Evaluation of Melatonin Secretion and Metabolism Exponents in Patients with Ulcerative and Lymphocytic Colitis
Abstract
:1. Introduction
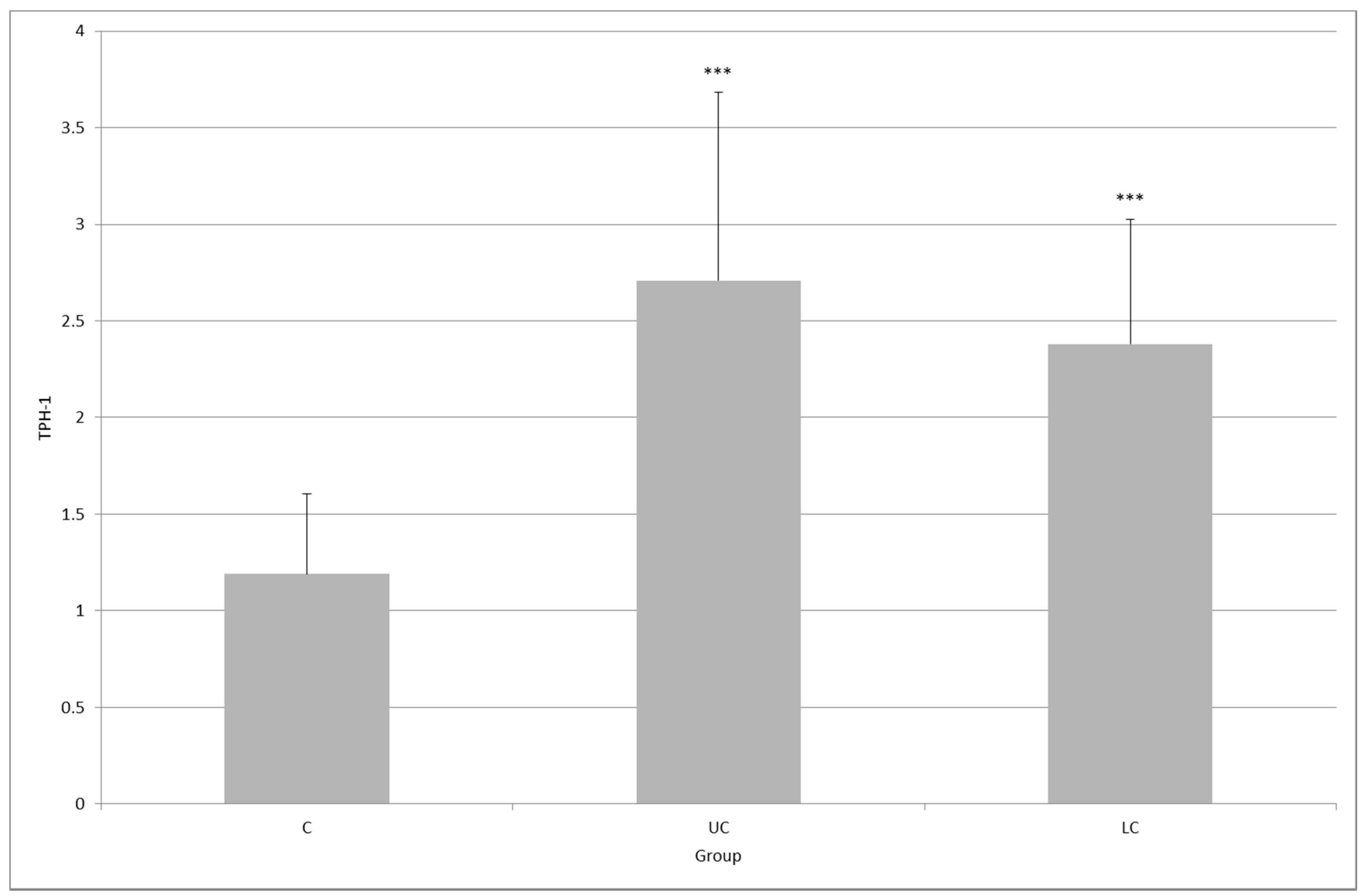
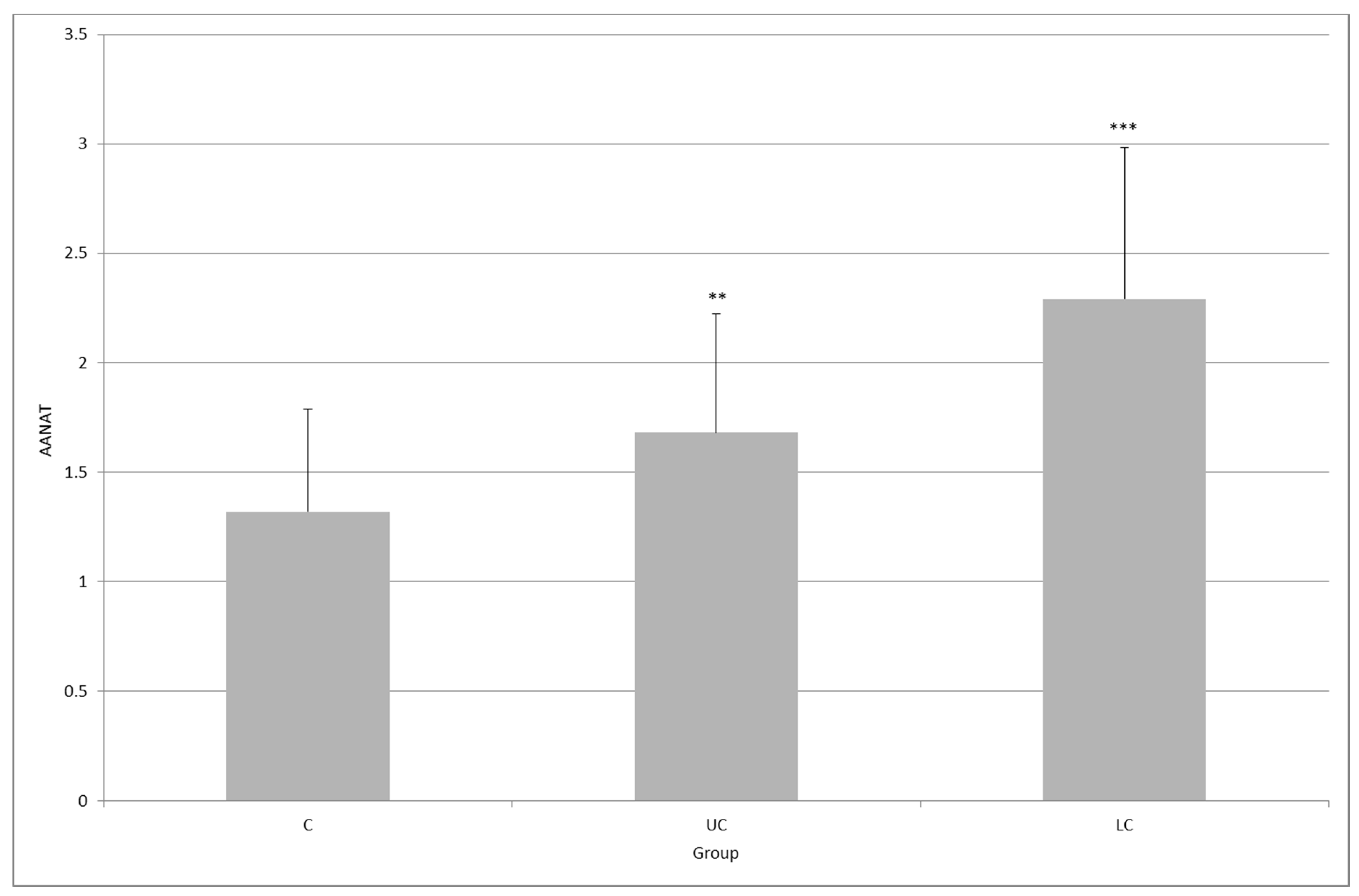
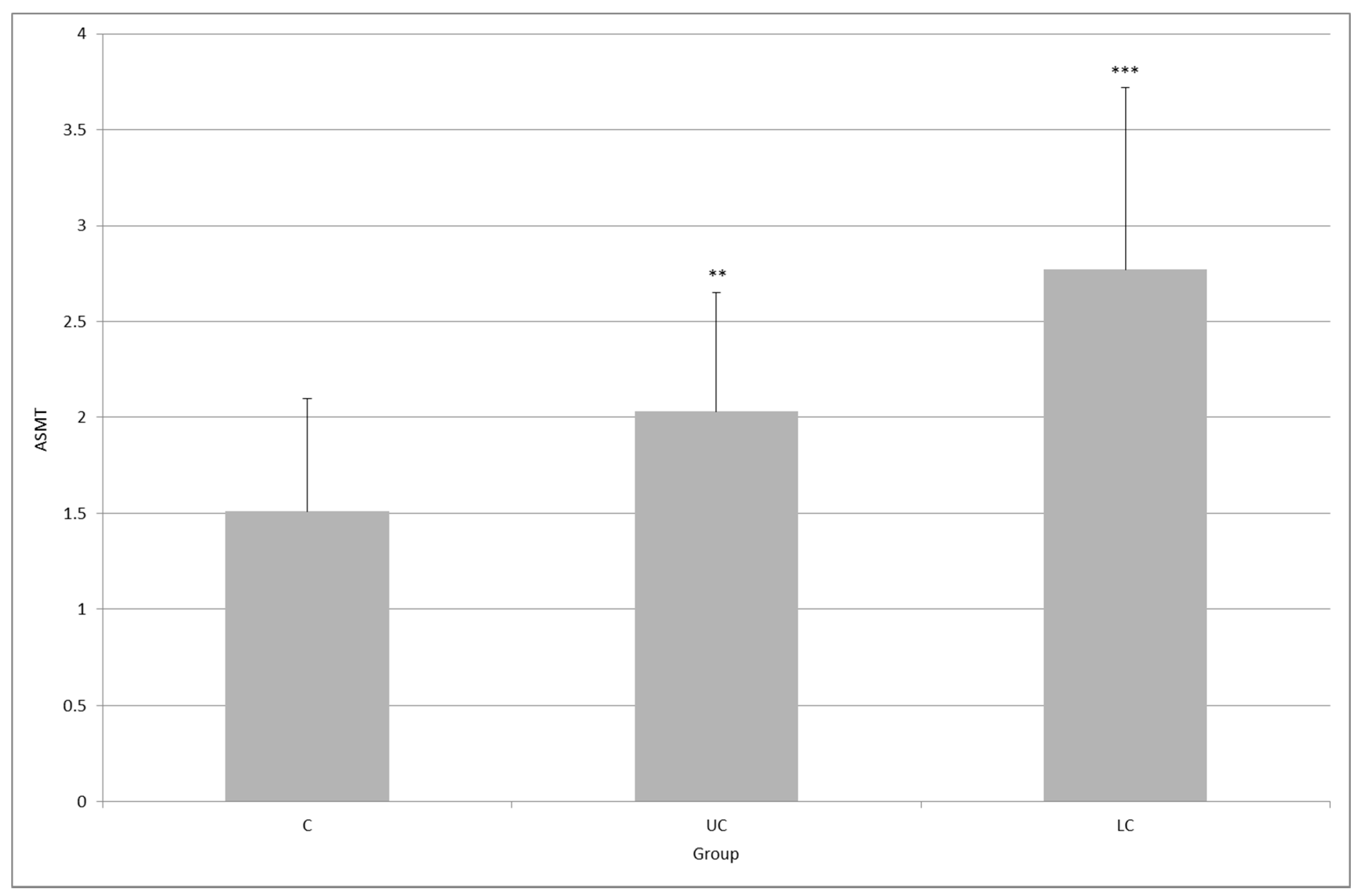
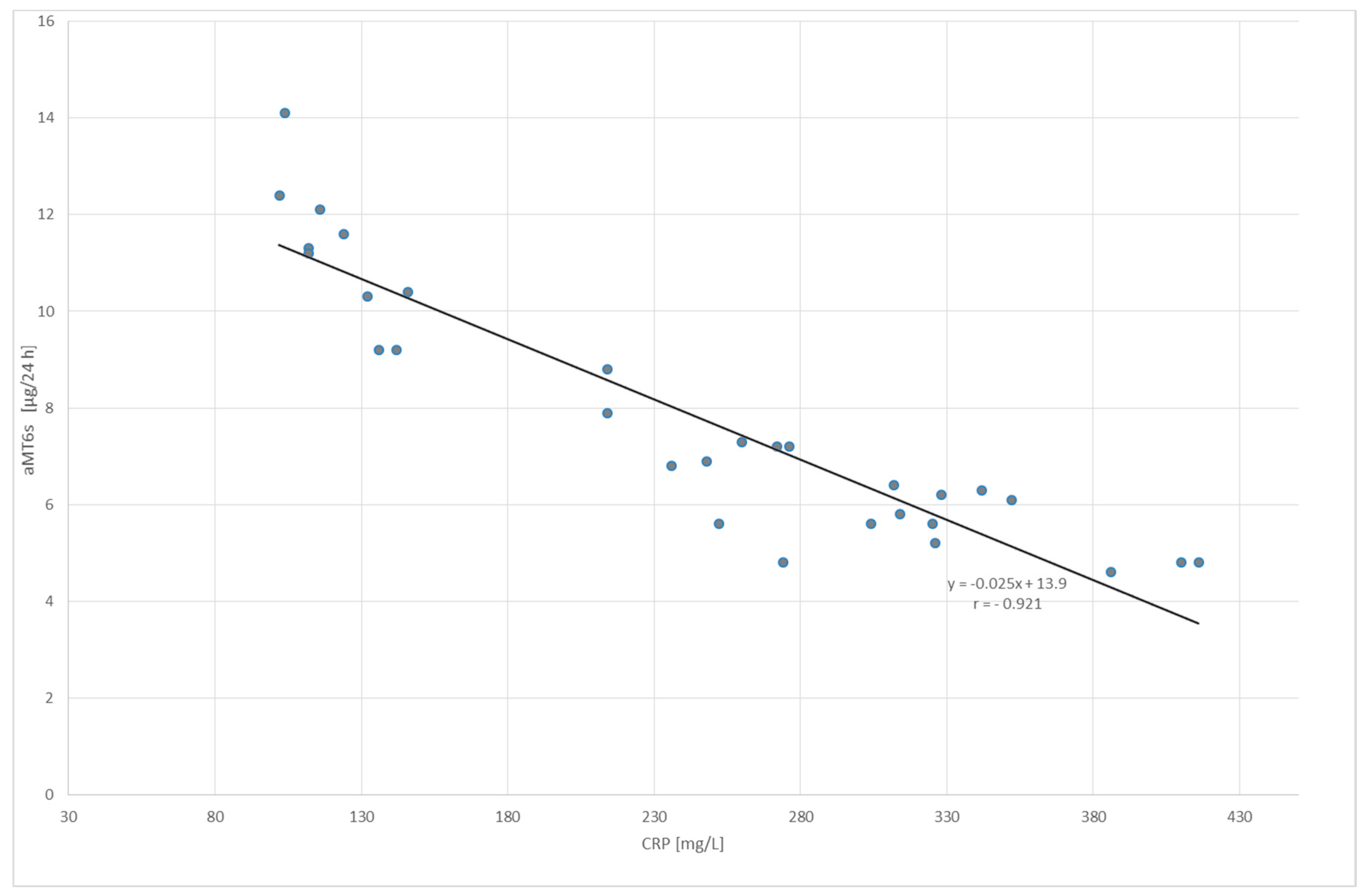
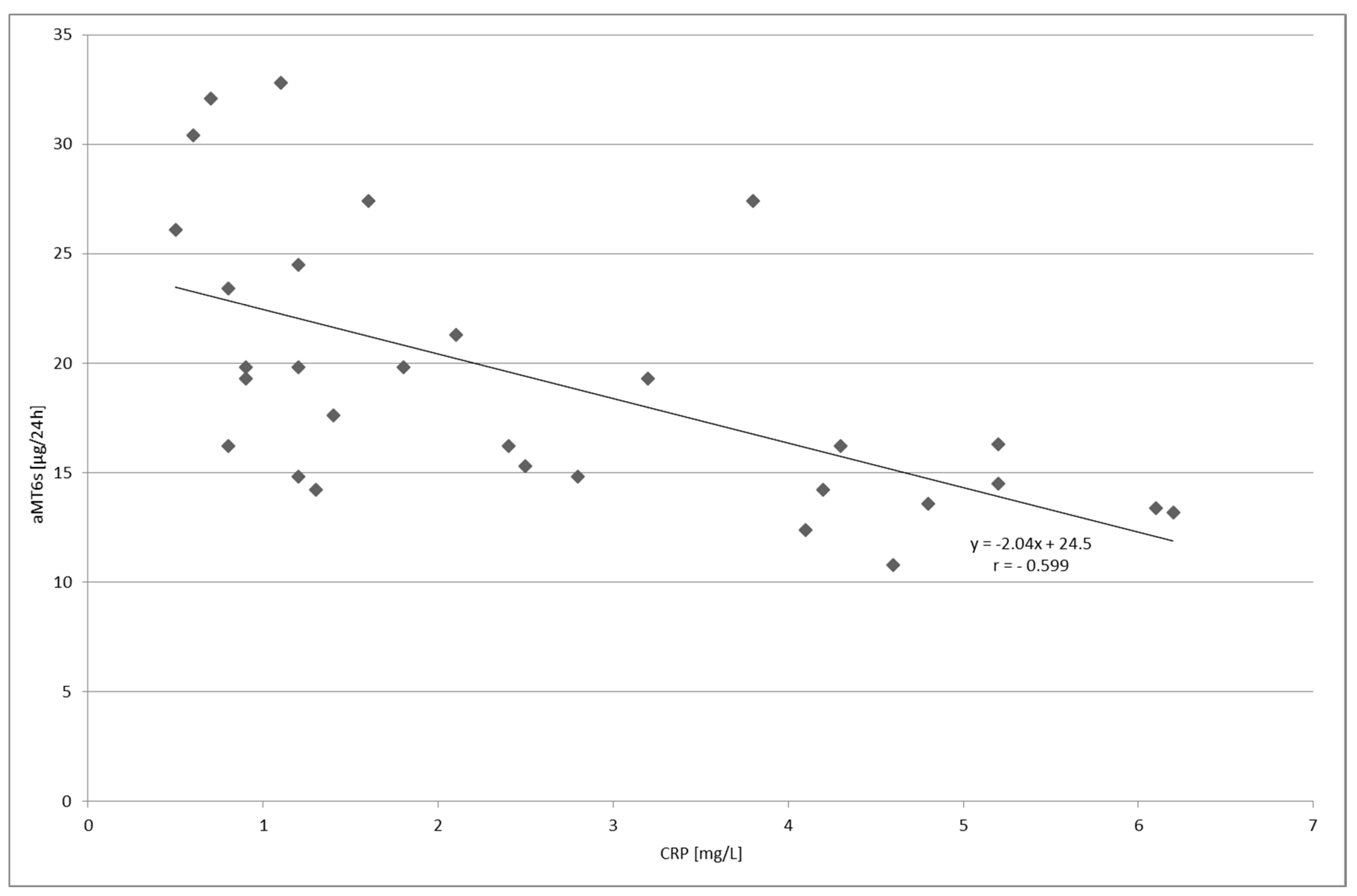
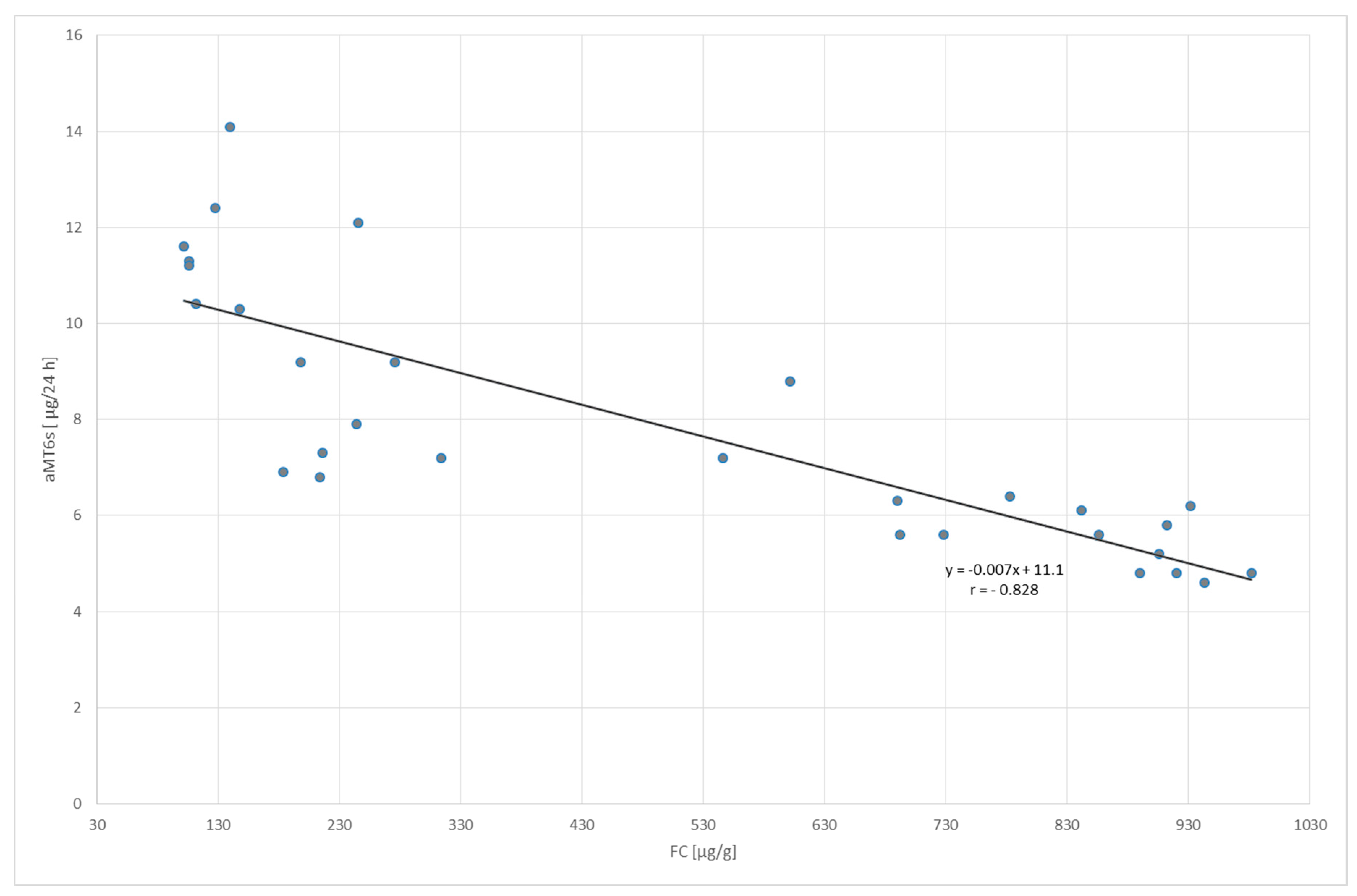
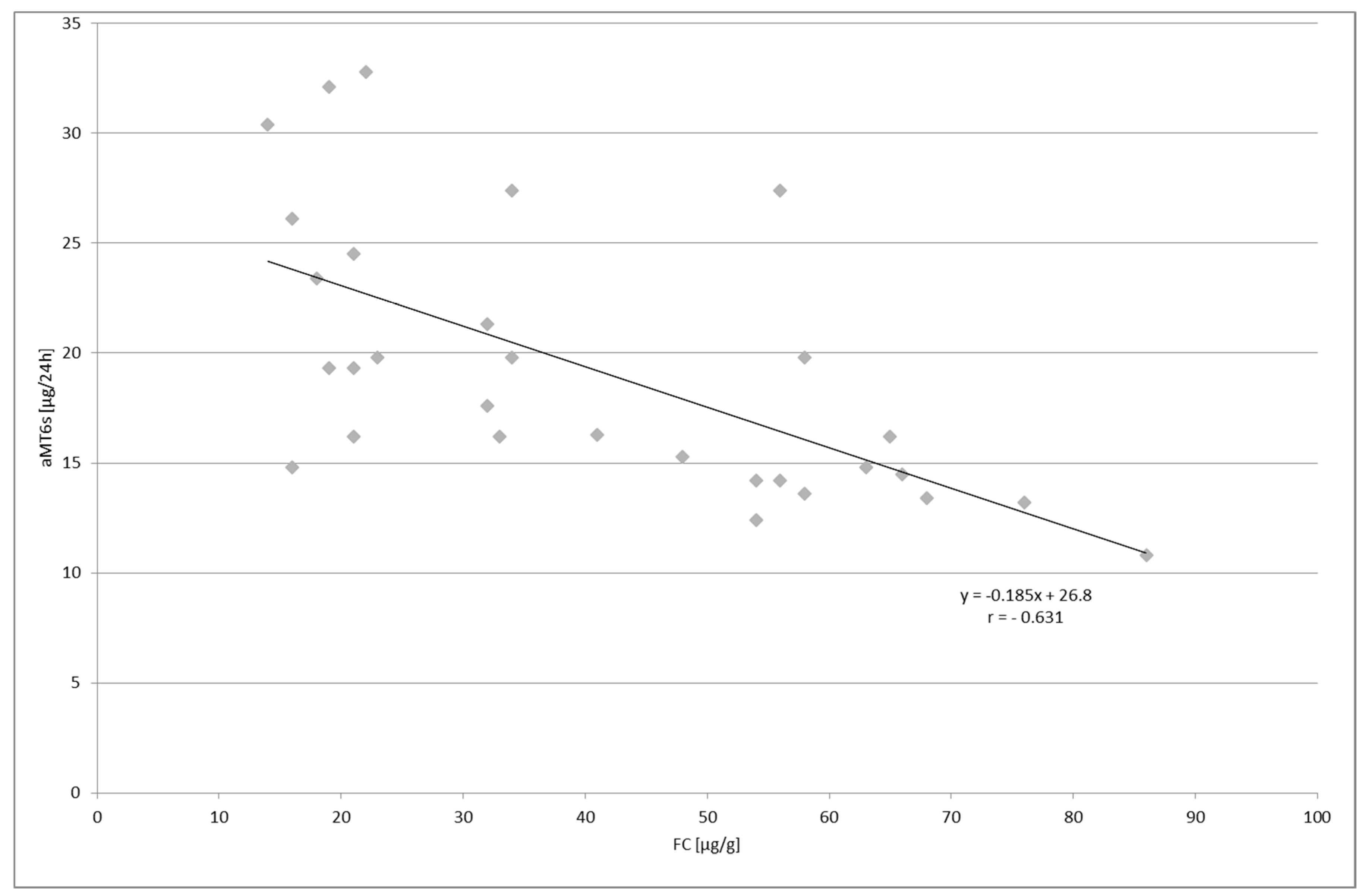
2. Results
3. Discussion
4. Material and methods
4.1. Patients
4.2. Study Design and Procedures
4.3. Ethics
4.4. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bubenik, G.A. Localization, physiological significance and possible clinical implication of gastrointestinal melatonin. Biol. Signals Recept. 2001, 10, 350–366. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.-X.; Mayo, J.C.; Sainz, R.M.; Leon, J.; Bandyopadhyay, D. Neurally-mediated and neurally-independent beneficial actions of melatonin in the gastrointestinal tract. J. Physiol. Pharmacol. 2003, 54 (Suppl. 4), 113–125. [Google Scholar] [PubMed]
- Chen, C.-Q.; Fichna, J.; Bashashati, M.; Li, Y.-Y.; Storr, M. Distribution, function and physiological role of melatonin in the lower gut. World J. Gastroenterol. 2011, 17, 3888–3898. [Google Scholar] [CrossRef] [PubMed]
- Velarde, E.; Cerdá-Reverter, J.M.; Alonso-Gómez, A.L.; Sánchez, E.; Isorna, E.; Delgado, M.J. Melatonin-synthesizing enzymes in pineal, retina, liver, and gut of the goldfish (carassius): MRNA expression pattern and regulation of daily rhythms by lighting conditions. Chronobiol. Int. 2010, 27, 1178–1201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zagajewski, J.; Drozdowicz, D.; Brzozowska, I.; Hubalewska-Mazgaj, M.; Stelmaszynska, T.; Laidler, P.M.; Brzozowski, T. Conversion l-tryptophan to melatonin in the gastrointestinal tract: The new high performance liquid chromatography method enabling simultaneous determination of six metabolites of l-tryptophan by native fluorescence and UV-VIS detection. J. Physiol. Pharmacol. 2012, 63, 613–621. [Google Scholar] [PubMed]
