Structure Characterization and Hypoglycaemic Activities of Two Polysaccharides from Inonotus obliquus
Abstract
:1. Introduction
2. Results
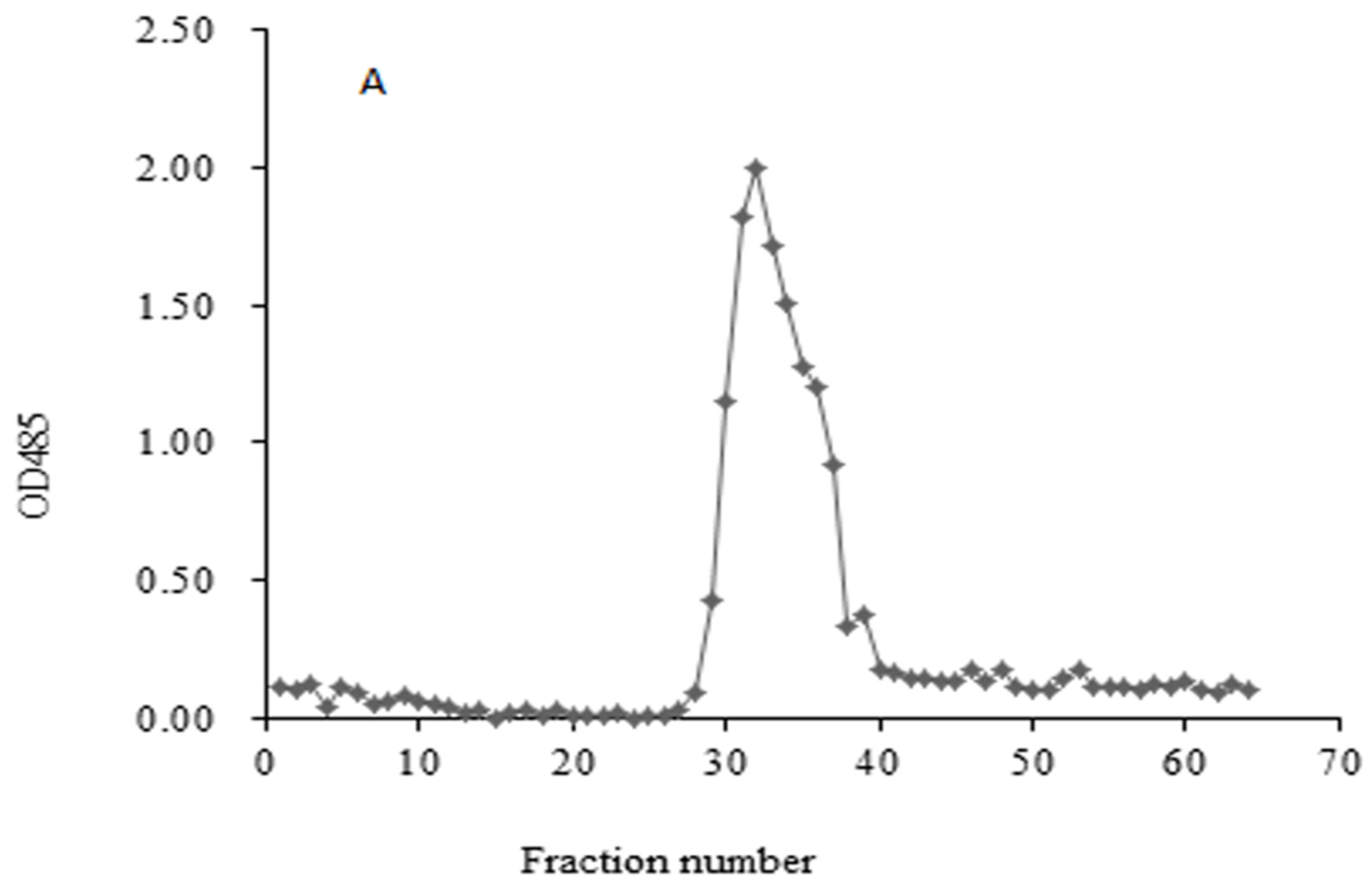
2.1. Homogeneity and Molecular Weight of HIOP1-S and HIOP2-S
2.2. Monosaccharide Composition of HIOP1-S and HIOP2-S
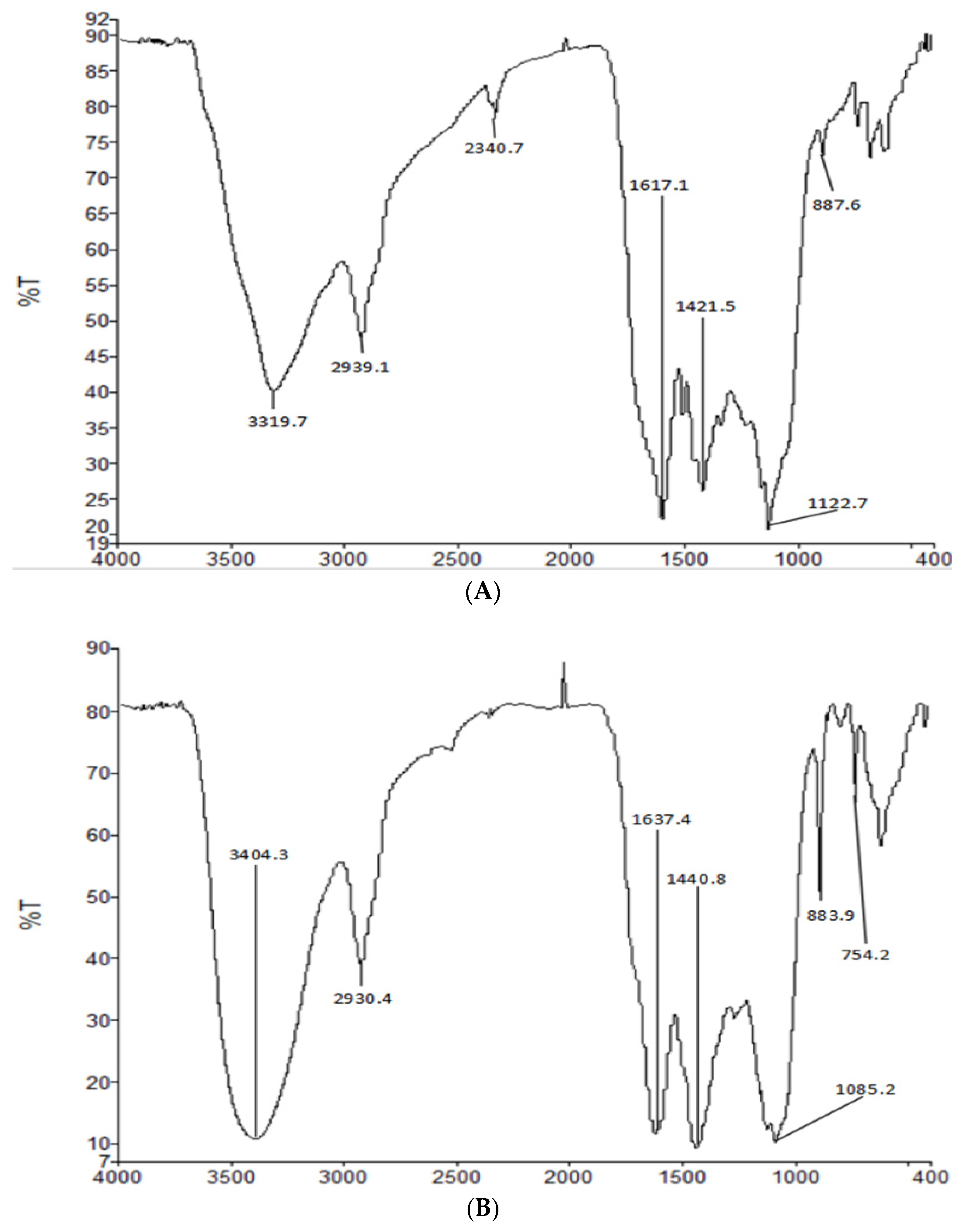
2.3. FT-IR of HIOP1-S and HIOP2-S
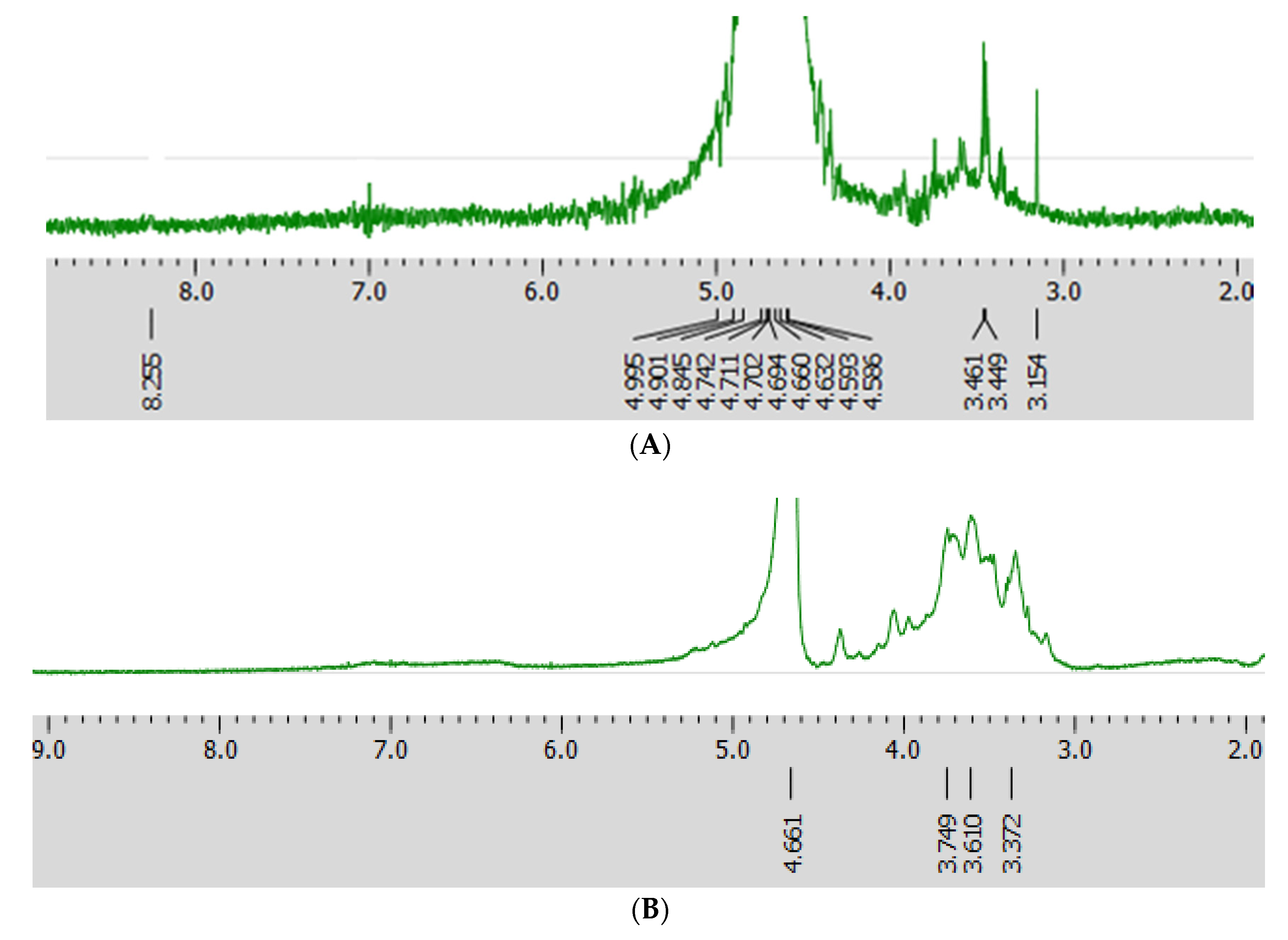
2.4. NMR of HIOP1-S and HIOP2-S
2.5. Ultraviolet Spectrum Scanning of HIOP1-S and HIOP2-S
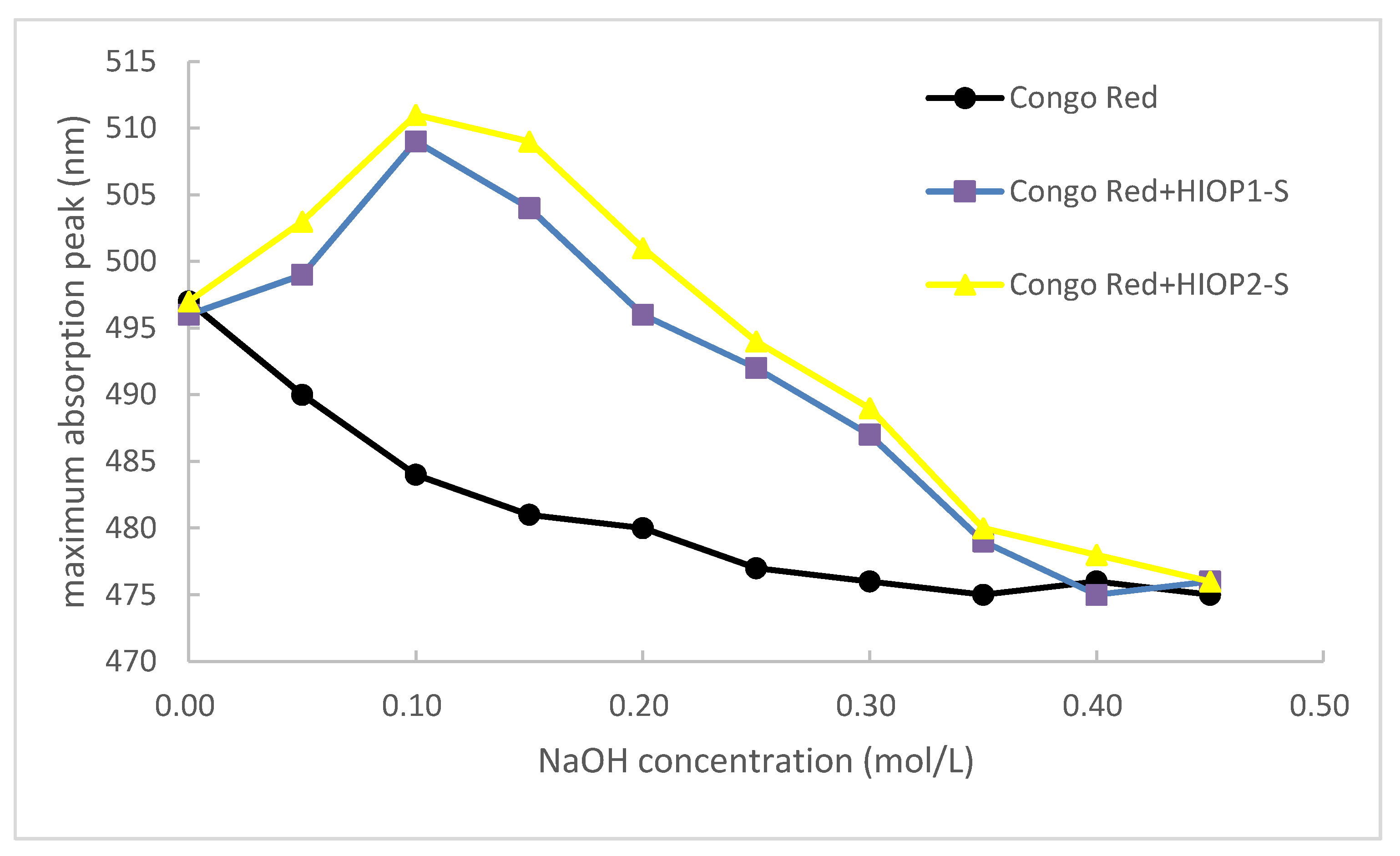
2.6. Congo Red Test of HIOP1-S and HIOP2-S
2.7. α-Glucosidase Inhibition Activity
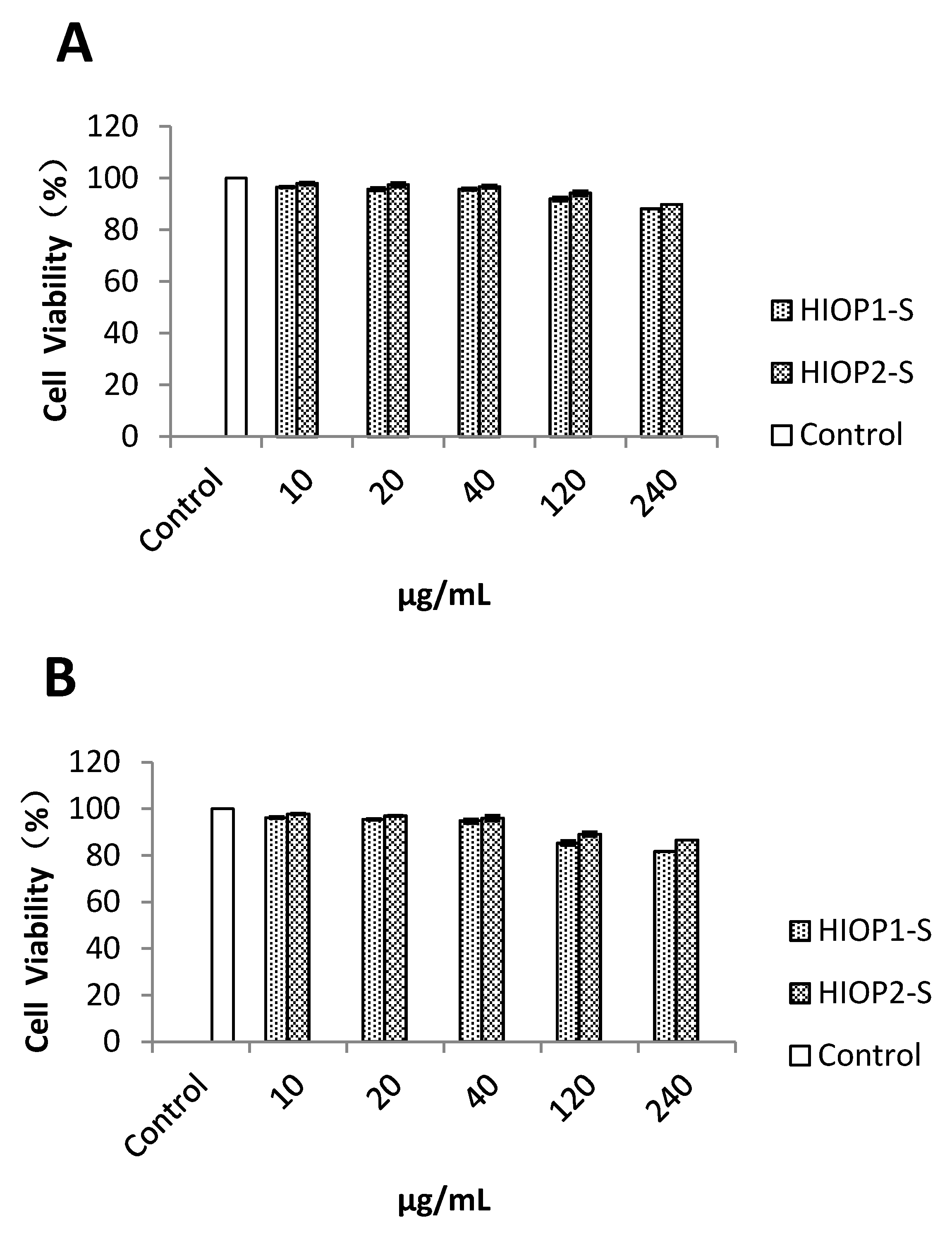
2.8. Cytotoxicity of the Polysaccharides at Different Concentrations on HepG2 Cells
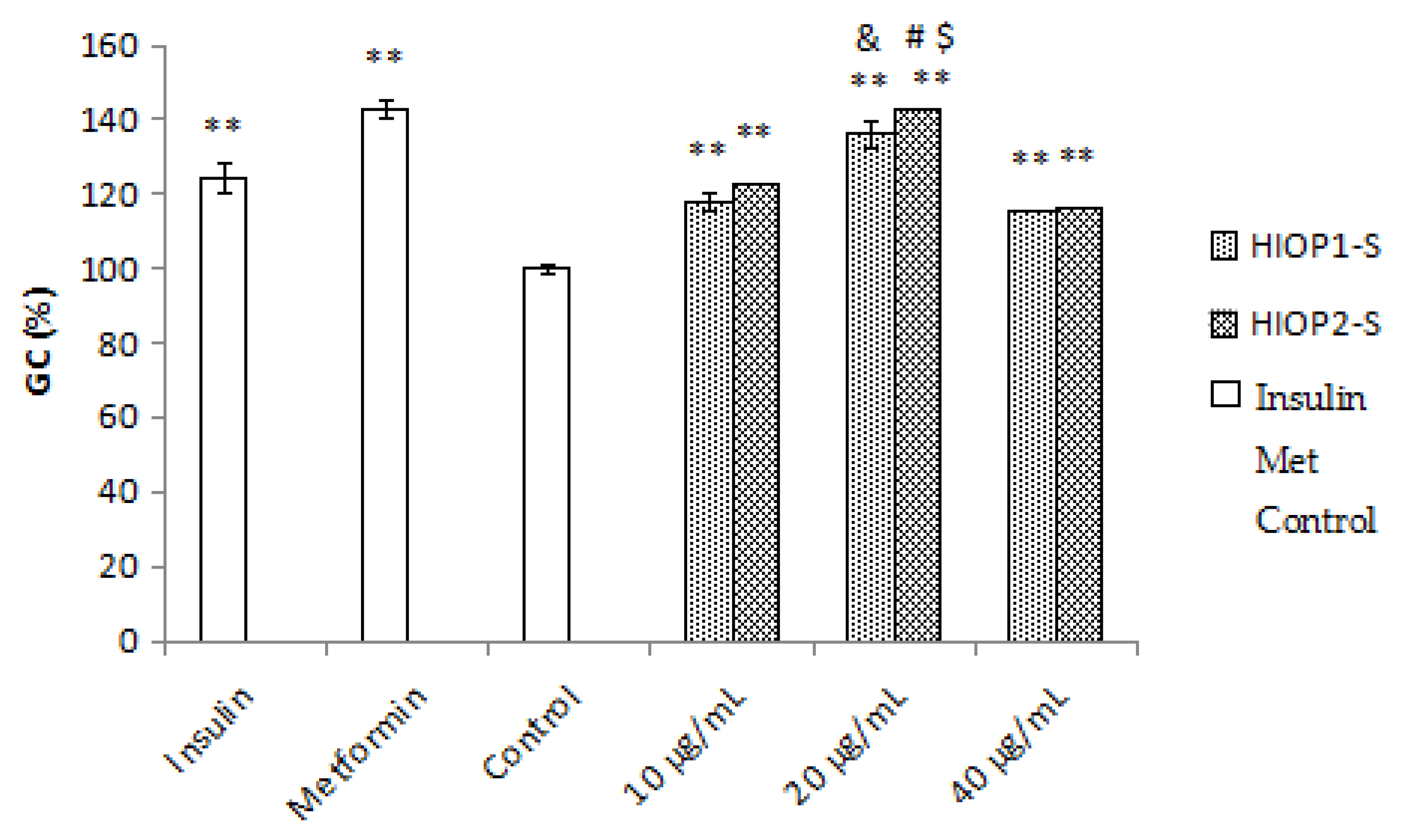
2.9. Glucose Consumption Assay
2.10. Effect of HIOP1-S and HIOP2-S on Fasting Blood Glucose in STZ-Induced Diabetic Mice
2.11. Effects of HIOP1-S and HIOP2-S on the Body Weights of STZ-Induced Diabetic Mice
3. Materials and Methods
3.1. Materials and Regents
3.2. Extraction, Isolation and Purification of Polysaccharides
3.3. Chemical Composition Analysis of HIOP1-S and HIOP2-S
3.4. Homogeneity and Molecular Weight Analysis
3.5. Monosaccharide Composition Analysis
3.6. Infrared Spectral Analysis (FT-IR)
3.7. Nuclear Magnetic Resonance (NMR) Spectroscopy
3.8. Ultraviolet Spectrum Scanning
3.9. Congo Red Test
3.10. Assay of α-glucosidase Inhibitory Activity
3.11. Cytotoxicity Assay to HepG2 Cells
