Antibacterial Activities of Azole Complexes Combined with Silver Nanoparticles
Abstract
1. Introduction
2. Discussion
2.1. Infrared Spectroscopy
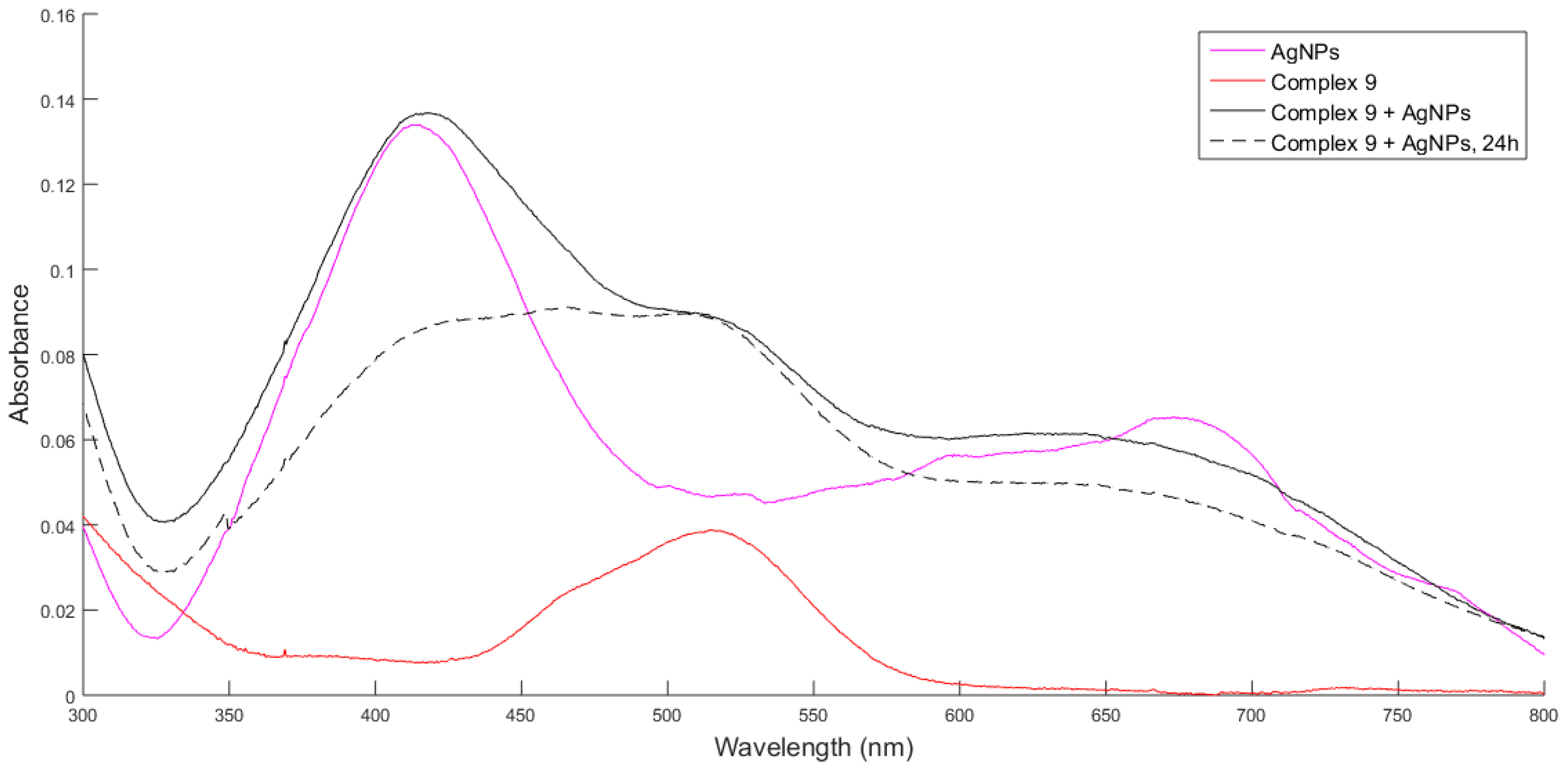
2.2. UV-Vis Spectroscopy
2.3. Theoretical Calculations
2.4. Thermal Analysis
2.5. Characterization of AgNPs
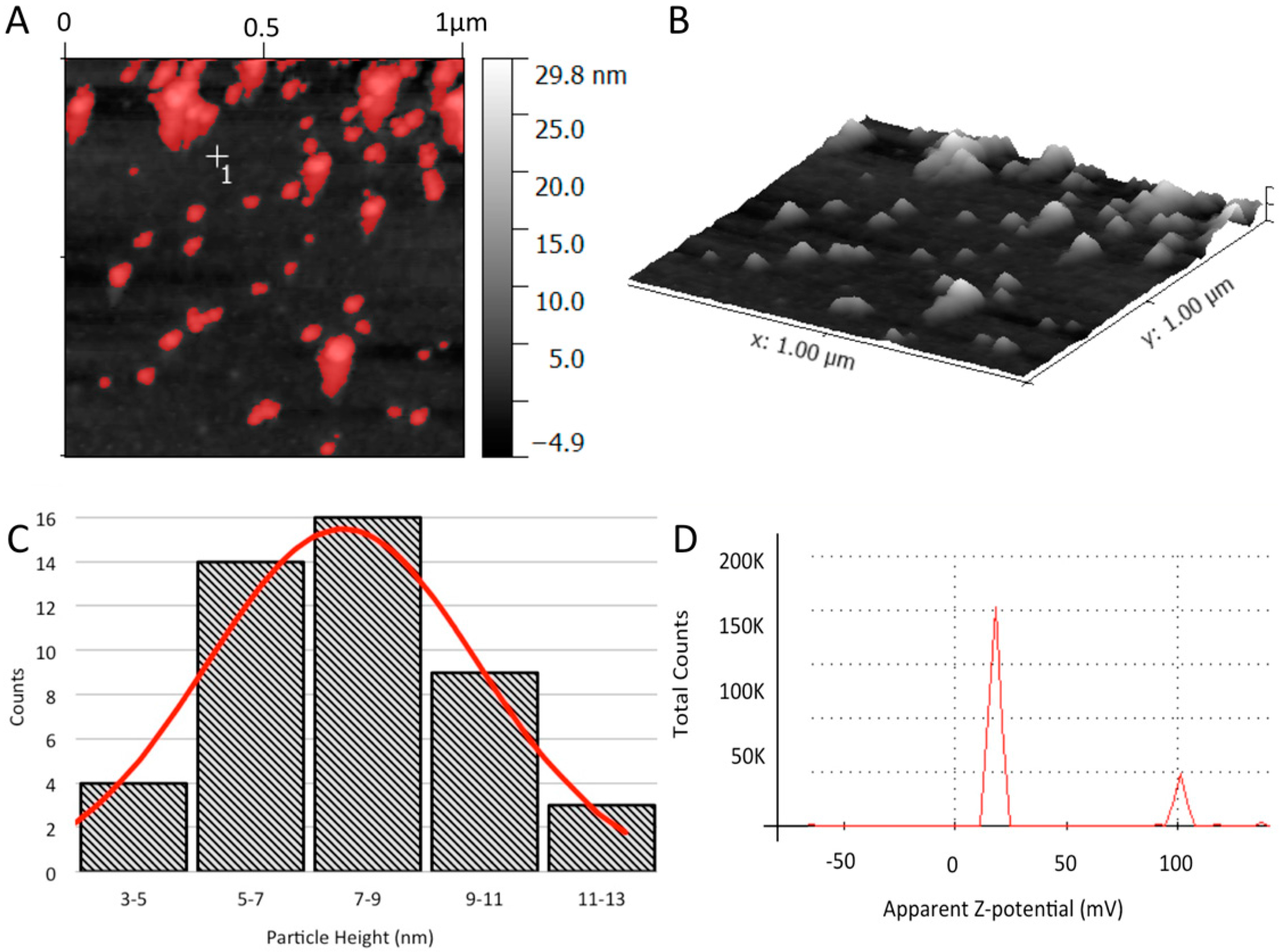
2.5.1. Atomic Force Microscopy (AFM)
2.5.2. Dynamic Light Scattering (DLS)
2.6. Biological Activity
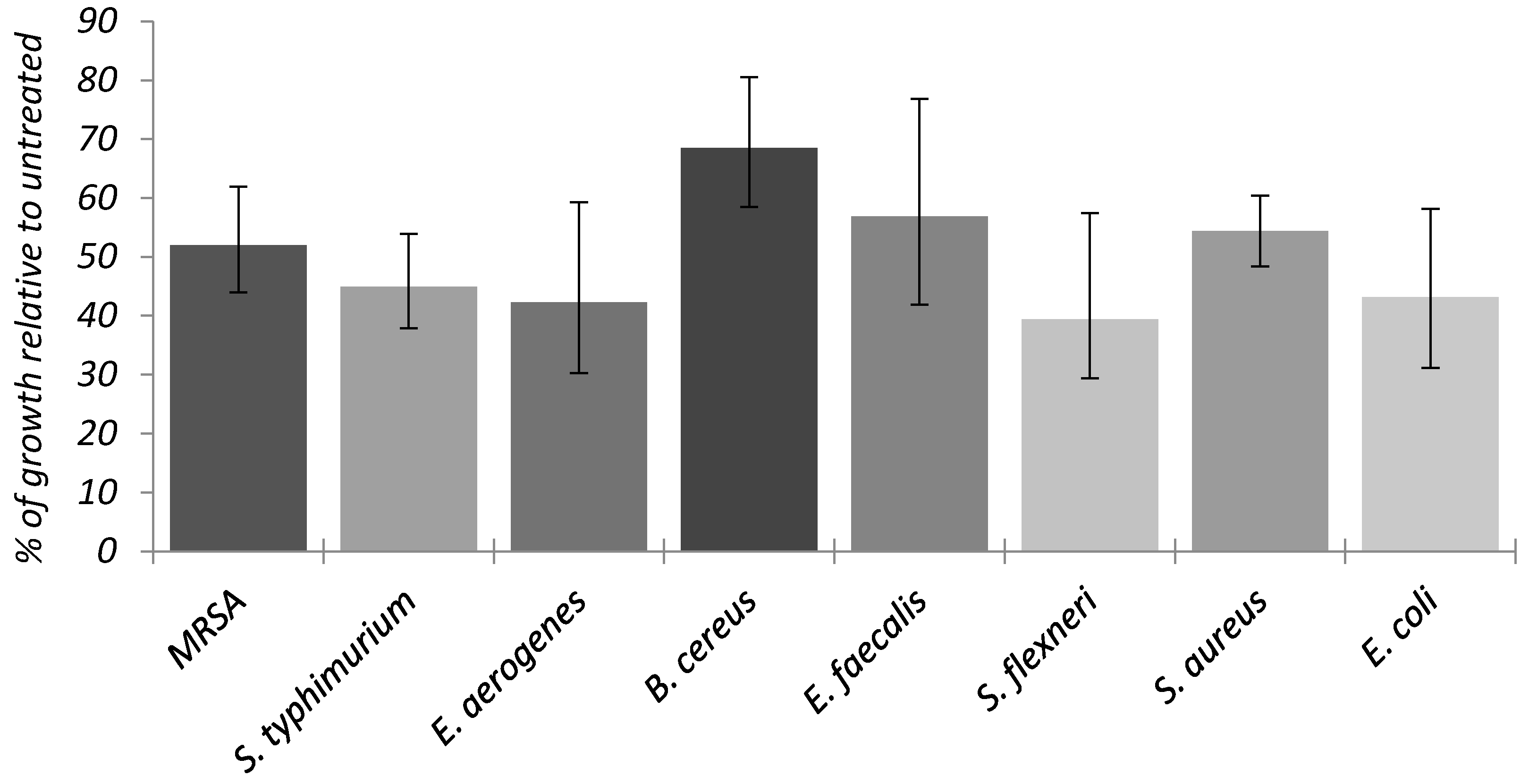
2.6.1. Antibacterial Activity of the AgNPs Only
2.6.2. Antibacterial Activity of the Azole Complexes with and without AgNPs
2.7. Physicochemical Characterization of the Colloids
2.8. Cytotoxicity
3. Materials and Methods
3.1. General Information
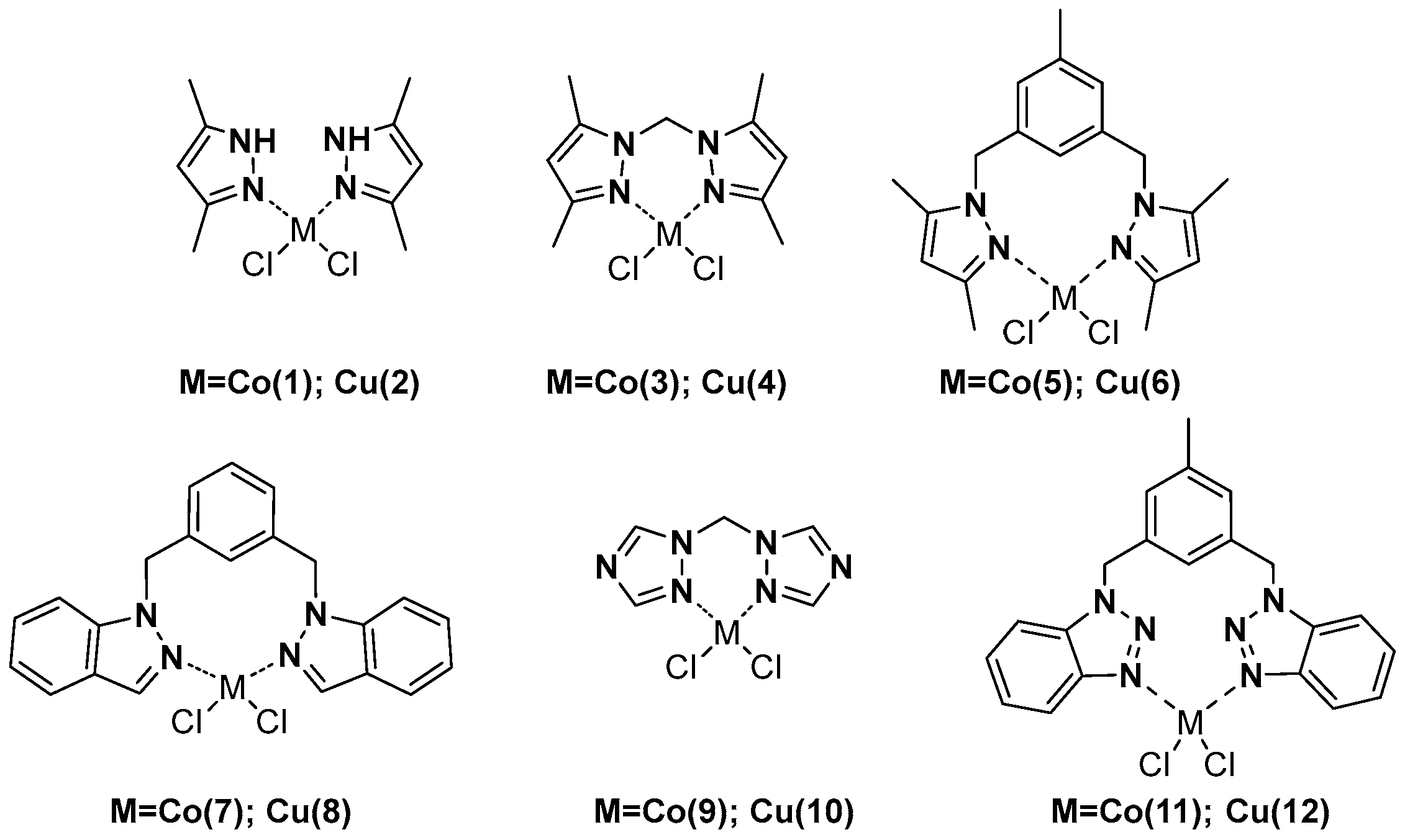
3.2. Synthesis of the Complexes
3.2.1. Dichloro[3,5-bis(3,5-dimethylpyrazol-1-ylmethyl)toluene-NN]cobalt(II) (5)
3.2.2. Dichloro[3,5-bis(3,5-dimethylpyrazol-1-ylmethyl)toluene-NN]copper(II) (6)
3.2.3. Dichloro[1,3-bis(indazol-1-ylmethyl)benzene-NN]cobalt(II) (7)
3.2.4. Dichloro[1,3-bis(indazol-1-ylmethyl)benzene-NN]copper(II) (8)
3.2.5. Dichloro[3,5-bis(benzotriazol-1-ylmethyl)toluene-NN]cobalt(II) (11)
3.2.6. Dichloro[3,5-bis(benzotriazol-1-ylmethyl)toluene-NN]copper(II) (12)
3.3. Characterization of AgNPs
3.3.1. Atomic Force Microscopy (AFM)
3.3.2. Physicochemical Characterization of the Colloids
3.4. In Vitro Antibacterial Activity
3.4.1. Reagents
3.4.2. Strains and Isolates
3.4.3. Antibacterial Activities
3.4.4. Mammalian-Cell Cytotoxicity Assay
3.4.5. Growth Inhibition of the Bacteria Strains
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AgNPs | Silver nanoparticles |
| WHO | World Health Organization; |
| CDC | Communicable Disease Center |
| FDA | Food and Drug Administration |
| MRSA | Methicillin-resistant Staphylococcus aureus |
| E. aerogenes | Enterobacter aerogenes |
| S. typhimurium | Salmonella typhimurium |
| S. aureus | Staphylococcus aureus |
| E. faecalis | Enterococcus faecalis |
| B. cereus | Bacillus cereus |
| E. coli | Escherichia coli |
| S. flexneri | Shigella flexneri |
| MBC | Minimum Bactericidal Concentration |
