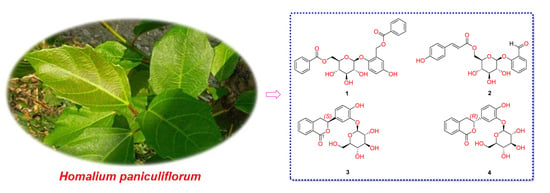
Bioactive Phenolic and Isocoumarin Glycosides from the Stems of Homalium paniculiflorum
Abstract
:1. Introduction
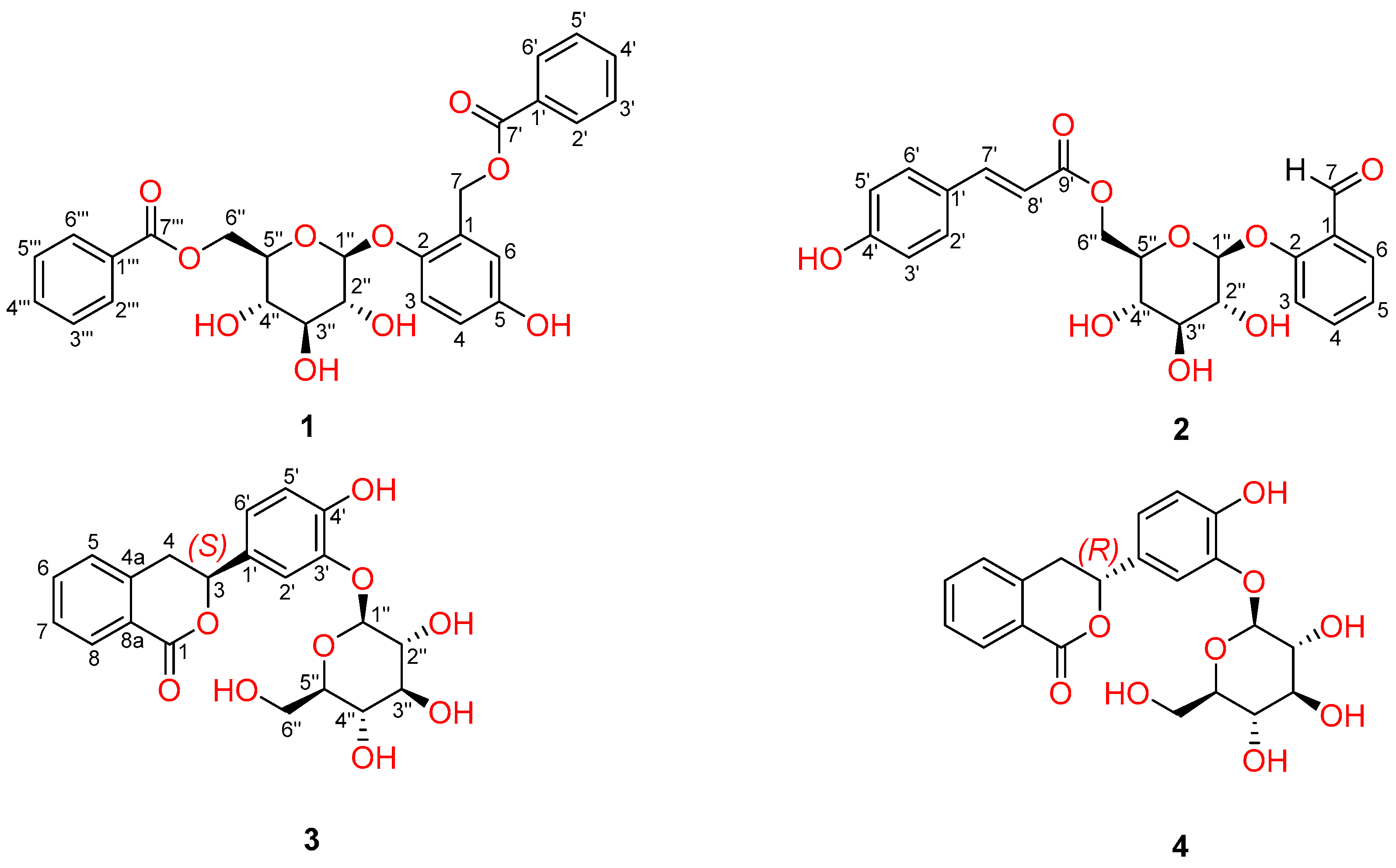
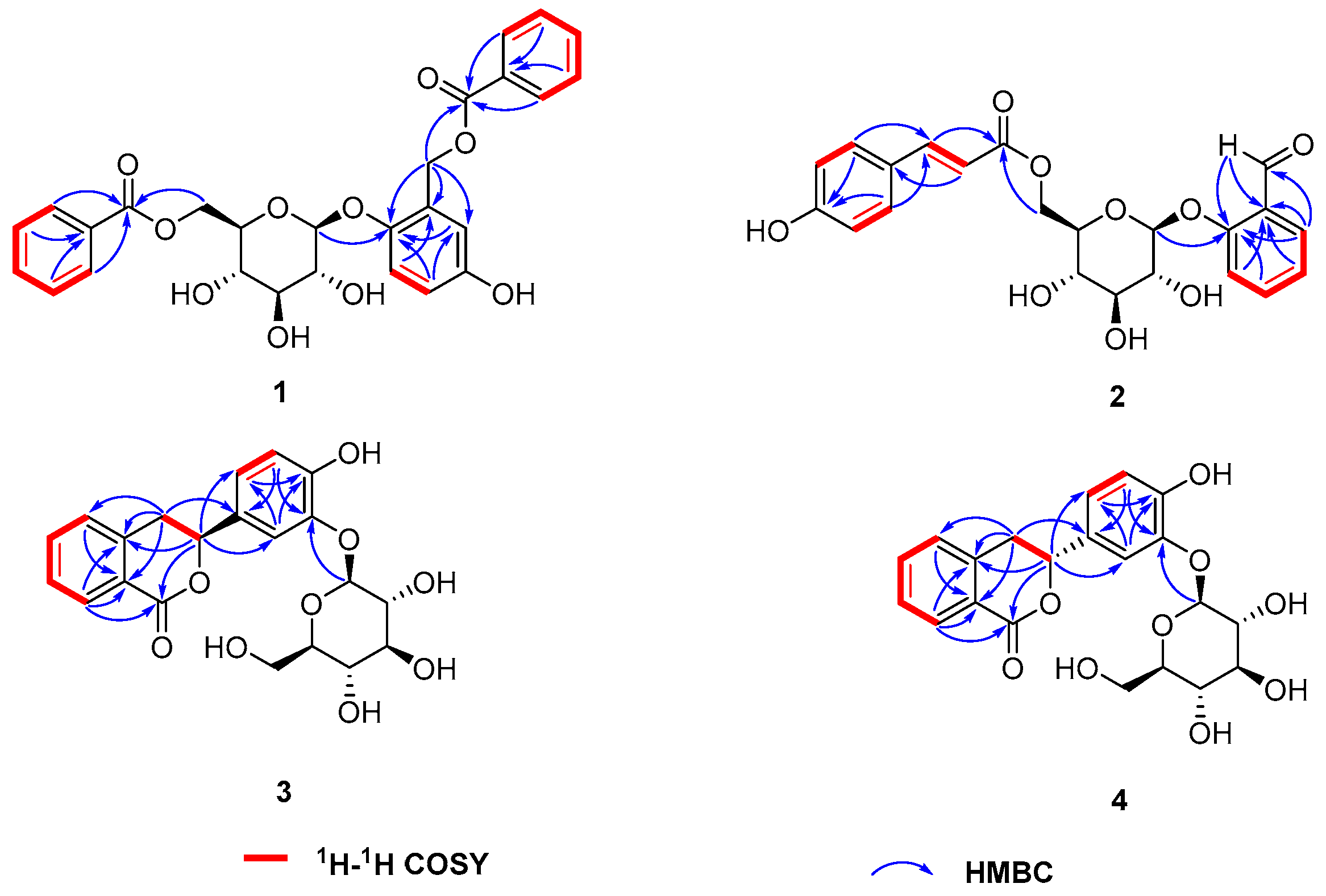
2. Results and Discussion
3. Experimental Section
3.1. General Experiment Procedure
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Acid Hydrolysis of Compounds 1–4
3.5. Inhibitory Assay of NO Production
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fan, G.S. A preliminary study on Flacourtiaceae from China. J. Wuhan Bot. Res. 1990, 8, 131–141. [Google Scholar]
- Ekabo, O.A.; Farnsworth, N.R.; Santisuk, T.; Reutrakul, V. A phytochemical investigation of Homalium ceylanicum. J. Nat. Prod. 1993, 56, 699–707. [Google Scholar] [CrossRef] [PubMed]
- Ekabo, O.A.; Farnsworth, N.R.; Santisuk, T.; Reutrakul, V. Phenolic, iridoid and ionyl glycosides from Homalium ceylanicum. Phytochemistry 1993, 32, 747–754. [Google Scholar] [CrossRef]
- Shaari, K.; Waterman, P.G. Glucosides of 2,5-dihydroxybenzyl alcohol from Homalium longifolium. Phytochemistry 1995, 39, 1415–1421. [Google Scholar] [CrossRef]
- Okokon, J.E.; Okokon, P.J.; Dar Farooq, A.; Choudhary, M.I. Anti-inflammatory and antinociceptive activities of Homalium letestui. Pharm. Biol. 2013, 51, 1459–1466. [Google Scholar] [CrossRef] [PubMed]
- Shaari, K.; Waterman, P.G. D: A-friedo-oleanane triterpenes from the stem of Homalium longifolium. Phytochemistry 1996, 41, 867–869. [Google Scholar] [CrossRef]
- Charubala, R.; Guggisberg, A.; Hesse, M.; Schmid, H. Natural occurrence of 3-phenyl-isocoumarin. Helv. Chim. Acta 1974, 57, 1096–1097. [Google Scholar] [CrossRef]
