Isolation and Purification of Potent Growth Inhibitors from Piper methysticum Root
Abstract
:1. Introduction
2. Results
2.1. Inhibitory Effect of Different Extracts of P. methysticum Root
2.2. Isolation and Purification of Plant Growth Inhibitors by Column Chromatography
2.3. Quantitative Analysis of Bioactive Compounds from P. methysticum Root
2.4. Inhibitory Activity of the Isolated Compounds on the Growth of R. sativus
3. Discussion
4. Materials and Methods
4.1. Plant Materials
4.1.1. Piper methysticum Root
4.1.2. Tested Plants
4.2. Preparation of Plant Extract
4.3. Inhibitory Activity of P. methysticum Root Crude Extracts
4.4. Isolation and Purification of Bioactive Compounds
4.4.1. General Experimental Procedures
4.4.2. Isolation, Purification and Identification by CC, GC-MS, EIS-MS, 1H- and 13C-NMR
4.4.3. Chemical Identification and Quantification
GC-MS Analysis
Quantification of Growth Inhibitors of P. methysticum Root
ESI/MS Analysis
NMR Analysis
4.5. Inhibitory Activity of Isolated Compounds in Bioassays
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rice, E.L. Allelopathy, 2nd ed.; Academic Press Inc.: Orlando, FL, USA, 1984; p. 422. ISBN 0125870558. [Google Scholar]
- Khanh, T.D.; Chung, I.M.; Xuan, T.D.; Tawata, S. The exploitation of crop allelopathy in sustainable agricultural production. J. Agron. Crop Sci. 2005, 191, 172–184. [Google Scholar] [CrossRef]
- Singh, Y.N. Kava: An overview. J. Ethnopharmacol. 1992, 37, 13–45. [Google Scholar] [CrossRef]
- Blumenthal, M. Kava safety questioned due to case reports of liver toxicity. Herbal Gram 2002, 55, 26–32. [Google Scholar]
- Meyer, H.J. Pharmacology of kava. In Ethnopharmacologic Search for Psychoactive Drugs, 2nd ed.; Efron, D.H., Holmstedt, B., Kline, N.S., Eds.; Raven Press: New York, NY, USA, 1979; ISBN 9780300052138. [Google Scholar]
- Lebot, V.; Merlin, M.; Lindstrom, L. Kava: The Pacific Drug; Yale University Press: New Haven, CT, USA, 1992; ISBN-10 0300052138, ISBN-13 978-0300052138. [Google Scholar]
- Hong, N.H.; Xuan, T.D.; Tsuzuki, E.; Matsuo, M.; Ogushi, Y. Evaluation of the allelopathic potential of kava (Piper methysticum L.) for weed control in rice (Oryza sativa). Weed Biol. Manag. 2002, 2, 143–147. [Google Scholar] [CrossRef]
- Xuan, T.D.; Eiji, T.; Hiroyuki, T.; Mitsuhiro, M.; Khanh, T.D. Identification of potential allelochemicals from kava (Piper methysticum L.) root. Allelopath. J. 2003, 12, 197–203. [Google Scholar]
