Biflavonoids Isolated from Selaginella tamariscina and Their Anti-Inflammatory Activities via ERK 1/2 Signaling
Abstract
:1. Introduction
2. Results
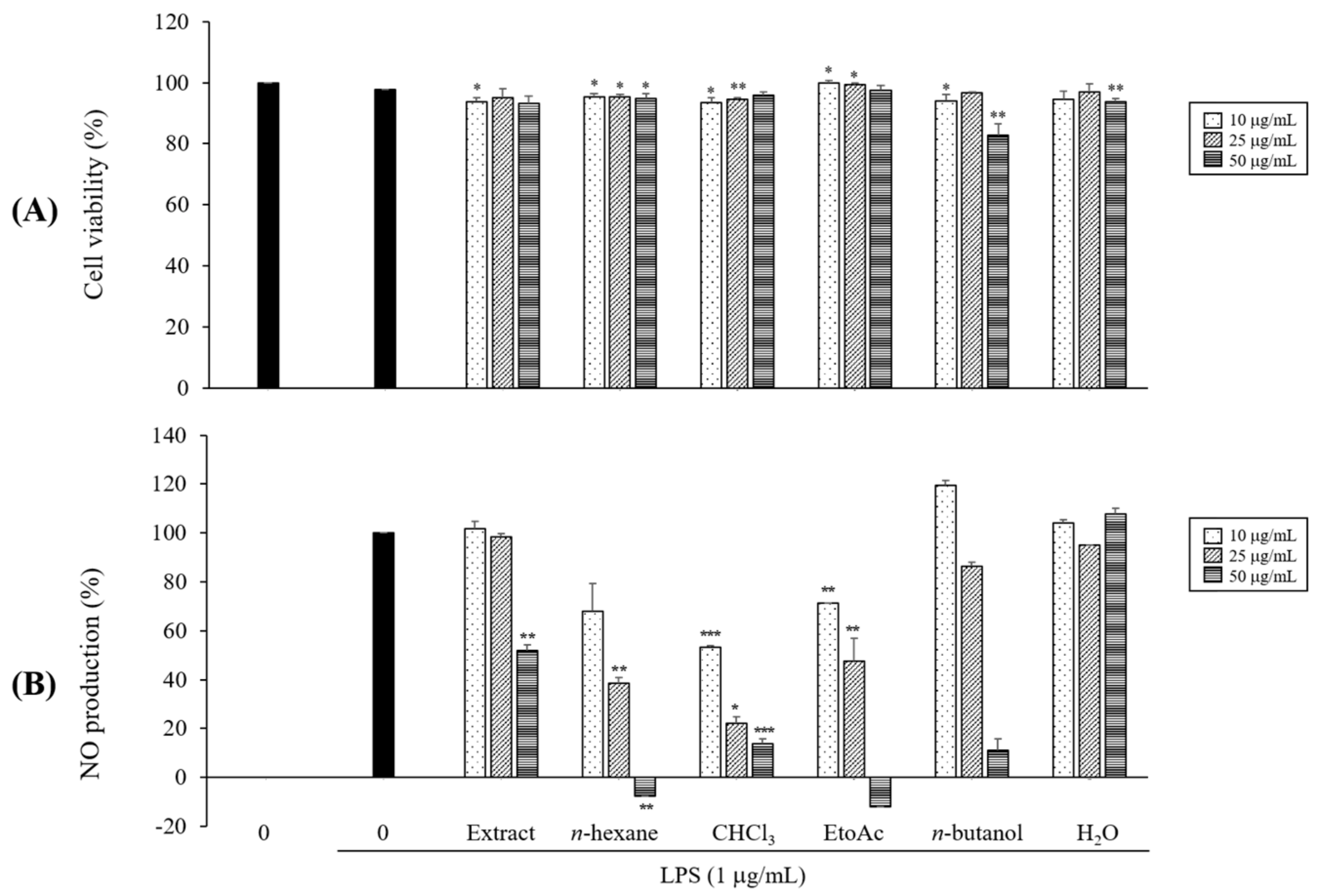
2.1. Preliminary Determination of the Effects on NO Production
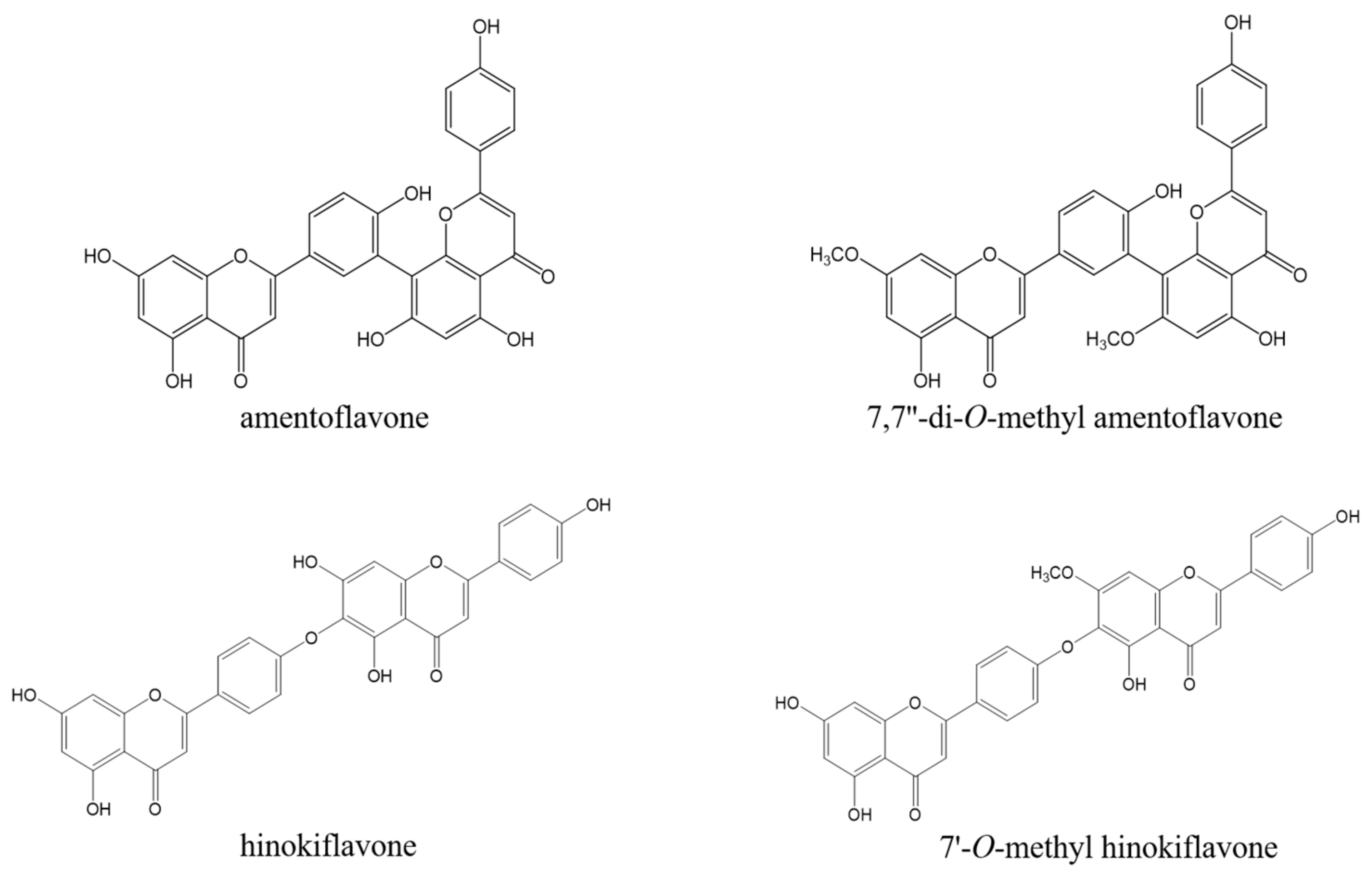
2.2. Isolation and Structural Determination of Biflavonoids from the EtOAc Fraction
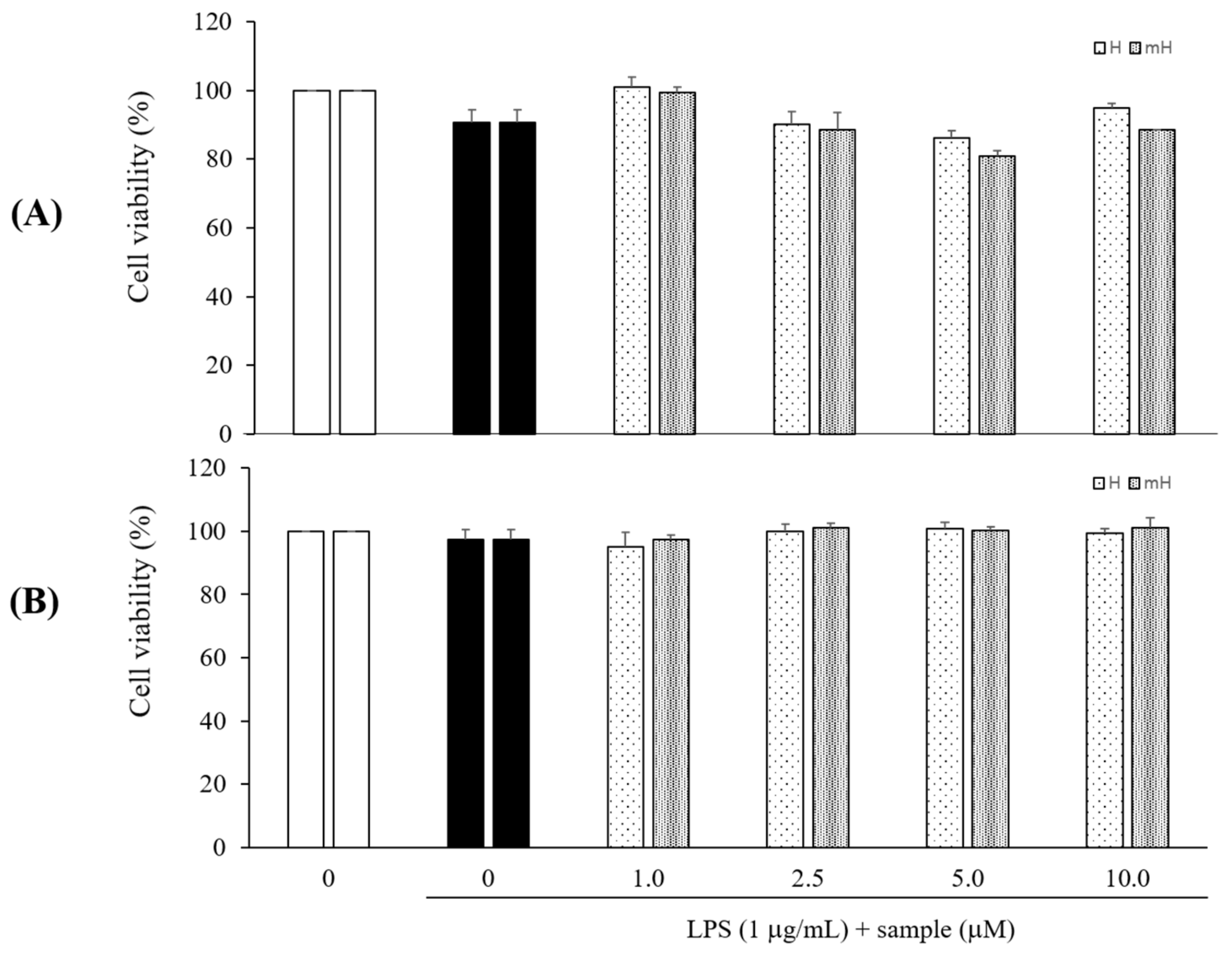
2.3. Effects on Cytotoxicity
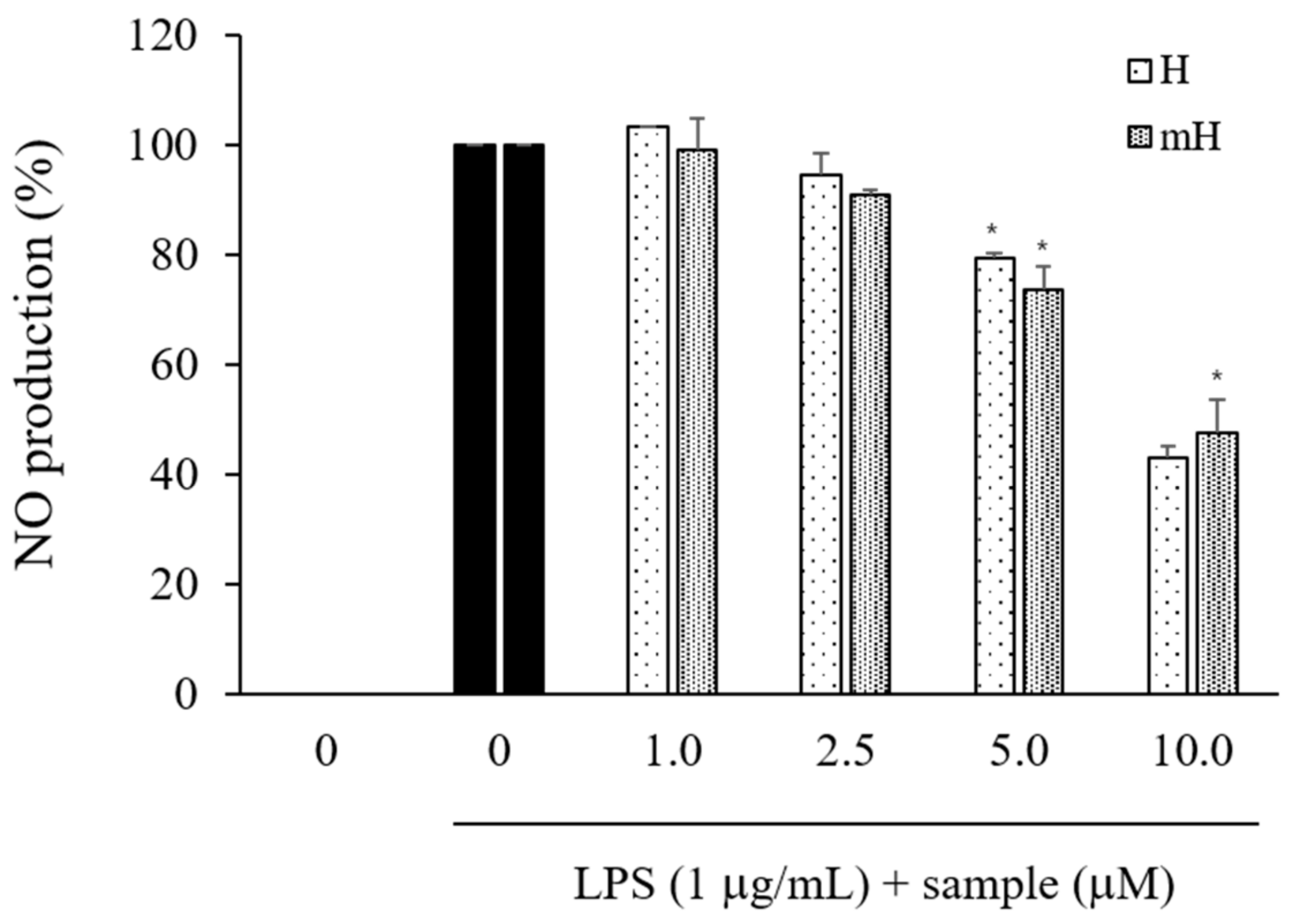
2.4. Effects on NO Production
2.5. Effects on iNOS and COX-2 Expression
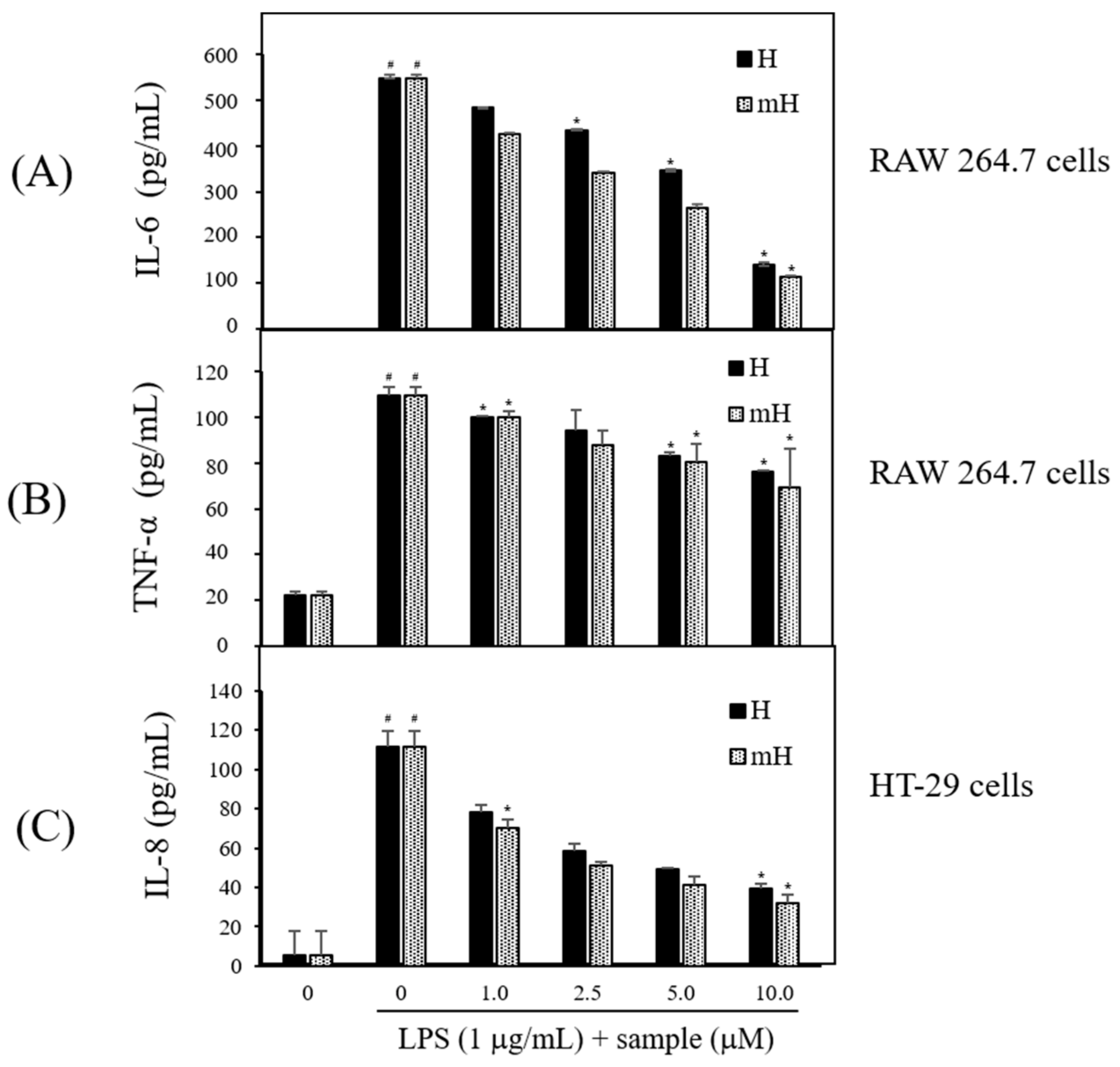
2.6. Effects on Proinflammatory Cytokines Production
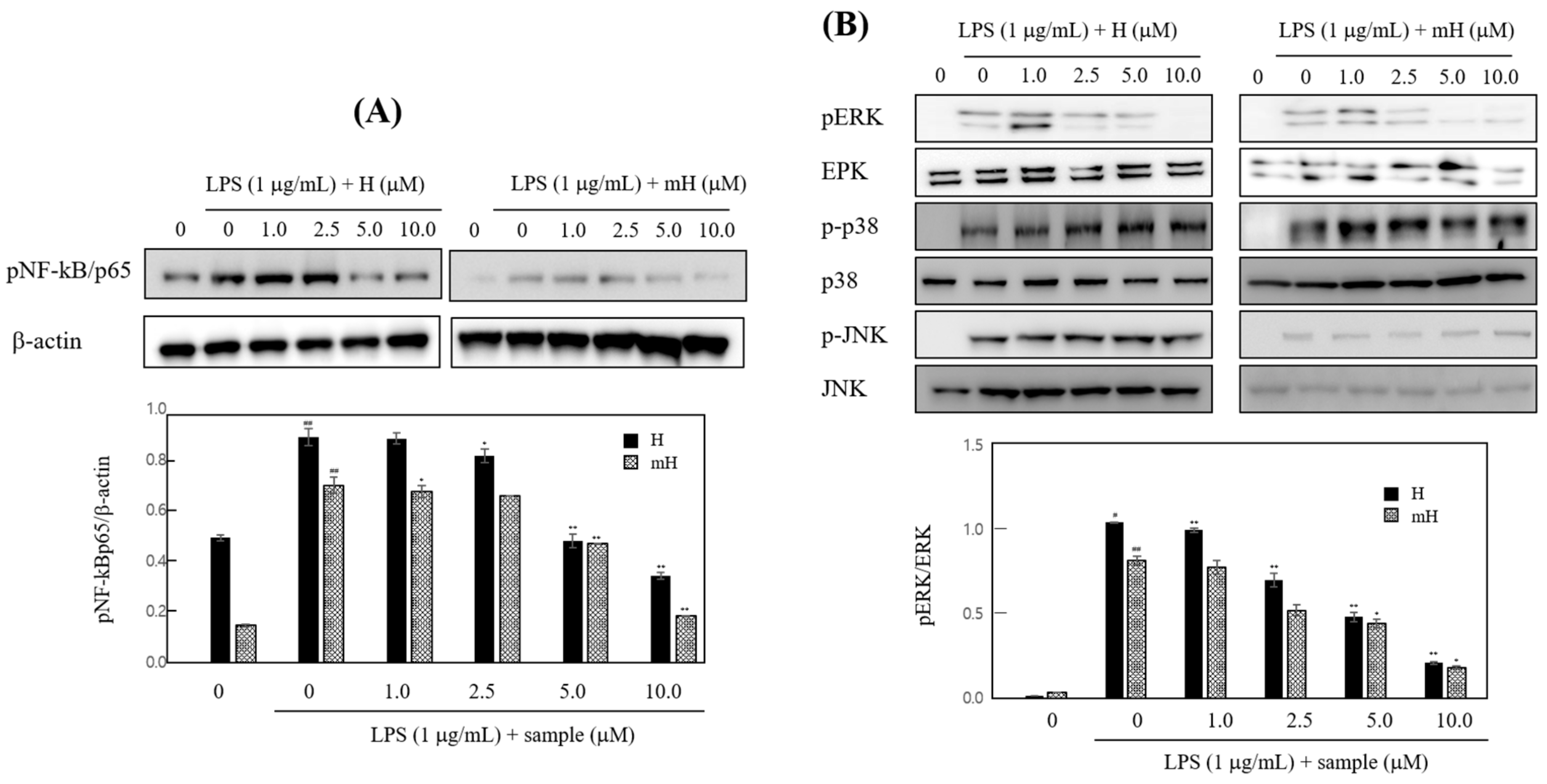
2.7. Effects on NF-κB Expression
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Extraction and Isolation
4.3. Cell Culture and Cytotoxicity Determination
4.4. Measurement of NO Production
4.5. Western Blot Analysis
4.6. Measurement of Cytokine Production
4.7. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ahmed, A.U. An overview of inflammation: Mechanism and consequences. Front Biol. 2011, 6, 274–281. [Google Scholar]
