Synthesis and Tuberculostatic Activity Evaluation of Novel Benzazoles with Alkyl, Cycloalkyl or Pyridine Moiety
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry
2.2. Tuberculostatic Activity
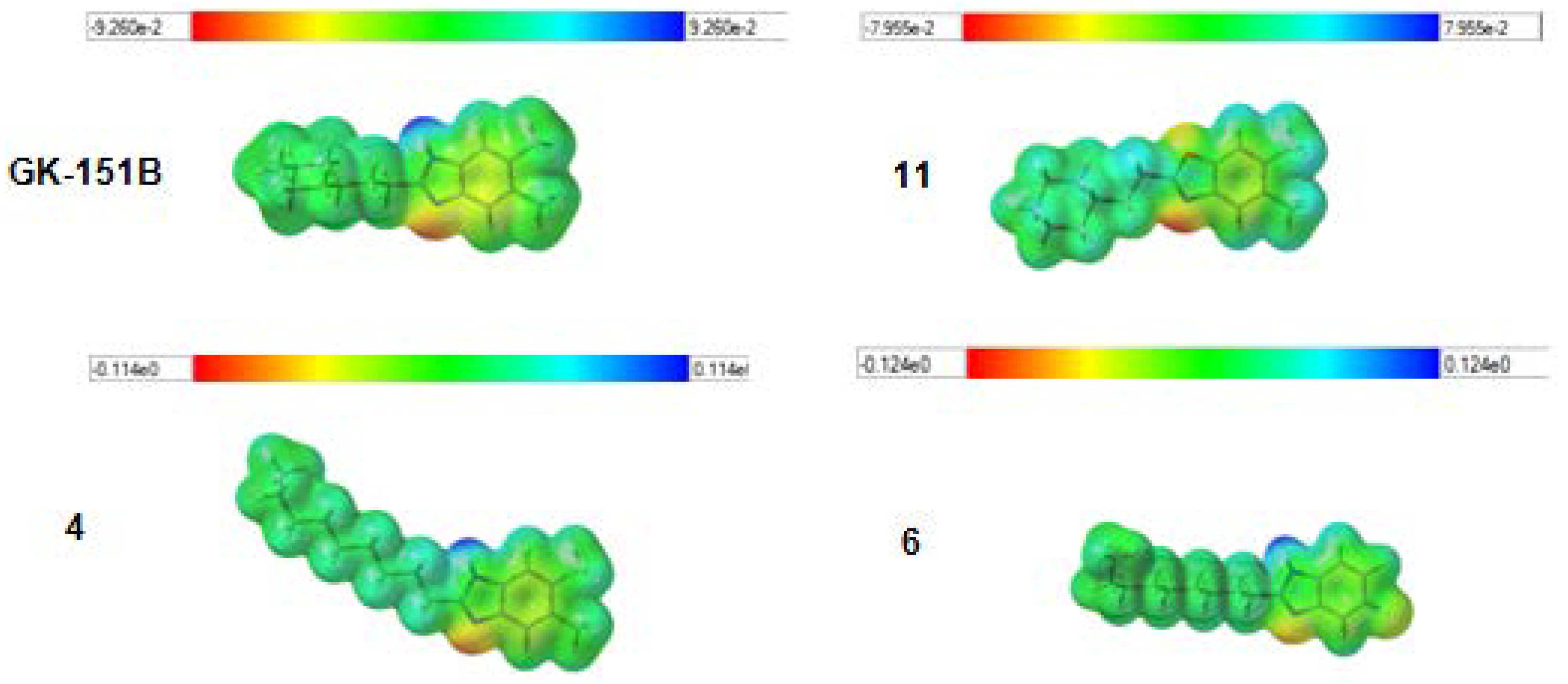
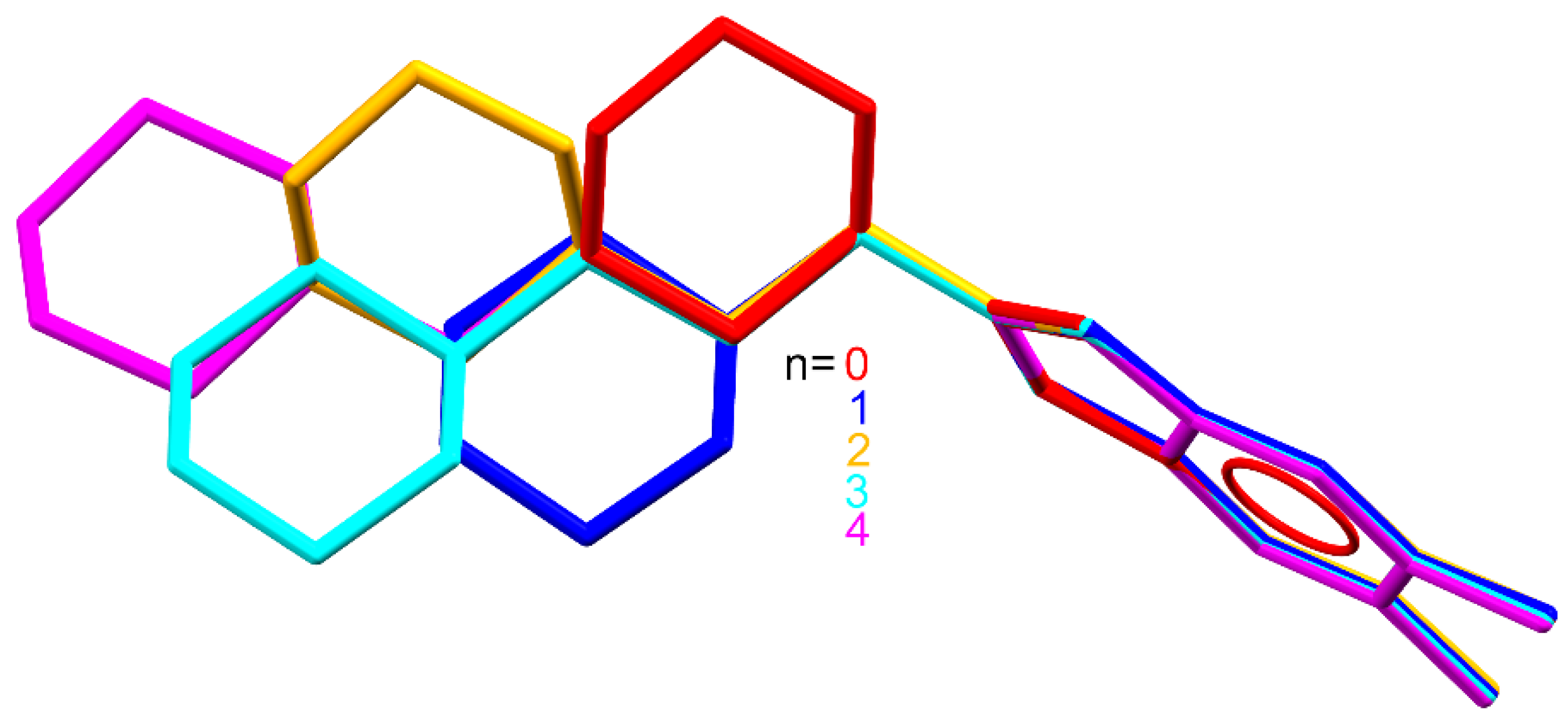
2.3. Quantum Chemical Calculations
3. Materials and Methods
3.1. Chemistry
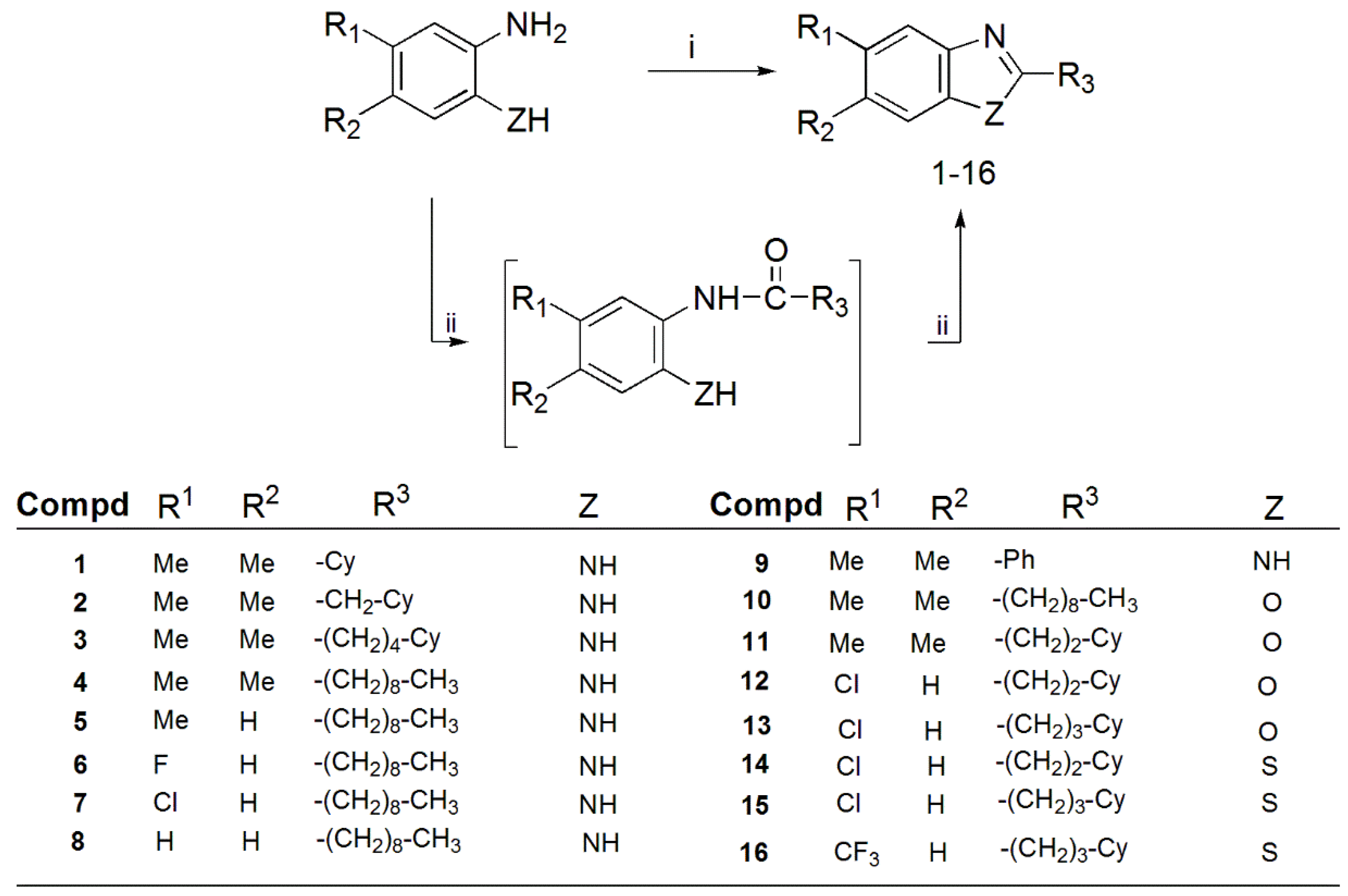
3.1.1. General Procedure for the Synthesis of Benzimidazoles 1–8
3.1.2. General Procedure for Synthesis of Benzimidazole 9
3.1.3. General Procedure for Synthesis of 4,5-Dimethylbenzoxazoles 10, 11
3.1.4. General Procedure for the Synthesis of 5-Chlorobenzoxazoles 12, 13
3.1.5. General Procedure for the Synthesis of Benzothiazoles 14, 15
3.1.6. General Method for the Synthesis of 5-Trifluoromethylbenzothiazoles 16
3.1.7. General Method for the Synthesis of 2-Pyridinebenzimidazoles 17–20
3.2. Antimycobacterial Activity Assay
3.3. Quantum Chemical Calculations
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Talbot, E.A.; Raffa, B.J. Mycobacterium tuberculosis. In Molecular Medical Microbiology, 2nd ed.; Academic Press: Boston, MA, USA, 2015; Volume 3, pp. 1637–1653. [Google Scholar]
- Krauss, M.R.; Harris, D.R.; Abreu, T.; Ferreira, F.G.; Ruz, N.P.; Worrell, C.; Hazra, R. Tuberculosis in HIV-infected infants, children and adolescents in Latin America. Braz. J. Infect. Dis. 2015, 19, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Post, F.A.; Grint, D.; Werlinrud, A.M.; Panteleev, A.; Riekstina, V.; Malashenkov, E.A.; Skrahina, A.; Duiculescu, D.; Podlekareva, D.; Karpov, I.; et al. Multi-drug-resistant tuberculosis in HIV positive patients in Eastern Europe. J. Infect. 2014, 68, 259–263. [Google Scholar] [CrossRef] [PubMed]
