Phenolic Composition and Bioactivity of Lavandula pedunculata (Mill.) Cav. Samples from Different Geographical Origin
Abstract
1. Introduction
2. Results
2.1. Phenolic Compounds
2.2. Antioxidant Potential
2.3. Anti-Inflammatory Activities
2.4. Cytotoxicity
2.5. Antimicrobial Activity
3. Materials and Methods
3.1. Samples and Samples Preparation
3.2. Standards and Reagents
3.3. Analysis of Phenolic Compounds
3.4. Evaluation of the Antioxidant Activity
3.5. Evaluation of the Cytotoxicity
3.5.1. General
3.5.2. In Tumor Cell Lines
3.5.3. In Non-Tumor Cells
3.6. Evaluation of the Anti-Inflammatory Activity
3.7. Evaluation of the Antimicrobial Activity
3.7.1. Antibacterial Activity
3.7.2. Antifungal Activity
3.8. Statistical Analysis
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Komes, D.; Belščak-cvitanović, A.; Horžić, D. Phenolic composition and antioxidant properties of some traditionally used medicinal plants affected by the extraction time and hydrolysis. Phytochem. Anal. 2011, 22, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Kaefer, C.M.; Milner, J.A. The role of herbs and spices in cancer prevention. J. Nutr. Biochem. 2008, 19, 347–361. [Google Scholar] [CrossRef] [PubMed]
- Costa, P.; Gonçalves, S.; Valentão, P.; Andrade, P.B.; Almeida, C.; Nogueira, J.M.F.; Romano, A. Metabolic profile and biological activities of Lavandula pedunculata subsp. lusitanica (Chaytor) Franco: Studies on the essential oil and polar extracts. Food Chem. 2013, 141, 2501–2506. [Google Scholar] [CrossRef] [PubMed]
- Port, S.; Chisté, R.C.; Godoy, H.T.; Prado, M.A. The phenolic compounds and the antioxidant potential of infusion of herbs from the Brazilian Amazonian region. Food Res. Int. 2013, 53, 875–881. [Google Scholar]
- Bajpai, V.K.; Agrawal, P.; Bang, B.H.; Park, Y.-H. Phytochemical analysis, antioxidant and antilipid peroxidation effects of a medicinal plant, Adhatoda vasica. Front. Life Sci. 2015, 8, 305–312. [Google Scholar] [CrossRef]
- Quer, P.F. Plantas Medicinales—El Dioscorides Renovado; Ediciones Península: Barcelona, Spain, 1999. [Google Scholar]
- Zuzarte, M.R.; Dinis, A.M.; Cavaleiro, C.; Salgueiro, L.R.; Canhoto, J.M. Trichomes, essential oils and in vitro propagation of Lavandula pedunculata (Lamiaceae). Ind. Crops Prod. 2010, 32, 580–587. [Google Scholar] [CrossRef]
- Lopes, V.; Barata, A.M. Ex situ morphological assessment of wild Lavandula populations in Portugal. Arab. J. Med. Aromat. Plants 2017, 3, 87–100. [Google Scholar]
- Morales, R. Lavandula L. In Flora Iberica; Castroviejo, S., Ed.; Real Jardín Botánico, CSIC: Madrid, Spain, 2010; Volume 12, pp. 484–496. [Google Scholar]
- Neves, J.M.; Matosa, C.; Moutinho, C.; Queiroz, G.; Gomes, L.R. Ethnopharmacological notes about ancient uses of medicinal plants in Trás-os-Montes (northern of Portugal). J. Ethnopharmacol. 2009, 124, 270–283. [Google Scholar] [CrossRef] [PubMed]
- Santos, E.S.; Abreu, M.M.; Saraiva, J.A. Mutielemental concentration and physiological responses of Lavandula pedunculata growing in soils developed on different mine. Environ. Pollut. 2016, 213, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Torras-Claveria, L.; Jauregui, O.; Bastida, J.; Codina, C.; Viladomat, F. Antioxidant Activity and Phenolic Composition of Lavandin (Lavandula x intermedia Emeric ex Loiseleur) Waste. J. Agric. Food Chem. 2007, 55, 8436–8443. [Google Scholar] [CrossRef] [PubMed]
- Costa, P.; Gonçalves, S.; Valentão, P.; Andrade, P.B.; Romano, A. Accumulation of phenolic compounds in in vitro cultures and wild plants of Lavandula viridis L’Hér and their antioxidant and anti-cholinesterase potential. Food Chem. Toxicol. 2013, 57, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Nunes, R.; Pasko, P.; Tyszka-Czochara, M.; Szewczyk, A.; Szlosarczyk, M.; Carvalho, I.S. Antibacterial, antioxidant and anti-proliferative properties and zinc content of five south Portugal herbs. Pharm. Biol. 2016, 55, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Pereira, F.; Baptista, R.; Ladeiras, D.; Madureira, A.M.; Teixeira, G.; Rosado, C.; Rijo, P. Production and characterization of nanoparticles containing methanol extracts of Portuguese Lavenders. Measurement 2015, 74, 170–177. [Google Scholar] [CrossRef]