- Guha, M.; Mackman, N. LPS induction of gene expression in human monocytes. Cell Signal. 2001, 13, 85–94. [Google Scholar] [CrossRef]
- Laroui, H.; Ingersoll, S.A.; Liu, H.C.; Baker, M.T.; Ayyadurai, S.; Charania, M.A.; Laroui, F.; Yan, Y.; Sitaraman, S.V.; Merlin, D. Dextran sodium sulfate (DSS) induces colitis in mice by forming nano-lipocomplexes with medium-chain-length fatty acids in the colon. PLoS ONE 2012, 7, e32084. [Google Scholar] [CrossRef] [PubMed]
- Marks-Konczalik, J.; Chu, S.C.; Moss, J. Cytokine-mediated transcriptional induction of the human inducible nitric oxide synthase gene requires both activator protein 1 and nuclear factor kappaB-binding sites. J. Biol. Chem. 1998, 273, 22201–22208. [Google Scholar] [CrossRef] [PubMed]
- Vane, J.R.; Mitchell, J.A.; Appleton, I.; Tomlinson, A.; Bishop-Bailey, D.; Croxtall, J.; Willoughby, D.A. Inducible isoforms of cyclooxygenase and nitric-oxide synthase in inflammation. Proc. Natl. Acad. Sci. USA 1994, 91, 2046–2050. [Google Scholar] [CrossRef] [PubMed]
