Chemical Constituents and Antioxidant, Anti-Inflammatory and Anti-Tumor Activities of Melilotus officinalis (Linn.) Pall
Abstract
:1. Introduction
2. Results and Discussion
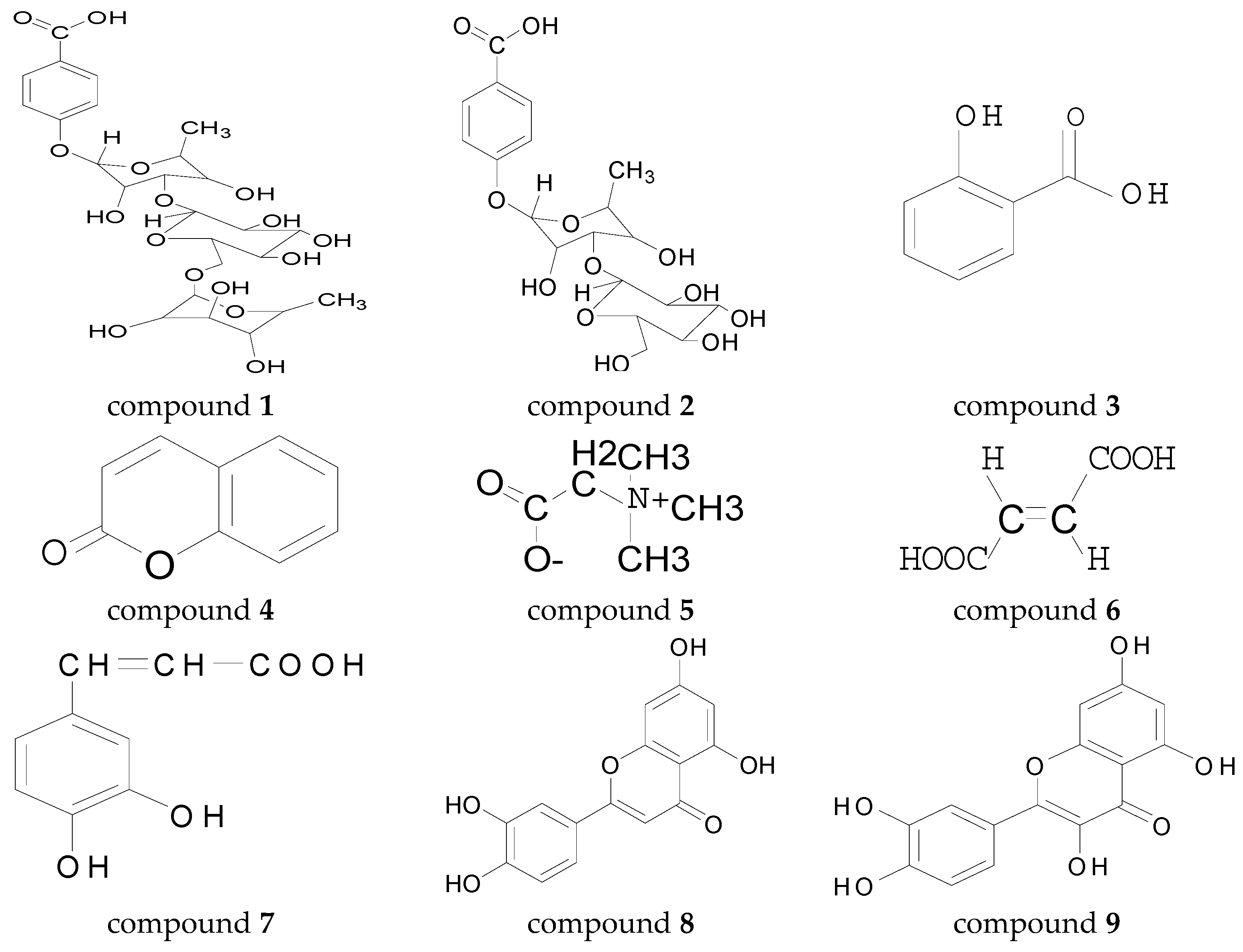
2.1. Chemical Components and Monosaccharide Compositions
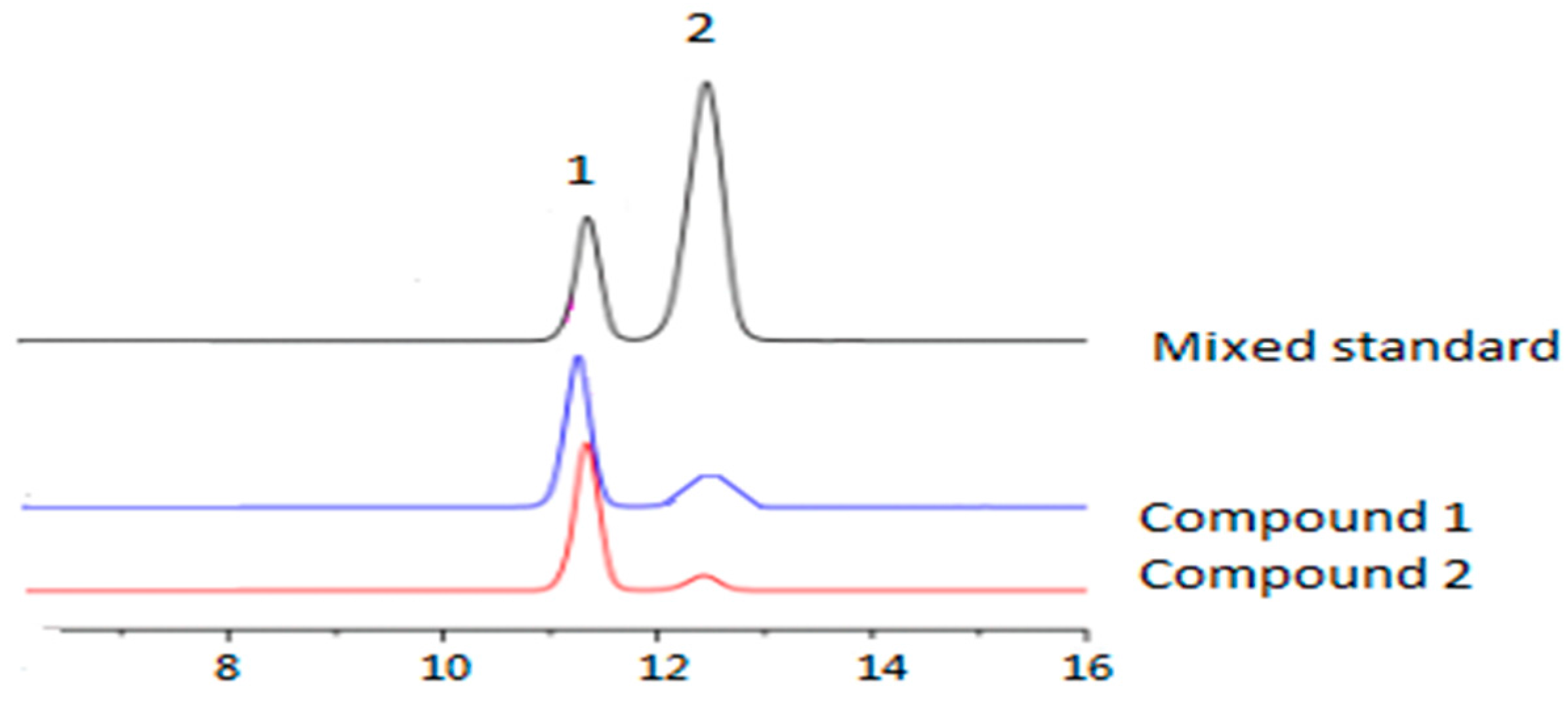
2.1.1. Identification of Chemical Composition
2.1.2. Monosaccharide Analysis
2.2. Biological Activity
2.2.1. Anti-Oxidative Activity
2.2.2. Anti-Inflammatory Activity
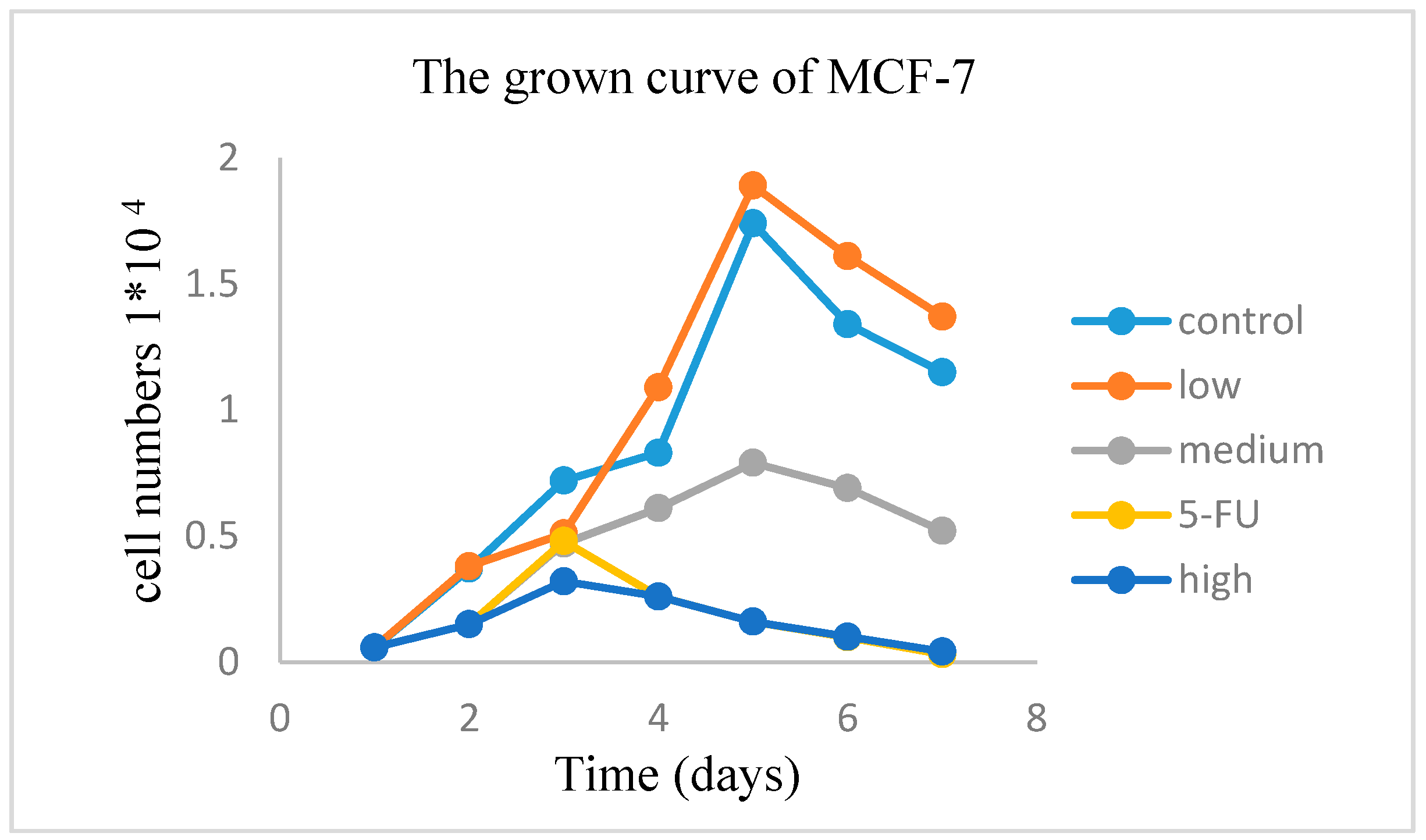