- Thwaites, G.E.; van Toorn, R.; Schoeman, J. Tuberculous meningitis: more questions, still too few answer. Lancet Neurol. 2013, 12, 999–1010. [Google Scholar] [CrossRef]
- Van der Werf, T.S.; van der Graaf, W.T.A.; Tappero, J.W.; Asiedu, K. Mycobacterium ulcerans infection. Lancet 1999, 354, 1013–1018. [Google Scholar] [CrossRef]
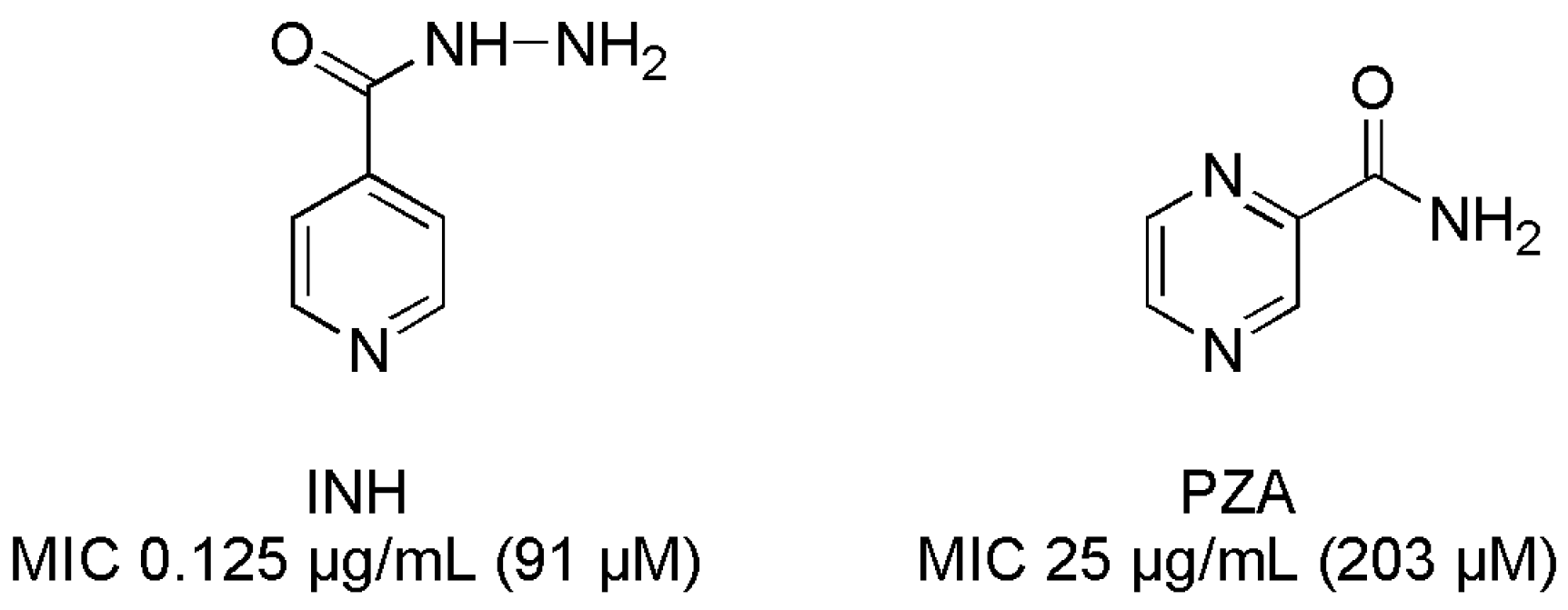
- Isoniazid. Tuberculosis 2008, 88, 112–116. [CrossRef]
- Pyrazinamide. Tuberculosis 2008, 88, 141–144. [CrossRef]
- Olczak, A.; Główka, M.L.; Gołka, J.; Szczesio, M.; Bojarska, J.; Kozłowska, K.; Foks, H.; Orlewska, C. Is planarity of pyridin-2-yl- and pyrazin-2-yl-formamide thiosemicarbazones related to their tuberculostatic activity? X-ray structures of two pyrazine-2-carboxamide-N’-carbonothioyl-hydrazones. J. Mol. Struct. 2007, 830, 171–175. [Google Scholar] [CrossRef]
- Olczak, A.; Szczesio, M.; Gołka, J.; Orlewska, C.; Gobis, K.; Foks, H.; Główka, M.L. Planarity of heteroaryldithiocarbazic acid derivatives showing tuberculostatic activity. II. Crystal structures of 3-[amino(pyrazin-2-yl)methylidene]-2-methylcarbazic acid esters. Acta Cryst. C 2011, C67, o37–o42. [Google Scholar] [CrossRef] [PubMed]
- Kazimierczuk, Z.; Andrzejewska, M.; Kaustova, J.; Klimešova, V. Synthesis and antimycobacterial activity of 2-substitute halogenobenzimidazoles. Eur. J. Med. Chem. 2005, 40, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Shingalapur, R.V.; Hosamani, K.M.; Keri, R.S. Derivatives of benzimidazole pharmacophore: Synthesis, anticonvulsant, antidiabetic and DNA cleavage studies. Eur. J. Med. Chem. 2010, 45, 1753–1759. [Google Scholar] [CrossRef] [PubMed]
- Jadhav, G.R.; Shaikh, M.U.; Kale, R.P.; Shiradkar, M.R.; Gill, C.H. SAR study of clubbed [1,2,4]-triazolyl with fluorobenzimidazoles as antimicrobial and antituberculosis agents. Eur. J. Med. Chem. 2009, 44, 2930–2935. [Google Scholar] [CrossRef] [PubMed]