- Xuan, T.D.; Elzaawely, A.A.; Fukuta, M.; Tawata, S. Herbicidal and fungicidal activities of lactones in Kava (Piper methysticum). J. Agric. Food Chem. 2006, 54, 720–725. [Google Scholar] [CrossRef] [PubMed]
- Xuan, T.D.; Tawata, S.; Khanh, T.D.; Chung, I.M. Decomposition of allelopathic plants in soil. J. Agron. Crop Sci. 2005, 191, 162–171. [Google Scholar] [CrossRef]
- Xuan, T.D.; Fukuta, M.; Wei, A.C.; Elzaawely, A.A.; Khanh, T.D.; Tawata, S. Efficacy of extracting solvents to chemical components of kava (Piper methysticum) roots. J. Nat. Med. 2008, 62, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Junior, R.W.A.; Gomes, D.B.; Zanchet, B.; Schönell, A.P.; Diel, K.A.; Banzato, T.P.; Ruizc, A.L.T.G.; Carvalhoc, J.E.; Neppel, A. Antiproliferative effects of pinostrobin and 5,6-dehydrokavain isolated from leaves of Alpinia zerumbet. Rev. Bras. Farmacogn. 2017, 27, 592–598. [Google Scholar] [CrossRef]
- Whitton, P.A.; Lau, A.; Salisbury, A.; Whitehouse, J.; Evans, C.S. Kava lactones and the kava-kava controversy. Phytochemistry 2003, 64, 673–679. [Google Scholar] [CrossRef]
- Dragul, K.; Yoshida, W.Y.; Tang, C. Piperidine alkaloids from Piper methysticum. Phytochemistry 2003, 63, 193–198. [Google Scholar] [CrossRef]
- Wu, D.; Yu, L.; Nair, M.G.; DeWitt, D.L.; Ramsewak, R.S. Cyclooxygenase enzyme inhibitory compounds with antioxidant activities from Piper methysticum (kava kava) roots. Phytomedicine 2002, 9, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Bilia, A.R.; Bergonzi, M.C.; Lazari, D.; Vinceiri, F.F. Characterization of commercial kava-kava herbal drug and herbal drug preparations by means of nuclear magnetic resonance spectroscopy. J. Agric. Food Chem. 2002, 50, 5016–5025. [Google Scholar] [CrossRef] [PubMed]
- Meissner, O.; Haberlein, H. HPLC analysis of flavokavins and kavapyrones from Piper methysticum Forst. J. Chromatogr. B 2005, 826, 46–49. [Google Scholar] [CrossRef] [PubMed]
- Asaduzzaman, M.D.; Islam, M.M.; Sultana, S. Allelopathy and allelochemicals in rice weed management. Bangladesh Res. Publ. J. 2010, 4, 1–14. [Google Scholar]
- Seigler, D.S. Chemistry and mechanisms of allelopathic interactions. Agron. J. 1996, 88, 876–885. [Google Scholar] [CrossRef]