- Govindachari, T.R.; Parthasarathy, P.C.; Desai, H.K.; Ramachandran, K.S. Homalicine and (-)-dihydrohomalicine, two new isocoumarin glucosides from Homalium zeylanicum. Indian J. Chem. 1975, 13, 537–540. [Google Scholar]
- Pais, M.; Sarfati, R.; Jarreau, F.X.; Goutarel, R. Homalium alkaloids: Structure of homaline, hopromine, hoprominol and hopromalinol. Tetrahedron 1973, 29, 1001–1010. [Google Scholar]
- Wu, S.Y.; Fu, Y.H.; Chen, G.Y.; Li, X.B.; Zhou, Q.; Han, C.R.; Du, X.J.; Xie, M.L.; Yao, G.G. Cytotoxic xanthene derivatives from Homalium paniculiflorum. Phytochem. Lett. 2015, 11, 236–239. [Google Scholar] [CrossRef]
- Song, X.P.; Han, C.R.; Yu, J.; Yin, Q.W.; Xu, J.F.; Song, H.W.; Song, X.M. Chemical constituents from the stems of Homalium paniculiflorum. Chin. J. Exp. Tradit. Med. Formulae 2013, 19, 85–88. [Google Scholar]
- Ghanadian, M.; Sadraei, H.; Yousuf, S.; Asghari, G.; Choudhary, M.I.; Jahed, M. New diterpene polyester and phenolic compounds from Pycnocycla spinosa Decne. Ex Boiss with relaxant effects on KCl-induced contraction in rat ileum. Phytochem. Lett. 2014, 7, 57–61. [Google Scholar] [CrossRef]
- Plourde, G.L.; Fisher, B.B. Synthesis of 6-methoxy-1-oxaspiro [4,5] deca-6,9-diene-8-ne. Molecules 2002, 7, 315–319. [Google Scholar] [CrossRef]
- Pettit, G.R.; Melody, N.; Thornhill, A.; Knight, J.C.; Groy, T.L.; Herald, C.L. Antineoplastic agents. 579. Synthesis and cancer cell growth evaluation of E-stilstatin 3: A resveratrol structural modification. J. Nat. Prod. 2009, 72, 1637–1642. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.R.; Jiang, H.E.; Zhao, Y.X.; Zhou, J.; Qian, J.F. Components of the heartwood of Populus euphratica from an ancient tomb. Chem. Nat. Compd. 2008, 44, 6–9. [Google Scholar] [CrossRef]