- Ceinos, R.M.; Chansard, M.; Revel, F.; Calgari, C.; Míguez, J.M.; Simonneaux, V. Analysis of adrenergic regulation of melatonin synthesis in Siberian hamster pineal emphasizes the role of HIOMT. Neurosignals 2004, 13, 308–317. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, P.A.; Tamura, E.K.; D’Argenio-Garcia, L.; Muxel, S.M.; da Silveira Cruz-Machado, S.; Marçola, M.; Carvalho-Sousa, C.E.; Cecon, E.; Ferreira, Z.S.; Markus, R.P. Dual Effect of Catecholamines and Corticosterone Crosstalk on Pineal Gland Melatonin Synthesis. Neuroendocrinology 2017, 104, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Lane, E.A.; Moss, H.B. Pharmacokinetics of Melatonin in Man: First Pass Hepatic Metabolism. J. Clin. Endocrinol. Metab. 1985, 61, 1214–1216. [Google Scholar] [CrossRef] [PubMed]
- Facciolá, G.; Hidestrand, M.; von Bahr, C.; Tybring, G. Cytochrome P450 isoforms involved in melatonin metabolism in human liver microsomes. Eur. J. Clin. Pharmacol. 2001, 56, 881–888. [Google Scholar] [CrossRef] [PubMed]
- Baskett, J.J.; Cockrem, J.F.; Antunovich, T.A. Sulphatoxymelatonin excretion in older people: Relationship to plasma melatonin and renal function. J. Pineal Res. 1998, 24, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Kovács, J.; Brodner, W.; Kirchlechner, V.; Arif, T.; Waldhauser, F. Measurement of urinary melatonin: A useful tool for monitoring serum melatonin after its oral administration. J. Clin. Endocrinol. Metab. 2000, 85, 666–670. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R. Antioxidative protection by melatonin: Multiplicity of mechanisms from radical detoxification to radical avoidance. Endocrine 2005, 27, 119–130. [Google Scholar] [CrossRef]
