Beneficial Effect of Herbal Formulation KM1608 on Inflammatory Bowl Diseases: A Preliminary Experimental Study
Abstract
:1. Introduction
2. Results and Discussion
2.1. HPLC Profile of KM1608
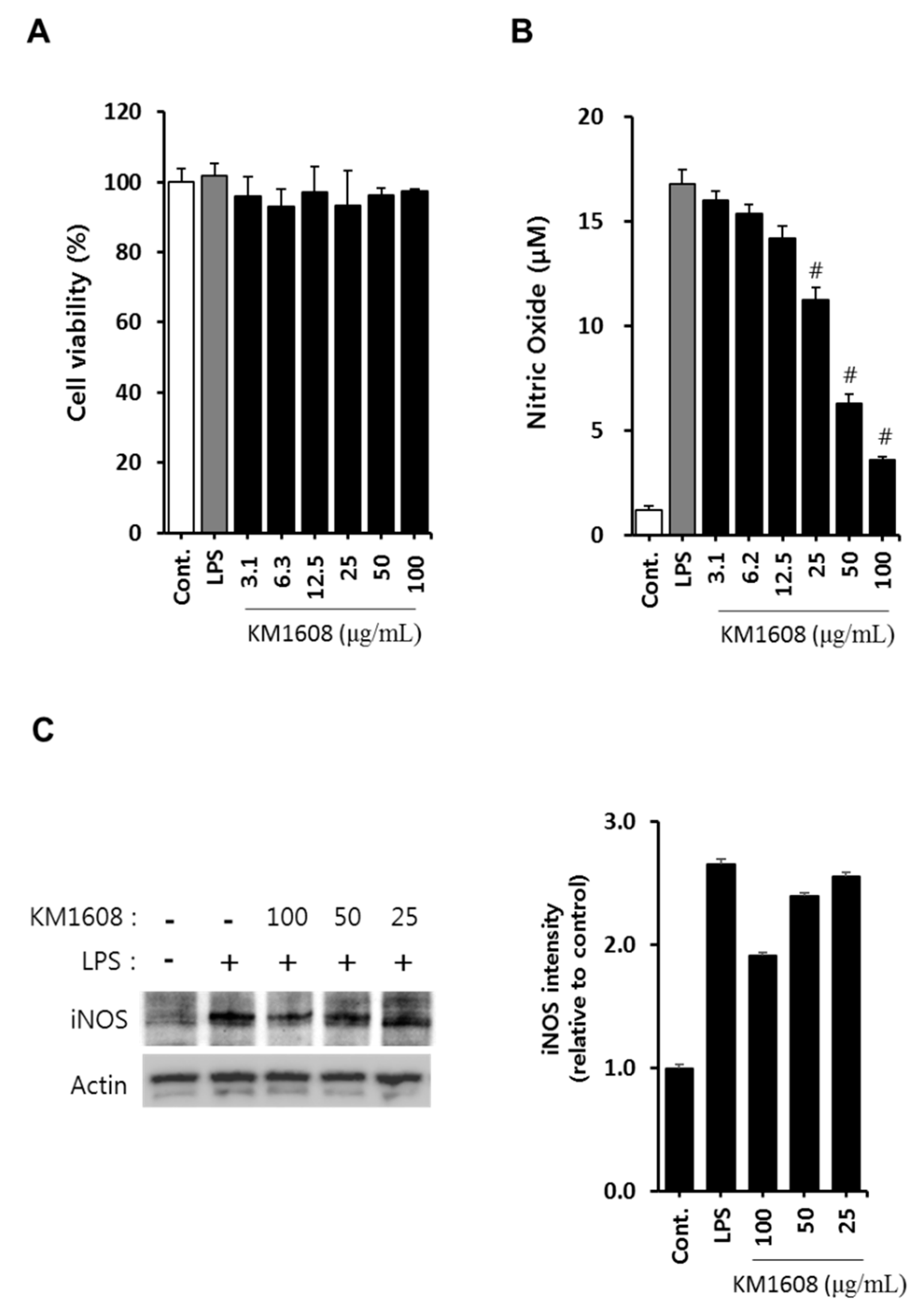
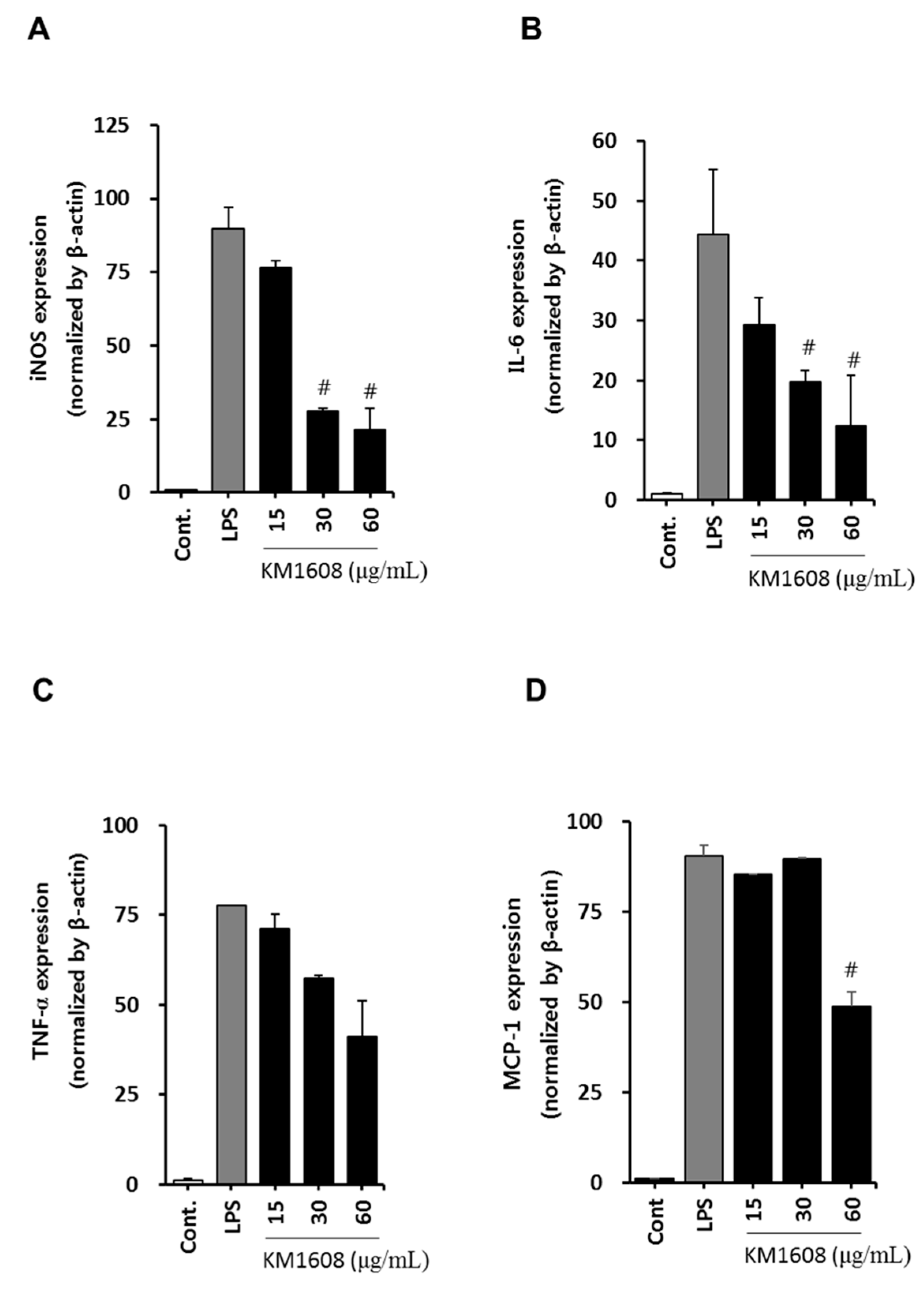
2.2. Anti-Inflammatory Activity of KM1608 in LPS-Induced RAW264.7 Cells
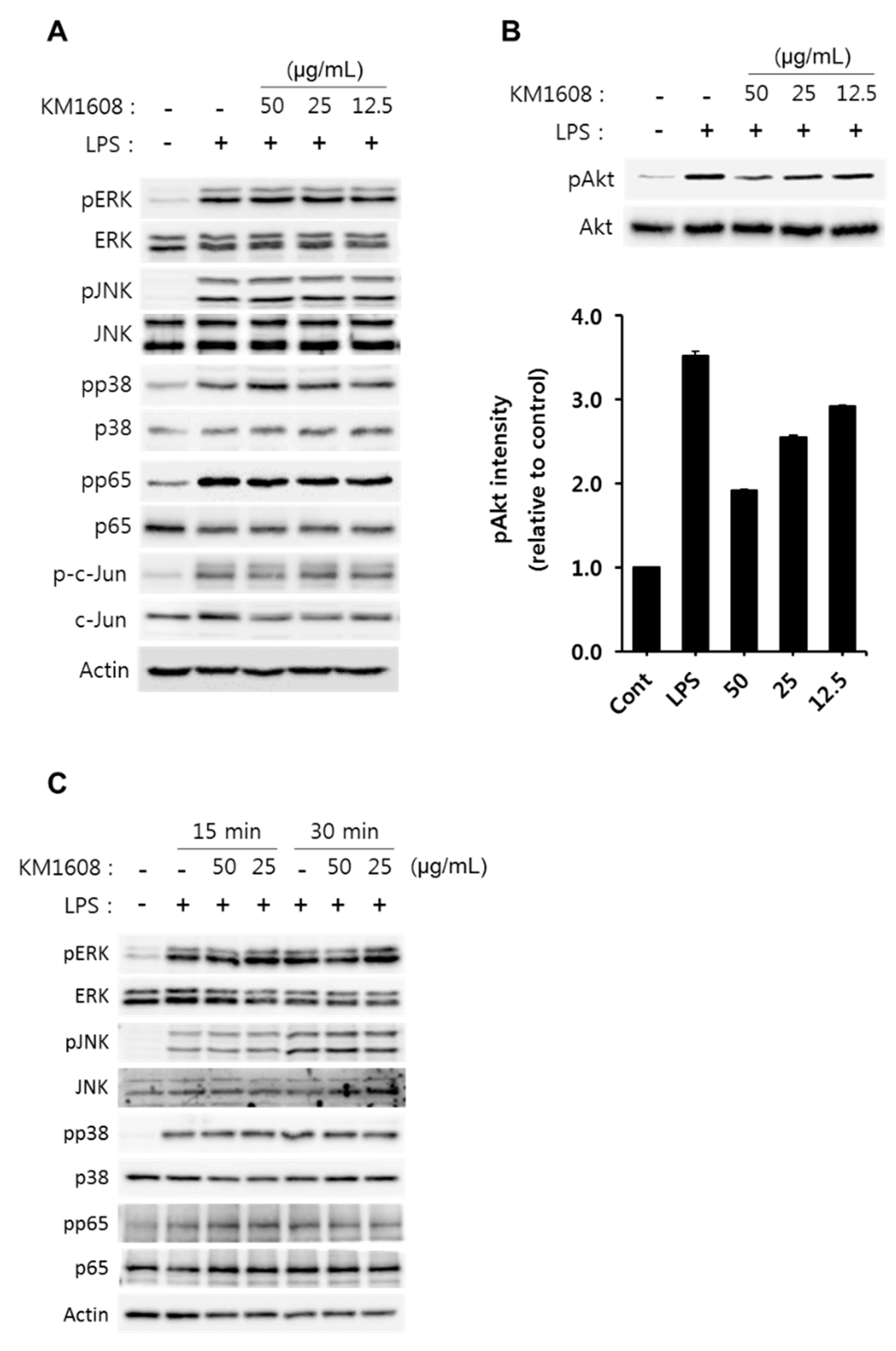
2.3. Effect of KM1608 on Akt Phosphorylation in LPS-Induced RAW264.7 Cells
2.4. Effect of KM1608 on Clinical Signs of DSS-Induced Colitis in Mice
3. Materials and Methods
3.1. Plant Materials and KM1608 Extraction
3.2. HPLC-UV/DAD Conditions
3.3. Antibodies and Reagents
3.4. Animal and Cell Culture
3.5. Induction of Colitis by DSS
3.6. Disease Activity Index (DAI)
3.7. Cell Viability
3.8. Determination of Nitrite, IL-6, MCP-1, TNF-a, and MPO Production
3.9. Cell Lysate Preparation and Immunoblotting
3.10. Real-Time Reverse Transcription PCR (qRT-PCR)
3.11. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bingham, C.O. The pathogenesis of rheumatoid arthritis: Pivotal cytokines involved in bone degradation and inflammation. J. Rheumatol. Suppl. 2002, 65, 3–9. [Google Scholar] [PubMed]