- Soltys, D.; Krasuska, U.; Bogatek, R.; Gniazdowska, A. Allelochemicals as bioherbicides—Present and perspectives. In Herbicides—Current Research and Case Studies in Use; Price, A.J., Kelton, J.A., Eds.; Intech: Rijeka, Croatia, 2013; ISBN 978-953-51-1112-2. [Google Scholar]
- Andriana, Y.; Xuan, T.D.; Quan, N.V.; Quy, T.N. Allelopathic potential of Tridax procumbens L. on radish and identification of allelochemicals. Allelopathy J. 2018, 43, 223–237. [Google Scholar] [CrossRef]
- Altemimi, A.; Lakhssassi, N.; Baharlouei, A.; Watson, D.G.; Lightfoot, D.A. Phytochemicals: Extraction, Isolation, and Identification of Bioactive Compounds from Plant Extracts. Plants 2017, 6, 42. [Google Scholar] [CrossRef] [PubMed]
- Singh, Y.N.; Blumenthal, M. Kava: An overview. Herbal Gram 1997, 39, 34–55. [Google Scholar] [CrossRef]
- Rowe, A.; Zhang, L.Y.; Ramzan, I. Toxicokinetics of kava. Adv. Pharmacol. Sci. 2010, 2011, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Teschke, R.; Lebot, V. Proposal for a kava quality standardization code. Food Chem. Toxicol. 2011, 49, 2503–2516. [Google Scholar] [CrossRef] [PubMed]
- Bhat, Z.S.; Rather, M.A.; Maqbool, M.; Lah, H.U.; Yousuf, S.K.; Ahmad, Z. α-pyrones: Small molecules with versatile structural diversity reflected in multiple pharmacological activities-an update. Biomed. Pharmacother. 2017, 91, 265–277. [Google Scholar] [CrossRef] [PubMed]
- Siméoni, P.; Lebot, V. Identification of factors determining kavalactone content and chemotype in kava (Piper methysticum Forst. f.). Biochem. Syst. Ecol. 2002, 30, 413–424. [Google Scholar] [CrossRef]
- Li, B.B.; Smith, B.; Hossain, M.d.M. Extraction of phenoloics from citrus peels I. Solvent extraction method. Sep. Purif. Technol. 2006, 48, 182–188. [Google Scholar] [CrossRef]
- Vera, N.; Solorzano, E.; Ordoñez, R.; Maldonado, L.; Bedascarrasbure, E.; Isla, M.I. Chemical composition of Argentinean propolis collected in extreme regions and its relation with antimicrobial and antioxidant activities. Nat. Prod. Commun. 2011, 6, 823–827. [Google Scholar] [PubMed]
- Wan, W.M.N.H.S.; Ahmad, F.; Yen, K.H. Essential oils, phytochemicals and bioactivities of Piper caninum Blume. In Natural Heterocycles: Extraction and Biological Activity; Ameta, K.L., Ed.; Nova Science Publishers Inc.: London, UK, 2015; ISBN 978-163-463-462-5. [Google Scholar]
