Oxadiazole-Based Highly Efficient Bipolar Fluorescent Emitters for Organic Light-Emitting Diodes
Abstract
:1. Introduction
2. Results and Discussion
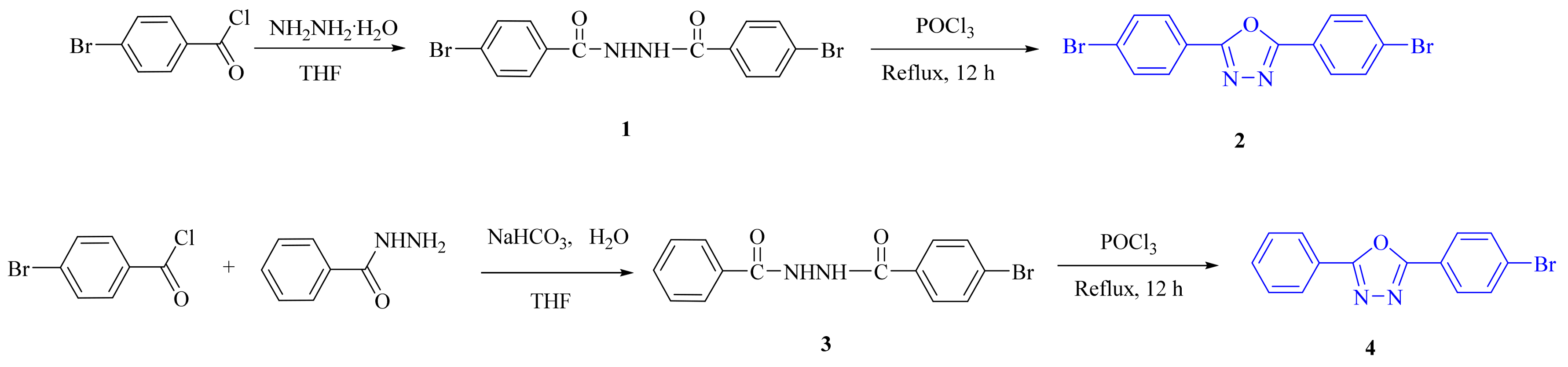
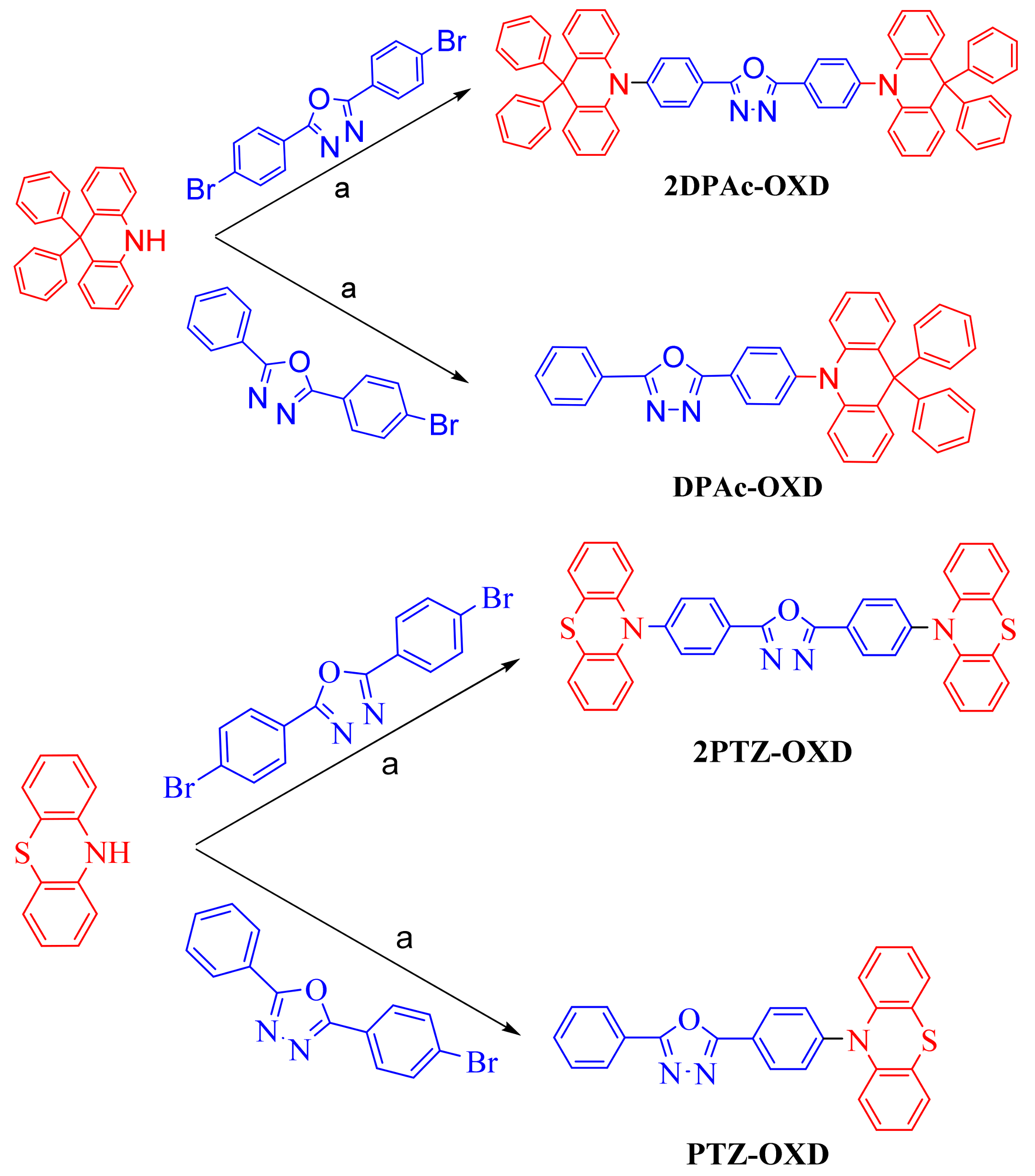
2.1. Synthesis
2.2. Thermal Properties
2.3. Photophysical and Electrochemical Properties
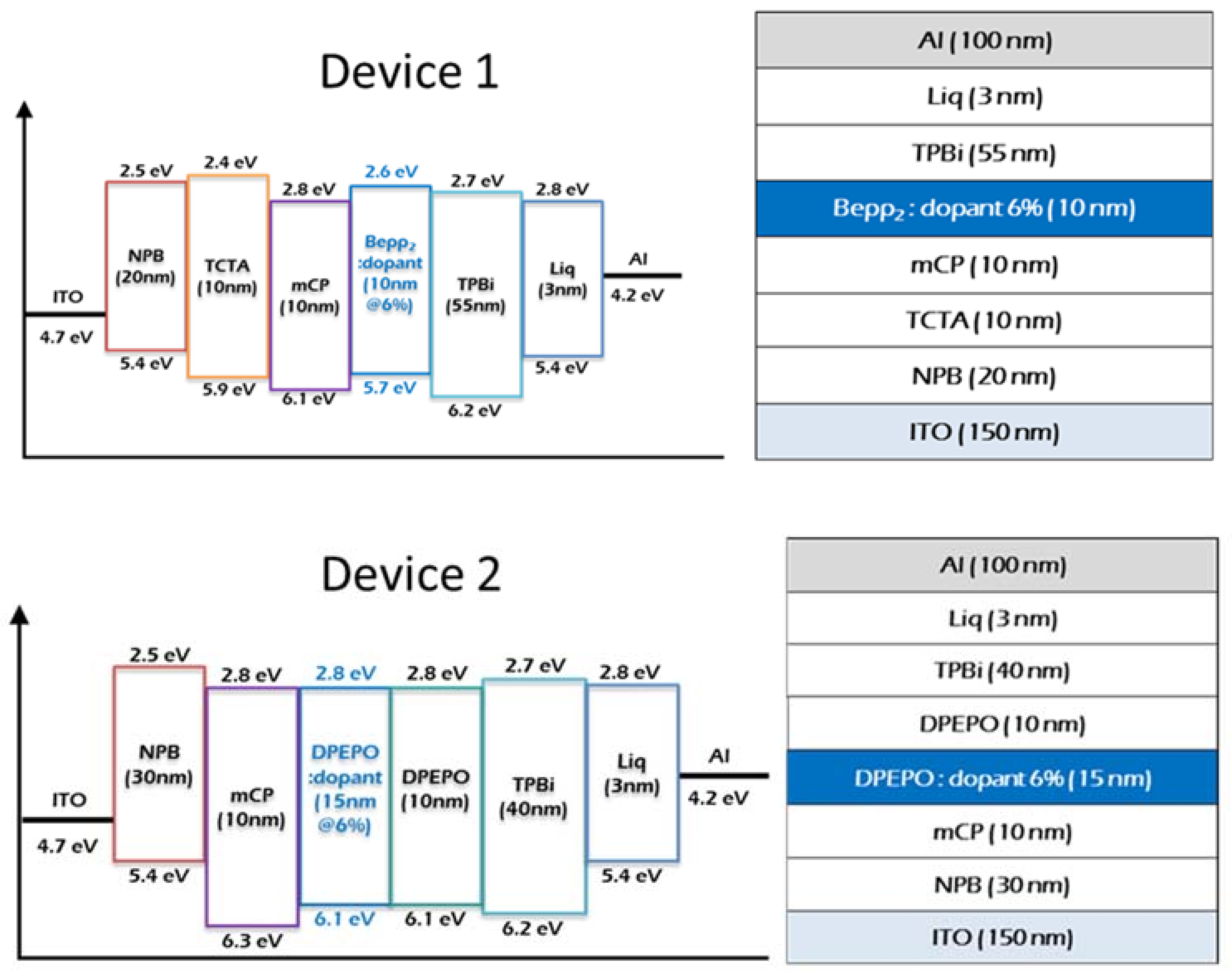
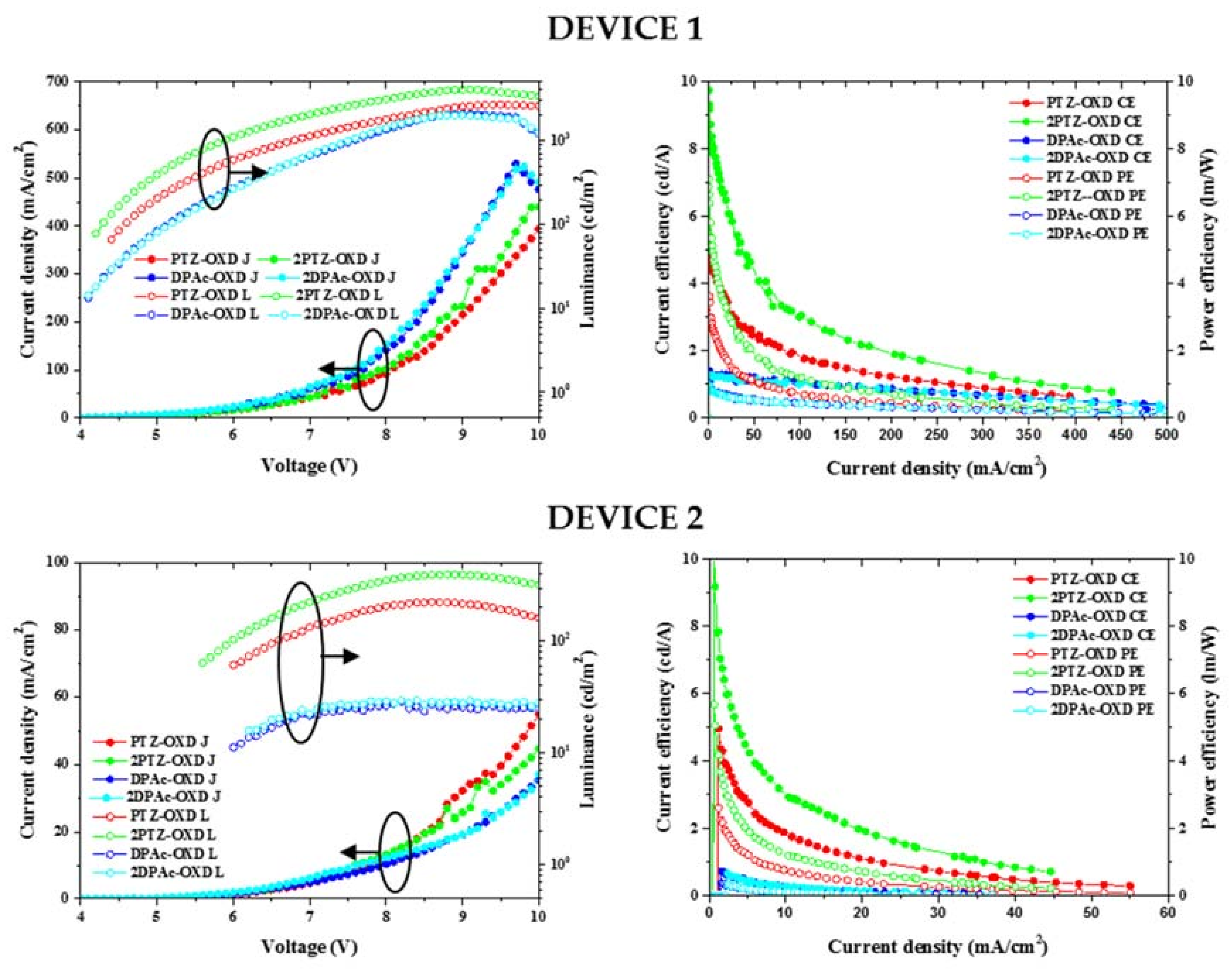
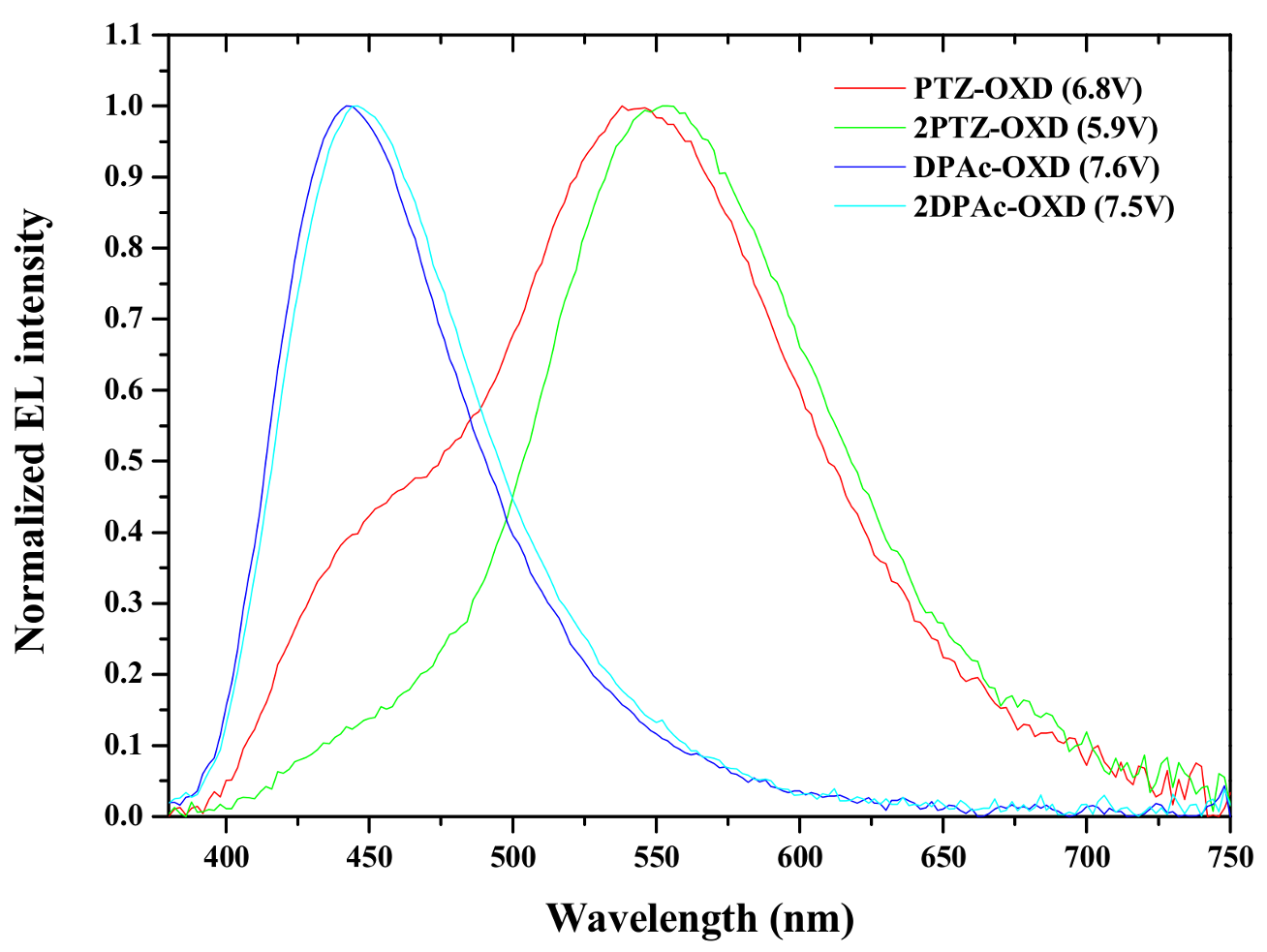
2.4. Device Characteristics
3. Materials and Methods
3.1. General Procedures
3.2. Synthetic Procedures
3.2.1. 4-Bromo-N′-(4-bromobenzoyl)benzohydrazide (1)
3.2.2. 2,5-Bis(4-bromophenyl)-1,3,4-oxadiazole (2)
3.2.3. 2-(4-Bromophenyl)-5-phenyl-1,3,4-oxadiazole (4)
3.2.4. 2,5-Bis(4-(9,9-diphenyl-9,10-dihydroacridine)phenyl)-1,3,4-oxadiazole (2DPAc-OXD)
3.2.5. 2-(4-(9,9-Diphenyl-9,10-dihydroacridine)phenyl)-1,3,4-oxadiazole (DPAc-OXD)
3.2.6. 2,5-Bis(4-(10H-phenothiazin-10-yl)-1,3,4-oxadizole (2PTZ-OXD)
3.2.7. 2-(4-(10H-phenothiazin-10-yl)-1,3,4-oxadiazole(PTZ-OXD)
3.3. OLED Fabrication and Characterization
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tang, C.W.; VanSlyke, S.A. Organic electroluminescent diodes. Appl. Phys. Lett. 1987, 51, 913–915. [Google Scholar] [CrossRef]
- D’Andrade, B.W.; Forrest, S.R. White organic light-emitting devices for solid-state lighting. Adv. Mater. 2004, 16, 1585–1595. [Google Scholar] [CrossRef]
- Reddy, S.S.; Sree, V.G.; Cho, W.; Jin, S.-H. Achieving pure deep-blue electroluminescence with CIE y ≤ 0.06 via a rational design approach for highly efficient non-doped solution-processed organic light-emitting diodes. Chem. Asian J. 2016, 11, 3275–3282. [Google Scholar] [CrossRef] [PubMed]