- Hacıbekiroğlu, I.; Kolak, U. Antioxidant and anticholinesterase constituents from the petroleum ether and chloroform extracts of Iris suaveolens. Phytother. Res. 2011, 25, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Bock, K.; Rein, S. The substrate specificity of amyloglucosidase (AMG). Part IV. Hydroxycyclohexyl glucosides. Acta Chem. Scand. 1989, 43, 373–380. [Google Scholar] [CrossRef]
- Kisiel, W.; Barszcz, B. Phenolics from Crepis rhoeadifolia. Fitoterapia 1996, 67, 284–285. [Google Scholar]
- Gibbons, S.; Gray, A.I.; Waterman, P.G. Poliothrysins: Novel hydroxycyclohexenone esters from the leaves of Poliothrysis sinensis. Nat. Prod. Lett. 1993, 3, 59–64. [Google Scholar] [CrossRef]
- Chai, X.Y.; Xu, Z.R.; Ren, H.Y. Itosides A–I, new phenolic glycosides from Itoa orientalis. Helv. Chim. Acta 2007, 90, 2176–2185. [Google Scholar] [CrossRef]
- Kim, C.S.; Subedi, L.; Park, K.J.; Kim, S.Y.; Choi, S.U.; Kim, K.H.; Lee, K.R. Salicin derivatives from Salix glandulosa and their biological activities. Fitoterapia 2015, 106, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, M.; Matsuda, H.; Shimoda, H.; Shimada, H.; Harada, E.; Naitoh, Y.; Miki, A.; Yamahara, J.; Murskami, N. Development of bioactive functions in hydrangeae dulcis folium. V. On the antiallergic and antimicrobial principles of hydrangeae dulcis folium. (2). Thunberginols C, D and E, thunberginol G 3′-O-glucoside, (-)-hydrangenol 4′-O-glucoside and (+)-hydrangenol 4′-O-glucoside. Chem. Pharm. Bull. 1996, 44, 1440–1447. [Google Scholar] [PubMed]
