Intraconversion of Polar Ginsenosides, Their Transformation into Less-Polar Ginsenosides, and Ginsenoside Acetylation in Ginseng Flowers upon Baking and Steaming
Abstract
:1. Introduction
2. Results and Discussion
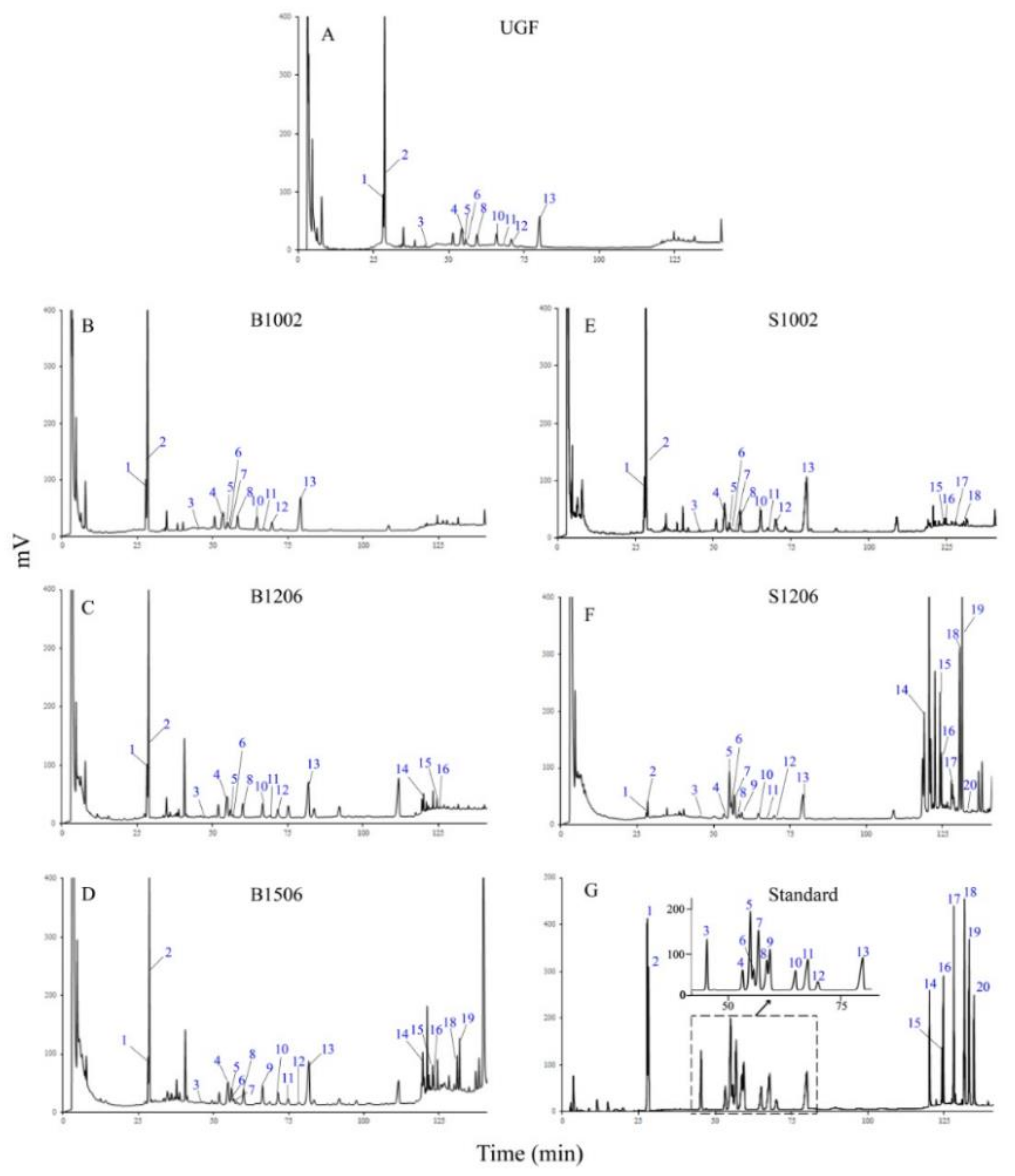
2.1. Effects of Baking and Steaming on HPLC Profiles of Ginsenosides in Ginseng Flowers
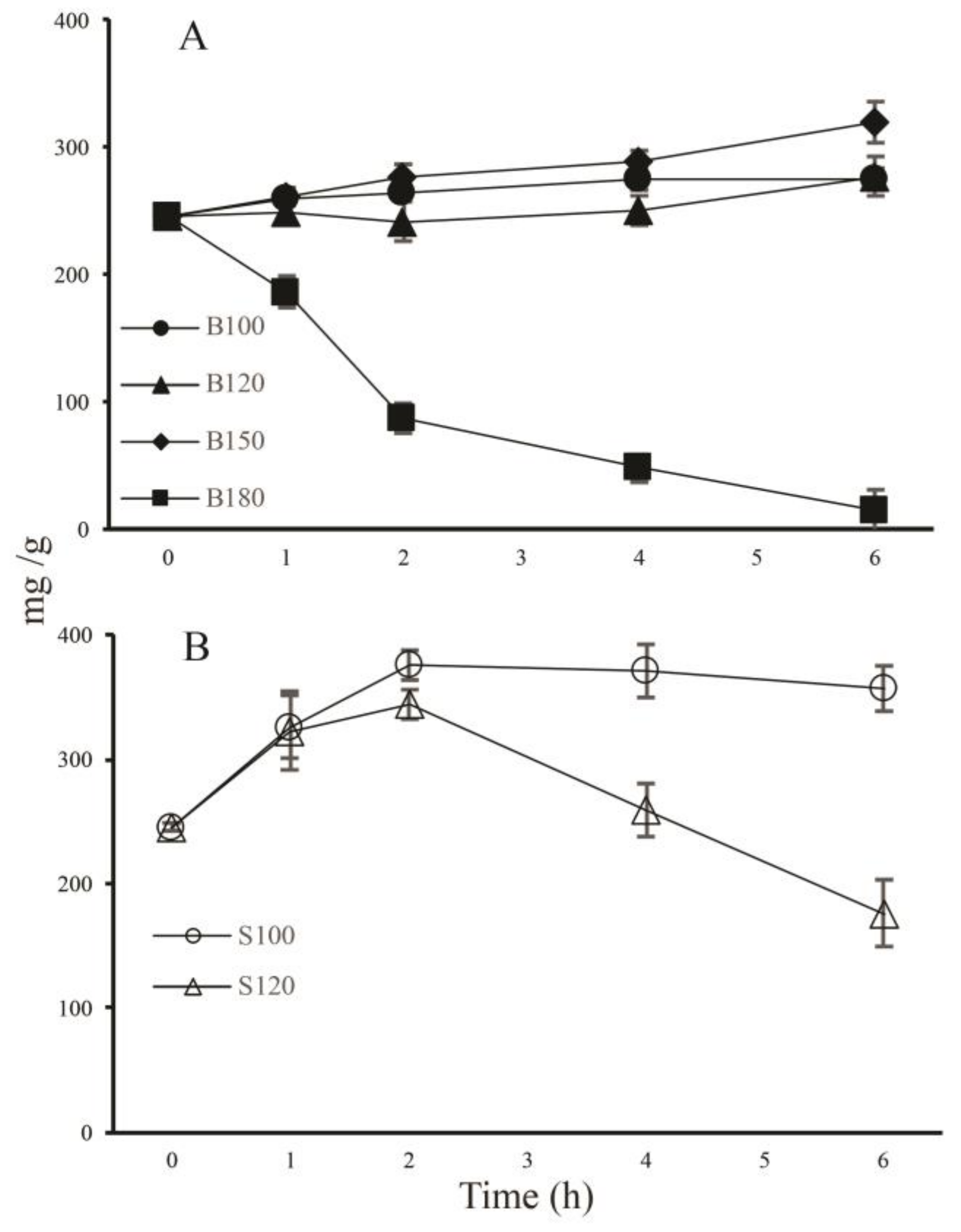
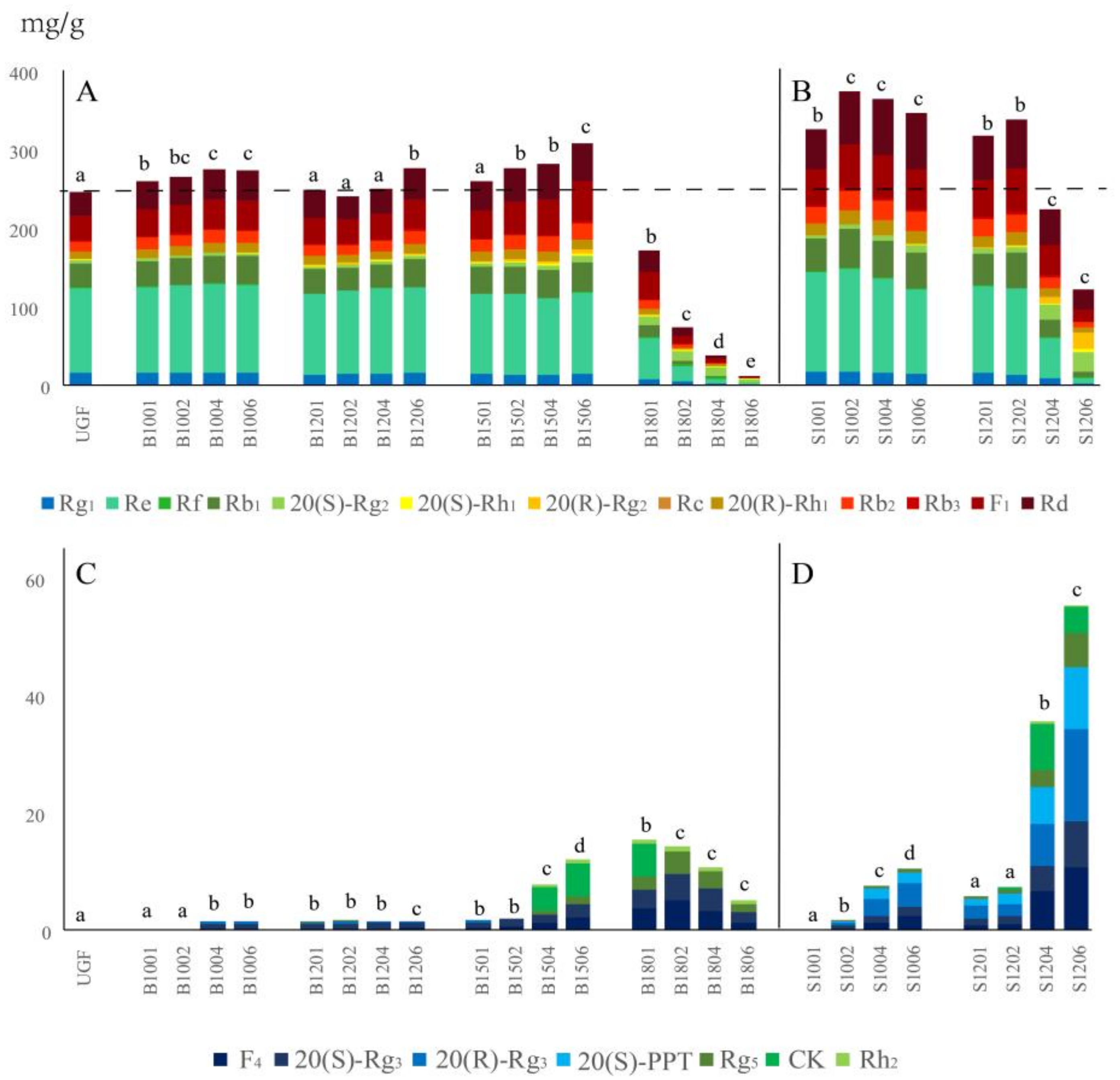
2.2. Effects of Baking and Steaming on 20 Representative Ginsenosides
2.2.1. Changes in Total Amount of the 20 Ginsenosides upon Baking and Steaming
2.2.2. Transformation of Polar Ginsenosides into Less Polar Ginsenosides upon Baking and Steaming
2.3. Identification of Ginsenosides in Ginseng Flowers Using UPLC-QTOF-MS/MS
2.3.1. Ginsenosides Originally Existing in UGF
2.3.2. Ginsenosides Newly Generated in Heated Ginseng Flowers
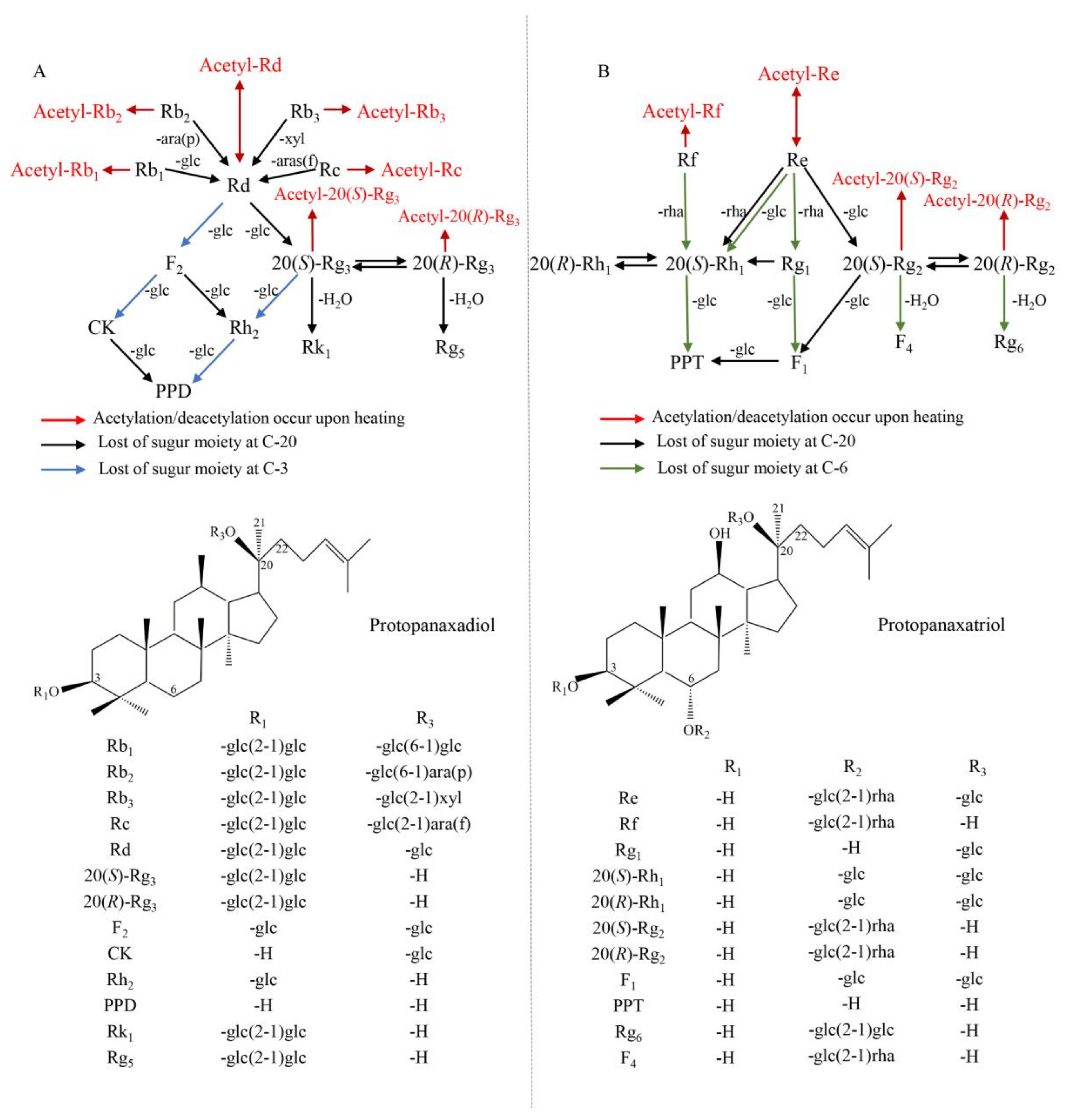
2.4. Transformation Mechanisms of Ginsenosides in Ginseng Flowers upon Heating
3. Materials and Methods
3.1. Chemicals and Plant Materials
3.2. Crude Extract Preparation
3.3. HPLC Analyses of Ginsenosides
3.4. UPLC-QTOF-MS/MS Analyses of Ginsenosides
3.5. Statistical Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ellis, J.M.; Reddy, P. The effects of Panax ginseng on quality of life. J. Clin. Pharm. Ther. 2003, 28, 5–15. [Google Scholar] [CrossRef] [PubMed]
- In, G.; Ahn, N.G.; Bae, B.S.; Lee, M.W.; Park, H.W.; Jang, K.H.; Cho, B.G.; Han, C.K.; Park, C.K.; Kwak, Y.S. In Situ analysis of chemical components induced by steaming between fresh ginseng, steamed ginseng, and red ginseng. J. Ginseng Res. 2017, 41, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.Y.; Luo, D.; Cheng, Y.J.; Ma, J.F.; Wang, Y.M.; Liang, Q.L.; Luo, G.A. Steaming-induced chemical transformations and holistic quality assessment of red ginseng derived from Panax ginseng by means of HPLC-ESI-MS/MS(n)-based multicomponent quantification fingerprint. J. Agric. Food Chem. 2012, 60, 8213–8224. [Google Scholar] [CrossRef] [PubMed]
- Christensen, L.P. Chapter 1 Ginsenosides. Adv. Food Nutr. Res. 2009, 55, 1–99. [Google Scholar] [PubMed]
- Qi, L.W.; Wang, C.Z.; Yuan, C.S. Isolation and analysis of ginseng: Advances and challenges. ChemInform 2011, 42, 467–495. [Google Scholar] [CrossRef]