- Barros, L.; Dueñas, M.; Dias, M.I.; Sousa, M.J.; Santos-Buelga, C.; Ferreira, I.C.F.R. Phenolic profiles of cultivated, in vitro cultured and commercial samples of Melissa officinalis L. infusions. Food Chem. 2013, 136, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Zhang, Q.; Wang, X.; Yang, J.; Wang, Q. Qualitative analysis and simultaneous quantification of phenolic compounds in the aerial Parts of Salvia miltiorrhiza by HPLCDAD and ESI/MSn. Phytochem. Anal. 2011, 22, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Zeng, G.; Xiao, H.; Liu, J.; Liang, X. Identification of phenolic constituents in Radix Salvia miltiorrhizae by liquid chromatography/electrospray ionization mass spectrometry. Rapid Commun. Mass Spectrom. 2006, 20, 499–506. [Google Scholar] [CrossRef] [PubMed]
- Ruan, M.; Li, Y.; Li, X.; Luo, J.; Kong, L. Qualitative and quantitative analysis of the major constituents in Chinese medicinal preparation Guan-Xin-Ning injection by HPLC-DAD-ESI-MSn. J. Pharm. Biomed. Anal. 2012, 59, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.; Barros, L.; Carvalho, A.M.; Santos-Buelga, C.; Ferreira, I.C.F.R. Infusions of artichoke and milk thistle represent a good source of phenolic acids and flavonoids. Food Funct. 2015, 6, 56. [Google Scholar] [CrossRef] [PubMed]
- Lech, K.; Witkoś, K.; Jaros, M. HPLC–UV–ESI MS/MS identification of the color constituents of sawwort (Serratula tinctoria L.). Anal. Bioanal. Chem. 2014, 406, 3703–3708. [Google Scholar] [CrossRef] [PubMed]
- Correa, R.C.G.; Souza, A.H.P.; Calhelha, R.C.; Barros, L.; Glamoclija, J.; Sokovic, M.; Peralta, R.M.; Bracht, M.; Ferreira, I.C.F.R. Bioactive formulations prepared from fruiting bodies and submerged culture mycelia of the Brazilian edible mushroom Pleurotus ostreatoroseus Singer. Food Funct. 2015, 6, 2155–2164. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, A.; Proença, C.; Serralheiro, M.L.M.; Araújo, M.E.M. The in vitro screening for acetylcholinesterase inhibition and antioxidant activity of medicinal plants from Portugal. J. Ethnopharmacol. 2006, 108, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Baptista, R.; Madureira, A.M.; Jorge, R.; Adão, R.; Duarte, A.; Duarte, N.; Lopes, M.M.; Teixeira, G. Antioxidant and Antimycotic Activities of Two Native Lavandula Species from Portugal. Evid.-Based Complement. Altern. Med. 2015, 2015, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Algieri, F.; Rodriguez-Nogales, A.; Vezza, T.; Garrido-Mesa, J.; Garrido-Mesa, N.; Utrilla, M.P.; González-Tejero, M.R.; Casares-Porcel, M.; Molero-Mesa, J.; Contreras, M.M.; et al. Anti-inflammatory activity of hydroalcoholic extracts of Lavandula dentata L. and Lavandula stoechas L. J. Ethnopharmacol. 2016, 190, 142–158. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Shia, J.; Liu, C.; Zhang, J.; Xang, N.; Liu, X.; Chen, Y.; You, J.; Shen, O.; Miao, M.; et al. Three new arylbenzofurans from Lavandula angustifolia and their bioactivities. Phytochem. Lett. 2017, 19, 60–63. [Google Scholar] [CrossRef]
- Carocho, M.; Calhelha, R.C.; Queirozc, M.J.R.Q.P.; Bento, A.; Morales, P.; Sokovic, M.; Ferreira, I.C.F.R. Infusions and decoctions of Castanea sativa flowers a seff ective antitumor and antimicrobial matrices. Ind. Crops Prod. 2014, 62, 42–46. [Google Scholar] [CrossRef]
- Rashed, K.; Ciric, A.; Glamoclijab, J.; Calhelha, R.C.; Ferreira, I.C.F.R.; Sokovic, M. Antimicrobial and cytotoxic activities of Alnus rugosa L. aerial parts and identification of the bioactive components. Ind. Crops Prod. 2014, 59, 189–196. [Google Scholar]