3.12. Glucose Consumption Assay
3.13. Animal Experiments
3.14. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Guariguata, L.; Whiting, D.R.; Hambleton, I.; Beagley, J.; Linnenkamp, U.; Shaw, J.E. Global estimates of diabetes prevalence for 2013 and projections for 2035. Diabetes Res. Clin. Pract. 2014, 103, 137–149. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Zhang, C.; Li, L.; Wang, Z.; Xiao, W.; Yang, Z. Phytomedicine. Int. J. Phytother. Phytopharmacol. 2014, 21, 807–814. [Google Scholar]
- Rangika, B.S.; Dayananda, P.D.; Peiris, D.C. Hypoglycemic and hypolipidemic activities of aqueous extract of flowers from Nycantus arbor-tristis L. in male mice. BMC Complement. Altern. Med. 2015, 15, 289. [Google Scholar] [CrossRef] [PubMed]
- Akindele, A.J.; Otuguor, E.; Singh, D.; Ota, D.; Benebo, A.S. Hypoglycemic, antilipidemic and antioxidant effects of valproic acid in alloxan-induced diabetic rats. Eur. J. Pharmacol. 2015, 762, 174–183. [Google Scholar] [CrossRef] [PubMed]
- Xiong, W.T.; Gu, L.; Wang, C.; Sun, H.X.; Liu, X. Anti-hyperglycemic and hypolipidemic effects of Cistanche tubulosa in type 2 diabetic db/db mice. J. Ethnopharmacol. 2013, 150, 935–945. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Hui, J.; Kou, W.; Xin, R.; Jia, F.; Wang, N. Identification of Inonotus obliquus and analysis of antioxidation and antitumor activities of polysaccharides. Curr. Microbiol. 2008, 57, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Ding, S.; Ai, L.; Deng, K. Antitumor and immunomodulatory activity of water-soluble polysaccharide from Inonotus obliquus. Carbohydr. Polym. 2012, 90, 870–874. [Google Scholar] [CrossRef] [PubMed]