- Tan, D.X.; Manchester, L.C.; Reiter, R.J.; Qi, W.B.; Karbownik, M.; Calvo, J.R. Significance of melatonin in antioxidative defense system: Reactions and products. Biol. Signals Recept. 2000, 9, 137–159. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.-X.; Manchester, L.C.; Terron, M.P.; Flores, L.J.; Reiter, R.J. One molecule, many derivatives: A never-ending interaction of melatonin with reactive oxygen and nitrogen species? J. Pineal Res. 2007, 42, 28–42. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Mayo, J.C.; Tan, D.-X.; Sainz, R.M.; Alatorre-Jimenez, M.; Qin, L. Melatonin as an antioxidant: Under promises but over delivers. J. Pineal Res. 2016, 61, 253–278. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Tan, D.X.; Reiter, R.J. On the free radical scavenging activities of melatonin’s metabolites, AFMK and AMK. J. Pineal Res. 2013, 54, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, C.; Mayo, J.C.; Sainz, R.M.; Antolín, I.; Herrera, F.; Martín, V.; Reiter, R.J. Regulation of antioxidant enzymes: A significant role for melatonin. J. Pineal Res. 2004, 36, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.L.M.; Tamura, E.K.; Macedo, S.M.D.; Cecon, E.; Bueno-Alves, L.; Farsky, S.H.P.; Ferreira, Z.S.; Markus, R.P. Melatonin inhibits nitric oxide production by microvascular endothelial cells in vivo and in vitro. Br. J. Pharmacol. 2007, 151, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Manikonda, P.K.; Jagota, A. Melatonin administration differentially affects age-induced alterations in daily rhythms of lipid peroxidation and antioxidant enzymes in male rat liver. Biogerontology 2012, 13, 511–524. [Google Scholar] [CrossRef] [PubMed]
- Chojnacki, J.; Śliwiński, T.; Wiśniewska-Jarosińska, M.; Turant, M.; Błasiak, J.; Kulig, A.; Majsterek, I.; Chojnacki, C. Evaluation of the number of enterochromaffin cells and melatonin metabolism exponents in subjects with ulcerative colitis. Clin. Exp. Med. Lett. 2011, 52, 19–23. [Google Scholar]
- Neurath, M.F.; Finotto, S. The many roads to inflammatory bowel diseases. Immunity 2006, 25, 189–191. [Google Scholar] [CrossRef] [PubMed]