| TGA | Thermogravimetric analysis |
| MIC50 | Minimum inhibitory concentration 50 |
| THF | Tetrahydrofuran |
| CH3CN | Acetonitrile |
| FTIR | Fourier-transform infrared spectroscopy |
| HCl | Hydrochloric acid |
| UV-Vis | Ultraviolet-visible spectroscopy |
| MeOH | Methanol |
| DMSO | Dimethylsulfoxide |
| AFM | Atomic force microscopy |
| PVP | Polyvinylpyrrolidone |
| MTT | (3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) tetrazolium |
| DLS | Dynamic Light Scattering |
| HHF | Human Foreskin Fibroblast |
| KBr | Potassium bromide |
| DCM | Dichloromethane |
| DMF | Dimethylformamide |
References
- Lok, C.-N.; Ho, C.-M.; Chen, R.; He, Q.-Y.; Yu, W.-Y.; Sun, H.; Tam, P.K.-H.; Chiu, J.-F.; Che, C.-M. Silver nanoparticles: Partial oxidation and antibacterial activities. J. Biol. Inorg. Chem. 2007, 12, 527–534. [Google Scholar] [CrossRef] [PubMed]
- Franci, G.; Falanga, A.; Galdiero, S.; Palomba, L.; Rai, M.; Morelli, G.; Galdiero, M. Silver Nanoparticles as Potential Antibacterial Agents. Molecules 2015, 20, 8856–8874. [Google Scholar] [CrossRef] [PubMed]
- Naqvi, S.Z.; Kiran, U. Combined efficacy of biologically synthesized silver nanoparticles and different antibiotics against multidrug-resistant bacteria. Int. J. Nanomed. 2013, 8, 3187–3189. [Google Scholar] [CrossRef] [PubMed]
- Allahverdiyev, A.M.; Kon, K.V.; Abamor, E.S.; Bagirova, M.; Rafailovich, M. Coping with antibiotic resistance: Combining nanoparticles with antibiotics and other antimicrobial agents. Expert Rev. Anti Infect. Ther. 2011, 9, 1035–1052. [Google Scholar] [CrossRef] [PubMed]
- Pelgrift, R.Y.; Friedman, A.J. Nanotechnology as a therapeutic tool to combat microbial resistance. Adv. Drug Deliv. Rev. 2013, 65, 1803–1815. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-F.; Choi, Y.-J.; Han, J.W.; Kim, E.; Park, J.H.; Gurunathan, S.; Kim, J.-H. Differential nanoreprotoxicity of silver nanoparticles in male somatic cells and spermatogonial stem cells. Int. J. Nanomed. 2015, 10, 1335–1357. [Google Scholar] [CrossRef]