- Živkovic, J.; Barreira, J.C.M.; Stojkovic, D.; Cebovic, T.; Santos-Buelga, C.; Maksimovic, Z.; Ferreira, I.C.F.R. Phenolic profile, antibacterial, antimutagenic and antitumour evaluation of Veronica urticifolia Jacq. J. Funct. Foods 2014, 9, 192–201. [Google Scholar] [CrossRef]
- Nikolic, M.; Glamoclija, J.; Ferreira, I.C.F.R.; Calhelha, R.C.; Fernandes, A.; Markovic, T.; Markovic, D.; Giwelie, A.; Sokovic, M. Chemical composition, antimicrobial, antioxidant and antitumor activity of Thymus serpyllum L., Thymus algeriensis Boiss. and Reut and Thymus vulgaris L. essential oils. Ind. Crops Prod. 2014, 52, 183–190. [Google Scholar] [CrossRef]
- Pereira, E.; Barros, L.; Calhelha, R.C.; Dueñas, M.; Carvalho, A.M.; Santos-Buelga, C.; Ferreira, I.C.F.R. Bioactivity and phytochemical characterization of Arenaria montana L. Food Funct. 2014, 5, 1848. [Google Scholar] [CrossRef] [PubMed]
- Bessada, S.M.F.; Barreira, J.C.M.; Barros, L.; Ferreira, I.C.F.R.; Oliveira, M.B.P.P. Phenolic profile and antioxidant activity of Coleostephus myconis (L.) Rchb.f.: An underexploited and highly disseminated species. Ind. Crops Prod. 2016, 89, 45–51. [Google Scholar] [CrossRef]
- Martins, N.; Barros, L.; Dueñas, M.; Santos-Buelga, C.; Ferreira, I.C.F.R. Characterization of phenolic compounds and antioxidant properties of Glycyrrhiza glabra L. rhizomes and roots. RSC Adv. 2015, 5, 26991–26997. [Google Scholar] [CrossRef]
- Guimarães, R.; Barros, L.; Dueñas, M.; Calhelha, R.C.; Carvalho, A.M.; Santos-Buelga, C.; Queiroz, M.J.R.P.; Ferreira, I.C.F.R. Infusion and decoction of wild German chamomile: Bioactivity and characterization of organic acids and phenolic compounds. Food Chem. 2013, 136, 947–954. [Google Scholar] [CrossRef] [PubMed]
- Taofiq, O.; Calhelha, R.C.; Heleno, S.; Barros, L.; Martins, A.A.; Santos-Buelga, C.; Queiroz, M.J.R.P.; Ferreira, I.C.F.R. The contribution of phenolic acids to the anti-inflammatory activity of mushrooms: Screening in phenolic extracts, individual parent molecules and synthesized glucuronated and methylated derivatives. Food Res. Int. 2015, 76, 821–827. [Google Scholar] [CrossRef] [PubMed]
- Carocho, M.; Barros, L.; Calhelha, R.C.; Ćirić, A.; Soković, M.; Santos-Buelga, C.; Ferreira, I.C.F.R. Melissa officinalis L. decoctions as functional beverages: A bioactive approach and chemical characterization. Food Funct. 2015, 6, 2240–2248. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
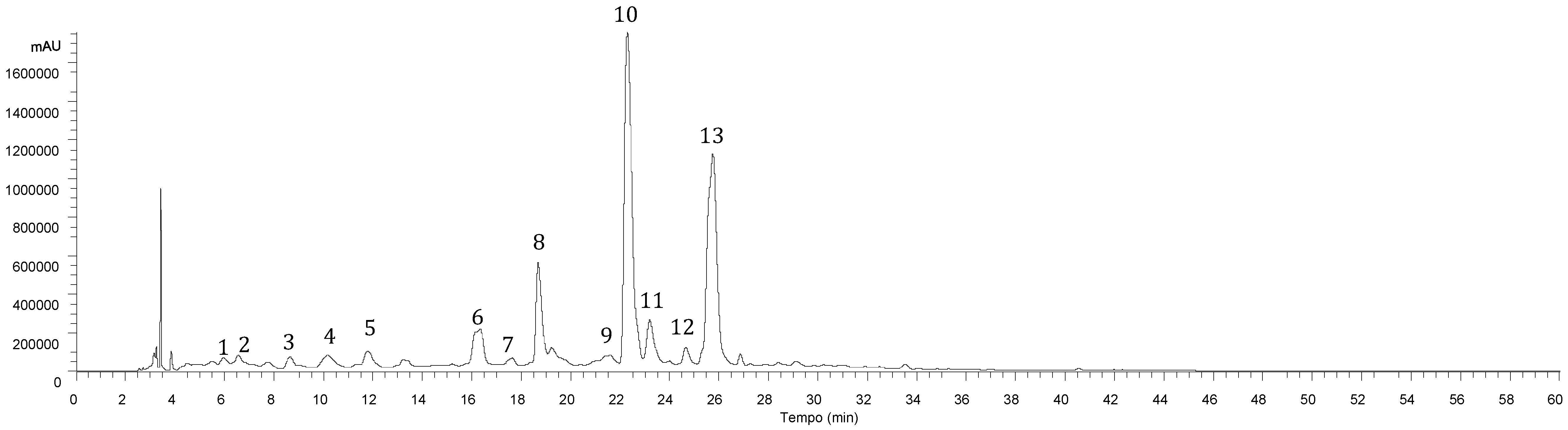
| Peak | Rt (min) | λmax (nm) | [M-H]− (m/z) | MS2 (m/z) | Tentative Identification | Identification Type |
|---|---|---|---|---|---|---|
| 1 | 5.97 | 320 | 341 | 179(100) | Caffeic acid hexoside | [12], DAD/MS |
| 2 | 6.57 | 320 | 341 | 179(100) | Caffeic acid hexoside | [12], DAD/MS |
| 3 | 8.65 | 310 | 325 | 163(100) | p-Coumaroyl hexoside | [12], DAD/MS |
| 4 | 10.16 | 320 | 179 | 119(100) | Caffeic acid | Standard/DAD/MS |
| 5 | 13.22 | 332 | 623 | 461(27), 285(100) | Luteolin-O-hexosyl-O-glucuronide | [20], DAD/MS |
| 6 | 16.34 | 284,330 sh | 463 | 287(100) | Eriodictyol-O-glucuronide | [21], DAD/MS |
| 7 | 17.64 | 320 | 521 | 359(100), 197(8), 179(14), 161(3), 135(5) | Rosmarinic acid hexoside | [22], DAD/MS |
| 8 | 18.68 | 347 | 461 | 285(100) | Luteolin-7-O-glucuronide | Standard/DAD/MS |
| 9 | 19.23 | 283,338 sh | 719 | 539(29), 521(18), 359(100), 179(32), 161(5), 135(5) | Sangerinic acid | [16], DAD/MS |
| 10 | 22.29 | 325 | 359 | 197(27), 179(37), 161(100), 135(5) | Rosmarinic acid | Standard/DAD/MS |
| 11 | 23.19 | 288,328 sh | 537 | 493(18), 359(100), 313(11), 295(5), 197(3), 179(3) | Lithospermic acid A | [17,18,19], DAD/MS |
| 12 | 24.65 | 340 | 475 | 299(100), 284(68) | Methylluteolin-O-glucuronide | [21], DAD/MS |
| 13 | 25.75 | 309,338sh | 717 | 537(47), 519(17), 493(40), 359(97), 339(10), 321(8), 313(17), 295(100), 197(7), 179(27) | Salvianolic acid B | [18,19], DAD/MS |
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hydroethanol Extract | |||||||||||||
| Peak 1 | 0.28 ± 0.01 f | 0.034 ± 0.003 k | 0.179 ± 0.001 i | tr | 0.210 ± 0.005 h | 0.23 ± 0.01 g | 0.11 ± 0.04 j | 2.44 ± 0.02 a | 0.37 ± 0.01 d | 0.44 ± 0.01 c | 0.277 ± 0.004 f | 0.59 ± 0.02 b | 0.33 ± 0.02 e |