- Mangoyi, R.; Midiwo, J.; Mukanganyama, S. Isolation and characterization of an antifungal compound 5-hydroxy-7,4’-dimethoxyflavone from Combretum zeyheri. BMC Complement. Altern. Med. 2015, 15. [Google Scholar] [CrossRef] [PubMed]
- Mierziak, J.; Kostyn, K.; Kulma, A. Flavonoids as important molecules of plant interactions with the environment. Molecules 2014, 19, 16240–16265. [Google Scholar] [CrossRef] [PubMed]
- Rouse, J. Kava: A South Pacific herb for anxiety, tension and insomnia. Clin. Nutr. Insights 1998, 96, 3900–3905. [Google Scholar]
- Teschke, R.; Sarris, J.; Schweitzer, I. Kava hepatotoxicity in traditional and modern use: The presumed Pacific kava paradox hypothesis revisited. Br. J. Clin. Pharmacol. 2012, 73, 170–174. [Google Scholar] [CrossRef] [PubMed]
- Rowe, A.; Ramzan, I. Are mould hepatotoxins responsible for kavahepatotoxicity? Phytother. Res. 2012, 26, 1768–1770. [Google Scholar] [CrossRef] [PubMed]
- Grace, R.F. Kava drinking in Vanuatu—A hospital based survey. Pac. Health Dialog. 2003, 10, 41–44. [Google Scholar] [PubMed]
- Zhou, P.; Gross, S.; Liu, J.H.; Yu, B.Y.; Feng, L.L.; Nolta, J.; Piwnica-Worms, D.; Qiu, S.X. Flavokawain B, the hepatotoxic constituent from kava root, induces GSH-sensitive oxidative stress through modulation of IKK/NF-κB and MAPK signaling pathways. FASEB J. 2010, 24, 4722–4732. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, Y.N.; Devkota, A.K. Aqueous kava extracts do not affect liver function tests in rats. Planta Med. 2003, 69, 496–499. [Google Scholar] [PubMed]
- DiSilvestro, R.A.; Zhang, W.; DiSilvestro, D.J. Kava feeding in rats does not cause liver injury nor enhance galactosamine-induced hepatitis. Food Chem. Toxicol. 2007, 45, 1293–1300. [Google Scholar] [CrossRef] [PubMed]
- Xuan, T.D.; Teschke, R. Dihydro-5,6-dehydrokavain (DDK) from Alpinia zerumbet: Its isolation, synthesis, and characterization. Molecules 2015, 20, 16306–16319. [Google Scholar] [CrossRef] [PubMed]
- Elzaawely, A.A.; Xuan, T.D.; Tawata, S. Changes in essential oils, kava pyrones and total phenolics of Alpinia zerumbet (Pers.) B.L. Burtt. & R.M. Sm. leaves exposed to copper sulphate. Environ. Exp. Bot. 2007, 59, 347–353. [Google Scholar]
- Elzaawely, A.A.; Xuan, T.D.; Tawata, S. Essential oils, kava pyrones and phenolic compounds from leaves and rhizomes of Alpinia zerumbet (Pers.) B.L. Burtt. & R.M. Sm. and their antioxidant activity. Food Chem. 2007, 103, 486–494. [Google Scholar]