- Niu, H.; Song, D.; Mu, H.; Zhang, W.; Sun, F.; Duan, J. Investigation of three lignin complexes with antioxidant and immunological capacities from Inonotus obliquus. Int. J. Biol. Macromol. 2016, 86, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Mai, Q.; Ma, J.; Xu, M.; Wang, X.; Cui, T.; Qiu, F.; Han, G. Triterpenoids from Inonotus obliquus and their antitumor activities. Fitoterapia 2015, 101, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Gu, X.; Huang, S.Q.; Li, J.; Wang, X.; Tang, J. Optimization of ultrasonic/microwave assisted extraction (UMAE) of polysaccharides from Inonotus obliquus and evaluation of its anti-tumor activities. Int. J. Biol. Macromol. 2010, 46, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.Q.; Ding, S.; Fan, L. Antioxidant activities of five polysaccharides from Inonotus obliquus. Int. J. Biol. Macromol. 2012, 50, 1183–1187. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; Mu, H.; Zhou, S.; Zhang, Y.; Zhu, X. Chemical analysis and antioxidant activity of polysaccharides extracted from Inonotus obliquus sclerotia. Int. J. Biol. Macromol. 2013, 62, 691–696. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Wu, Y.; Chen, H. Comparative antioxidative characteristics of polysaccharide-enriched extracts from natural sclerotia and cultured mycelia in submerged fermentation of Inonotus obliquus. Food Chem. 2011, 127, 74–79. [Google Scholar] [CrossRef]