- Yang, H.; Lee, D.Y.; Kang, K.B.; Kim, J.Y.; Kim, S.O.; Yoo, Y.H.; Sung, S.H. Identification of ginsenoside markers from dry purified extract of Panax ginseng by a dereplication approach and UPLC-QTOF/MS analysis. J. Pharm. Biomed. Anal. 2015, 109, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Jin, Z.; Yuan, Y.; Wei, H.; Xu, X.; He, S.; Chen, S.; Hou, W.; Guo, Q.; Hua, B. Ginsenoside Rg3 serves as an adjuvant chemotherapeutic agent and VEGF inhibitor in the treatment of non-small cell lung cancer: A meta-analysis and systematic review. Evid.-Based Complement. Altern. Med. 2016. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.D.; Yang, Y.Y.; Ouyang, D.S.; Yang, G.P. A review of biotransformation and pharmacology of ginsenoside compound K. Fitoterapia 2015, 100, 208–220. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.M.; Li, S.L.; Zhang, H.; Wang, Y.; Zhao, Z.L.; Chen, S.L.; Xu, H.X. Holistic quality evaluation of commercial white and red ginseng using a UPLC-QTOF-MS/MS-based metabolomics approach. J. Pharm. Biomed. Anal. 2012, 62, 258–273. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.Z.; Aung, H.H.; Ni, M.; Wu, J.A.; Tong, R.B.; Wicks, S.; He, T.C.; Yuan, C.S. Red American ginseng: Ginsenoside constituents and antiproliferative activities of heat-processed Panax quinquefolius roots. Planta Med. 2007, 73, 669–674. [Google Scholar] [CrossRef] [PubMed]
- Yun, T.K.; Lee, Y.S.; Lee, Y.H. Anticarcinogenic effect of Panax ginseng C.A. Meyer and identification of active compounds. J. Korean Med. Sci. 2001, 16, S6–S18. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.E.; Lyu, S.Y. Anti-inflammatory and anti-oxidative effects of Korean red ginseng extract in human keratinocytes. Immune Netw. 2011, 11, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Lu, Z.; Teng, Y.; Guo, Y.; Liu, S. Structural characterization of ginsenosides from flower buds of Panax ginseng by RRLC-Q-TOF MS. J. Chromatogr. Sci. 2016, 54, 136–143. [Google Scholar] [PubMed]
- Nguyen, H.T.; Song, G.Y.; Kim, J.A.; Hyun, J.H.; Kang, H.K.; Kim, Y.H. Dammarane-type saponins from the flower buds of Panax ginseng and their effects on human leukemia cells. Bioorg. Med. Chem. Lett. 2010, 20, 309–314. [Google Scholar] [PubMed]