- Klimešova, V.; Koči, J.; Waisser, K.; Kaustova, J. New benzimidazole derivatives as antimycobacterial agent. Farmaco 2002, 57, 259–265. [Google Scholar] [CrossRef]
- Foks, H.; Pancechowska-Ksepko, D.; Kuźmierkiewicz, W.; Zwolska, Z.; Augustynowicz-Kopeć, E.; Janowiec, M. Synthesis and tuberculostatic activity of new benzimidazole derivatives. Chem. Heterocycl. Compd. 2006, 42, 611–614. [Google Scholar] [CrossRef]
- Gobis, K.; Foks, H.; Bojanowski, K.; Augustynowicz-Kopeć, E.; Napiórkowska, A. Synthesis of novel 3-cyclohexylpropanoic acid-derived nitrogen heterocyclic compounds and their evaluation for tuberculostatic activity. Bioorg. Med. Chem. 2012, 20, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Gobis, K.; Foks, H.; Serocki, M.; Augustynowicz-Kopeć, E.; Napiórkowska, A. Synthesis and evaluation of in vitro antimycobacterial activity of novel 1H-benzo[d]imidazole derivatives and analogues. Eur. J. Med. Chem. 2015, 89, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Gobis, K.; Foks, H.; Suchan, K.; Augustynowicz-Kopeć, E.; Napiórkowska, A.; Bojanowski, K. Novel 2-(2-phenalkyl)-1H-benzo[d]imidazoles as antitubercular agents. Synthesis, biological evaluation and structure–activity relationship. Bioorg. Med. Chem. 2015, 23, 2112–2120. [Google Scholar] [CrossRef] [PubMed]
- Koci, J.; Klimešova, V.; Waisser, K.; Kaustova, J.; Dahsec, H.M.; Mollmann, U. Heterocyclic benzazole derivatives with antimycobacterial in vitro activity. Bioorg. Med. Chem. Lett. 2002, 12, 3275–3278. [Google Scholar] [CrossRef]