- Sartor, R.B. Mechanisms of Disease: Pathogenesis of Crohn’s disease and ulcerative colitis. Nat. Clin. Pract. Gastroenterol. Hepatol. 2006, 3, 390–407. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, A.; Calpena, A.; Clares, B. Evaluating the Oxidative Stress in Inflammation: Role of Melatonin. Int. J. Mol. Sci. 2015, 16, 16981–17004. [Google Scholar] [CrossRef] [PubMed]
- Nemeth, Z.H.; Bogdanovski, D.A.; Barratt-Stopper, P.; Paglinco, S.R.; Antonioli, L.; Rolandelli, R.H. Crohn’s Disease and Ulcerative Colitis Show Unique Cytokine Profiles. Cureus 2017, 9, e1177. [Google Scholar] [CrossRef] [PubMed]
- Chojnacki, C.; Wiśniewska-Jarosińska, M.; Kulig, G.; Majsterek, I.; Reiter, R.J.; Chojnacki, J. Evaluation of enterochromaffin cells and melatonin secretion exponents in ulcerative colitis. World J. Gastroenterol. 2013, 19, 3602–3607. [Google Scholar] [CrossRef] [PubMed]
- El-Salhy, M.; Danielsson, A.; Stenling, R.; Grimelius, L. Colonic endocrine cells in inflammatory bowel disease. J. Intern. Med. 1997, 242, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Rybakova, M.G.; Botina, A.V.; Solov’eva, O.I. Immunomorphological characteristics of mucosal and endocrine cells of the colon in patients with chronic ulcerative colitis. Arkhiv Patol. 2005, 67, 30–33. [Google Scholar]
- Ingle, S.B.; Adgaonkar, B.D.; Ingle, C.R.H. Microscopic colitis: Common cause of unexplained nonbloody diarrhea. World J. Gastrointest. Pathophysiol. 2014, 5, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Zabana, Y.; Ferrer, C.; Aceituno, M.; Salas, A.; Fernández-Bañares, F. Advances for improved diagnosis of microscopic colitis in patients with chronic diarrhoea. Gastroenterol. Hepatol. 2017, 40, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Bañares, F.; Esteve, M.; Espinós, J.C.; Rosinach, M.; Forné, M.; Salas, A.; Viver, J.M. Drug Consumption and the Risk of Microscopic Colitis. Am. J. Gastroenterol. 2007, 102, 324–330. [Google Scholar] [CrossRef] [PubMed]