- Wang, C.; Chen, Z.; Pan, Y.; Gao, X.; Chen, H. Effects of polysaccharides from Inonotus obliquus and its chromium (III) complex on advanced glycation end-products formation, α-amylase, α-glucosidase activity and H2O2-induced oxidative damage in hepatic L02 cells. Food Chem. Toxicol. 2018, 116, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, C.; Jiang, H.; Zhou, H.; Li, P.; Wang, F. Isolation, structural characterization and neurotrophic activity of a polysaccharide from Phellinus ribis. Carbohydr. Polym. 2015, 127, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Ye, H.; Zhou, C.; Li, W.; Hu, B.; Wang, X.; Zeng, X. Structural elucidation of polysaccharide fractions from brown seaweed Sargassum pallidum. Carbohydr. Polym. 2013, 97, 659–664. [Google Scholar] [CrossRef] [PubMed]
- Figueiro, S.; Goes, J.; Moreira, R.; Sombra, A. On the physico-chemical and dielectric properties of glutaraldehyde crosslinked galactomannan–collagen films. Carbohydr. Polym. 2004, 56, 313–320. [Google Scholar] [CrossRef]
- Li, S.; Pan, C.; Xia, W.; Zhang, W.; Wu, S. Structural characterization of the polysaccharide moiety of an aqueous glycopeptide from mannatide. Int. J. Biol. Macromol. 2014, 67, 351–359. [Google Scholar] [CrossRef] [PubMed]