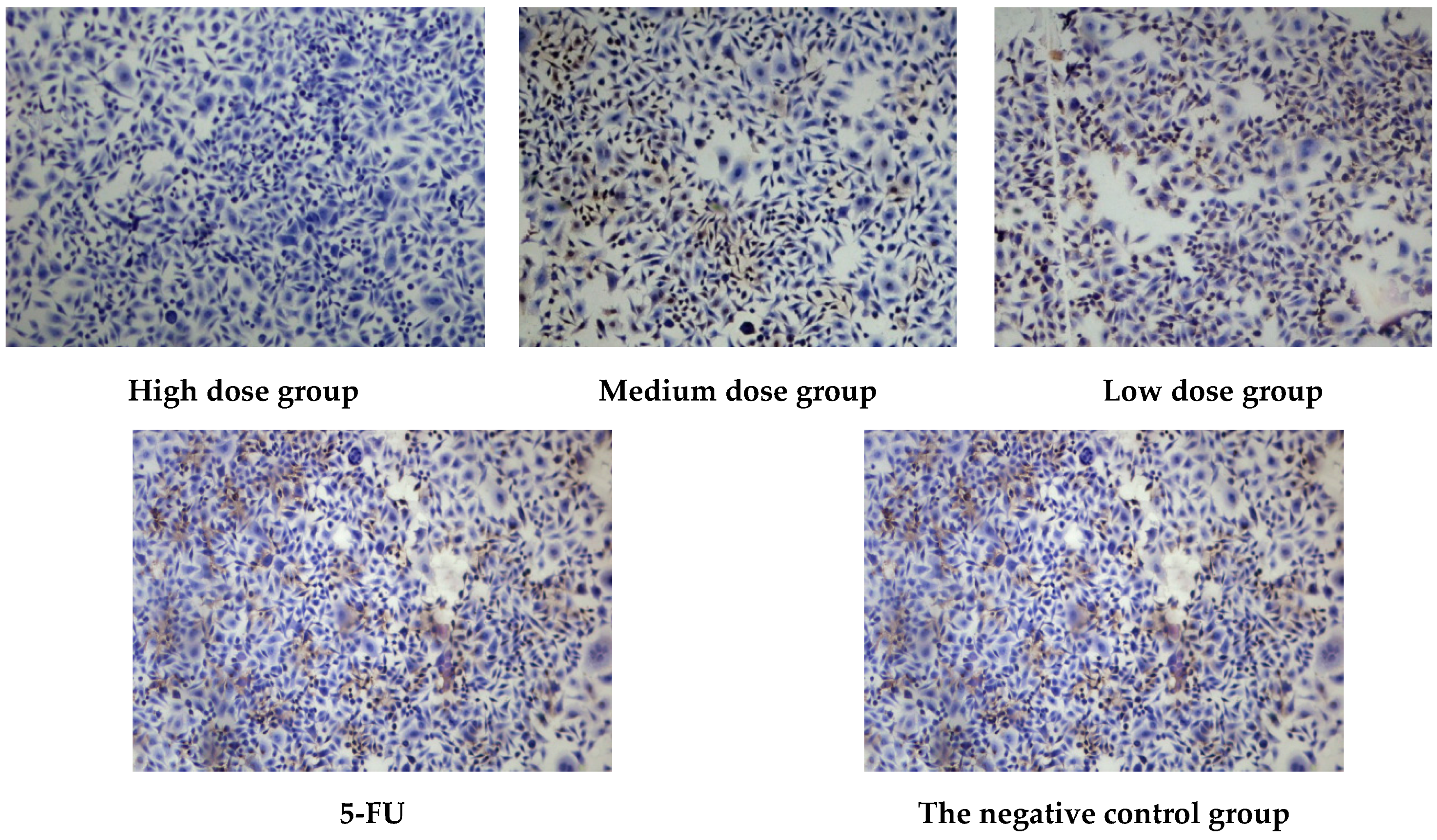
2.2.3. Antitumor Activity
2.3. Discussion
3. Experimental Section
3.1. Materials
3.1.1. Chemicals and Reagents
3.1.2. Cell-Lines
3.1.3. Plant Materials
3.2. Methods
3.2.1. Extraction and Isolation
3.2.2. p-Hydroxybenzoic Acid-4-O-α-d-manopyranosyl-(1 → 3)-α-l-rhamnopyranoside (1)
3.2.3. p-Hydroxybenzoic acid-4-O-α-l-rhamnopyranosyl-(1 → 6)-α-d-manopyranosyl-(1 → 3)-α-l-rhamnopyranoside (2)
3.2.4. Salicylic Acid
3.2.5. Coumarin
3.2.6. Betaine
3.2.7. Fumalic Acid
3.2.8. Caffeic Acid