- Shirota, Y.; Kinoshita, M.; Noda, T.; Okumoto, K.; Ohara, T. A novel class of emitting amorphous molecular materials as bipolar radical formants: 2-{4-[bis(4-methylphenyl)amino]phenyl}-5-(dimesitylboryl)thiophene and 2-{4-[bis(9,9-dimethylfluorenyl)amino]phenyl}-5-(dimesitylboryl)thiophene. J. Am. Chem. Soc. 2000, 122, 11021–11022. [Google Scholar] [CrossRef]
- Scott, J.C.; Karg, S.; Carter, S.A. Bipolar charge and current distributions in organic light-emitting diodes. J. Appl. Phys. 1997, 82, 1454–1460. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Fujiyama, T.; Tanaka, H.; Yokoyama, M. Material design of organic thin films for bipolar charge transport. Chem. Mater. 1990, 2, 341–342. [Google Scholar] [CrossRef]
- Woo, S.-J.; Kim, Y.; Kim, M.-J.; Baek, J.Y.; Kwon, S.K.; Kim, Y.H.; Kim, J.J. Strategies for the molecular design of donor–acceptor-type fluorescent emitters for efficient deep blue organic light emitting diodes. Chem. Mater. 2018, 30, 857–863. [Google Scholar] [CrossRef]
- Reddy, S.S.; Sree, V.G.; Gunasekar, K.; Cho, W.; Gal, Y.S.; Song, M.; Kang, J.W.; Jin, S.H. Highly efficient bipolar deep-blue fluorescent emitters for solution-processed non-doped organic light-emitting diodes based on 9,9-dimethyl-9,10-dihydroacridine/phenanthroimadazole derivatives. Adv. Opt. Mater. 2016, 4, 1236–1246. [Google Scholar] [CrossRef]
- Thirion, D.; Rault-Berthelot, J.; Vignau, L.; Poriel, C. Synthesis and properties of a blue bipolar indenofluorene emitter based on a D-π-A design. Org. Lett. 2011, 13, 4418–4421. [Google Scholar] [CrossRef] [PubMed]