- Kurosaki, Y.; Fukuda, T.; Iwao, M. Asymmetric synthesis of 3-substituted 3,4-dihydroisocoumarins via stereoselective addition of laterally lithiated chiral 2-(O-tolyl) oxazolines to aldehydes followed by diastereomer-selective lactonization. Tetrahedron 2005, 61, 3289–3303. [Google Scholar] [CrossRef]
- Chen, J.; Zhou, L.; Tan, C.K.; Yeung, Y.Y. An enantioselective approach toward 3,4-dihydroisocoumarin through the bromocyclization of styrene-type carboxylic acids. J. Org. Chem. 2011, 77, 999–1009. [Google Scholar] [CrossRef] [PubMed]
- Reif, D.W.; Mccreedy, S.A. N-nitro-l-arginine and N-monomethyl-l-arginine exhibit a different pattern of inactivation toward the three nitric oxide synthases. Arch. Biochem. Biophys. 1995, 320, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 1–18 are available from the authors. |
| Position | Compound 1 | Compound 2 | ||
|---|---|---|---|---|
| δHa | δCb | δHa | δCb | |
| 1 | 129.4 s | 127.5 s | ||
| 2 | 149.7 s | 161.3 s | ||
| 3 | 7.07 (1H, d, 8.0) | 119.9 d | 7.31 (1H, d, 7.6) | 118.2 d |
| 4 | 6.55 (1H, dd, 8.0, 2.0) | 116.4 d | 7.50 (1H, dd, 7.6, 7.6) | 137.1 d |
| 5 | 154.3 s | 7.08 (1H, dd, 7.6, 7.6) | 123.8 d | |
| 6 | 6.83 (1H, d, 2.0) | 116.2 d | 7.76 (1H, d, 7.6) | 128.7 d |
| 7α | 5.46 (1H, d, 13.2) | 63.1 t | 10.50 (1H, s) | 191.8 d |
| 7β | 5.41 (1H, d, 13.2) | |||
| 1′ | 131.3 s | 127.1 s | ||
| 2′ | 7.98 (1H, d, 7.6) | 130.6 d | 7.46 (1H, d, 8.4) | 131.2 d |
| 3′ | 7.45 (1H, dd, 7.6, 7.6) | 129.6 d | 6.82 (1H, d, 8.4) | 116.9 d |
| 4′ | 7.59 (1H, dd, 7.6, 7.6) | 134.2 d | 161.5 s | |