- Joung, E.J.; Lee, B.; Gwon, W.G.; Shin, T.; Jung, B.M.; Yoon, N.Y.; Choi, J.S.; Oh, C.W.; Kim, H.R. Sargaquinoic acid attenuates inflammatory responses by regulating NF-κB and Nrf2 pathways in lipopolysaccharide-stimulated RAW 264.7 cells. Int. Immunopharmacol. 2015, 29, 693–700. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.F.; LeBel, C.P.; Bondy, S.C. Reactive oxygen species formation as a biomarker of methylmercury and trimethyltin neurotoxicity. Neurotoxicology 1992, 13, 637–648. [Google Scholar] [PubMed]
- Chen, Z.; Hagler, J.; Palombella, V.J.; Melandri, F.; Scherer, D.; Ballard, D.; Maniatis, T. Signal-induced site-specific phosphorylation targets I kappa B alpha to the ubiquitin-proteasome pathway. Genes Dev. 1995, 9, 1586–1597. [Google Scholar] [CrossRef] [PubMed]
- Henkel, T.; Machleidt, T.; Alkalay, I.; Krönke, M.; Ben-Neriah, Y.; Baeuerle, P.A. Rapid proteolysis of I kappa B-alpha is necessary for activation of transcription factor NF-kappa B. Nature 1993, 365, 182–185. [Google Scholar] [CrossRef] [PubMed]
- Abarikwu, S.O. Kolaviron, a natural flavonoid from the seeds of Garcinia kola, reduces LPS-induced inflammation in macrophages by combined inhibition of IL-6 secretion, and inflammatory transcription factors, ERK1/2, NF-κB, p38, Akt, p-c-JUN and JNK. Biochim. Biophys. Acta 2014, 1840, 2373–2381. [Google Scholar] [CrossRef] [PubMed]