- Sass, J.; Heine, L.; Hwang, N. Use of a modified GreenScreen tool to conduct a screening-level comparative hazard assessment of conventional silver and two forms of nanosilver. Environ. Health 2016, 15, 105. [Google Scholar] [CrossRef] [PubMed]
- Castillo, K.F.; Bello-Vieda, N.J.; Nuñez-Dallos, N.G.; Pastrana, H.F.; Celis, A.M.; Restrepo, S.; Hurtado, J.J.; Ávila, A.G. Metal Complex Derivatives of Azole: A Study on Their Synthesis, Characterization, and Antibacterial and Antifungal Activities. J. Braz. Chem. Soc. 2016, 27, 1–14. [Google Scholar] [CrossRef]
- Singh, K.; Kumar, Y.; Puri, P.; Kumar, M.; Sharma, C. Cobalt, nickel, copper and zinc complexes with 1,3-diphenyl-1H-pyrazole-4-carboxaldehyde Schiff bases: Antimicrobial, spectroscopic, thermal and fluorescence studies. Eur. J. Med. Chem. 2012, 52, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Yousef, T.A.; Abu El-Reash, G.M.; Al-Jahdali, M.; El-Rakhawy, E.-B.R. Synthesis, spectral characterization and biological evaluation of Mn(II), Co(II), Ni(II), Cu(II), Zn(II) and Cd(II) complexes with thiosemicarbazone ending by pyrazole and pyridyl rings. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2014, 129, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Pahontu, E.; Fala, V.; Gulea, A.; Poirier, D.; Tapcov, V.; Rosu, T. Synthesis and Characterization of Some New Cu(II), Ni(II) and Zn(II) Complexes with Salicylidene Thiosemicarbazones: Antibacterial, Antifungal and in Vitro Antileukemia Activity. Molecules 2013, 18, 8812–8836. [Google Scholar] [CrossRef] [PubMed]
- Montazerozohori, M.; Mojahedi Jahromi, S.; Masoudiasl, A.; McArdle, P. Nano structure zinc (II) Schiff base complexes of a N3-tridentate ligand as new biological active agents: Spectral, thermal behaviors and crystal structure of zinc azide complex. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2015, 138, 517–528. [Google Scholar] [CrossRef] [PubMed]
- Atkins, P.W.; Overton, T.; Rourke, J.; Weller, M.; Armstrong, F. Shriver and Atkins’ Inorganic Chemistry; Oxford University Press: Oxford, UK, 2010; ISBN 9780199236176. [Google Scholar]
- Cotton, F.A.; Wilkinson, G.; Murillio, C.A.; Bochmann, M. Advanced Inorganic Chemistry; Wiley: Hoboken, NJ, USA, 1999; ISBN 9788126513383. [Google Scholar]