- He, S.D.; wang, X.; Zhang, Y.; Wang, J.; Sun, H.J.; Wang, J.H.; Cao, X.D.; Ye, Y.K. Isolation and prebiotic activity of water-soluble polysaccharides fractions from the bamboo shoots (Phyllostachys praecox). Carbohydr. Polym. 2016, 151, 295–304. [Google Scholar] [CrossRef] [PubMed]
- HARA, C.; Kiho, T.; Ukai, S. Anti-inflammatory(1-3)β-d-Glucan. Carbohydr. Res. 1983, 117, 201–213. [Google Scholar] [CrossRef]
- Deng, Y.T.; Linshiau, S.Y.; Shyur, L.F.; Lin, J.K. Pu-erh tea polysaccharides decrease blood sugar by inhibition of α-glucosidase activity in vitro and in mice. Food Funct. 2015, 6, 1539–1546. [Google Scholar] [CrossRef] [PubMed]
- Bressler, R.; Johnson, D. New Pharmacological Approaches to Therapy of NIDDM. Diabetes Care 1992, 15, 792–805. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.; Wu, J.; Zhang, Y.; Chen, H.; Wang, Y. Physicochemical characterization of puerh tea polysaccharides and their antioxidant and α-glycosidase inhibition. J. Funct. Foods. 2014, 6, 545–554. [Google Scholar] [CrossRef]
- Liu, W.; Zheng, Y.; Zhang, Z.; Yao, W.; Gao, X. Hypoglycemic, hypolipidemic and antioxidant effects of Sarcandra glabra polysaccharide in type 2 diabetic mice. Food Funct. 2014, 5, 2850–2860. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Liu, W.; Yu, J.; Zou, S.; Wang, J.; Yao, W.; Gao, X. Characterization and hypoglycemic effect of a polysaccharide extracted from the fruit of Lycium barbarum L. Carbohydr. Polym. 