- Li, S.S.; Li, K.K.; Xu, F.; Tao, L.; Yang, L.; Chen, S.X. A strategy for simultaneous isolation of less polar ginsenosides, including a pair of new 20-methoxyl isomers, from flower buds of Panax ginseng. Molecules 2017, 22, 442. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, M.; Sugimoto, S.; Nakamura, S. Medicinal flowers. XI. Structures of new dammarane-type triterpene diglycosides with hydroperoxide group from flower buds of Panax ginseng. Chem. Pharm. Bull. 2007, 55, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Ko, S.K.; Cho, O.S.; Bae, H.M.; Im, B.O.; Lee, O.H.; Lee, B.Y. Quantitative analysis of ginsenosides composition in flower buds of various ginseng plants. J. Korean Soc. Appl. Biol. 2011, 54, 154–157. [Google Scholar] [CrossRef]
- Cho, K.; Song, S.B.; Tung, N.H.; Kim, K.E.; Kim, Y.H. Inhibition of TNF-α-Mediated NF-ĸB transcriptional activity by dammarane-type ginsenosides from steamed flower buds of Panax ginseng in HepG2 and SK-Hep1 cells. Biomol. Ther. 2014, 22, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Tung, N.H.; Quang, T.H.; Son, J.H.; Koo, J.E.; Hong, H.J.; Koh, Y.S.; Song, G.Y.; Kim, Y.H. Inhibitory effect of ginsenosides from steamed ginseng-leaves and flowers on the LPS-stimulated IL-12 production in bone marrow-derived dendritic cells. Food Sci. Biotechnol. 2011, 34, 681–685. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.F.; Gao, Y.; Xu, S.Y.; Liu, H.; Xue, X.; Zhang, Y.; Zhang, H.; Liu, M.N.; Xiong, H.; Lin, R.C.; et al. Remarkable impact of steam temperature on ginsenosides transformation from fresh ginseng to red ginseng. J. Ginseng Res. 2017. [Google Scholar] [CrossRef]
- Zhou, Q.L.; Zhu, D.N.; Yang, Y.F.; Xu, W.; Yang, X.W. Simultaneous quantification of twenty-one ginsenosides and their three aglycones in rat plasma by a developed UFLC-MS/MS assay: Application to a pharmacokinetic study of red ginseng. J. Pharm. Biomed. Anal. 2017, 137, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.P.; Zhang, Y.B.; Yang, X.W.; Zhao, D.Q.; Wang, Y.P. Rapid characterization of ginsenosides in the roots and rhizomes of Panax ginseng by UPLC-DAD-QTOF-MS/MS and simultaneous determination of 19 ginsenosides by HPLC-ESI-MS. J. Ginseng Res. 2016, 40, 382–394. [Google Scholar] [CrossRef] [PubMed]
- Du, Q.Q.; Liu, S.Y.; Xu, R.F.; Li, M.; Song, F.R.; Liu, Z.Q. Studies on structures and activities of initial maillard reaction products by electrospray ionisation mass spectrometry combined with liquid chromatography in processing of red ginseng. Food Chem. 2012, 135, 832–838. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, X.; Meng, L.; Wang, Y. Qualitative and quantitative analysis of furosine in fresh and processed ginsengs. J. Ginseng Res. 2018, 42, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Qiu, S.; Yang, W.Z.; Yao, C.L. Malonyl ginsenosides with potential antidiabetic activities from the flower buds of Panax ginseng. J. Nat. Prod. 2017, 80, 899–908. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Xia, J.; Wang, C.Z.; Zhang, J.Q.; Ruan, C.C.; Sun, G.Z.; Yuan, C.S. Remarkable impact of acidic ginsenosides and organic acids on ginsenoside transformation from fresh ginseng to red ginseng. J. Agric. Food. Chem. 2016, 64, 5389–5399. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.Y.; Kim, J.M.; Han, S.B.; Lee, S.K.; Kim, N.D.; Park, M.K. Steaming of ginseng at high temperature enhances biological activity. J. Nat. Prod. 2000, 63, 1702–1704. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.Y.; Luo, G.A. Chemical reactions of ginsenosides in red ginseng processing by HPLC/MS/MS. Chin. Tradit. Herbal Drugs 2005, 1, 18. [Google Scholar]
- Qi, L.W.; Wang, H.Y.; Zhang, H.; Wang, C.Z.; Li, P.; Yuan, C.S. Diagnostic ion filtering to characterize ginseng saponins by rapid liquid chromatography with time-of-flight mass spectrometry. J. Chromatogr. A 2012, 1230, 93–99. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
| No. | Rt (min) | Compound Name | Error (ppm) | Molecular Formula | Measured Value (m/z) | MS/MS Fragments m/z | Sample Items |
|---|---|---|---|---|---|---|---|
| 1 | 20.96 | Rg1 | 0.8 | C42H72O14 | 799.48 | 637.43[M-(Glu-H2O)-H]−/475.38[M-2(Glu-H2O)-H]− | All |
| 2 | 21.19 | Re | 1.1 | C48H82O18 | 945.57 | 783.49[M-(Glu-H2O)-H]−/637.43[M-(Glu-H2O)-(Rha-H2O)-H]−/475.38[M-2(Glu-H2O)-(Rha-H2O)-H]− | All |
| 3 | 22.98 | G-Ia | −1.9 | C42H72O14 | 799.49 | 637.43[M-(Glu-H2O)-H]−/475.32[M-2(Glu-H2O)-H]− | S1206 |
| 4 | 24.16 | Re isomer | −0.6 | C48H82O18 | 945.55 | 799.49[M-(Rha-H2O)-H]−/637.43[M-(Glu-H2O)-(Rha-H2O)-H]−/475.33[M-2(Glu-H2O)-(Rha-H2O)-H]− | S1206 |
| 5 | 26.69 | Floral G-P | −2.4 | C53H90O23 | 1093.58 | 961.53[M-(Ara-H2O)-H]−/799.48[M-(Ara-H2O)-(Glu-H2O)-H]−/637.43[M-(Ara-H2O)-2(Glu-H2O)-H]−/ 475.37[M-(Ara-H2O)-3(Glu-H2O)-H]− | UPG, B1206, B1506 |
| 6 | 27.80 | Acetyl-Re isomer I | 0.4 | C50H84O19 | 987.60 | 945.54[M-Ac-H]−/799.48[M-Ac-(Rha-H2O)-H]−/637.43[M-Ac-(Glu-H2O)-(Rha-H2O)-H]−/ 475.38[M-Ac-2(Glu-H2O)-(Rha-H2O)-H]− | B1506 |
| 7 | 28.52 | Floral G-P isomer | −1.3 | C53H90O23 | 1093.58 | 961.54[M-(Ara-H2O)-H]−/799.48[M-(Ara-H2O)-(Glu-H2O)-H]−/637.41[M-(Ara-H2O)-2(Glu-H2O)-H]- | UPG, S1204 |
