | 22.0 ± 0.50 | 18.0 ± 0.00 | - |
| Amphotericin B | - | - | - | - | 21.0 ± 0.00 |
| Samples | MICs (µg/mL) | ||||
|---|---|---|---|---|---|
| S.enteritidis | E. coli | L.monocytogenes | S. aureus | A. niger | |
| O. vulgare ssp. vulgare | 78.13 | 156.25 | 156.25 | 78.13 | 19.53 |
| 70% Ethanol | 625 | 2500 | 1250 | 625 | 2500 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oniga, I.; Pușcaș, C.; Silaghi-Dumitrescu, R.; Olah, N.-K.; Sevastre, B.; Marica, R.; Marcus, I.; Sevastre-Berghian, A.C.; Benedec, D.; Pop, C.E.; et al. Origanum vulgare ssp. vulgare: Chemical Composition and Biological Studies. Molecules 2018, 23, 2077. https://doi.org/10.3390/molecules23082077
Oniga I, Pușcaș C, Silaghi-Dumitrescu R, Olah N-K, Sevastre B, Marica R, Marcus I, Sevastre-Berghian AC, Benedec D, Pop CE, et al. Origanum vulgare ssp. vulgare: Chemical Composition and Biological Studies. Molecules. 2018; 23(8):2077. https://doi.org/10.3390/molecules23082077
Chicago/Turabian StyleOniga, Ilioara, Cristina Pușcaș, Radu Silaghi-Dumitrescu, Neli-Kinga Olah, Bogdan Sevastre, Raluca Marica, Ioan Marcus, Alexandra Cristina Sevastre-Berghian, Daniela Benedec, Carmen Elena Pop, and et al. 2018. "Origanum vulgare ssp. vulgare: Chemical Composition and Biological Studies" Molecules 23, no. 8: 2077. https://doi.org/10.3390/molecules23082077
APA StyleOniga, I., Pușcaș, C., Silaghi-Dumitrescu, R., Olah, N.-K., Sevastre, B., Marica, R., Marcus, I., Sevastre-Berghian, A. C., Benedec, D., Pop, C. E., & Hanganu, D. (2018). Origanum vulgare ssp. vulgare: Chemical Composition and Biological Studies. Molecules, 23(8), 2077. https://doi.org/10.3390/molecules23082077