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, I.E. The role of cytokines, chemokines, and adhesion molecules in the pathogenesis of idiopathic inflammatory myopathies. Curr. Rheumatol. Rep. 2000, 2, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Payne, D.N.R. Nitric oxide in allergic airway inflammation. Curr. Opin. Allergy Clin. Immunol. 2003, 3, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Baumgart, D.C.; Carding, S.R. Inflammatory bowel disease: Cause and immunobiology. Lancet 2007, 369, 1627–1640. [Google Scholar] [CrossRef]
- Kornbluth, A.; Sachar, D.B. Ulcerative colitis practice guidelines in adults (update): American college of gastroenterology, practice parameters committee. Am. J. Gastroenterol. 2004, 99, 1371–1385. [Google Scholar] [CrossRef] [PubMed]
- Choi, C.H.; Kim, Y.H.; Kim, Y.S.; Ye, B.D.; Lee, K.M.; Lee, B.I.; Jung, S.A.; Kim, W.H.; Lee, H. IBD Study Group of the Korean Association for the Study of Intestinal Diseases. Guidelines for the management of ulcerative colitis. Korean J. Gastroenterol. 2012, 59, 118–140. [Google Scholar] [CrossRef] [PubMed]
- Jeen, Y.T.; Kim, J.H. Advances in ulcerative colitis therapy. Korean J. Med. 2009, 76, 654–660. [Google Scholar]
- Kim, S.Y.; Park, J.W.; Ryu, B.H. Effects of Auklandia Lappa on dextran sulfate sodium-induced murine colitis. Korean J. Orient. Int. Med. 2013, 34, 134–146. [Google Scholar]