- Abdurahman, A.; Obolda, A.; Peng, Q.; Li, F. Efficient deep blue fluorescent oleds with ultra-low efficiency roll-off based on 4h-1,2,4-triazole cored D-A and D-A-D type emitters. Dyes Pigm. 2018, 153, 10–17. [Google Scholar] [CrossRef]
- Liu, X.K.; Zheng, C.J.; Lo, M.F.; Xiao, J.; Chen, Z.; Liu, C.L.; Lee, C.S.; Fung, M.K.; Zhang, X.H. Novel blue fluorophor with high triplet energy level for high performance single-emitting-layer fluorescence and phosphorescence hybrid white organic light-emitting diodes. Chem. Mater. 2013, 25, 4454–4459. [Google Scholar] [CrossRef]
- Lin, S.L.; Chan, L.H.; Lee, R.H.; Yen, M.Y.; Kuo, W.J.; Chen, C.T.; Jeng, R.J. Highly efficient carbazole-π-dimesitylborane bipolar fluorophores for nondoped blue organic light-emitting diodes. Adv. Mater. 2008, 20, 3947–3952. [Google Scholar] [CrossRef]
- Fisher, A.L.; Linton, K.E.; Kamtekar, K.T.; Pearson, C.; Bryce, M.R.; Petty, M.C. Efficient deep-blue electroluminescence from an ambipolar fluorescent emitter in a single-active-layer device. Chem. Mater. 2011, 23, 1640–1642. [Google Scholar] [CrossRef]
- Jeong, S.; Kim, M.K.; Kim, S.H.; Hong, J.I. Efficient deep-blue emitters based on triphenylamine-linked benzimidazole derivatives for nondoped fluorescent organic light-emitting diodes. Org. Electron. 2013, 14, 2497–2504. [Google Scholar] [CrossRef]
- Ramaiah, D.; Thurakkal, S.; Sanju, K.S.; Soman, A.; Unni, N.; Joseph, J. Design and synthesis of solution processable green fluorescent D-π-A dyads for oled applications. New J. Chem. 2018, 42, 5456–5464. [Google Scholar]