2013, 98, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Jie, Z.; Fei, G.; Zhou, J.; Xian, Z.; Zhou, C.; Wan, L.; Chen, J. Structure features and in vitro hypoglycemic activities of polysaccharides from different species of Maidong. Carbohydr. Polym. 2017, 173, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric Method for Determination of Sugars and Related Substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Chem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Blumenkrantz, N.; Asboe-Hansen, G. New method for quantitative determination of uronic acids. Anal. Biochem. 1973, 54, 484–489. [Google Scholar] [CrossRef]
- Wei, Y.A.; Fang, J.N. Determination of purity and molecular weight of polysaccharide by high performance gel permeation chromatography. Acta Pharm. Sin. 1989, 24, 532–536. [Google Scholar]
- Zhang, S.; Li, X.Z. Inhibition of α-glucosidase by polysaccharides from the fruit hull of Camellia oleifera Abel. Carbohydr. Polym. 2015, 115, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.; Qin, T.; Qiu, F.; Song, Y.; Lin, D.; Ma, Y.; Li, J.; Huang, Y. Immunomodulatory effects of hydroxyethylated Hericium erinaceus polysaccharide on macrophages RAW264.7. Int. J. Biol. Macromol. 2017, 105, 879–885. [Google Scholar] [CrossRef] [PubMed]
- Kojima, T.; Tabata, K.; Itoh, W.; Yanaki, T. Molecular weight dependence of the antitumor activity of schizophyllan. Agric. Biol. Chem. 1986, 50, 231–232. [Google Scholar]