| Peak 2 | 0.18 ± 0.02 h | 0.079 ± 0.003 j | 0.31 ± 0.001 d | 0.120 ± 0.003 i | 0.22 ± 0.02 g | 0.295 ± 0.001 e | 0.24 ± 0.01 f | 1.87 ± 0.02 a | 0.40 ± 0.01 c | 0.31 ± 0.02 de | 0.22 ± 0.01 fg | 0.54 ± 0.02 b | 0.115 ± 0.001 i |
| Peak 3 | 0.77 ± 0.01 f | 1.002 ± 0.001 d | 0.80 ± 0.002 f | 0.50 ± 0.01 h | 0.95 ± 0.02 e | 0.98 ± 0.01 de | 1.48 ± 0.03 a | 1.27 ± 0.07 b | 1.16 ± 0.03 c | 0.98 ± 0.03 de | 0.80 ± 0.01 f | 1.26 ± 0.01 b | 0.57 ± 0.03 g |
| Peak 4 | 0.63 ± 0.02 i | 0.805 ± 0.002 f | 0.88 ± 0.001 d | 0.639 ± 0.002 hi | 1.01 ± 0.02 c | 0.85 ± 0.02 e | 1.14 ± 0.02 b | 1.49 ± 0.03 a | 1.00 ± 0.04 c | 0.802 ± 0.001 f | 0.72 ± 0.03 g | 1.12 ± 0.04 b | 0.65 ± 0.02 h |
| Peak 5 | 2.05 ± 0.03 f | 2.33 ± 0.03 c | 2.68 ± 0.01 b | 1.37 ± 0.01 k | 1.846 ± 0.001 g | 1.84 ± 0.01 g | 1.599 ± 0.004 i | 1.54 ± 0.01 j | 2.22 ± 0.01 d | 2.695 ± 0.003 a | 2.09 ± 0.01 e | 2.06 ± 0.01 f | 1.698 ± 0.003 h |
| Peak 6 | 0.137 ± 0.003 h | tr | 0.40 ± 0.01 c | 0.74 ± 0.01 b | 0.082 ± 0.003 i | tr | 0.060 ± 0.001 j | 0.782 ± 0.003 a | tr | 0.299 ± 0.005 d | 0.150 ± 0.003 g | 0.231 ± 0.004 e | 0.17 ± 0.01 f |
| Peak 7 | 1.18 ± 0.01 g | 1.379 ± 0.001 de | 1.52 ± 0.01 b | 0.93 ± 0.02 i | 1.40 ± 0.05 cd | 1.355 ± 0.004 ef | 1.39 ± 0.04 cde | 1.78 ± 0.06 a | 1.51 ± 0.09 b | 1.32 ± 0.01 f | 1.38 ± 0.05 de | 1.43 ± 0.03 c | 1.047 ± 0.002 h |
| Peak 8 | 13.10 ± 0.01 l | 19.93 ± 0.02 e | 21.68 ± 0.03 c | 22.35 ± 0.06 b | 12.15 ± 0.02 m | 17.37 ± 0.02 i | 18.62 ± 0.01 g | 20.66 ± 0.01 d | 19.7 ± 0.1 f | 15.60 ± 0.02 k | 17.57 ± 0.01 h | 16.54 ± 0.02 j | 24.36 ± 0.04 a |
| Peak 9 | 1.99 ± 0.01 e | 2.34 ± 0.05 c | 2.25 ± 0.04 d | 2.90 ± 0.01 a | 1.77 ± 0.08 g | 1.97 ± 0.01 ef | 2.38 ± 0.03 bc | 2.4 ± 0.1 b | 2.84 ± 0.08 a | 2.20 ± 0.05 d | 1.93 ± 0.01 f | 1.13 ± 0.04 i | 1.41 ± 0.03 h |
| Peak 10 | 21.4 ± 0.40 j | 34.4 ± 0.4 e | 38.5 ± 0.60 b | 16.9 ± 0.5 k | 36.0 ± 0.2 c | 32.5 ± 0.6 f | 26.11 ± 0.05 h | 50.9 ± 0.9 a | 30.4 ± 0.4 g | 35.2 ± 0.6 d | 23.5 ± 0.2 i | 17.0 ± 0.6 k | 7.5 ± 0.2 l |
| Peak 11 | 2.89 ± 0.01 k | 4.27 ± 0.06 f | 5.4 ± 0.4 c | 4.00 ± 0.09 gh | 3,52 ± 0.08 j | 3.546 ± 0.002 ij | 3.69 ± 0.09 i | 6.4 ± 0.1 a | 5.15 ± 0.04 d | 4.4 ± 0.1 e | 3.95 ± 0.1 h | 4.1 ± 0.6 fg | 6.0 ± 0.01 b |
| Peak 12 | 2.520 ± 0.001 i | 2.73 ± 0.02 h | 5.09 ± 0.02 a | 3.57 ± 0.01 c | 1.78 ± 0.01 k | 2.41 ± 0.01 j | 2.76 ± 0.02 g | nd | 3.14 ± 0.01 f | 2.41 ± 0.01 j | 5.06 ± 0.01 b | 3.41 ± 0.03 e | 3.52 ± 0.001 d |
| Peak 13 | 20.2 ± 0.50 h | 36.8 ± 0.3 b | 30.9 ± 0.5 d | 14.89 ± 0.04 j | 30.27 ± 0.04 e | 30.9 ± 0.8 d | 29.8 ± 0.4 f | 44.3 ± 0.2 a | 34.1 ± 0.5 c | 30.6 ± 0.4 de | 25.9 ± 0.4 g | 16.7 ± 0.5 i | 8.7 ± 0.01 k |
| TPA | 50 ± 1 h | 81 ± 1 b | 81 ± 2 b | 40.8 ± 0.4 j | 75.4 ± 0.3 d | 72.6 ± 0.2 e | 66 ± 1 f | 113 ± 1 a | 77 ± 1 c | 76.351 ± 0.003 c | 59 ± 1 g | 43.97 ± 0.04 i | 26.3 ± 0.5 k |
| TF | 17.81 ± 0.01 l | 25.0 ± 0.01 e | 29.85 ± 0.03 a | 28.04 ± 0.04 c | 15.86 ± 0.03 m | 21.62 ± 0.01 j | 23.04 ± 0.01 g | 22.988 ± 0.002 h | 25.1 ± 0.1 d | 21.00 ± 0.02 k | 24.86 ± 0.01 f | 22.2 ± 0.1 i | 29.75 ± 0.02 b |
| TPC | 68 ± 1 k | 106 ± 1 c | 111 ± 2 b | 68.9 ± 0.5 i | 91.3 ± 0.3 g | 94.2 ± 0.2 f | 89 ± 1 h | 136 ± 1 a | 102 ± 1 d | 97.36 ± 0.02 e | 84 ± 1 i | 66.17 ± 0.02 l | 56.1 ± 0.5 m |
| Aqueous Extract | |||||||||||||
| Peak 1 | 1.18 ± 0.06 d | 0.87 ± 0.04 g | 1.02 ± 0.02 e | 0.59 ± 0.02 h | 1.31 ± 0.01 b | 1.20 ± 0.06 cd | 1.23 ± 0.01 c | 7.9 ± 0.1 a | 1.19 ± 0.06 d | 0.91 ± 0.05 fg | 0.96 ± 0.04 f | 1.02 ± 0.05 e | 0.28 ± 0.04 i |
| Peak 2 | 2.36 ± 0.02 h | 1.97 ± 0.04 j | 2.65 ± 0.01 f | 2.55 ± 0.08 g | 2.31 ± 0.01 hi | 3.8 ± 0.1 c | 3.83 ± 0.03 c | 5.03 ± 0.07 a | 2.37 ± 0.05 h | 2.28 ± 0.05 i | 4.3 ± 0.01 b | 3.19 ± 0.03 d | 3.06 ± 0.08 e |
| Peak 3 | 3.5 ± 0.1 d | 3.18 ± 0.03 g | 3.002 ± 0.002 h | 1.52 ± 0.06 l | 3.22 ± 0.02 f | 3.83 ± 0.02 c | 4.19 ± 0.06 a | 4.13 ± 0.03 b | 2.98 ± 0.04 h | 3.30 ± 0.03 e | 2.77 ± 0.08 j | 2.19 ± 0.09 k | 2.90 ± 0.05 i |