- Klimešova, V.; Koci, J.; Waisser, K.; Kaustova, J.; Mollmann, U. Preparation and in vitro evaluation of benzylsulfanyl benzoxazole derivatives as potential antituberculosis agents. Eur. J. Med. Chem. 2009, 44, 2286–2293. [Google Scholar] [CrossRef]
- Zahajska, L.; Klimešova, V.; Koci, J.; Waisser, K.; Kaustova, J. Synthesis and antimycobacterial activity of pyridylmethylsulfanyl and naphthylmethylsulfanyl derivatives of benzazoles, 1, 2, 4-triazole, and pyridine-2-carbothioamide/-2-carbonitrile. Arch. Pharm. Pharm. Med. Chem. 2004, 337, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Arisoy, M.; Temiz-Arpaci, O.; Kaynak-Onurdag, F.; Ozgen, S. Antimycobacterial properties of some 2,5-disubstituted-benzoxazole derivatives. J. Fac. Pharm. Ankara 2010, 39, 155–162. [Google Scholar]
- Pichota, A.; Duraiswamy, J.; Yin, Z.; Keller, T.H.; Alam, J.; Liung, S.; Lee, G.; Ding, M.; Wang, G.; Chan, W.L.; et al. Peptide deformylase inhibitors of Mycobacterium tuberculosis: Synthesis, structural investigations, and biological results. Bioorg. Med. Chem. Lett. 2008, 18, 6568–6572. [Google Scholar] [CrossRef] [PubMed]
- Telvekar, V.N.; Bairwa, V.K.; Satardekar, K.; Bellubi, A. Novel N′-benzylidene benzofuran-3-carbohydrazide derivatives as antitubercular and antifungal agents. Bioorg. Med. Chem. Lett. 2012, 22, 2343–2346. [Google Scholar] [CrossRef] [PubMed]
- Sidoova, E.; Odlerova, Z. 2-Alkylthio-6-(3-nitrobenzoylamino)benzothiazoles and their antimycobacterial activity. Chem. Pap. 1986, 40, 801–806. [Google Scholar]
- Cho, Y.; Ioerger, T.R.; Sacchettini, J.C. Discovery of novel nitrobenzothiazole inhibitors for Mycobacterium tuberculosis ATP phosphoribosyl transferase (HisG) through virtual screening. J. Med. Chem. 2008, 51, 5984–5992. [Google Scholar] [CrossRef] [PubMed]