- Park, T.; Cave, D.; Marshall, C. Microscopic colitis: A review of etiology, treatment and refractory disease. World J. Gastroenterol. 2015, 21, 8804–8810. [Google Scholar] [CrossRef] [PubMed]
- Pisani, L.F.; Tontini, G.E.; Vecchi, M.; Pastorelli, L. Microscopic Colitis. Inflamm. Bowel Dis. 2016, 22, 450–458. [Google Scholar] [CrossRef] [PubMed]
- Law, E.H.; Badowski, M.; Hung, Y.T.; Weems, K.; Sanchez, A.; Lee, T.A. Association between Proton Pump Inhibitors and Microscopic Colitis. Ann. Pharmacother. 2017, 51, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Macaigne, G.; Lahmek, P.; Locher, C.; Boivin, J.F.; Lesgourgues, B.; Yver, M.; Costes, L.; Alsamad, I.A.; Cucherousset, J.; Charpignon, C.; et al. Over 90% of cases of Microscopic Colitis can be diagnosed by performing a short colonoscopy. Clin. Res. Hepatol. Gastroenterol. 2017, 41, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Pardi, D.S. Diagnosis and management of microscopic colitis. Am. J. Gastroenterol. 2017, 112, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Platz-Baudin, C.; Katzenberger, T.; Eck, M. Microscopic colitis: Histopathological review with a clinicopathological correlation. Pathologe 2011, 32, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Chatelain, D.; Mokrani, N.; Fléjou, J.F. Microscopic colitis: Collagenous colitis and lymphocytic colitis. Ann. Pathol. 2007, 27, 448–458. [Google Scholar] [CrossRef]
- El-Salhy, M.; Gundersen, D.; Hatlebakk, J.G.; Hausken, T. High densities of serotonin and peptide YY cells in the colon of patients with lymphocytic colitis. World J. Gastroenterol. 2012, 18, 6070–6075. [Google Scholar] [CrossRef] [PubMed]
- Boznańska, P.; Wichan, P.; Stepień, A.; Wiśniewska-Jarosńska, M.; Smigielski, J.; Szadkowski, K.; Stec-Michalska, K. 24-hour urinary 6-hydroxymelatonin sulfate excretion in patients with ulcerative colitis. Pol. Merkur. Lekarski 2007, 22, 369–372. [Google Scholar] [PubMed]
- Chen, M.; Mei, Q.; Xu, J.; Lu, C.; Fang, H.; Liu, X. Detection of melatonin and homocysteine simultaneously in ulcerative colitis. Clin. Chim. Acta 2012, 413, 30–33. [Google Scholar] [CrossRef] [PubMed]