- Turner, M.D.; Nedjai, B.; Hurst, T.; Pennington, D.J. Cytokines and chemokines: At the crossroads of cell signalling and inflammatory disease. Biochim. Biophys. Acta 2014, 1843, 2563–2582. [Google Scholar] [CrossRef] [PubMed]
- Medzhitov, R.; Horng, T. Transcriptional control of the inflammatory response. Nat. Rev. Immunol. 2009, 9, 692–703. [Google Scholar] [CrossRef] [PubMed]
- Kuo, Y.C.; Sun, C.M.; Tsai, W.J.; Ou, J.C.; Chen, W.P.; Lin, C.Y. Chinese herbs as modulators of human mesangial cell proliferation: Preliminary studies. J. Lab. Clin. Med. 1998, 132, 76–85. [Google Scholar] [CrossRef]
- Lee, I.S.; Nishikawa, A.; Furukawa, F.; Kasahara, K.; Kim, S.U. Effects of Selaginella tamariscina on in vitro tumor cell growth, p53 expression, G1 arrest and in vivo gastric cell proliferation. Cancer Lett. 1999, 144, 93–99. [Google Scholar] [CrossRef]
- Dai, Y.; But, P.P.; Chu, L.M.; Chan, Y.P. Inhibitory effects of Selaginella tamariscina on immediate allergic reactions. Am. J. Chin. Med. 2005, 33, 957–966. [Google Scholar] [CrossRef] [PubMed]