- Zhang, G.; Auer-Berger, M.; Gehrig, D.; Blom, P.; Baumgarten, M.; Schollmeyer, D.; List-Kratochvil, E.; Müllen, K. Blue light emitting polyphenylene dendrimers with bipolar charge transport moieties. Molecules 2016, 21, 1400. [Google Scholar] [CrossRef] [PubMed]
- Uoyama, H.; Goushi, K.; Shizu, K.; Nomura, H.; Adachi, C. Highly efficient organic light-emitting diodes from delayed fluorescence. Nature 2012, 492, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Kuwabara, H.; Potscavage, W.J.; Huang, S.; Hatae, Y.; Shibata, T.; Adachi, C. Anthraquinone-based intramolecular charge-transfer compounds: Computational molecular design, thermally activated delayed fluorescence, and highly efficient red electroluminescence. J. Am. Chem. Soc. 2014, 136, 18070–18081. [Google Scholar] [CrossRef] [PubMed]
- Yook, K.S.; Lee, J.Y.; Yook, K.S. Bipolar host materials for organic light-emitting diodes. Chem. Rec. 2016, 16, 159–172. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Kulkarni, A.P.; Jenekhe, S.A. Phenoxazine-based emissive donor−acceptor materials for efficient organic light-emitting diodes. Chem. Mater. 2005, 17, 5225–5227. [Google Scholar] [CrossRef]
- Goes, M.; Verhoeven, J.W.; Hofstraat, H.; Brunner, K. Oled and pled devices employing electrogenerated, intramolecular charge-transfer fluorescence. ChemPhysChem 2003, 4, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, A.P.; Kong, X.; Jenekhe, S.A. High-performance organic light-emitting diodes based on intramolecular charge-transfer emission from donor–acceptor molecules: Significance of electron-donor strength and molecular geometry. Adv. Funct. Mater. 2006, 16, 1057–1066. [Google Scholar] [CrossRef]