- Kim, J.-S.; Hyun, T.K.; Kim, M.-J. The inhibitory effects of ethanol extracts from sorghum, foxtail millet and proso millet on α-glucosidase and α-amylase activities. Food Chem. 2011, 124, 1647–1651. [Google Scholar] [CrossRef]
- Gu, J.F.; Zheng, Z.Y.; Yuan, J.R.; Zhao, B.J.; Wang, C.F.; Zhang, L.; Xu, Q.Y.; Yin, G.W.; Feng, L.; Jia, X.B. Comparison on hypoglycemic and antioxidant activities of the fresh and dried Portulaca oleracea L. in insulin-resistant HepG2 cells and streptozotocin-induced C57BL/6J diabetic mice. J. Ethnopharmacol. 2015, 161, 214–223. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds HIOP1-S and HIOP2-S are available from the authors. |



| Sample | HIOP1-S (%) | HIOP2-S (%) |
|---|---|---|
| Carbohydrate | 82.73 ± 3.01 a | 84.62 ± 1.09 a |
| Protein | 1.79 ± 0.74 a | 1.93 ± 0.32 a |
| Uronic acid | 9.98 ± 1.34 a | 11.45 ± 0.98 a |
| Mannose | 1.75 | 9.714 |
| Rhamnose | 12.233 | 15.331 |
| Glucose | 29.673 | 49.881 |
| Galactose | 20.547 | 15.321 |
| Xylose | 2.386 | 4.675 |
| Arabinose | 15.786 | 5.078 |
| Fucose | 17.626 | - |
| Polysaccharides | IC50 (µg/mL) |
|---|---|
| HIOP1-S | 7.875 |
| HIOP2-S | 3.841 |
| acarbose | 2306.018 |
| Groups | Numbers | Dose (mg/kg bw) | Blood Glucose (mmol L−1) | |||
|---|---|---|---|---|---|---|
| 0 day | 7 days | 14 days | 21 days | |||
| Blank control | 12 | 0 | 7.74 ± 5.30 | 8.72 ± 1.24 | 9.96 ± 1.8 | 9.70 ± 1.70 |
| Model group | 12 | 0 | 19.02 ± 7.05 * | 22.81 ± 2.02 | 34.25 ± 1.94 | 19.50 ± 1.86 |
| Met | 12 | 125 | 19.95 ± 6.23 * | 18.24 ± 3.95 ** | 25.92 ± 5.98 ** | 16.62 ± 3.2 ** |