| 8 | 28.72 | Acetyl-Re isomer II | −3.2 | C50H84O19 | 987.56 | 945.54[M-Ac-H]−/799.48[M-Ac-(Ara-H2O)-(Glu-H2O)-H]−/637.43[M-Ac-(Glu-H2O)-(Rha-H2O)-H]−/ 475.30[M-Ac-(Glu-H2O)-(Rha-H2O)-H]− | B1206, B1506 |
| 9 | 28.89 | Acetyl-Re isomer III | −1.4 | C50H84O19 | 987.56 | 945.54[M-Ac-H]−/799.48[M-(Ara-H2O)-(Glu-H2O)-H]−/637.43[M-Ac-(Glu-H2O)-(Rha-H2O)-H]−/ 475.38[M-Ac-(Glu-H2O)-(Rha-H2O)-H]− | B1506 |
| 10 | 29.73 | Acetyl- Rg1I/isomer | −1.5 | C44H74O15 | 841.56 | 799.48[M-Ac-H]−/637.43[M-Ac-(Glu-H2O)-H]−/475.37[M-Ac-(Glu-H2O)-(Rha-H2O)-H]− | B1506 |
| 11 | 30.57 | Acetyl-Re isomer IV | −0.7 | C50H84O19 | 987.58 | 945.54[M-Ac-H]−/799.48[M-(Ara-H2O)-(Glu-H2O)-H]−/637.43[M-Ac-(Glu-H2O)-(Rha-H2O)-H]−/ 475.38[M-Ac-(Glu-H2O)-(Rha-H2O)-H]− | B1506 |
| 12 | 31.55 | G-Re1/2/3/NG-N isomer | 0.9 | C48H82O19 | 961.56 | 799.49[M-(Glu-H2O)-H]−/637.43[M-2(Glu-H2O)-H]−/475.38[M-3(Glu-H2O)-H]− | All |
| 13 | 32.89 | Acetyl-Re | 0.2 | C50H84O19 | 987.55 | 945.54[M-Ac-H]−/927.5[M-Ac-OH]−/799.48[M-(Ara-H2O)-(Glu-H2O)-H]−/637.43[M-(Ara-H2O)-2(Glu-H2O)-H]− | All |
| 14 | 33.18 | Acetyl-Rg1 II | 1 | C44H74O15 | 841.56 | 799.48[M-Ac-H]−/637.43[M-Ac-(Glu-H2O)-H]−/475.37[M-Ac-(Ara-H2O)-(Glu-H2O)-H]− | B1506 |
| 15 | 36.52 | Rf | −0.1 | C42H72O14 | 799.49 | 637.43[M-(Glu-H2O)-H]−/475.38[M-2(Glu-H2O)-H]− | All |
| 16 | 39.86 | NG-R2 | −1.2 | C41H70O13 | 769.48 | 637.43[M-(Ara-H2O)-H]−/475.38[M-(Ara-H2O)-(Glu-H2O)-H]− | All |
| 17 | 41.00 | F5/F3 | −1.2 | C41H70O13 | 769.47 | 637.43[M-(Ara-H2O)-H]−/475.38[M-(Ara-H2O)-(Glu-H2O)-H]− | All |
| 18 | 42.90 | Rb1 | −0.3 | C54H92O23 | 1107.62 | 945.54[M-(Glu-H2O)-H]−/783.48[M-2(Glu-H2O)-H]−/621.43[M-3(Glu-H2O)-H]−/459.38[M-4(Glu-H2O)-H]− | All |
| 19 | 42.89 | F5/F3 | 0.4 | C41H70O13 | 769.48 | 637.42[M-(Rha-H2O)-H]−/475.37[M-(Rha-H2O)-(Glu-H2O)-H]− | S1204, B1206, B1506 |
| 20 | 43.23 | 20(S)-Rg2 | 0.8 | C42H71O13 | 783.49 | 637.43[M-(Rha-H2O)-H]−/475.38[M-(Rha-H2O)-(Glu-H2O)-H]− | All |
| 21 | 44.57 | 20(S)-Rh1 | −0.6 | C36H62O9 | 637.43 | 475.38[M-(Glu-H2O)-H]− | S1204, S1206, B1206 |
| 22 | 45.37 | 20(R)-Rg2 | 0.8 | C42H71O13 | 783.49 | 637.43[M-(Rha-H2O)-H]−/475.38[M-(Rha-H2O)-(Glu-H2O)-H]− | S1204, S1206 |
| 23 | 47.01 | Rc | −0.8 | C53H90O22 | 1077.60 | 945.54[M-(Ara-H2O)-H]−/783.49[M-(Ara-H2O)-(Glu-H2O)-H]−/621.44[M-(Ara-H2O)-2(Glu-H2O)-H]−/ 459.38[M-(Ara-H2O)-3(Glu-H2O)-H]− | All |