- Arab Ahmadi, R.; Hasanvand, F.; Bruno, G.; Amiri Rudbari, H.; Amani, S. Synthesis, Spectroscopy, and Magnetic Characterization of Copper(II) and Cobalt(II) Complexes with 2-Amino-5-bromopyridine as Ligand. ISRN Inorg. Chem. 2013, 2013, 1–7. [Google Scholar] [CrossRef]
- Navarro-Ranninger, M.C.; Alvarez-Valdés, A.; Camazón, M.J.; Masaguer, J.R. Complexes of copper(II) with substituted benzylideneamines. Inorg. Chim. Acta 1987, 132, 7–10. [Google Scholar] [CrossRef]
- Zhou, Y.; Kong, Y.; Kundu, S.; Cirillo, J.D.; Liang, H. Antibacterial activities of gold and silver nanoparticles against Escherichia coli and bacillus Calmette-Guérin. J. Nanobiotechnol. 2012, 10, 19. [Google Scholar] [CrossRef] [PubMed]
- Rai, M.; Yadav, A.; Gade, A. Silver nanoparticles as a new generation of antimicrobials. Biotechnol. Adv. 2009, 27, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yang, M.; Portney, N.G.; Cui, D.; Budak, G.; Ozbay, E.; Ozkan, M.; Ozkan, C.S. Zeta potential: A surface electrical characteristic to probe the interaction of nanoparticles with normal and cancer human breast epithelial cells. Biomed. Microdevices 2008, 10, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Honary, S.; Zahir, F. Effect of Zeta Potential on the Properties of Nano-Drug Delivery Systems—A Review (Part 2). Trop. J. Pharm. Res. 2013, 12, 265. [Google Scholar] [CrossRef]
- Gottenbos, B.; Grijpma, D.W.; van der Mei, H.C.; Feijen, J.; Busscher, H.J. Antimicrobial effects of positively charged surfaces on adhering Gram-positive and Gram-negative bacteria. J. Antimicrob. Chemother. 2001, 48, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Agnihotri, S.; Mukherji, S.; Mukherji, S. Size-controlled silver nanoparticles synthesized over the range 5–100 nm using the same protocol and their antibacterial efficacy. RSC Adv. 2014, 4, 3974–3983. [Google Scholar] [CrossRef]
- Elechiguerra, J.L.; Burt, J.L.; Morones, J.R.; Camacho-Bragado, A.; Gao, X.; Lara, H.H.; Yacaman, M.J. Interaction of silver nanoparticles with HIV-1. J. Nanobiotechnol. 2005, 3, 6. [Google Scholar] [CrossRef] [PubMed]