- Tawata, S.; Fukuta, M.; Xuan, T.D.; Deba, F. Total utilization of tropical plants Leucaena leucocephala and Alpinia zerumbet. J. Pest. Sci. 2008, 33, 40–43. [Google Scholar] [CrossRef]
- Teschke, R.; Xuan, T.D. Viewpoint: A contributory role of shell ginger (Alpinia zerumbet) for human longevity in Okinawa, Japan? Nutrients 2018, 10, 166. [Google Scholar] [CrossRef] [PubMed]
- Suyatno, S.; Syarief, S.H.; Hidajati, N.; Rinningsih, R.; Tori, M.; Shimizu, K. Phenolic compounds isolated from the fern Chingia sakayensis (Zeiller). J. ILMU DASAR 2009, 10, 22–29. [Google Scholar]
- Wongsa, N.; Kanokmedhakul, K.; Boonmak, J.; Youngme, S.; Kanokmedhakul, S. Bicyclic lactones and racemic mixtures of dimeric styrylpyrones from the leaves of Miliusa velutina. RSC Adv. 2017, 7, 25285–25297. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |
| Extract | Concentration (µg/mL) | Shoot Inhibition (%) | Root Inhibition (%) |
|---|---|---|---|
| Hexane | 50 | 22.62 ± 3.15 d | 28.00 ± 3.73 i |
| 250 | 55.33 ± 3.06 c | 70.54 ± 3.82 d | |
| 500 | 68.67 ± 3.06 b | 89.52 ± 3.36 bc | |
| Chloroform | 50 | 25.40 ± 2.75 d | 36.67 ± 3.51 g |
| 250 | 74.00 ± 2.61 ab | 88.48 ± 3.94 bc | |
| 500 | 81.00 ± 3.61 a | 98.57 ± 3.43 a | |
| EtOAc | 50 | 24.79 ± 3.39 d | 39.33 ± 3.76 fg |
| 250 | 78.33 ± 2.08 ab | 94.87 ± 3.75 ab | |
| 500 | 83.67 ± 4.51 a | 99.05 ± 3.83 a | |
| Acetone | 50 | 24.13 ± 4.29 d | 36.11 ± 3.47 gh |
| 250 | 68.67 ± 3.12 b | 85.62 ± 2.72 c | |
| 500 | 78.00 ± 2.00 ab | 95.86 ± 3.21 ab | |
| Water | 50 | 23.29 ± 4.02 d | 29.00 ± 3.00 hi |
| 250 | 30.67 ± 2.31 d | 44.83 ± 3.16 ef | |
| 500 | 51.33 ± 7.02 c | 50.48 ± 3.18 e |
| Extracts | Inhibitory Levels, IC50 (µg/mL) | |
|---|---|---|
| Shoot | Root | |
| Hexane | 278.60 ± 19.30 b | 173.70 ± 15.44 b |
| Chloroform | 181.09 ± 8.64 c | 81.40 ± 26.80 c |
| Ethyl acetate | 172.00 ± 25.20 c | 51.31 ± 0.23 c |
| Acetone | 206.36 ± 1.88 bc | 90.50 ± 21.00 c |
| Water | 483.80 ± 71.70 a | 420.30 ± 53.20 a |
| Fraction | Solvent Combination and Fraction | Weight (mg) |
|---|---|---|
| C1 | Crystal in H-E 9:1 (fractions 14–17) | 7.7 |
| C2 | Crystal in H-E 9:1 (fractions 20–26) | 54.7 |
| C3 | Crystal in H-E 8:2 (fractions 11–15) | 174.4 |
| C4 | Crystal in H-E 8:2 (fractions 32–47) | 518.0 |
| C5 | Crystal in H-E 8:2 (fractions 48–55) | 70.3 |
| C6 | Crystal in H-E 8:2 (fractions 69–74) | 49.0 |