| 24 | 47.29 | 20(R)-Rh1 | 2.2 | C36H62O9 | 637.44 | 475.3811[M-(Glu-H2O)-H]− | S1204, S1206 |
| 25 | 49.47 | PQ-R1 | −1.5 | C56H94O24 | 1149.76 | 1107.59[M-Ac-H]−/945.54[M-Ac-(Glu-H2O)-H]−/783.49[M-Ac-2(Glu-H2O)-H]−/ 621.43[M-Ac-3(Glu-H2O)-H]−/459.38[M-Ac-4(Glu-H2O)-H]− | B1506 |
| 26 | 52.17 | Rb2 | −0.7 | C53H90O22 | 1077.60 | 945.54[M-(Ara(p)-H2O)-H]−/783.49[M-(Ara(p)-H2O)-(Glu-H2O)-H]−/ 621.44[M-(Ara(p)-H2O)-2(Glu-H2O)-H]−/459.38[M-(Ara(p)-H2O)-3(Glu-H2O)-H]− | All |
| 27 | 54.08 | Rb3 | −1.5 | C53H90O22 | 1077.59 | 945.54[M-(Xyl-H2O)-H]−/783.49[M-(Xyl(p)-H2O)-(Glu-H2O)-H]−/ 621.44[M-(Xyl(p)-H2O)-2(Glu-H2O)-H]−/459.37[M-(Xyl(p)-H2O)-3(Glu-H2O)-H]− | All |
| 28 | 56.46 | F1 | 1.6 | C36H62O9 | 637.44 | 475.37[M-(Glu-H2O)-H]− | All |
| 29 | 57.91 | Acetyl-Re isomer V | −1.4 | C51H84O21 | 987.56 | 945.54[M-Ac-H]−/783.48[M-Ac-(Glu-H2O)-H]−/637.43[M-(Glu-H2O)-(Rha-H2O)-H]−/ 475.38[M-2(Glu-H2O)-(Rha-H2O)-H]− | UPG |
| 30 | 59.09 | PQ-R1 isomer | −0.3 | C56H94O24 | 1149.81 | 1107.59[M-Ac-H]−/945.54[M-Ac-(Glu-H2O)-H]−/783.49[M-Ac-2(Glu-H2O)-H]−/ 621.43[M-Ac-3(Glu-H2O)-H]−/475.3767[M-Ac-(Rha-H2O)-3(Glu-H2O)-H]− | S1204, S1206, B1206, B1506 |
| 31 | 61.46 | Acetyl-Rb2 isomer | −0.1 | C55H92O23 | 1119.71 | 1077.58[M-Ac-H]−/945.54[M-Ac-(Ara(p)-H2O)-H]−/783.48[M-Ac-(Ara(p)-H2O)-(Glu-H2O)-H]−/ 621.43[M-Ac-(Ara(p)-H2O)-2(Glu-H2O)-H]−/459.38[M-Ac-(Ara(p)-H2O)-3(Glu-H2O)-H]− | B1506 |
| 32 | 64.88 | Rd | −0.3 | C48H82O18 | 945.56 | 783.49[M-(Glu-H2O)-H]−/621.44/459.38[M-2(Glu-H2O)-H]− | All |
| 33 | 65.51 | Rs1/Rs2 | −2.8 | C55H92O23 | 1119.75 | 1077.5[M-Ac-H]−/945.53[M-Ac-(Ara(p)-H2O)-H]−/783.48[M-Ac-(Ara(p)-H2O)-(Glu-H2O)-H]−/ 621.43[M-Ac-(Ara(p)-H2O)-2(Glu-H2O)-H]−/459.38[M-Ac-(Ara(p)-H2O)-3(Glu-H2O)-H]− | B1206, B1506 |
| 34 | 70.40 | Acetyl-Rg3 isomer | 0.1 | C44H74O14 | 825.51 | 783.49[M-Ac-H]−/637.43[M-Ac-(Rha-H2O)-H]−/475.37[M-Ac-(Rha-H2O)-(Glu-H2O)-H]− | B1506 |
| 35 | 73.77 | Rs1/Rs2 | −0.8 | C55H92O23 | 1119.78 | 1077.58[M-Ac-H]−/945.54[M-Ac-(Ara(p)-H2O)-H]−/783.49[M-Ac-(Ara(p)-(Glu-H2O)-H2O)-H]−/ 621.43[M-Ac-(Ara(p)-2(Glu-H2O)-H2O)-H]−/459.37[M-Ac-(Ara(p)-3(Glu-H2O)-H2O)-H]− | S1204, S1206, B1206, B1506 |
| 36 | 73.96 | PQ-R1 isomer | −1.5 | C56H94O24 | 1149.78 | 1107.59[M-Ac-H]−/1089.58[M-Ac-OH]−/945.53[M-Ac-(Glu-H2O)-H]−/783.49[M-Ac-2(Glu-H2O)-H]−/ 621.43[M-Ac-3(Glu-H2O)-H]−/459.38[M-Ac-4(Glu-H2O)-H]− | B1206 |