- Cotter, T.G.; Binder, M.; Loftus, E.V., Jr.; Abboud, R.; McNally, M.A.; Smyrk, T.C.; Tremaine, W.J.; Sandborn, W.J.; Pardi, D.S. Development of a Microscopic Colitis Disease Activity Index: A prospective cohort study. Gut 2016. [Google Scholar] [CrossRef] [PubMed]
- Kane, J.S.; Sood, R.; Law, G.R.; Gracie, D.J.; To, N.; Gold, M.J.; Ford, A.C. Validation and modification of a diagnostic scoring system to predict microscopic colitis. Scand. J. Gastroenterol. 2016, 51, 1206–1212. [Google Scholar] [CrossRef] [PubMed]
- Liszka, Ł.; Woszczyk, D.; Pajak, J. Histopathological diagnosis of microscopic colitis. J. Gastroenterol. Hepatol. 2006, 21, 792–997. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.S.; Kim, T.K. Is microscopic colitis really microscopic? Gut Liver 2015, 9, 137–138. [Google Scholar] [CrossRef] [PubMed]
- Park, H.S.; Han, D.S.; Ro, Y.O.; Eun, C.S.; Yoo, K.S. Does lymphocytic colitis always present with normal endoscopic findings? Gut Liver 2015, 9, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Aust, D.E. Histopathology of microscopic colitis. Pathologe 2012, 33 (Suppl. 2), 221–224. [Google Scholar] [CrossRef] [PubMed]
- Langner, C.; Aust, D.; Ensari, A.; Villanacci, V.; Becheanu, G.; Miehlke, S.; Geboes, K.; Münch, A. Histology of microscopic colitis-review with a practical approach for pathologists. Histopathology 2015, 66, 613–626. [Google Scholar] [CrossRef] [PubMed]
- Pisani, L.F.; Tontini, G.E.; Marinoni, B.; Villanacci, V.; Bruni, B.; Vecchi, M.; Pastorelli, L. Biomarkers and Microscopic Colitis: An Unmet Need in Clinical Practice. Front. Med. 2017, 4, 54. [Google Scholar] [CrossRef] [PubMed]
- El-Salhy, M.; Lomholt-Beck, B.; Gundersen, T.D. High chromogranin A cell density in the colon of patients with lymphocytic colitis. Mol. Med. Rep. 2011, 4, 603–605. [Google Scholar] [CrossRef] [PubMed]