- Zhong, H.; Lai, H.; Fang, Q. New conjugated triazine based molecular materials for application in optoelectronic devices: Design, synthesis, and properties. J. Phys. Chem. C 2011, 115, 2423–2427. [Google Scholar] [CrossRef]
- Linton, K.E.; Fisher, A.L.; Pearson, C.; Fox, M.A.; Palsson, L.O.; Bryce, M.R.; Petty, M.C. Colour tuning of blue electroluminescence using bipolar carbazole-oxadiazole molecules in single-active-layer organic light emitting devices (oleds). J. Mater. Chem. 2012, 22, 11816–11825. [Google Scholar] [CrossRef]
- Duan, L.; Qiao, J.; Sun, Y.; Qiu, Y. Strategies to design bipolar small molecules for oleds: Donor-acceptor structure and non-donor-acceptor structure. Adv. Mater. 2011, 23, 1137–1144. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, A.Y.; Li, B.-X.; Huang, J.; Zhao, L.; Wang, B.-Z.; Li, J.-W.; Zhu, X.-H.; Peng, J.; Cao, Y.; et al. Asymmetrically 4,7-disubstituted benzothiadiazoles as efficient non-doped solution-processable green fluorescent emitters. Org. Lett. 2009, 11, 5318–5321. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ye, J.; Chen, Z.; Fung, M.K.; Zheng, C.; Ou, X.; Zhang, X.; Yuan, Y.; Lee, C.S. Carbazole/sulfone hybrid D-π-A-structured bipolar fluorophores for high-efficiency blue-violet electroluminescence. Chem. Mater. 2013, 25, 2630–2637. [Google Scholar] [CrossRef]
- Zhang, M.; Xue, S.; Dong, W.; Wang, Q.; Fei, T.; Gu, C.; Ma, Y. Highly-efficient solution-processed oleds based on new bipolar emitters. ChemComm 2010, 46, 3923–3925. [Google Scholar] [CrossRef] [PubMed]
- Antoniadis, H.; Inbasekaran, M.; Woo, E.P. Blue-green organic light-emitting diodes based on fluorene-oxadiazole compounds. Appl. Phys. Lett. 1998, 73, 3055–3057. [Google Scholar] [CrossRef]