| 37 | 82.72 | Vinaginsenoside R16 | −2.5 | C47H80O17 | 915.53 | 753.48[M-(Glu-H2O)-H]−/621.44[M-(Ara(p)-H2O)-(Glu-H2O)-H]−/ 475.37[M-(Ara(p)-H2O)-(Glu-H2O)-(Rha-H2O)-H]− | UPG, S1204 |
| 38 | 75.82 | Acetyl-Rb2 | −2 | C55H92O23 | 1119.77 | 1077.58[M-Ac-H]−/945.54[M-Ac-(Ara(p)-H2O)-H]−/783.48[M-Ac-(Ara(p)-H2O)-(Glu-H2O)-H]−/ 621.43[M-Ac-(Ara(p)-H2O)-2(Glu-H2O)-H]−/459.38[M-Ac-(Ara(p)-H2O)-3(Glu-H2O)-H]− | B1206, B1506 |
| 39 | 78.02 | Acetyl-Rd isomer I | −1.1 | C50H84O19 | 987.67 | 945.54[M-Ac-H]−/783.48[M-Ac-(Glu-H2O)-H]−/621.43[M-Ac-2(Glu-H2O)-H]−/459.38[M-Ac-3(Glu-H2O)-H]− | B1206, B1506 |
| 40 | 79.54 | Acetyl-Rb3 | −1.5 | C55H92O23 | 1119.76 | 1077.58[M-Ac-H]−/1059.57[M-Ac-OH]−/945.54[M-Ac-(Ara(p)-H2O)-H]−/ 783.49[M-Ac-(Ara(p)-H2O)-2(Glu-H2O)-H]−/621.43[M-Ac-(Ara(p)-H2O)-3(Glu-H2O)-H]−/ 459.38[M-Ac-(Ara(p)-H2O)-4(Glu-H2O)-H]− | B1206, B1506 |
| 41 | 81.22 | Acetyl-Rd isomer II | −3.9 | C50H84O19 | 987.50 | 945.54[M-Ac-H]−/799.48[M-Ac-(Rha(p)-H2O)-H]−/621.43[M-Ac-(Rha(p)-H2O)-(Glu-H2O)-OH]−/ 459.38[M-Ac-(Rha(p)-H2O)-2(Glu-H2O)-OH]− | B1206, B1506 |
| 42 | 92.13 | Acetyl-Rd | 1.4 | C50H84O19 | 987.67 | 945.54[M-Ac-H]−/927.52[M-Ac-OH]−/783.49[M-Ac-(Glu-H2O)-H]−/621.44[M-Ac-2(Glu-H2O)-H]−/ 459.37[M-Ac-3(Glu-H2O)-H]− | All |
| 43 | 95.90 | NG-Fe | −0.4 | C47H80O17 | 915.53 | 783.43[M-(Ara-H2O)-H]−/621.43[M-(Ara-H2O)-(Glu-H2O)-H]−/459.37[M-(Ara-H2O)-2(Glu-H2O)-H]− | UPG, S1204, S1206 |
| 44 | 98.60 | Acetyl-Rd isomer III | −1.1 | C50H84O19 | 987.71 | 945.54[M-Ac-H]−/783.43[M-Ac-(Glu-H2O)-H]−/621.43[M-Ac-2(Glu-H2O)-H]−/459.38[M-Ac-3(Glu-H2O)-H]− | B1206, B1506 |
| 45 | 100.46 | Rg6 isomer | 0 | C42H69O12 | 765.48 | 619.42[M-(Rha-H2O)-H]−/457.36[M-(Rha-H2O)-(Glu-H2O)-H]− | B1506 |
| 46 | 101.46 | Rg6 | −0.8 | C42H69O12 | 765.48 | 619.42[M-(Rha-H2O)-H]−/457.37[M-(Rha-H2O)-(Glu-H2O)-H]− | S1204, S1206, B1506 |
| 47 | 104.98 | Pseudo-G-RC1 | −1 | C50H84O19 | 987.43 | 945.54[M-Ac-H]−/783.48[M-Ac-(Glu-H2O)-H]−/621.43[M-Ac-2(Glu-H2O)-H]−/459.38[M-Ac-3(Glu-H2O)-H]− | S1204, S1206, B1206, B1506 |
| 48 | 109.06 | F4 | −1.2 | C42H69O12 | 765.48 | 619.41[M-(Rha-H2O)-H]−/457.37[M-(Rha-H2O)-(Glu-H2O)-H]− | S1204, S1206, B1206, B1506 |
| 49 | 109.23 | Rk3 | 1.1 | C36H59O8 | 619.42 | 457.36[M-(Glu-H2O)-H]− | S1206 |
| 50 | 114.46 | Rg3 isomer | 1.3 | C42H72O13 | 783.69 | 621.44[M-(Glu-H2O)-H]−/459.38[M-2(Glu-H2O)-H]− | S1204 |
| 51 | 116.00 | G-La | 0.3 | C42H69O13 | 781.47 | 619.42[M-(Glu-H2O)-H]−/455.34[M-2(Glu-H2O)-H]− | S1206 |