- Nair, L.S.; Laurencin, C.T. Silver Nanoparticles: Synthesis and Therapeutic Applications. J. Biomed. Nanotechnol. 2007, 3, 301–316. [Google Scholar] [CrossRef]
- Van den Bossche, H.; Willemsens, G.; Cools, W.; Marichal, P.; Lauwers, W. Hypothesis on the molecular basis of the antifungal activity of N-substituted imidazoles and triazoles. Biochem. Soc. Trans. 1983, 11, 665–667. [Google Scholar] [CrossRef] [PubMed]
- Herbrecht, R. Voriconazole: Therapeutic review of a new azole antifungal. Expert Rev. Anti Infect. Ther. 2004, 2, 485–497. [Google Scholar] [CrossRef] [PubMed]
- Fang, B.; Zhou, C.-H.; Rao, X.-C. Synthesis and biological activities of novel amine-derived bis-azoles as potential antibacterial and antifungal agents. Eur. J. Med. Chem. 2010, 45, 4388–4398. [Google Scholar] [CrossRef] [PubMed]
- Shelke, S.; Mhaske, G.; Gadakh, S.; Gill, C. Green synthesis and biological evaluation of some novel azoles as antimicrobial agents. Bioorg. Med. Chem. Lett. 2010, 20, 7200–7204. [Google Scholar] [CrossRef] [PubMed]
- Khabnadideh, S.; Rezaei, Z.; Khalafi-Nezhad, A.; Bahrinajafi, R.; Mohamadi, R.; Farrokhroz, A.A. Synthesis of N-Alkylated derivatives of imidazole as antibacterial agents. Bioorg. Med. Chem. Lett. 2003, 13, 2863–2865. [Google Scholar] [CrossRef]
- Bayrak, H.; Demirbas, A.; Demirbas, N.; Karaoglu, S.A. Synthesis of some new 1,2,4-triazoles starting from isonicotinic acid hydrazide and evaluation of their antimicrobial activities. Eur. J. Med. Chem. 2009, 44, 4362–4366. [Google Scholar] [CrossRef] [PubMed]
- Heerding, D.A.; Chan, G.; DeWolf, W.E.; Fosberry, A.P.; Janson, C.A.; Jaworski, D.D.; McManus, E.; Miller, W.H.; Moore, T.D.; Payne, D.J.; et al. 1,4-Disubstituted imidazoles are potential antibacterial agents functioning as inhibitors of enoyl acyl carrier protein reductase (FabI). Bioorg. Med. Chem. Lett. 2001, 11, 2061–2065. [Google Scholar] [CrossRef]
- Kljun, J.; Scott, A.J.; Lanišnik Rižner, T.; Keiser, J.; Turel, I. Synthesis and Biological Evaluation of Organoruthenium Complexes with Azole Antifungal Agents. First Crystal Structure of a Tioconazole Metal Complex. Organometallics 2014, 33, 1594–1601. [Google Scholar] [CrossRef]