3.2.9. Luteolin
3.2.10. Quercetin
3.2.11. Monosaccharide Analysis
3.2.12. Anti-Oxidative Activity
3.2.13. Anti-Inflammatory Activity
3.2.14. Cytotoxicity Assay
Acknowledgments
Author Contributions
Conflicts of Interest
References
- European Medicines Quality Board. European Pharmacopoeia, 8th ed.; European Medicines and Quality Administration: Strasbourg, France, 2013; pp. 1317–1318. [Google Scholar]
- Tang, C.N. Study on the extraction process of total flavonoids from Melilotus officinalis medicinal plant. J. Anhui Agric. Sci. 2012, 3, 23–25. [Google Scholar] [CrossRef]
- Chen, H.F. European Pharmacopoeia: Melilot. Foreign Med. (Plant Med.) 2006, 21, 184. [Google Scholar]
- Bisby, F.A.; Buckingham, J.; Harborue, J.B. Phytochemical Dictionary of the Leguminosae; Chapman & Hall Press: London, UK, 1994; pp. 472–475. ISBN 978-0412397707. [Google Scholar]
- China Ministry of Health. People’s Republic of China Ministry of Health Drug Standard Tibetan Medicine; Chemical Industry Press: Beijing, China, 1995; Volume 1, p. 65.