| Fraction | Retention Time | Peak Area (%) | Compounds | Chemical Formula | Molecular Weight | Chemical Class |
|---|---|---|---|---|---|---|
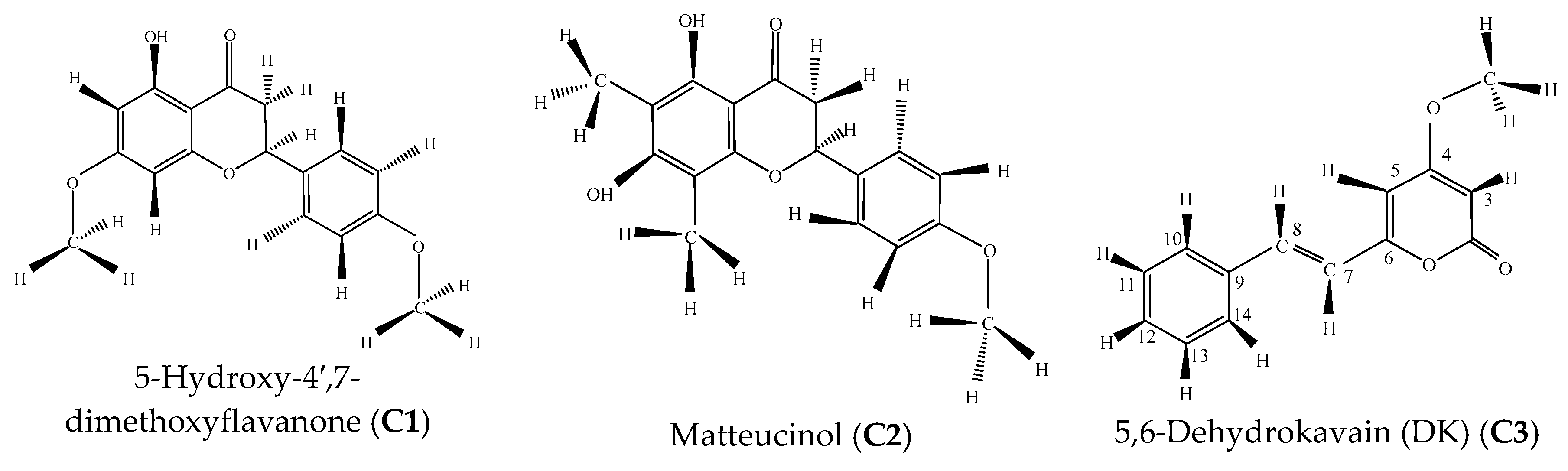
| C1 | 23.40 | 93.28 | 5-Hydroxy-4′,7-dimethoxyflavanone | C17H16O5 | 300 | Flavanone |
| C2 | 25.37 | 90.00 | Matteucinol | C18H18O5 | 314 | Flavanone |
| C3 | 20.80 | 94.66 | 5,6-Dehydrokavain (DK) | C14H12O3 | 228 | Kavalactone |
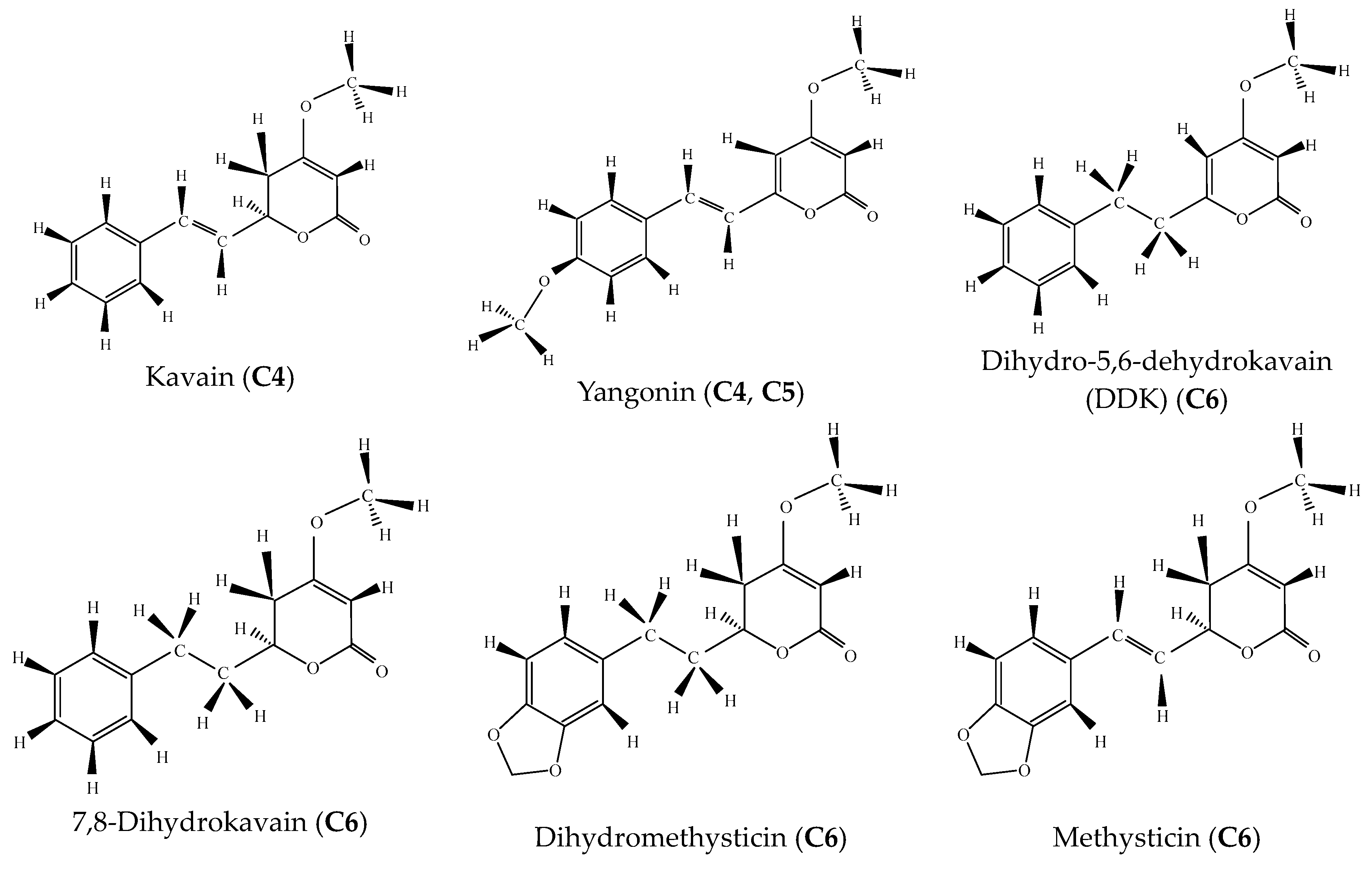
| C4 | 20.07 | 59.42 | Kavain | C14H14O3 | 230 | Kavalactone |
| 23.37 | 40.58 | Yangonin | C15H14O4 | 258 | Kavalactone | |
| C5 | 23.34 | 93.64 | Yangonin | C15H14O4 | 258 | Kavalactone |
| C6 | 16.25 | 12.78 | Dihydro-5,6-dehydrokavain (DDK) | C14H14O3 | 230 | Kavalactone |
| 18.35 | 20.44 | 7,8-Dihydrokavain | C14H16O3 | 232 | Kavalactone | |
| 22.45 | 54.77 | Dihydromethysticin | C15H16O5 | 276 | Kavalactone | |
| 23.40 | 8.90 | Methysticin | C15H14O5 | 274 | Kavalactone |
| Fractions | Retention Time | Compounds | mg/g DW |
|---|---|---|---|
| C1 | 23.38 ± 0.01 | 5-Hydroxy-4′,7-dimethoxyflavanone | 0.76 |
| C2 | 25.36 ± 0.02 | Matteucinol | 2.50 |
| C3 | 20.65 ± 0.01 | 5,6-Dehydrokavain | 2.75 |
| C5 | 23.34 ± 0.01 | Yangonin | 2.09 |
| Fractions | Compounds | IC50 (µg/mL) | |
|---|---|---|---|
| Shoot | Root | ||
| C1 | 5-Hydroxy-4′,7-dimethoxyflavanone | 120.22 ± 14.64 e | - |
| C2 | Matteucinol | 248.03 ± 5.43 d | - |
| C3 | 5,6-Dehydrokavain (DK) | 265.88 ± 19.78 c,d | 375.33 ± 11.93 a |
| C4 | Kavain; yangonin | 457.18 ± 28.64 a | 7.70 ± 1.57 b |
| C5 | Yangonin | 313.82 ± 0.68 b,c | 365.00 ± 44.00 a |
| C6 | Dihydro-5,6-dehydrokavain (DDK); 7,8-dihydrokavain; dihydromethysticin; methysticin | 360.19 ± 20.37 b | 15.67 ± 5.13 b |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Van, T.M.; Xuan, T.D.; Minh, T.N.; Quan, N.V. Isolation and Purification of Potent Growth Inhibitors from Piper methysticum Root. Molecules 2018, 23, 1907. https://doi.org/10.3390/molecules23081907
Van TM, Xuan TD, Minh TN, Quan NV. Isolation and Purification of Potent Growth Inhibitors from Piper methysticum Root. Molecules. 2018; 23(8):1907. https://doi.org/10.3390/molecules23081907
Chicago/Turabian StyleVan, Truong Mai, Tran Dang Xuan, Truong Ngoc Minh, and Nguyen Van Quan. 2018. "Isolation and Purification of Potent Growth Inhibitors from Piper methysticum Root" Molecules 23, no. 8: 1907. https://doi.org/10.3390/molecules23081907