- Kabbani, A.T.; Hammud, H.H.; Ghannoum, A.M. Preparation and antibacterial activity of copper and cobalt complexes of 4-chloro-3-nitrobenzoate with a nitrogen donor ligand. Chem. Pharm. Bull. (Tokyo) 2007, 55, 446–450. [Google Scholar] [CrossRef] [PubMed]
- Silver, S. Bacterial resistances to toxic metal ions—A review. Gene 1996, 179, 9–19. [Google Scholar] [CrossRef]
- Chandra, S.; Raizada, S.; Tyagi, M.; Gautam, A. Synthesis, spectroscopic, and antimicrobial studies on bivalent nickel and copper complexes of bis(thiosemicrbazone). Bioinorg. Chem. Appl. 2007, 2007, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Atria, A.M.; Cortés-Cortés, P.; Garland, M.T.; Baggio, R.; Morales, K.; Soto, M.; Corsini, G. X-ray studies and antibacterial activity in copper and cobalt complexes with imidazole derivative ligands. J. Chil. Chem. Soc. 2011, 56, 786–792. [Google Scholar] [CrossRef]
- Collin, F.; Karkare, S.; Maxwell, A. Exploiting bacterial DNA gyrase as a drug target: Current state and perspectives. Appl. Microbiol. Biotechnol. 2011, 92, 479–497. [Google Scholar] [CrossRef] [PubMed]
- Boni, A.; Pampaloni, G.; Peloso, R.; Belletti, D.; Graiff, C.; Tiripicchio, A. Synthesis of copper(I) bis(3,5-dimethylpyrazolyl)methane olefin complexes and their reactivity towards carbon monoxide. J. Organomet. Chem. 2006, 691, 5602–5609. [Google Scholar] [CrossRef]
- Juliá, S.; Sala, P.; Del Mazo, J.; Sancho, M.; Ochoa, C.; Elguero, J.; Fayet, J.-P.; Vertut, M.-C. N-polyazolylmethanes. 1. Synthesis and nmr study of N,N′-diazolylmethanes. J. Heterocycl. Chem. 1982, 19, 1141–1145. [Google Scholar] [CrossRef]
- Hurtado, J.; Portaluppi, M.; Quijada, R.; Rojas, R.; Valderrama, M. Synthesis, characterization, and reactivity studies in ethylene polymerization of cyclometalated palladium(II) complexes containing terdentate ligands with N,C,N-donors. J. Coord. Chem. 2009, 62, 2772–2781. [Google Scholar] [CrossRef]
- Hurtado, J.; Rojas, R.S.; Pérez, E.G.; Valderrama, M. Palladium complex bearing 3,5-bis(benzotriazol-1-ylmethyl)toluene ligand catalyzes oxidative amination of allyl butyl ether. J. Chil. Chem. Soc. 2013, 58, 1534–1536. [Google Scholar] [CrossRef]