- Kim, D.S.; Kim, S.H.; Kee, J.Y.; Han, Y.H.; Park, J.; Mun, J.G.; Joo, M.-J.; Jeon, Y.-D.; Park, S.-H.; Park, S.-J.; et al. Eclipta prostrata improves dss-induced colitis through regulation of inflammatory response in intestinal epithelial cells. Am. J. Chin. Med. 2017, 45, 1047–1060. [Google Scholar] [CrossRef] [PubMed]
- Yoon, W.H.; Lee, K.H. Anti-inflammatory, anti-arthritic and analgesic effect of the herbal extract made from bacopa monnieriis, cassia fistula and phyllanthus polyphyllus. Nat. Prod. Sci. 2017, 23, 108–112. [Google Scholar] [CrossRef]
- Shin, M.S.; Park, J.Y.; Lee, J.; Yoo, H.H.; Hahm, D.H.; Lee, S.C.; Lee, S.; Hwang, G.S.; Jung, K.; Kang, K.S. Anti-inflammatory effects and corresponding mechanisms of cirsimaritin extracted from Cirsium japonicum var. maackii Maxim. Bioorg. Med. Chem. Lett. 2017, 27, 3076–3080. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.H.; Yoon, W.H. Preventive effects of co-treatment with fucoidan and lutein on the development of Inflammatory Bowel Disease in DSS mouse model. Nat. Prod. Sci. 2011, 17, 234–238. [Google Scholar]
- Jang, S.E.; Choi, J.R.; Han, M.J.; Kim, D.H. The preventive and curative effect of cyanidin-3β-d-glycoside and its metabolite protocatechuic acid against TNBS-induced colitis in mice. Nat. Prod. Sci. 2016, 22, 282–286. [Google Scholar] [CrossRef]
- Kang, C.S.; Tae, J.; Ham, S.H.; Kim, D.K.; Lee, Y.M.; Lee, K.S.; Yun, Y.G. Administration of aqueous extract of Schizandra chinensis fruit inhibits the experimental colitis in mice. Nat. Prod. Sci. 2007, 13, 78–84. [Google Scholar]