- El-Salhy, M.; Gundersen, D.; Hatlebakk, J.G.; Hausken, T. Chromogranin A cell density as a diagnostic marker for lymphocytic colitis. Dig. Dis. Sci. 2012, 57, 3154–3159. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spiller, R.C.; Jenkins, D.; Thornley, J.P.; Hebden, J.M.; Wright, T.; Skinner, M.; Neal, K.R. Increased rectal mucosal enteroendocrine cells, T lymphocytes, and increased gut permeability following acute Campylobacter enteritis and in post-dysenteric irritable bowel syndrome. Gut 2000, 47, 804–811. [Google Scholar] [CrossRef] [PubMed]
- Dunlop, S.P.; Jenkins, D.; Neal, K.R.; Spiller, R.C. Relative importance of enterochromaffin cell hyperplasia, anxiety, and depression in postinfectious IBS. Gastroenterology 2003, 125, 1651–1659. [Google Scholar] [CrossRef] [PubMed]
- Carrillo-Vico, A.; Calvo, J.R.; Abreu, P.; Lardone, P.J.; Garcia-Maurino, S.; Reiter, R.J.; Guerro, J.M. Evidence of melatonin synthesis by human lymphocytes and its physiological significance: Possible role as intacrine, autocrine, and/or paracrine substance. FASEB J. 2004, 18, 537–539. [Google Scholar] [CrossRef] [PubMed]
- Lin, G.J.; Huang, S.H.; Chen, S.J.; Wang, C.H.; Chang, D.M.; Sytwu, H.K. Modulation by melatonin of the pathogenesis of inflammatory autoimmune diseases. Int. J. Mol. Sci. 2013, 14, 11742–11766. [Google Scholar] [CrossRef] [PubMed]
- Ren, W.; Liu, G.; Chen, S.; Yin, J.; Wang, J.; Tan, B.; Wu, G.; Bazer, F.W.; Peng, Y.; Li, T.; et al. Melatonin signaling in T cells: Functions and applications. J. Pineal Res. 2017, 62. [Google Scholar] [CrossRef] [PubMed]
- Chojnacki, C.; Wisniewska-Jarosinska, M.; Walecka-Kapica, E.; Klupinska, G.; Jaworek, J.; Chojnacki, J. Evaluation of melatonin effectiveness in the adjuvant treatment of ulcerative colitis. J. Physiol. Pharmacol. 2011, 62, 327–334. [Google Scholar] [PubMed]
- Jena, G.; Trivedi, P.P. A review of the use of melatonin in ulcerative colitis: Experimental evidence and new approaches. Inflamm. Bowel Dis. 2014, 20, 553–563. [Google Scholar] [CrossRef] [PubMed]