- Johnson, P.B.; Christy, R.W. Optical constants of the noble metals. Phys. Rev. B 1972, 6, 4370–4379. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
Sample Availability: Samples of all the compounds are available from the authors. |


| Compound | Wavenumber ν (cm−1) | ||||
|---|---|---|---|---|---|
| (N-H) | (C-H) | (C-H)Pyr | (C-N) | (M-Cl) | |
| 1 | 3344s | 3142w | 1568vs | 1470m | 427m |
| 2 | 3265vs | 3147vs | 1570vs | 1472m | 431m |
| (C-H) | (C-CH3) | (C=N) | (C-C)Pyr | (M-Cl) | |
| 3 | 3011w | 1465s | 1051m | 1003m | 493w |
| 4 | 3027w | 1467s | 1044m | 1001m | 492w |
| (C-H) | (C-CH3) | (C-C)Pyr | (C-C-N) | (M-Cl) | |
| 5 | 3127m | 1468m | 1608m | 729m | 419w |
| 6 | 3137w | 1469m | 1608m | 738m | 420w |
| (C-H) | (C-C)Ind | (C-N)Ind | (C-H)Ind | (M-Cl) | |
| 7 | 3094m | 1628s | 1519s | 1478m | 490w |
| 8 | 3098m | 1628s | 1519s | 1477m | 497w |
| (C-H) | (H2C-N) | (N-N) | (C-N) | (M-Cl) | |
| 9 | 3115w | 1518s | 1458m | 1362w | 507vw |
| 10 | 3137s | 1525s | 1460m | 1357w | 503vw |
| (C-H) | (C-C)Tol | (N-N) | (N=N) | (M-Cl) | |
| 11 | 2970w | 1610m | 1284m | 1229s | 419w |
| 12 | 2968m | 1610m | 1288m | 1233s | 419w |
| Complex Bacteria Strain | 3 | 4 | 5 | 8 | 9 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Without AgNPs | With AgNPs * | Without AgNPs | With AgNPs * | Without AgNPs | With AgNPs * | Without AgNPs | With AgNPs * | Without AgNPs | With AgNPs * | |
| S. aureus (+) | >500 | >500 | 200 ¶ | 200 | 250 | 25 | 200 | 62.5 | 400 ¶ | 250 ¶ |
| S. flexneri (−) | 200 | 125 | 200 | 100 | 200 | 62.5 | 125 | 100 | 250 | 250 |
| MRSA (+) | 62.5 | 62.5 | 250 | 200 | 200 ¶ | 200 | 200 | 250 | >500 | N/A |
| E. faecalis (+) | 62.5 ¶ | 100 | 400 | 200 | 250 ¶ | 250 ¶ | 400 | 400 | >500 | N/A |
| E. aerogenes (−) | >500 | >500 | >500 | 400 | 400 | 250 | 400 | 250 | >500 | 500 ¶ |
| B. cereus (+) | >500 | >500 | 400 | 400 | >500 | >500 | 500 ¶ | 400 ¶ | >500 | >500 |
| E. coli (−) | 125 | 125 | >500 | 500 | 400 | 400 | 500 | 500 | >500 | 250 |
| S. typhymurium (−) | 250 ¶ | 250 ¶ | N/A | 250 ¶ | 400 | 250 | 400 ¶ | 400 ¶ | N/A | 500 ¶ |