- Lian, M.; Yu, Y.; Zhao, J.; Huang, Z.; Yang, X.; Zhou, G.; Wu, Z.; Wang, D. Novel phosphorescent polymers containing both ambipolar segments and functionalized IrIII phosphorescent moieties: Synthesis, photophysical, redox, and electrophosphorescence investigation. J. Mater. Chem. C 2014, 2, 9523–9535. [Google Scholar] [CrossRef]
- Wróblowska, M.; Kudelko, A.; Kuźnik, N.; Łaba, K.; Łapkowski, M. Synthesis of extended 1,3,4-oxadiazole and 1,3,4-thiadiazole derivatives in the Suzuki cross-coupling reactions. J. Heterocycl. Chem. 2017, 54, 1550–1557. [Google Scholar] [CrossRef]
- Kwon, W.; Ahn, B.; Kim, D.M.; Ko, Y.-G.; Hahm, S.G.; Kim, Y.; Kim, H.; Ree, M. Morphology-dependent electrical memory characteristics of a well-defined brush polymer bearing oxadiazole-based mesogens. J. Phys. Chem. C 2011, 115, 19355–19363. [Google Scholar] [CrossRef]
- Mi, B.; Gao, Z.; Liao, Z.; Huang, W.; Chen, C.H. Molecular hosts for triplet emitters in organic light-emitting diodes and the corresponding working principle. Sci. China Chem. 2010, 53, 1679–1694. [Google Scholar] [CrossRef]
- Zhao, F.; Zhang, Z.; Liu, Y.; Dai, Y.; Chen, J.; Ma, D. A hybrid white organic light-emitting diode with stable color and reduced efficiency roll-off by using a bipolar charge carrier switch. Org. Electron. 2012, 13, 1049–1055. [Google Scholar] [CrossRef]
- Zhang, J.; Ding, D.; Wei, Y.; Xu, H. Extremely condensing triplet states of DPEPO-type hosts through constitutional isomerization for high-efficiency deep-blue thermally activated delayed fluorescence diodes. Chem. Sci. 2016, 7, 2870–2882. [Google Scholar] [CrossRef]