- Esteban-Zubero, E.; López-Pingarrón, L.; Alatorre-Jiménez, M.A.; Ochoa-Moneo, P.; Buisac-Ramón, C.; Rivas-Jiménez, M.; Castán-Ruiz, S.; Antoñanzas-Lombarte, Á.; Tan, D.X.; García, J.J.; et al. Melatonin’s role as a co-adjuvant treatment in colonic diseases: A review. Life Sci. 2017, 170, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Najeeb, S.; Khurshid, Z.; Zohaib, S.; Zafar, M.S. Therapeutic potential of melatonin in oral medicine and peridontology. Kaohsiung J. Med. Sci. 2016, 32, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Werbach, M.R. Melatonin for the treatment of gastroesophageal reflux disease. Altern. Ther. Health Med. 2008, 14, 534–558. [Google Scholar]
Sample Availability: Samples of the compounds are available from the authors. |
| Features | Control Group (n = 30) | Ulcerative Colitis (n = 30) | Lymphocytic Colitis (n = 30) | |
|---|---|---|---|---|
| Age—years | 38.9 ± 9.4 | 41.4 ± 10.2 | 43.7 ± 12.4 | |
| Gender | M | 16 | 13 | 14 |
| K | 14 | 17 | 16 | |
| CRP (mg/L) | 1.97 ± 1.05 | 242.9 ± 99.4 | 2.58 ± 1.81 | |
| FC (µ/g) | 25.8 ± 9.72 | 498.6 ± 338.3 | 40.8 ± 21.0 | |
| aMT6s (µg/24 h) | 13.4 ± 4.87 | 7.86 ± 2.69 | 19.2 ± 6.14 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chojnacki, C.; Błasiak, J.; Fichna, J.; Chojnacki, J.; Popławski, T. Evaluation of Melatonin Secretion and Metabolism Exponents in Patients with Ulcerative and Lymphocytic Colitis. Molecules 2018, 23, 272. https://doi.org/10.3390/molecules23020272
Chojnacki C, Błasiak J, Fichna J, Chojnacki J, Popławski T. Evaluation of Melatonin Secretion and Metabolism Exponents in Patients with Ulcerative and Lymphocytic Colitis. Molecules. 2018; 23(2):272. https://doi.org/10.3390/molecules23020272
Chicago/Turabian StyleChojnacki, Cezary, Janusz Błasiak, Jakub Fichna, Jan Chojnacki, and Tomasz Popławski. 2018. "Evaluation of Melatonin Secretion and Metabolism Exponents in Patients with Ulcerative and Lymphocytic Colitis" Molecules 23, no. 2: 272. https://doi.org/10.3390/molecules23020272