| Complex Bacteria Strain | 3 | 4 | 5 | 8 | 9 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Without AgNPs | With AgNPs * | Without AgNPs | With AgNPs * | Without AgNPs | With AgNPs * | Without AgNPs | With AgNPs * | Without AgNPs | With AgNPs * | |
| S. aureus (+) | 100 | 100 | 100 | 100 | 250 | 250 | 200 | 200 | 400 ¶ | 200 ¶ |
| S. flexneri (−) | 500 | 250 | 100 | 50 | 200 | 125 | 100 | 50 | 200 | 200 |
| MRSA (+) | 250 | 250 | 250 | 250 | 200 | 200 | 200 | 250 | 500 ¶ | 500 ¶ |
| E. faecalis (+) | 400 | 200 | 400 | 200 | 200 | 200 | 400 | 400 | >500 | >500 |
| E. aerogenes (−) | 200 | 200 | >500 | 250 | 500 | 500 | 400 | 400 | >500 | >500 |
| B. cereus (+) | 400 | 400 | >500 | >500 | >500 | >500 | >500 | >500 | >500 | >500 |
| E. coli (−) | >500 | 400 | 200 | 200 | 400 | 400 | 400 | 400 | 500 ¶ | 250 |
| S. typhymurium (−) | 125 ¶ | 125 ¶ | >500 | 250 | 400 | 400 | 400 | 400 | >500 | >500 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bello-Vieda, N.J.; Pastrana, H.F.; Garavito, M.F.; Ávila, A.G.; Celis, A.M.; Muñoz-Castro, A.; Restrepo, S.; Hurtado, J.J. Antibacterial Activities of Azole Complexes Combined with Silver Nanoparticles. Molecules 2018, 23, 361. https://doi.org/10.3390/molecules23020361
Bello-Vieda NJ, Pastrana HF, Garavito MF, Ávila AG, Celis AM, Muñoz-Castro A, Restrepo S, Hurtado JJ. Antibacterial Activities of Azole Complexes Combined with Silver Nanoparticles. Molecules. 2018; 23(2):361. https://doi.org/10.3390/molecules23020361
Chicago/Turabian StyleBello-Vieda, Nestor J., Homero F. Pastrana, Manuel F. Garavito, Alba G. Ávila, Adriana M. Celis, Alvaro Muñoz-Castro, Silvia Restrepo, and John J. Hurtado. 2018. "Antibacterial Activities of Azole Complexes Combined with Silver Nanoparticles" Molecules 23, no. 2: 361. https://doi.org/10.3390/molecules23020361
APA StyleBello-Vieda, N. J., Pastrana, H. F., Garavito, M. F., Ávila, A. G., Celis, A. M., Muñoz-Castro, A., Restrepo, S., & Hurtado, J. J. (2018). Antibacterial Activities of Azole Complexes Combined with Silver Nanoparticles. Molecules, 23(2), 361. https://doi.org/10.3390/molecules23020361