| Peak 4 | 3.25 ± 0.006 f | 3.46 ± 0.08 e | 2.917 ± 0.004 h | 2.80 ± 0.03 i | 3.29 ± 0.04 f | 3.14 ± 0.08 g | 3.99 ± 0.08 b | 3.87 ± 0.05 c | 3.18 ± 0.04 g | 2.80 ± 0.04 i | 2.61 ± 0.05 j | 3.63 ± 0.01 d | 4.35 ± 0.04 a |
| Peak 5 | 6.49 ± 0.02 c | 6.71 ± 0.02 b | 8.76 ± 0.01 a | 3.681 ± 0.003 k | 5.18 ± 0.04 f | 5.28 ± 0.01 d | 4.1 ± 0.10 i | 4.45 ± 0.02 g | 3.32 ± 0.02 l | 3.79 ± 0.01 j | 4.34 ± 0.01 h | 2.95 ± 0.03 m | 5.22 ± 0.03 e |
| Peak 6 | 12.83 ± 0.04 d | 9.36 ± 0.02 h | 8.87 ± 0.03 i | 16.73 ± 0.04 a | 8.6 ± 0.1 k | 10.8 ± 0.1 g | 11.6 ± 0.10 f | 12.9 ± 0.1 d | 8.17 ± 0.02 l | 12.97 ± 0.04 c | 13.31 ± 0.05 b | 8.8 ± 0.1 j | 12.26 ± 0.02 e |
| Peak 7 | 2.52 ± 0.01 cd | 2.41 ± 0.09 e | 3.33 ± 0.01 a | 1.6 ± 0.2 h | 2.45 ± 0.08 de | 2.26 ± 0.04 f | 2.38 ± 0.07 e | 2.83 ± 0.01 b | 2.61 ± 0.03 c | 3.3 ± 0.2 a | 2.16 ± 0.09 g | 2.74 ± 0.01 b | 2.11 ± 0.06 g |
| Peak 8 | 70.81 ± 0.04 g | 64.31 ± 0.02 k | 76.4 ± 0.1 d | 101.5 ± 0.1 a | 42.12 ± 0.01 m | 66.8 ± 0.1 i | 65.9 ± 0.1 j | 84.1 ± 0.1 c | 73.56 ± 0.09 f | 67.35 ± 0.08 h | 73.8 ± 0.1 e | 53.6 ± 0.2 l | 99.43 ± 0.02 b |
| Peak 9 | 4.4 ± 0.1 ef | 3.26 ± 0.03 i | 4.4 ± 0.4 ef | 6.6 ± 0.1 a | 3.73 ± 0.03 h | 4.3 ± 0.5 g | 4.6 ± 0.1 e | 5.2 ± 0.1 d | 6.2 ± 0.5 b | 5.7 ± 0.4 c | 3.30 ± 0.01 i | 4.3 ± 0.30 g | 5.8 ± 0.3 c |
| Peak 10 | 290.0 ± 0.4 e | 293 ± 3 d | 353.0 ± 0.3 b | 190 ± 2 j | 288.6 ± 0.2 f | 318.0 ± 0.7 c | 211 ± 1 i | 550 ± 3 a | 266.3 ± 0.7 g | 291 ± 4 e | 218.4 ± 0.5 h | 52.2 ± 0.6 k | 27.1 ± 0.3 l |
| Peak 11 | 11.5 ± 0.3 i | 13.9 ± 0.4 g | 16.37 ± 0.06 c | 15.1 ± 0.4 d | 9.8 ± 0.3 j | 12.2 ± 0.6 h | 9.63 ± 0.02 j | 16.8 ± 0.2 b | 14.83 ± 0.07 de | 14.7 ± 0.4 e | 14.14 ± 0.09 f | 16.9 ± 0.1 b | 26.2 ± 0.6 a |
| Peak 12 | 11.25 ± 0.03 e | 7.75 ± 0.02 j | 16.08 ± 0.01 b | 15.1 ± 0.1 c | 4.45 ± 0.04 k | 8.54 ± 0.04 i | 8.9 ± 0.1 h | nd | 10.9 ± 0.1 f | 8.51 ± 0.02 i | 19.8 ± 0.1 a | 9.73 ± 0.02 g | 13.7 ± 0.1 d |
| Peak 13 | 381 ± 5 d | 390 ± 2 c | 366.7 ± 0.9 e | 212.9 ± 0.4 k | 309.9 ± 0.4 h | 405 ± 3 b | 279 ± 2 j | 582 ± 1 a | 343 ± 2 f | 340 ± 4 g | 305.7 ± 0.5 i | 62.8 ± 0.3 l | 47.7 ± 0.8 m |
| TPA | 699 ± 5 d | 712 ± 1 c | 753.4 ± 0.7 b | 433 ± 3 j | 621.6 ± 0.8 g | 754 ± 3 b | 519 ± 3 i | 1177 ± 3 a | 643 ± 2 f | 664 ± 9 e | 554.3 ± 0.1 h | 148.9 ± 0.6 k | 119 ± 1 l |
| TF | 101.4 ± 0.2 e | 88.13 ± 0.05 j | 110.06 ± 0.01 d | 137.0 ± 0.2 a | 60.3 ± 0.1 l | 91.4 ± 0.1 h | 90.4 ± 0.2 i | 101.4 ± 0.2 e | 96.0 ± 0.2 f | 92.6 ± 0.1 g | 111.2 ± 0.1 c | 75.1 ± 0.3 k | 130.6 ± 0.2 b |
| TPC | 801 ± 5 d | 801 ± 1 d | 863.5 ± 0.7 b | 570 ± 3 j | 681.9 ± 0.7 g | 845 ± 3 c | 610 ± 3 i | 1278 ± 3 a | 739 ± 2 f | 756 ± 9 e | 665.6 ± 0.2 h | 224 ± 1 l | 250 ± 1 k |
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hydroethanolic Extracts | |||||||||||||
| Antioxidant Activity (EC50 Values, μg/mL) | |||||||||||||
| DPPH scavenging activity | 142 ± 6 ef | 139 ± 3 ef | 139 ± 2 ef | 212 ± 8 b | 137 ± 3 f | 139 ± 1 f | 150 ± 3 d | 87 ± 2 g | 150 ± 2 d | 146 ± 8 de | 181 ± 2 c | 140 ± 5 ef | 257 ± 7 a |
| Reducing power | 110 ± 1 f | 98 ± 1 g | 117.9 ± 0.5 e | 149 ± 2 b | 97 ± 2 g | 107 ± 2 f | 129 ± 1 d | 72 ± 1 h | 67 ± 1 i | 135 ± 9 c | 133 ± 3 cd | 133 ± 1 cd | 216 ± 6 a |
| β-carotene bleaching inhibition | 1009 ± 88 d | 821 ± 34 e | 1062 ± 65 d | 516 ± 75 f | 1176 ± 40 c | 1578 ± 77 b | 541 ± 43 f | 1833 ± 178 a | 372 ± 18 g | 252 ± 7 h | 190 ± 20 h | 223 ± 3 h | 214 ± 10 h |
| TBARS inhibition | 28 ± 1 g | 17 ± 1 i | 26 ± 1 h | 35 ± 1 e | 38.25 ± 0.01 d | 63.5 ± 0.1 a | 44.56 ± 0.01 c | 25.57 ± 0.02 h | 38.2 ± 0.3 d | 48 ± 2 b | 62 ± 4 a | 27.7 ± 0.05 gh | 31.4 ± 0.1 f |
| Anti-Inflammatory Potential (EC50 Values, µg/mL) | |||||||||||||
| Nitric oxide (NO) production | 190 ± 3 c | 198 ± 5 b | 190 ± 6 c | >400 | >400 | 124 ± 8 f | >400 | >400 | >400 | 216 ± 12 a | 171 ± 3 d | 162 ± 4 e | >400 |
| Aqueous Extracts | |||||||||||||
| Antioxidant Activity (EC50 Values. μg/mL) | |||||||||||||