| 5′ | 7.45 (1H, dd, 7.6, 7.6) | 129.6 d | 6.82 (1H, d, 8.4) | 116.9 d |
| 6′ | 7.98 (1H, d, 7.6) | 130.6 d | 7.46 (1H, d, 8.4) | 131.2 d |
| 7′ | 167.8 s | 7.61 (1H, d, 16.0) | 146.8 d | |
| 8′ | 6.33 (1H, d, 16.0) | 114.9 d | ||
| 9′′ | 168.9 s | |||
| 1′′ | 4.79 (1H, d, 7.6) | 104.7 d | 5.05 (1H, d, 7.6) | 102.7 d |
| 2′′ | 3.53–3.49 (1H, m) | 75.0 d | 3.61–3.57 (1H, m) | 74.8 d |
| 3′′ | 3.49–3.46 (1H, m) | 78.0 d | 3.55–3.50 (1H, m) | 77.9 d |
| 4′′ | 3.45–3.40 (1H, m) | 72.1 d | 3.47–3.43 (1H, m) | 71.7 d |
| 5′′ | 3.71–3.67 (1H, m) | 75.5 d | 3.78–3.74 (1H, m) | 75.8 d |
| 6′′α | 4.69 (1H, dd, 11.6, 1.6) | 65.3 t | 4.53 (1H, dd, 11.6, 1.6) | 64.5 t |
| 6′′β | 4.40 (1H, dd, 11.6, 7.6) | 4.39 (1H, dd, 11.6, 6.8) | ||
| 1′′′ | 131.5 s | |||
| 2′′′ | 8.02 (1H, d, 7.6) | 130.6 d | ||
| 3′′′ | 7.48 (1H, dd, 7.6, 7.6) | 129.6 d | ||
| 4′′′ | 7.61 (1H, dd, 7.6, 7.6) | 134.3 d | ||
| 5′′′ | 7.48 (1H, dd, 7.6, 7.6) | 129.6 d | ||
| 6′′′ | 8.02 (1H, d, 7.6) | 130.6 d | ||
| 7′′′ | 168.0 s | |||
| Position | Compound 3 | Compound 4 | ||
|---|---|---|---|---|
| δHa | δCb | δHa | δCb | |
| 1 | 164.9 s | 164.9 s | ||
| 2 | ||||
| 3 | 6.04 (1H, dd, 11.6, 3.2) | 74.2 d | 5.94 (1H, dd, 11.6, 3.2) | 74.3 d |
| 4α | 3.27–3.32 (1H, m) | 33.7 t | 3.40–3.43 (1H, m) | 33.2 t |
| 4β | 3.12–3.15 (1H, m) | 3.15–3.17 (1H, m) | ||
| 4a | 140.0 s | 140.0 s | ||
| 5 | 7.45 (1H, d, 7.6) | 127.5 d | 7.39 (1H, d, 7.6) | 127.5 d |
| 6 | 7.65 (1H, dd, 7.6, 7.6) | 133.9 d | 7.64 (1H, dd, 7.6, 7.6) | 133.9 d |
| 7 | 7.48 (1H, dd, 7.6, 7.6) | 127.8 d | 7.47 (1H, dd, 7.6, 7.6) | 128.0 d |
| 8 | 7.99 (1H, d, 7.6) | 129.3 d | 7.97 (1H, d, 7.6) | 129.2 d |
| 8a | 124.8 s | 124.7 s | ||