- Singh, P.; Malhotra, H. Terminalia chebula: A review pharmacognistic and phytochemical studies. Int. J. Recent Sci. Res. 2017, 8, 21496–21507. [Google Scholar]
- Li, A.; Sun, A.; Liu, R. Preparative isolation and purification of costunolide and dehydrocostuslactone from Aucklandia lappa Decne by high-speed counter-current chromatography. J. Chromatogr. A 2005, 1076, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Ali, B.H.; Blunden, G.; Tanira, M.O.; Nemmar, A. Some phytochemical, pharmacological and toxicological properties of ginger (Zingiber officinale Roscoe): A review of recent research. Food Chem. Toxicol. 2008, 46, 409–420. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Kim, N.Y.; Jang, M.K.; Son, H.J.; Kim, K.M.; Sohn, D.H.; Lee, S.H.; Ryu, J.H. A sesquiterpene, dehydrocostus lactone, inhibits the expression of inducible nitric oxide synthase and TNF-α in LPS-activated macrophages. Planta Med. 1999, 65, 104–108. [Google Scholar] [CrossRef] [PubMed]
- Pandey, M.M.; Rastogi, S.; Rawat, A.K.S. Saussurea costus: Botanical, chemical and pharmacological review of an ayurvedic medicinal plant. J. Ethnopharmacol. 2007, 110, 379–390. [Google Scholar] [CrossRef] [PubMed]