| DPPH scavenging activity | 109 ± 4 f | 99 ± 2 g | 99 ± 1 g | 68.0 ± 0.5 i | 99 ± 2 g | 115 ± 1 e | 125 ± 2 d | 69 ± 3 h | 98 ± 3 g | 115 ± 2 e | 133 ± 3 c | 144 ± 6 b | 191 ± 2 a |
| Reducing power | 101 ± 2 f | 51 ± 1 i | 113 ± 4 e | 137 ± 4 b | 99 ± 2 f | 93 ± 3 g | 111 ± 2 e | 73 ± 1 h | 110 ± 2 e | 125 ± 4 cd | 122 ± 6 d | 130 ± 6 c | 167 ± 1 a |
| β-carotene bleaching inhibition | 274 ± 10 f | 253 ± 14 fg | 267 ± 9 f | 385 ± 13 d | 472 ± 14 b | 475 ± 19 b | 422 ± 25 c | 388 ± 21 d | 452 ± 12 b | 236 ± 14 g | 305 ± 4 e | 391 ± 25 d | 530 ± 21 a |
| TBARS inhibition | 14 ± 1 i | 24.8 ± 0.4 de | 16 ± 1 h | 29 ± 1 c | 21.8 ± 0.4 fg | 22 ± 1 f | 21.15 ± 0.01 g | 21.2 ± 0.2 g | 21 ± 1 g | 39.1 ± 0.1 a | 25 ± 1 d | 24.1 ± 0.4 e | 36 ± 1 b |
| Anti-Inflammatory Potential (EC50 Values. µg/mL) | |||||||||||||
| Nitric oxide (NO) production | >400 | >400 | >400 | 301 ± 7 a | 237 ± 7 b | >400 | 205 ± 7 c | 140 ± 5 d | >400 | >400 | >400 | >400 | >400 |
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hydroethanolic Extracts | |||||||||||||
| Tumor Cell Lines (GI50 Values, µg/mL) | |||||||||||||
| MCF-7 | 61 ± 2 f | 57 ± 3 fg | 58 ± 2 fg | 115 ± 9 c | 82 ± 8 d | 53 ± 5 g | 70 ± 2 e | 61 ± 4 f | 60 ± 4 f | 53 ± 3 g | 61 ± 3 f | 236 ± 8 a | 212 ± 1 b |
| NCI-H460 | 285 ± 22 bc | 275 ± 22 c | 277 ± 10 c | >400 | >400 | 340 ± 21 a | >400 | >400 | >400 | >400 | 297 ± 6 b | 119 ± 7 e | 241 ± 23 d |
| HeLa | 70 ± 5 de | 65.0 ± 0.2 fgh | 69 ± 2 def | 200 ± 14 c | 216 ± 7 b | 66.2 ± 0.2 efgh | 67 ± 2 efgh | 63.3 ± 0.1 gh | 73.1 ± 0.3 d | 62.2 ± 0.6 h | 65 ± 2 fgh | 68 ± 3 efg | 222 ± 6 a |
| HepG2 | 82 ± 4 e | 67 ± 1 f | 94 ± 6 d | 144 ± 10 c | 191 ± 16 b | 34 ± 3 g | 212 ± 19 a | 67 ± 10 f | 100 ± 3 d | 62 ± 5 f | 65 ± 1 f | 203 ± 14 a | 204 ± 4 a |
| Non-Tumor Cells (GI50 Values, µg/mL) | |||||||||||||
| PLP2 | >400 | >400 | >400 | >400 | >400 | >400 | >400 | >400 | >400 | >400 | >400 | 291 ± 11 | >400 |
| Aqueous Extracts | |||||||||||||
| Tumor Cell Lines (GI50 Values, µg/mL) | |||||||||||||
| MCF-7 | >400 | 270 ± 14 c | 267 ± 2 c | 287 ± 8 b | 267.7 ± 0.3 c | 222 ± 14 e | 223 ± 6 e | 185 ± 16 g | 150 ± 9 h | 290 ± 12 b | 256.2 ± 0.6 d | 270 ± 14 f | 342 ± 14 a |
| NCI-H460 | 329 ± 15 c | 256 ± 5 e | 334 ± 15 c | 113 ± 9 i | 142 ± 12 h | 226 ± 14 f | 188 ± 12 g | 183 ± 11 g | 258 ± 14 e | 245 ± 13 e | 349 ± 5 b | 293 ± 12 d | 374 ± 7 a |
| HeLa | >400 | >400 | 310.2 ± 0.4 b | 343.92 ± 0.01 a | 253 ± 23 d | 262 ± 14 d | 199 ± 12 f | 159 ± 16 g | 286 ± 4 c | 195 ± 11 f | 224 ± 11 e | 82 ± 14 h | 298 ± 17 bc |
| HepG2 | 315 ± 6 a | 254 ± 14 c | 251 ± 14 c | 293 ± 10 b | >400 | 211 ± 17 d | 194 ± 15 e | 179 ± 14 f | 118 ± 8 h | 256 ± 4 c | 191 ± 143 ef | 148 ± 1 g | 324 ± 6 a |
| Non-Tumor Cells (GI50 Values, µg/mL) | |||||||||||||
| PLP2 | >400 | >400 | >400 | >400 | >400 | >400 | 349 ± 12 b | 362 ± 21 a | 240 ± 16 c | >400 | >400 | >400 | >400 |
| Antibacterial Activity | |||||||||||||||
| Hydroethanolic Extracts | |||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | S | A | |
| MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | |
| MBC | MBC | MBC | MBC | MBC | MBC | MBC | MBC | MBC | MBC | MBC | MBC | MBC | MBC | MBC | |
| B.c. | 50 | 75 | 75 | 75 | 50 | 75 | 75 | 75 | 75 | 75 | 75 | 75 | 200 | 100 | 250 |
| 75 | 150 | 150 | 150 | 75 | 150 | 150 | 150 | 150 | 150 | 150 | 150 | 300 | 200 | 400 | |
| M.f. | 75 | 100 | 100 | 100 | 75 | 100 | 100 | 100 | 100 | 100 | 100 | 300 | 200 | 200 | 250 |
| 150 | 150 | 150 | 150 | 150 | 150 | 150 | 150 | 150 | 150 | 150 | 450 | 300 | 300 | 400 | |
| S.a. | 20 | 40 | 30 | 150 | 100 | 150 | 150 | 150 | 200 | 150 | 200 | 450 | 200 | 40 | 250 |
| 40 | 75 | 40 | 300 | 150 | 300 | 300 | 300 | 150 | 300 | 300 | 600 | 300 | 100 | 450 | |
| L.m. | 100 | 150 | 100 | 150 | 100 | 150 | 150 | 150 | 150 | 150 | 200 | 450 | 200 | 200 | 400 |
| 150 | 300 | 150 | 300 | 150 | 300 | 300 | 300 | 300 | 300 | 300 | 600 | 300 | 300 | 500 | |
| E.c. | 75 | 100 | 100 | 100 | 75 | 100 | 100 | 100 | 100 | 100 | 100 | 200 | 450 | 200 | 400 |