- Patel, R.V.; Patel, P.K.; Kumari, P.; Rajani, D.P.; Chikhalia, K.H. Synthesis of benzimidazolyl-1,3,4-oxadiazol-2ylthio-N-phenyl (benzothiazolyl) acetamides as antibacterial, antifungal and antituberculosis agents. Eur. J. Med. Chem. 2012, 53, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Kaul, S.; Kumar, A.; Sain, B.; Bhatnagar, A.K. Simple an convenient one-pot synthesis of benzimidazoles and benzoxazoles using N,N-dimethylchlorosulfitemethaniminium chloride as condensing agent. Synth. Commun. 2007, 37, 2457–2460. [Google Scholar] [CrossRef]
- Sharghi, H.; Asemani, O. Methanesulfonic acid/SiO2 as an efficient combination for the synthesis of 2-substituted aromatic and aliphatic benzothiazoles from carboxylic acids. Synth. Commun. 2009, 39, 860–867. [Google Scholar] [CrossRef]
- Jerchel, D.; Fischer, H.; Kracht, M. Zur darstellung der benzimidazole. Justus Liebigs Annalen der Chemie 1952, 575, 162–173. [Google Scholar] [CrossRef]
- Martinez-Palou, R.; Zepeda, L.G.; Hoepfl, H.; Montoya, A.; Guzman-Lucero, D.J.; Guzman, J. Parallel and automated library synthesis of 2-long alkyl chain benzoazoles and azole[4,5-b]pyridines under microwave irradiation. Mol. Divers 2005, 9, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Yalcin, I.; Oren, I.; Sener, E.; Akin, A.; Ucarturk, N. The synthesis and the structure-activity relationships of some substituted benzoxazoles, oxazolo(4,5-b)pyridines, benzothiazoles and benzimidazoles as antimicrobial agents. Eur. J. Med. Chem. 1992, 27, 401–406. [Google Scholar] [CrossRef]
- Di, A.L.; Kerns, E.H. Profiling drug-like properties in discovery research. Curr. Opin. Chem. Biol. 2003, 7, 402–408. [Google Scholar] [CrossRef]
- Di, L.; Kerns, E.H. Lipophilicity. In Drug-Like Properties: Concepts, Structure Design and Methods from ADME to Toxicity Optimization, 2nd ed.; Academic Press: Boston, MA, USA, 2016; Volume 5, pp. 43–47. [Google Scholar]