| HIOP1-S | 12 | 4.5 | 18.24 ± 7.49 * | 21.79 ± 5.02 | 28.04 ± 7.32 | 18.35 ± 3.02 |
| HIOP2-S | 12 | 4.5 | 18.53 ± 7.21 * | 18.59 ± 7.69 | 28.25 ± 10.78 | 14.9 ± 5.26 ** |
| Groups | Numbers | Dose (mg/kg) | bw/g | |||
|---|---|---|---|---|---|---|
| 0 day | 7 days | 14 days | 21 days | |||
| Blank control | 12 | 0 | 24.89 ± 1.35 | 30.05 ± 2.87 | 32.23 ± 2.83 | 34.84 ± 2.87 |
| Model group | 12 | 0 | 23.41 ± 1.78 | 25.15 ± 1.73 * | 27.51 ± 1.92 * | 29.22 ± 2.55 * |
| Met | 12 | 125 | 24.41 ± 1.63 | 26.75 ± 2.01 * | 28.39 ± 2.15 * | 30.25 ± 2.74 * |
| HIOP1-S | 12 | 4.5 | 24.50 ± 2.39 | 27.58 ± 2.34 * | 28.29 ± 2.38 * | 28.41 ± 2.61 * |
| HIOP2-S | 12 | 4.5 | 23.95 ± 1.69 | 25.59 ± 3.29 * | 26.97 ± 2.75 * | 28.31 ± 2.92 * |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, P.; Xue, J.; Tong, S.; Dong, W.; Wu, P. Structure Characterization and Hypoglycaemic Activities of Two Polysaccharides from Inonotus obliquus. Molecules 2018, 23, 1948. https://doi.org/10.3390/molecules23081948
Liu P, Xue J, Tong S, Dong W, Wu P. Structure Characterization and Hypoglycaemic Activities of Two Polysaccharides from Inonotus obliquus. Molecules. 2018; 23(8):1948. https://doi.org/10.3390/molecules23081948
Chicago/Turabian StyleLiu, Ping, Jiao Xue, Shisheng Tong, Wenxia Dong, and Peipei Wu. 2018. "Structure Characterization and Hypoglycaemic Activities of Two Polysaccharides from Inonotus obliquus" Molecules 23, no. 8: 1948. https://doi.org/10.3390/molecules23081948