- Braveenth, R.; Bae, I.-J.; Han, J.-H.; Qiong, W.; Seon, G.; Raagulan, K.; Yang, K.; Park, Y.H.; Kim, M.; Chai, K.Y. Utilizing a Spiro Core with Acridine- and Phenothiazine-Based New Hole Transporting Materials for Highly Efficient Green Phosphorescent Organic Light-Emitting Diodes. Molecules 2018, 23, 713. [Google Scholar] [CrossRef] [PubMed]
- Song, D.; Zhao, S.; Luo, Y.; Aziz, H. Causes of efficiency roll-off in phosphorescent organic light emitting devices: Triplet-triplet annihilation versus triplet-polaron quenching. Appl. Phys. Lett. 2010, 97, 268. [Google Scholar] [CrossRef]
- Zheng, T.; Choy, W.C.; Ho, C.L.; Wong, W.Y. Improving efficiency roll-off in organic light emitting devices with a fluorescence-interlayer-phosphorescence emission architecture. Appl. Phys. Lett. 2009, 95, 264. [Google Scholar] [CrossRef] [Green Version]
Sample Availability: Samples of the compounds are not available from the authors. |
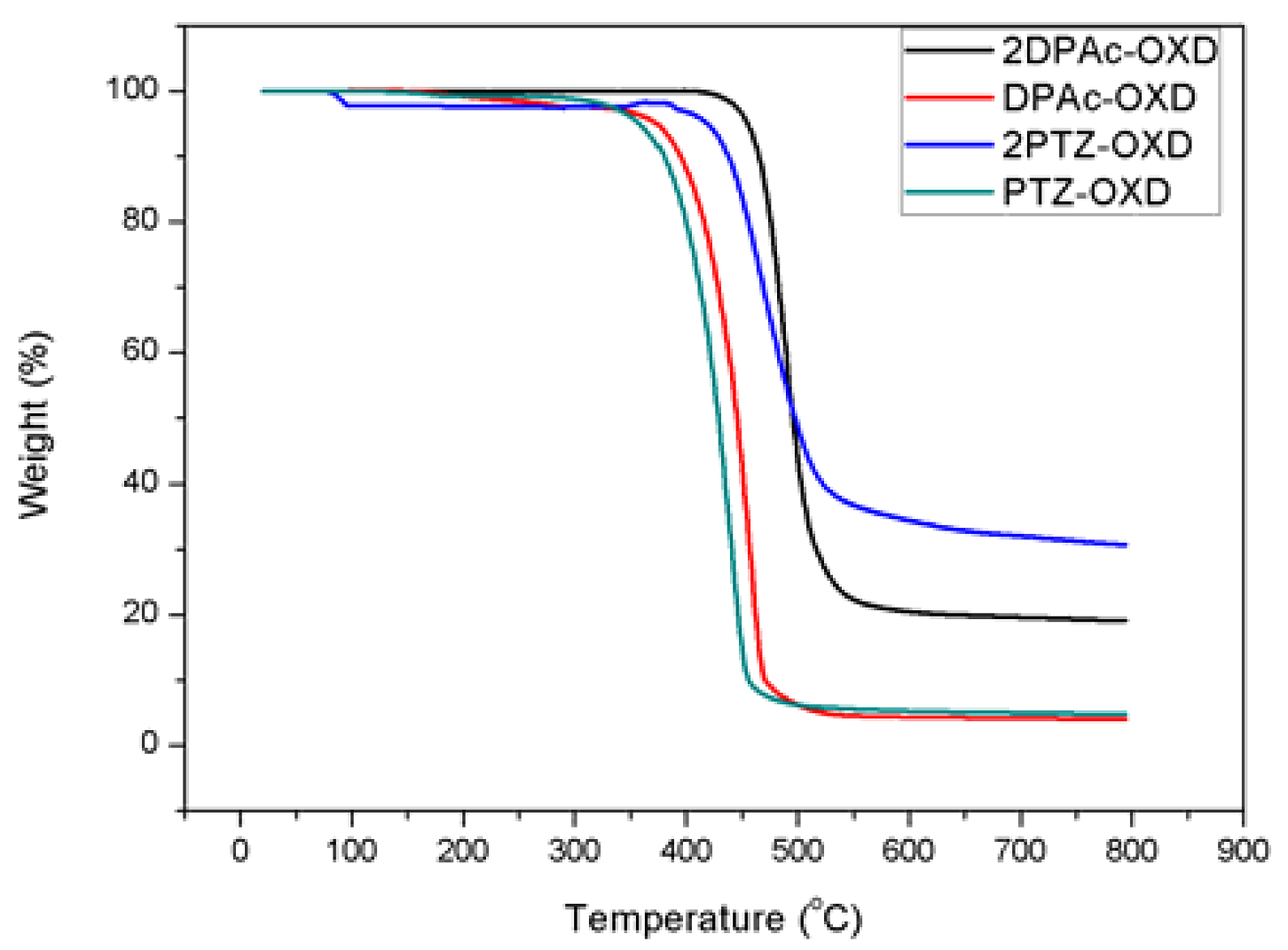
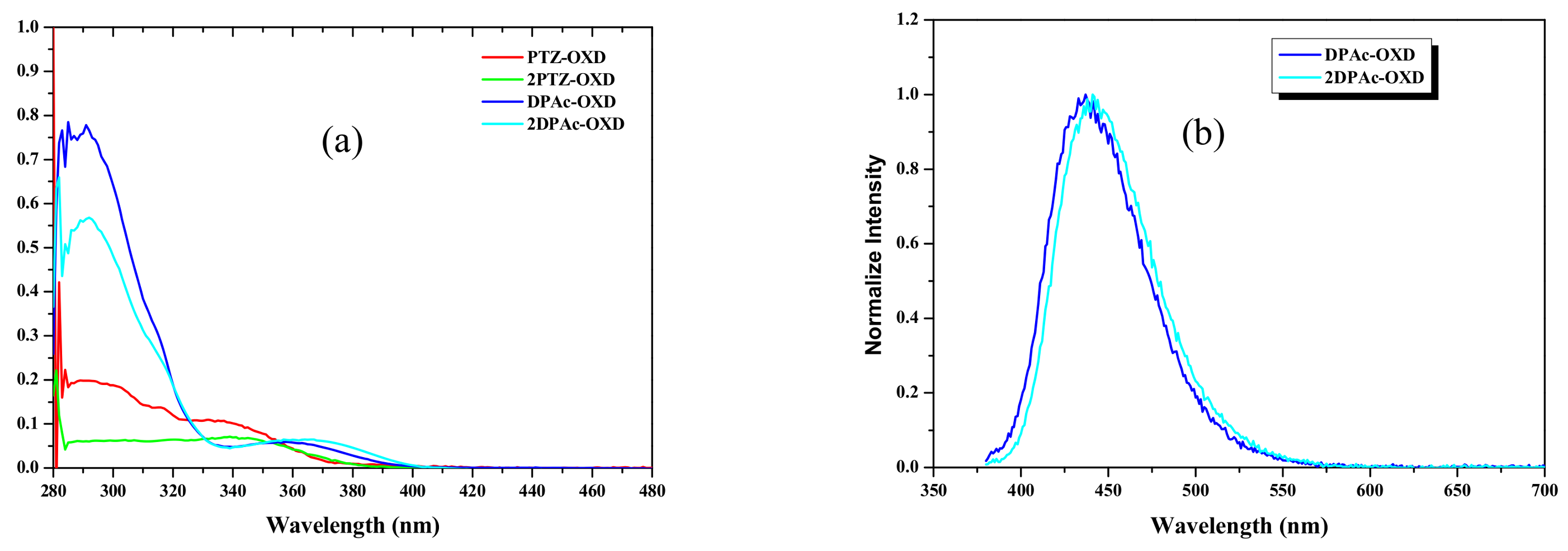
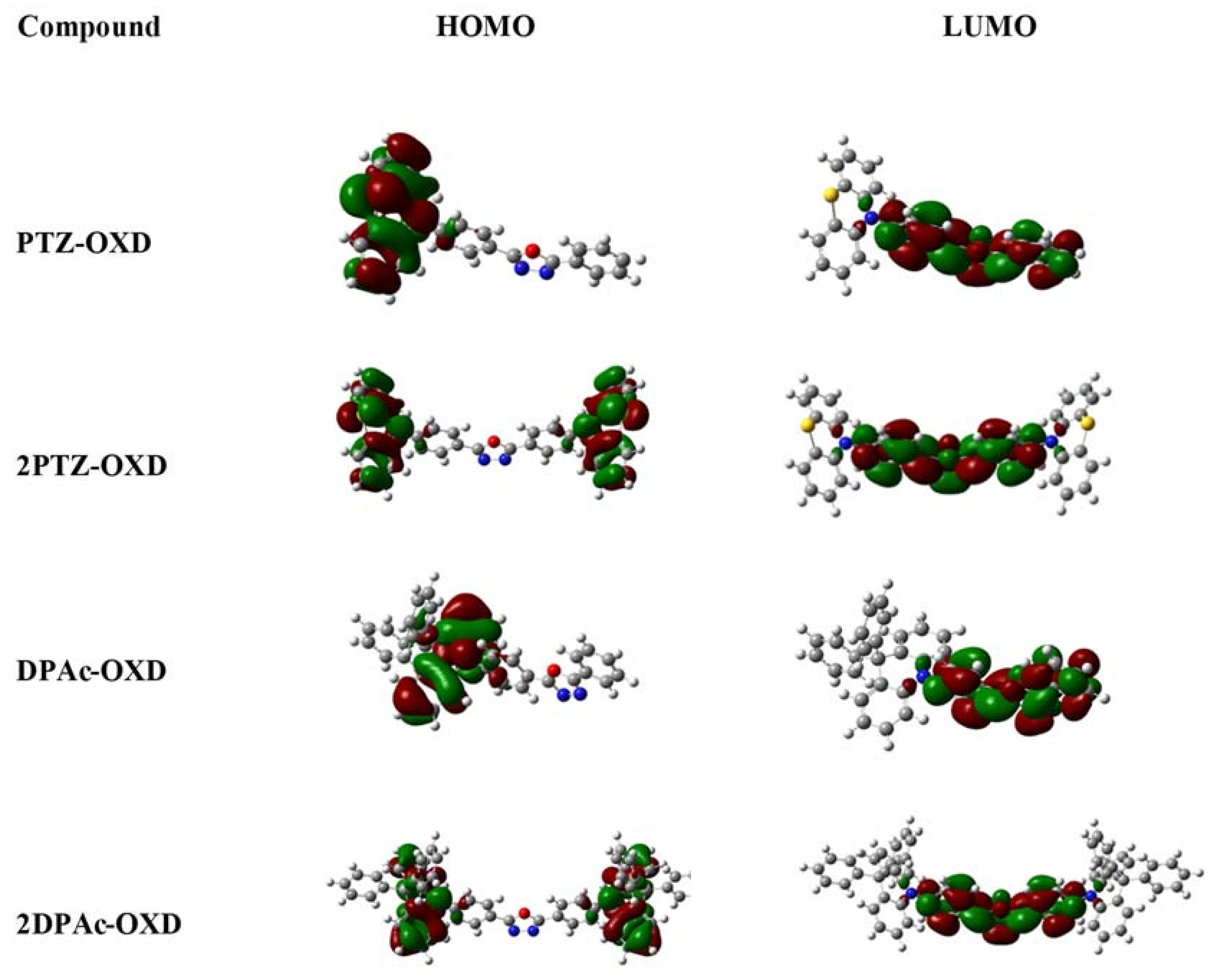
| Dopant | Td a (°C) | UV-vis b (nm) | PL max c (nm) | HOMO d (eV) | LUMO e (eV) | Eg f (eV) | ET g (eV) |
|---|---|---|---|---|---|---|---|
| PTZ-OXD 2PTZ-OXD DPAc-OXD 2DPAc-OXD | 358 419 375 455 | 376 384 396 402 | 500 512 435 442 | −5.48 −5.48 −5.69 −5.68 | −2.18 −2.25 −2.56 −2.60 | 3.30 3.23 3.13 3.08 | 2.48 2.42 2.85 2.81 |
| Calculation Values | PTZ-OXD | 2PTZ-OXD | DPAc-OXD | 2DPAc-OXD |
|---|---|---|---|---|
| S1 (eV) a T1 (eV) b ΔEST (eV) c D-A rotation(°) HOMO (eV) LUMO (eV) | 3.71 3.36 0.35 77.05 −5.975 −2.185 | 3.63 3.34 0.29 78.91 −5.485 −2.255 | 3.83 3.37 0.46 81.26 −5.695 −2.565 | 3.78 3.34 0.44 81.70 −5.685 −2.605 |