| 150 | 150 | 150 | 150 | 150 | 150 | 150 | 150 | 150 | 150 | 150 | 300 | 600 | 300 | 500 | |
| En.cl. | 40 | 40 | 40 | 50 | 40 | 40 | 100 | 75 | 75 | 75 | 75 | 200 | 450 | 200 | 250 |
| 75 | 75 | 75 | 75 | 75 | 75 | 150 | 150 | 150 | 150 | 150 | 300 | 600 | 300 | 500 | |
| P.a. | 100 | 150 | 100 | 150 | 100 | 150 | 150 | 150 | 150 | 150 | 150 | 300 | 150 | 200 | 750 |
| 150 | 300 | 150 | 300 | 150 | 300 | 300 | 300 | 300 | 300 | 300 | 450 | 300 | 300 | 1200 | |
| S.t. | 150 | 150 | 150 | 200 | 100 | 200 | 200 | 150 | 200 | 200 | 200 | 150 | 150 | 250 | 400 |
| 300 | 300 | 300 | 300 | 150 | 300 | 300 | 300 | 300 | 300 | 300 | 300 | 300 | 500 | 750 | |
| Aqueous Extracts | |||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | S | A | |
| MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | |
| MBC | MBC | MBC | MBC | MBC | MBC | MBC | MBC | MBC | MBC | MBC | MBC | MBC | MBC | MBC | |
| B.c. | 200 | 300 | 150 | 300 | 75 | 200 | 150 | 200 | 150 | 150 | 150 | 300 | 150 | 100 | 250 |
| 300 | 450 | 300 | 450 | 150 | 300 | 300 | 300 | 300 | 300 | 300 | 450 | 300 | 200 | 400 | |
| M.f. | 200 | 300 | 200 | 300 | 200 | 300 | 200 | 200 | 200 | 150 | 200 | 450 | 200 | 200 | 250 |
| 300 | 450 | 300 | 450 | 300 | 450 | 300 | 300 | 300 | 300 | 300 | 600 | 300 | 300 | 400 | |
| S.a. | 300 | 300 | 300 | 300 | 450 | 300 | 200 | 200 | 150 | 300 | 300 | 300 | 300 | 40 | 250 |
| 450 | 450 | 600 | 450 | 600 | 450 | 300 | 300 | 300 | 450 | 450 | 600 | 600 | 100 | 450 | |
| L.m. | 300 | 300 | 300 | 300 | 450 | 300 | 300 | 300 | 150 | 300 | 300 | 300 | 300 | 200 | 400 |
| 600 | 600 | 600 | 600 | 600 | 450 | 450 | 450 | 300 | 450 | 450 | 600 | 600 | 300 | 500 | |
| E.c. | 200 | 200 | 200 | 200 | 200 | 200 | 200 | 100 | 200 | 450 | 200 | 450 | 200 | 200 | 40 |
| 300 | 300 | 300 | 300 | 300 | 300 | 300 | 150 | 300 | 600 | 300 | 900 | 300 | 300 | 500 | |
| En.cl. | 150 | 200 | 200 | 200 | 200 | 150 | 200 | 150 | 150 | 200 | 150 | 200 | 150 | 200 | 250 |
| 300 | 300 | 300 | 300 | 300 | 300 | 300 | 300 | 300 | 300 | 300 | 300 | 300 | 300 | 500 | |
| P.a. | 300 | 450 | 300 | 300 | 300 | 300 | 300 | 150 | 150 | 300 | 300 | 300 | 300 | 200 | 750 |
| 450 | 600 | 600 | 600 | 600 | 450 | 600 | 450 | 300 | 450 | 600 | 600 | 600 | 300 | 1200 | |
| S.t. | 450 | 450 | 300 | 300 | 300 | 300 | 300 | 300 | 150 | 300 | 300 | 300 | 300 | 250 | 400 |
| 600 | 600 | 600 | 600 | 600 | 600 | 600 | 450 | 300 | 450 | 600 | 600 | 600 | 500 | 750 | |
| Antifungal Activity | |||||||||||||||
| Hydroethanolic Extracts | |||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | K | B | |
| MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | |
| MFC | MFC | MFC | MFC | MFC | MFC | MFC | MFC | MFC | MFC | MFC | MFC | MFC | MFC | MFC | |
| A.fum. | 75 | 150 | 75 | 75 | 100 | 40 | 150 | 75 | 100 | 150 | 150 | 200 | 200 | 250 | 150 |
| 150 | 300 | 150 | 150 | 150 | 75 | 300 | 150 | 150 | 300 | 300 | 300 | 300 | 500 | 200 | |
| A.v. | 40 | 30 | 40 | 40 | 75 | 40 | 40 | 40 | 75 | 150 | 75 | 150 | 150 | 200 | 100 |
| 75 | 75 | 150 | 75 | 150 | 75 | 75 | 75 | 150 | 300 | 150 | 300 | 300 | 500 | 200 | |
| A.o. | 40 | 40 | 50 | 40 | 75 | 40 | 50 | 40 | 20 | 75 | 40 | 100 | 200 | 150 | 150 |
| 75 | 75 | 75 | 75 | 150 | 75 | 75 | 100 | 40 | 150 | 75 | 300 | 300 | 2000 | 200 | |
| A.n. | 50 | 75 | 40 | 75 | 150 | 75 | 150 | 75 | 150 | 150 | 200 | 150 | 200 | 200 | 150 |
| 75 | 150 | 75 | 150 | 300 | 150 | 300 | 150 | 300 | 300 | 300 | 300 | 300 | 500 | 200 | |
| T.v. | 20 | 40 | 20 | 30 | 75 | 30 | 30 | 50 | 15 | 50 | 30 | 75 | 100 | 1000 | 150 |
| 40 | 75 | 40 | 40 | 150 | 40 | 40 | 75 | 20 | 75 | 40 | 150 | 150 | 1000 | 200 | |
| P.f. | 75 | 75 | 40 | 75 | 200 | 75 | 75 | 40 | 75 | 75 | 150 | 200 | 200 | 200 | 200 |
| 150 | 150 | 75 | 150 | 300 | 15 | 150 | 75 | 150 | 150 | 300 | 300 | 300 | 500 | 250 | |
| P.o. | 40 | 40 | 30 | 40 | 100 | 40 | 40 | 150 | 40 | 75 | 75 | 30 | 30 | 2500 | 200 |