- Zha, X.; Xu, Z.; Liu, Y.; Xu, L.; Huang, H.; Zhang, J.; Cui, L.; Zhou, C.; Xu, D. Amentoflavone enhances osteogenesis of human mesenchymal stem cells through JNK and p38 MAPK pathways. J. Nat. Med. 2016, 70, 634–644. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.J.; Park, K.; Lee, I.S.; Kim, H.S.; Yeo, S.H.; Woo, E.R.; Lee, D.G. S-phase accumulation of Candida albicans by anticandidal effect of amentoflavone isolated from Selaginella tamariscina. Biol. Pharm. Bull. 2007, 30, 1969–1971. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kim, H.; Kang, J.W.; Kim, J.H.; Lee, D.H.; Kim, M.S.; Yang, Y.; Woo, E.R.; Kim, Y.M.; Hong, J.; et al. The biflavonoid amentoflavone induces apoptosis via suppressing E7 expression, cell cycle arrest at sub-G1 phase, and mitochondria-emanated intrinsic pathways in human cervical cancer cells. J. Med. Food 2011, 14, 808–816. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.H.; Zhao, B.T.; Ali, M.Y.; Choi, J.S.; Rhyu, D.Y.; Min, B.S.; Woo, M.H. Insulin-mimetic selaginellins from Selaginella tamariscina with protein tyrosine phosphatase 1B (PTP1B) inhibitory activity. J. Nat. Prod. 2015, 78, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Woo, E.R.; Lee, J.Y.; Cho, I.J.; Kim, S.G.; Kang, K.W. Amentoflavone inhibits the induction of nitric oxide synthase by inhibiting NF-kappaB activation in macrophages. Pharmacol. Res. 2005, 51, 539–546. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.W.; Pokharel, Y.R.; Kim, M.R.; Woo, E.R.; Choi, H.K.; Kang, K.W. Inhibition of inducible nitric oxide synthase by sumaflavone isolated from Selaginella tamariscina. J. Ethnopharmacol. 2006, 105, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.Y.; Tao, W.W.; Duan, J.A. Antithrombotic flavonoids from the faeces of Trogopterus xanthipes. Nat. Prod. Res. 2010, 24, 1843–1849. [Google Scholar] [CrossRef] [PubMed]
- Krauze-Baranowska, M.; Pobłocka, L.; El-Hela, A.A. Biflavones from Chamaecyparis obtusa. Z. Naturforsch. C 2005, 60, 679–685. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Shi, S.; Wang, Y.; Huang, K. Target-guided isolation and purification of antioxidants from Selaginella sinensis by offline coupling of DPPH-HPLC and HSCCC experiments. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2011, 879, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Son, K.H.; Chang, H.W.; Kang, S.S.; Kim, H.P. Inhibition of arachidonate release from rat peritoneal macrophage by biflavonoids. Arch. Pharm. Res. 1997, 20, 533–538. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Oh, W.K.; Kim, B.Y.; Ahn, S.C.; Kang, D.O.; Shin, D.I.; Kim, J.; Mheen, T.I.; Ahn, J.S. Inhibition of phospholipase C gamma 1 activity by amentoflavone isolated from Selaginella tamariscina. Planta Med. 1996, 62, 293–296. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.; Rho, H.S.; Yang, Y.; Yoon, J.Y.; Lee, J.; Hong, Y.D.; Kim, H.C.; Choi, S.S.; Kim, T.W.; Shin, S.S.; et al. Extracellular signal-regulated kinase is a direct target of the anti-inflammatory compound amentoflavone derived from Torreya nucifera. Mediat. Inflamm. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Vezza, T.; Rodríguez-Nogales, A.; Algieri, F.; Utrilla, M.P.; Rodriguez-Cabezas, M.E.; Galvez, J. Flavonoids in Inflammatory Bowel Disease: A Review. Nutrients 2016, 8, 211. [Google Scholar] [CrossRef] [PubMed]
- Johnson, G.L.; Lapadat, R. Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science 2002, 298, 1911–1912. [Google Scholar] [CrossRef] [PubMed]
- Schottelius, A.; Baldwin, A., Jr. A role for transcription factor NF-kappa B in intestinal inflammation. Int. J. Colorectal Dis. 1999, 14, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Atreya, I.; Atreya, R.; Neurath, M. NF-kappaB in inflammatory bowel disease. J. Intern. Med. 2008, 263, 591–596. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds used in this research are available from the authors. |

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shim, S.-Y.; Lee, S.-g.; Lee, M. Biflavonoids Isolated from Selaginella tamariscina and Their Anti-Inflammatory Activities via ERK 1/2 Signaling. Molecules 2018, 23, 926. https://doi.org/10.3390/molecules23040926
Shim S-Y, Lee S-g, Lee M. Biflavonoids Isolated from Selaginella tamariscina and Their Anti-Inflammatory Activities via ERK 1/2 Signaling. Molecules. 2018; 23(4):926. https://doi.org/10.3390/molecules23040926
Chicago/Turabian StyleShim, Sun-Yup, Seul-gi Lee, and Mina Lee. 2018. "Biflavonoids Isolated from Selaginella tamariscina and Their Anti-Inflammatory Activities via ERK 1/2 Signaling" Molecules 23, no. 4: 926. https://doi.org/10.3390/molecules23040926