| Device properties | PTZ-OXD | 2PTZ-OXD | DPAc-OXD | 2DPAc-OXD |
|---|---|---|---|---|
| Turn on voltage (V) | 4.4 a 6.0 b | 4.2 a 5.6 b | 4.1 a 6.0 b | 4.1 a 6.2 b |
| Driving voltage (V) | 6.8 a | 5.9 a | 7.6 a | 7.5 a |
| Current (mA) | 0.05 a 0.05 b | 0.03 a 0.03 b | 0.04 a 0.06 b | 0.05 a 0.10 b |
| Current efficiency (cd/A) | 5.03 a 4.97 b | 9.20 a 10.10 b | 1.30 a 0.72 b | 1.28 a 0.66 b |
| Power efficiency (Lm/W) | 3.59 a 2.60 b | 6.88 a 5.67 b | 0.99 a 0.38 b | 0.98 a 0.33 b |
| EQE (%) | 1.94 a | 3.38 a | 1.84 a | 1.81 a |
| 2.26 b | 3.99 b | 1.08 b | 0.88 b | |
| Luminance (at 1000 cd/m2) | 1008 | 1020 | 1029 | 992 |
| CIE (x,y) | (0.38, 0.50) a (0.43, 0.14) b | (0.40, 0.53) a (0.31, 0.49) b | (0.16, 0.12) a (0.18, 0.12) b | (0.17, 0.14) a (0.18, 0.15) b |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, Q.; Braveenth, R.; Zhang, H.Q.; Bae, I.-J.; Kim, M.; Chai, K.Y. Oxadiazole-Based Highly Efficient Bipolar Fluorescent Emitters for Organic Light-Emitting Diodes. Molecules 2018, 23, 843. https://doi.org/10.3390/molecules23040843
Wu Q, Braveenth R, Zhang HQ, Bae I-J, Kim M, Chai KY. Oxadiazole-Based Highly Efficient Bipolar Fluorescent Emitters for Organic Light-Emitting Diodes. Molecules. 2018; 23(4):843. https://doi.org/10.3390/molecules23040843
Chicago/Turabian StyleWu, Qiong, Ramanaskanda Braveenth, Heng Qiang Zhang, Il-Ji Bae, Miyoung Kim, and Kyu Yun Chai. 2018. "Oxadiazole-Based Highly Efficient Bipolar Fluorescent Emitters for Organic Light-Emitting Diodes" Molecules 23, no. 4: 843. https://doi.org/10.3390/molecules23040843