| 75 | 75 | 40 | 75 | 150 | 75 | 75 | 200 | 75 | 150 | 150 | 450 | 450 | 3500 | 250 | |
| P.v.c. | 75 | 150 | 50 | 100 | 200 | 75 | 150 | 150 | 150 | 150 | 150 | 300 | 300 | 200 | 100 |
| 150 | 300 | 75 | 150 | 300 | 150 | 300 | 300 | 300 | 300 | 300 | 450 | 450 | 300 | 200 | |
| Aqueous Extracts | |||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | K | B | |
| MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | MIC | |
| MFC | MFC | MFC | MFC | MFC | MFC | MFC | MFC | MFC | MFC | MFC | MFC | MFC | MFC | MFC | |
| A.fum. | 300 | 300 | 600 | - | 600 | 300 | - | - | 600 | 600 | 300 | 300 | 450 | 250 | 150 |
| 600 | 600 | 1200 | - | 1200 | 600 | - | - | 900 | 1200 | 450 | 600 | 900 | 500 | 200 | |
| A.v. | 300 | 300 | 300 | 600 | 300 | 300 | 300 | 300 | 300 | 200 | 100 | 150 | 200 | 200 | 100 |
| 600 | 600 | 600 | 1200 | 600 | 600 | 600 | 600 | 600 | 450 | 200 | 300 | 300 | 500 | 200 | |
| A.o. | 600 | 900 | - | - | - | 600 | 300 | - | 200 | 300 | 150 | 300 | 200 | 1500 | 150 |
| 1200 | 1200 | - | - | - | 1200 | 600 | - | 450 | 600 | 300 | 450 | 450 | 2000 | 200 | |
| A.n. | - | 300 | - | - | - | 600 | - | - | 300 | - | 200 | 450 | 450 | 200 | 150 |
| - | 600 | - | - | - | 1200 | - | - | 450 | - | 450 | 900 | 900 | 500 | 200 | |
| T.v. | 600 | 300 | 600 | 600 | - | 600 | - | - | 100 | 300 | 200 | 450 | 450 | 1000 | 150 |
| 1200 | 600 | 1200 | 1200 | - | 1200 | - | - | 300 | 600 | 450 | 600 | 600 | 1000 | 200 | |
| P.f. | 600 | 300 | 600 | - | 600 | - | - | - | - | - | 450 | 450 | 450 | 200 | 200 |
| 1200 | 600 | 1200 | - | 1200 | - | - | - | - | - | 600 | 900 | 900 | 500 | 250 | |
| P.o. | 900 | 450 | 600 | - | 600 | - | - | - | 200 | - | 450 | 450 | 450 | 2500 | 200 |
| 1200 | 600 | 1200 | - | 1200 | - | - | - | 450 | - | 600 | 900 | 900 | 3.50 | 0.25 | |
| P.v.c. | - | 600 | 600 | - | - | - | - | - | 200 | 450 | 450 | 450 | 450 | 200 | 100 |
| - | 1200 | 1200 | - | - | - | - | - | 450 | 900 | 600 | 900 | 900 | 300 | 200 | |
| Samples | BPGV Accession Number | Origin | Site Data Altitude (m) |
|---|---|---|---|
| 1 | 10378 | Marvão. Portalegre | 333 |
| 2 | 09845 | Évora | 198 |
| 3 | 09838 | Vila Viçosa. Évora | 420 |
| 4 | 11290 | Bragança | 810 |
| 5 | 10372 | Arronches. Portalegre | 375 |
| 6 | 10400 | Portalegre | 375 |
| 7 | 10418 | Nisa. Portalegre | 291 |
| 8 | 10387 | Ponte de Sôr. Portalegre | 175 |
| 9 | 10391 | Évora | 155 |
| 10 | 10412 | Castelo de Vide. Portalegre | 358 |
| 11 | 10369 | Elvas. Portalegre | 267 |
| 12 | 10379 | Castelo de Vide. Portalegre | 353 |
| 13 | 11308 | Bragança | 667 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lopes, C.L.; Pereira, E.; Soković, M.; Carvalho, A.M.; Barata, A.M.; Lopes, V.; Rocha, F.; Calhelha, R.C.; Barros, L.; Ferreira, I.C.F.R. Phenolic Composition and Bioactivity of Lavandula pedunculata (Mill.) Cav. Samples from Different Geographical Origin. Molecules 2018, 23, 1037. https://doi.org/10.3390/molecules23051037
Lopes CL, Pereira E, Soković M, Carvalho AM, Barata AM, Lopes V, Rocha F, Calhelha RC, Barros L, Ferreira ICFR. Phenolic Composition and Bioactivity of Lavandula pedunculata (Mill.) Cav. Samples from Different Geographical Origin. Molecules. 2018; 23(5):1037. https://doi.org/10.3390/molecules23051037
Chicago/Turabian StyleLopes, Catarina L., Eliana Pereira, Marina Soković, Ana Maria Carvalho, Ana Maria Barata, Violeta Lopes, Filomena Rocha, Ricardo C. Calhelha, Lillian Barros, and Isabel C.F.R. Ferreira. 2018. "Phenolic Composition and Bioactivity of Lavandula pedunculata (Mill.) Cav. Samples from Different Geographical Origin" Molecules 23, no. 5: 1037. https://doi.org/10.3390/molecules23051037
APA StyleLopes, C. L., Pereira, E., Soković, M., Carvalho, A. M., Barata, A. M., Lopes, V., Rocha, F., Calhelha, R. C., Barros, L., & Ferreira, I. C. F. R. (2018). Phenolic Composition and Bioactivity of Lavandula pedunculata (Mill.) Cav. Samples from Different Geographical Origin. Molecules, 23(5), 1037. https://doi.org/10.3390/molecules23051037