- Maged, R.; Nordin, N.; Abdulla, M.S. Anti-inflammatory effects of zingiber officinale roscoe involve suppression of nitric oxide and prostaglandin E2 production. Zanco J. Med. Sci. 2013, 17, 349–356. [Google Scholar] [CrossRef]
- Mansouri, M.T.; Hemmati, A.A.; Naghizadeh, B.; Mard, S.A.; Rezaie, A.; Ghorbanzadeh, B. A study of the mechanisms underlying the anti-inflammatory effect of ellagic acid in carrageenan-induced paw edema in rats. Indian J. Pharmacol. 2015, 47, 292–298. [Google Scholar] [PubMed]
- Pandurangan, A.K.; Mohebali, N.; Norhaizan, M.E.; Looi, C.Y. Gallic acid attenuates dextran sulfate sodium-induced experimental colitis in BALB/c mice. Drug Des. Devel. Ther. 2015, 9, 3923–3934. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.T.; Lin, C.Y.; Ho, D.H.; Peng, J.; Chen, Y.; Tsang, S.W.; Bian, Z.X. Inhibitory effect of the gallotannin corilagin on dextran sulfate sodium-induced murine ulcerative colitis. J. Nat. Prod. 2013, 76, 2120–2125. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Li, L.; Kim, S.H.; Hagerman, A.E.; Lü, J. Anti-cancer, anti-diabetic and other pharmacologic and biological activities of penta-galloyl-glucose. J. Pharm. Res. Int. Pharm. Res. 2009, 26, 2066–2080. [Google Scholar] [CrossRef] [PubMed]