| 52 | 118.43 | 20(S)-Rg3 | 0.1 | C42H72O13 | 783.49 | 621.44[M-(Glu-H2O)-H]−/459.38[M-2(Glu-H2O)-H]− | S1204, S1206, B1206, B1506 |
| 53 | 119.01 | 20(R)-Rg3 | 0.1 | C42H72O13 | 783.49 | 621.44[M-(Glu-H2O)-H]−/459.38[M-2(Glu-H2O)-H]− | S1204, S1206, B1206 |
| 54 | 121.37 | 20(S)-PPT | 0.6 | C30H52O4 | 475.38 | 475.38 | S1204, S1206 |
| 55 | 121.94 | 20(R)-PPT | −1.9 | C30H52O4 | 475.37 | 475.37 | S1204, S1206 |
| 56 | 122.26 | Acetyl-20(S)-Rg3 | −0.1 | C44H74O14 | 825.50 | 783.49[M-Ac-H]−/765.48[M-Ac-OH]−/621.44[M-Ac-(Glu-H2O)-H]−/459.38[M-Ac-2(Glu-H2O)-H]− | S1204, S1206, B1506 |
| 57 | 122.75 | Acetyl-20(R)-Rg3 | −0.4 | C44H74O14 | 825.50 | 783.49[M-Ac-H]−/765.48[M-Ac-OH]−/621.44[M-Ac-(Glu-H2O)-H]−/459.38[M-Ac-2(Glu-H2O)-H]− | S1204, S1206, B1506 |
| 58 | 123.72 | Acetyl-Rg3 isomer | 0.2 | C44H74O14 | 825.50 | 783.49[M-Ac-H]−/459.36[M-Ac-2(Glu-H2O)-H]− | S1204, S1206 |
| 59 | 124.73 | Rk1 | 0.5 | C42H69O12 | 765.49 | 603.44[M-(Glu-H2O)-H]− | S1204, S1206, B1206, B1506 |
| 60 | 125.41 | Rg5 | 0.5 | C42H69O12 | 765.49 | 603.43[M-(Glu-H2O)-H]- | S1204, S1206, B1506 |
| 61 | 126.59 | CK | 0.1 | C36H62O8 | 621.44 | 459.38[M-(Glu-H2O)-H]− | S1204, S1206, B1506 |
| 62 | 127.99 | Rh2 | 2.4 | C36H62O8 | 621.44 | 459.38[M-(Glu-H2O)-H]− | S1204, S1206, B1506 |
| 63 | 130.30 | Rs5 | −0.2 | C44H71O13 | 807.50 | 765.48[M-Ac-H]−/603.43[M-Ac-(Glu-H2O)-H]− | S1204, S1206, B1506 |
| 64 | 131.31 | Rs4 | −0.4 | C44H71O13 | 807.49 | 765.48[M-Ac-H]−/603.43[M-Ac-(Glu-H2O)-H]− | S1204, S1206, B1506 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, X.; Yao, F.; Fan, H.; Li, K.; Sun, L.; Liu, Y. Intraconversion of Polar Ginsenosides, Their Transformation into Less-Polar Ginsenosides, and Ginsenoside Acetylation in Ginseng Flowers upon Baking and Steaming. Molecules 2018, 23, 759. https://doi.org/10.3390/molecules23040759
Li X, Yao F, Fan H, Li K, Sun L, Liu Y. Intraconversion of Polar Ginsenosides, Their Transformation into Less-Polar Ginsenosides, and Ginsenoside Acetylation in Ginseng Flowers upon Baking and Steaming. Molecules. 2018; 23(4):759. https://doi.org/10.3390/molecules23040759
Chicago/Turabian StyleLi, Xiang, Fan Yao, Hang Fan, Ke Li, Liwei Sun, and Yujun Liu. 2018. "Intraconversion of Polar Ginsenosides, Their Transformation into Less-Polar Ginsenosides, and Ginsenoside Acetylation in Ginseng Flowers upon Baking and Steaming" Molecules 23, no. 4: 759. https://doi.org/10.3390/molecules23040759