- Oyaga, S.C.; Valdes, J.C.; Paez, S.B.; Marquez, K.H. DFT description of intermolecular forces between 9-aminoacridines and DNA base pairs. J. Theor. Comput. Chem. 2013, 2013. [Google Scholar] [CrossRef]
- Youmans, G.P. Test tube evaluation of tuberculostatic agents. Am. Rev. Tuberc. 1947, 56, 376. [Google Scholar] [PubMed]
- Youmans, G.P.; Youmans, A.S. A method for the determination of the rate of growth of Tubercle bacilli by the use of small inocula. J. Bactriol. 1949, 58, 247–255. [Google Scholar]
- Atlas, R.M.; Snyder, J.W. Handbook of Media for Clinical Microbiology, 2nd ed.; CRC Press: Boca Raton, FL, USA, 1995. [Google Scholar]
- Foks, H.; Buraczewska, M.; Manowska, W.; Sawlewicz, J. Investigation on pyrazine derivatives. Part 1. Synthesis and tuberculostatic action of pyrazinamidrazone 6-chloropyrazinamidrazone and their condensation products with aldehydes and ketones. Diss. Pharm. Pharmacol. 1971, 23, 49–58. [Google Scholar]
- Dykstra, C.; Frenking, G.; Kim, K.; Scuseria, G. Advances in electronic structure theory: GAMESS a decade later. In Theory and Applications of Computational Chemistry: The First Forty Years, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2005; pp. 1167–1189. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |


| Compd. | MIC a [µg/mL (µM)] | Compd. | MIC a [µg/mL (µM)] | ||
|---|---|---|---|---|---|
| M. tuberculosisc | M. tuberculosisc | ||||
| H37Rv | Spec. 210 | H37Rv | Spec. 210 | ||
| 1 | 3.1 (13) | 3.1 (13) | 13 | 100 (361) | 100 (361) |
| 2 | 6.25 (25) | 6.25 (25) | 14 | 100 (358) | 100 (358) |
| 3 | 1.5 (5) | 1.5 (5) | 15 | 100 (341) | 100 (341) |
| 4 | 3.1 (11) | 3.1 (11) | 16 | 100 (306) | 100 (306) |
| 5 | 25 (96) | 12.5 (48) | 17 | 12.5 (56) | 12.5 (56) |