- Hossen, M.J.; Hong, Y.D.; Baek, K.S.; Yoo, S.; Hong, Y.H.; Kim, J.H.; Lee, J.O.; Kim, D.; Park, J.; Cho, J.Y. In vitro antioxidative and anti-inflammatory effects of the compound K-rich fraction BIOGF1K, prepared from Panax ginseng. J. Ginseng Res. 2017, 41, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Hommes, D.W.; Peppelenbosch, M.P.; Van Deventer, S.J.H. Mitogen activated protein (MAP) kinase signal transduction pathways and novel anti-inflammatory targets. Gut 2003, 52, 144–151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murthy, S.N.S.; Cooper, H.S.; Shim, H.; Shah, R.S.; Ibrahim, S.A.; Sedergran, D.J. Treatment of dextran sulfate sodium-induced murine colitis by intracolonic cyclosporin. Digest. Dis. Sci. 1993, 38, 1722–1734. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.S.; Song, J.H.; Choi, P.; Lee, J.H.; Kim, S.Y.; Shin, K.S.; Kang, K.S. Stimulation of innate immune function by panax ginseng after heat processing. J. Agric. Food Chem. 2018, 66, 4652–4659. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Kim, J.; Park, J.Y.; Kang, K.S.; Park, J.H.; Hwang, G.S. Processed Panax ginseng, sun ginseng, inhibits the differentiation and proliferation of 3T3-L1 preadipocytes and fat accumulation in Caenorhabditis elegans. J. Ginseng Res. 2017, 41, 257–267. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
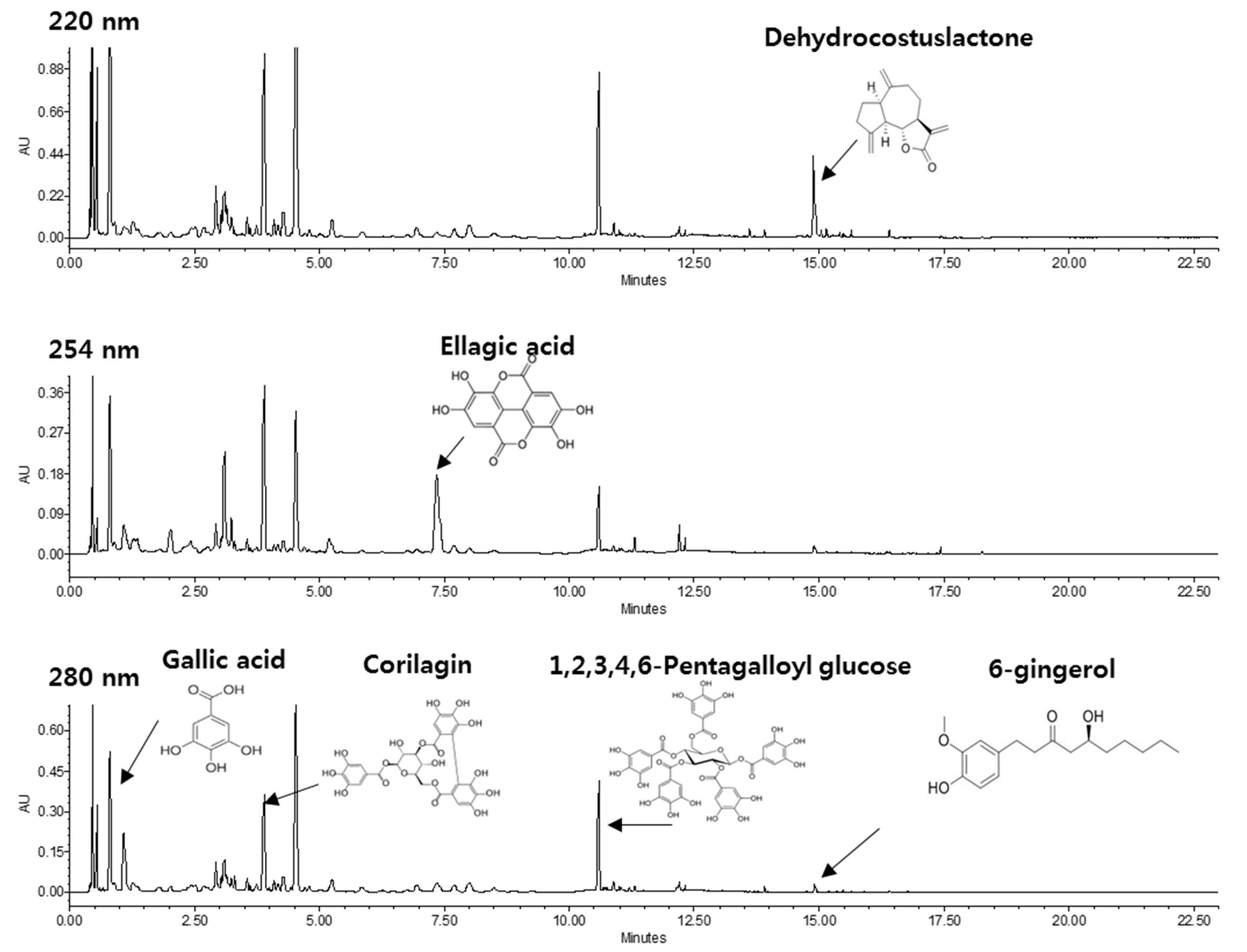



| Plant | Active Compound | Content (%) |
|---|---|---|
| Aucklandia lappa DC. | dehydrocostus lactone | 0.68–1.2 |