| 1′ | 129.9 s | 129.3 s | ||
| 2′ | 6.92 (1H, d, 2.0) | 112.8 d | 6.92 (1H, d, 2.0) | 112.7 d |
| 3′ | 146.9 s | 147.1 s | ||
| 4′ | 152.7 s | 152.5 s | ||
| 5′ | 7.02 (1H, d, 8.2) | 118.2 d | 7.09 (1H, d, 8.2) | 117.6 d |
| 6′ | 6.72 (1H, dd, 8.2, 2.0) | 115.7 d | 6.70 (1H, dd, 8.2, 2.0) | 115.5 d |
| 1′′ | 4.59 (1H, d, 7.6) | 103.5 d | 4.65 (1H, d, 7.6) | 102.6 d |
| 2′′ | 3.15–3.13 (1H, m) | 73.3 d | 3.16–3.14 (1H, m) | 73.4 d |
| 3′′ | 3.22–3.19 (1H, m) | 77.1 d | 3.23–3.19 (1H, m) | 77.1 d |
| 4′′ | 3.10–3.05 (1H, m) | 69.9 d | 3.11–3.04 (1H, m) | 69.8 d |
| 5′′ | 3.26–3.23 (1H, m) | 76.3 d | 3.26–3.24 (1H, m) | 76.6 d |
| 6′′α | 3.74 (1H, dd, 11.6, 5.2) | 61.0 t | 3.69 (1H, dd, 11.6, 5.2) | 60.8 t |
| 6′′β | 3.43 (1H, dd, 11.6, 6.0) | 3.47 (1H, dd, 11.6, 6.0) | ||
| 4′-OH | 9.28 (1H, s) | 9.27 (1H, s) | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, S.-Y.; Fu, Y.-H.; Zhou, Q.; Bai, M.; Chen, G.-Y.; Zhao, S.-Y.; Han, C.-R.; Song, X.-P. Bioactive Phenolic and Isocoumarin Glycosides from the Stems of Homalium paniculiflorum. Molecules 2018, 23, 472. https://doi.org/10.3390/molecules23020472
Wu S-Y, Fu Y-H, Zhou Q, Bai M, Chen G-Y, Zhao S-Y, Han C-R, Song X-P. Bioactive Phenolic and Isocoumarin Glycosides from the Stems of Homalium paniculiflorum. Molecules. 2018; 23(2):472. https://doi.org/10.3390/molecules23020472
Chicago/Turabian StyleWu, Shou-Yuan, Yan-Hui Fu, Qi Zhou, Meng Bai, Guang-Ying Chen, Si-Yu Zhao, Chang-Ri Han, and Xiao-Ping Song. 2018. "Bioactive Phenolic and Isocoumarin Glycosides from the Stems of Homalium paniculiflorum" Molecules 23, no. 2: 472. https://doi.org/10.3390/molecules23020472