| 6 | 50 (190) | 25 (95) | 18 | 12.5 (43) | 25 (86) |
| 7 | 25 (89) | 12.5 (44) | 19 | 25 (81) | 25 (81) |
| 8 | >100 (>409) | >100 (>409) | 20 | 12.5 (33) | 12.5 (33) |
| 9 | 50 (225) | 50 (225) | INHb | 0.125 (0.91) | 12.5 (91) |
| 10 | 25 (91) | 25 (91) | PZA | 25 (203) | >400 (3249) |
| 11 | 50 (194) | 50 (194) | RMP | 1.2 (1.4) | 2.5 (2.8) |
| 12 | 50 (190) | 50 (190) | |||
| Compd. | MIC (µg/mL) | µ (Debye) | log P |
|---|---|---|---|
| GK-151B | 0.75 | 3.28 | 4.95 |
| 4 | 3.1 | 3.57 | 5.95 |
| 6 | 25-50 | 4.92 | 5.13 |
| 11 | 50 | 0.93 | 5.02 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krause, M.; Foks, H.; Augustynowicz-Kopeć, E.; Napiórkowska, A.; Szczesio, M.; Gobis, K. Synthesis and Tuberculostatic Activity Evaluation of Novel Benzazoles with Alkyl, Cycloalkyl or Pyridine Moiety. Molecules 2018, 23, 985. https://doi.org/10.3390/molecules23040985
Krause M, Foks H, Augustynowicz-Kopeć E, Napiórkowska A, Szczesio M, Gobis K. Synthesis and Tuberculostatic Activity Evaluation of Novel Benzazoles with Alkyl, Cycloalkyl or Pyridine Moiety. Molecules. 2018; 23(4):985. https://doi.org/10.3390/molecules23040985
Chicago/Turabian StyleKrause, Malwina, Henryk Foks, Ewa Augustynowicz-Kopeć, Agnieszka Napiórkowska, Małgorzata Szczesio, and Katarzyna Gobis. 2018. "Synthesis and Tuberculostatic Activity Evaluation of Novel Benzazoles with Alkyl, Cycloalkyl or Pyridine Moiety" Molecules 23, no. 4: 985. https://doi.org/10.3390/molecules23040985
APA StyleKrause, M., Foks, H., Augustynowicz-Kopeć, E., Napiórkowska, A., Szczesio, M., & Gobis, K. (2018). Synthesis and Tuberculostatic Activity Evaluation of Novel Benzazoles with Alkyl, Cycloalkyl or Pyridine Moiety. Molecules, 23(4), 985. https://doi.org/10.3390/molecules23040985