| Terminalia chebula Retz | ellagic acid | 0.94–1.5 |
| Zingiber officinale Roscoe | 6-gingerol | 0.1–0.2 |
| Score | Weight Loss | Stool Consistency | Occult/Gross Rectal Bleeding |
|---|---|---|---|
| 0 | None | Normal | Normal |
| 1 | 1–5% | - | - |
| 2 | 5–10% | Loose stool | Hemoccult |
| 3 | 10–20% | - | - |
| 4 | >20% | Diarrhea | Gross bleeding |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shin, M.-S.; Kim, S.-B.; Lee, J.; Choi, H.-S.; Park, J.; Park, J.Y.; Lee, S.; Hwang, G.S.; Koo, B.A.; Kang, K.S. Beneficial Effect of Herbal Formulation KM1608 on Inflammatory Bowl Diseases: A Preliminary Experimental Study. Molecules 2018, 23, 2068. https://doi.org/10.3390/molecules23082068
Shin M-S, Kim S-B, Lee J, Choi H-S, Park J, Park JY, Lee S, Hwang GS, Koo BA, Kang KS. Beneficial Effect of Herbal Formulation KM1608 on Inflammatory Bowl Diseases: A Preliminary Experimental Study. Molecules. 2018; 23(8):2068. https://doi.org/10.3390/molecules23082068
Chicago/Turabian StyleShin, Myoung-Sook, Sang-Back Kim, Jaemin Lee, Han-Seok Choi, Jimin Park, Jun Yeon Park, Sullim Lee, Gwi Seo Hwang, Bon Am Koo, and Ki Sung Kang. 2018. "Beneficial Effect of Herbal Formulation KM1608 on Inflammatory Bowl Diseases: A Preliminary Experimental Study" Molecules 23, no. 8: 2068. https://doi.org/10.3390/molecules23082068