- Macias, F.A.; Simonet, A.M.; Galindo, J.C.G.; Pacheco, P.C.; Sánchez, J.A. Bioactive polar triterpenoids from Melilotus messanensis. Phytochemistry 1998, 49, 709–717. [Google Scholar] [CrossRef]
- Yang, J.; Wang, L.L.; Zhang, T.J. Research progress on chemical constituents in plants of Melilotus Linn. and their pharmacological activities. Chin. Tradit. Herb. Drugs 2014, 45, 447–454. [Google Scholar] [CrossRef]
- Gupta, A.K.; Grasdalen, H. A D-galacto-D-mannan from Melilotus officinalis seed. Carbohydr. Res. 1988, 173, 159–168. [Google Scholar] [CrossRef]
- Luminiţa, P.M.; Pârvu, A.E.; Pârvu, M.; Taămaş, M.; Buia, R.; Puia, M. Effects of Melilotus officinalis on acute inflammation. Phytother. Res. 2002, 16, 316–319. [Google Scholar] [CrossRef]
- Gu, B.Q. Herbal rhinoceros extract immersion tablets plus traditional Chinese medicine fumigation treatment of mixed hemorrhoids complications. Zhejiang JITCWM 2006, 16, 607–608. [Google Scholar]
- Yan, M.M.; Wu, C.Y.; Wei, Z.X.; Fu, M.L.; Liu, C.; Tian, S.; Shao, S. Anti-cancer effect of external saponins from Melilotus officinalis L. Jilin J. Tradit. Chin. Med. 2015, 35, 191–192. [Google Scholar]
- Yan, M.M.; Yang, Z.; Wang, Y.S.; Zhao, D.Q.; Wu, Y.; Zhang, Y.L.; Yu, H.W.; Zhou, Y. The Preparation Method and Drug Use of Total Saponin from Melilotus officinalis. CN Patent CN102178725 A, 14 September 2011. [Google Scholar]
- Cheng, Z.L.; Shi, Y.P.; Chong, X.T.; Yao, Q.Q. Study on Chemical Constituents of Asteris souliei. Food Drug 2009, 11, 33–35. [Google Scholar]
- Kang, S.S.; Lim, C.H.; Lee, S.Y. Soyasapogenols B and E from Melilotus officinalis. Arch. Pharm. Res. 1987, 10, 9–13. [Google Scholar] [CrossRef]
- Liu, X.Y.; Ma, L.; Du, N.S.; Qu, S.H.; Qiao, J. Extraction and isolation of betaine from waste honey produced by sugar production from Beta vulgaris. Northwest Pharm. J. 2004, 19, 63–64. [Google Scholar]
- Pan, J.B.; Liao, S.Y.; Shen, L.B.; Ma, R.; Lu, W.; He, M.S. Study on the constituents of organic acid compounds from Melilotus. Hubei J. TCM 2009, 31, 58. [Google Scholar]
- Ling, Y.; Bao, Y.; Zhu, L. Chemical constituents of Taraxacum mongolicum. Chin. Pharm. J. 1997, 32, 584–586. [Google Scholar]
- Kang, S.S.; Lee, Y.S.; Lee, E.B. Isolation of azukisaponin V possessing leucocyte migration inhibitory activity from Melilotus officinalis. Korean J. Pharmacogn. 1987, 18, 89–93. [Google Scholar]
- Zhou, L.Y.; Zhang, X.H.; Chen, C.X. Chemical study on Rhodiola from Lijiang. Nat. Prod. Res. Dev. 2004, 16, 410–414. [Google Scholar]
- Yu, D.Q.; Yang, J.S. Analytic Chemistry Handbook; Chemical Industry Press: Beijing, China, 1999; Volume 5, p. 902. [Google Scholar]
- Hasegawa, T.; Takano, F.; Takata, T.; Niiyama, M.; Ohta, T. Bioactive monoterpene glycosides conjugated with gallic acid from the leaves of Eucalyptus globulus. Phytochemistry 2008, 69, 747–753. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.H.; Chang, F.R.; Lu, M.C.; Hsieh, P.W.; Wu, M.J.; Du, Y.C.; Wu, Y.C. New benzoyl glucosides and cytotoxic pterosin sesquiterpenes from Pteris ensiformis Burm. Molecules 2008, 13, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Khadem, S.; Marles, R.J. Monocyclic Phenolic Acids; Hydroxy- and Polyhydroxybenzoic Acids: Occurrence and Recent Bioactivity Studies. Molecules 2010, 15, 7985–8005. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Ren, G. Determination of d- chiro-inositol in tartary buckwheat using high-performance liquid chromatography with an evaporative light-scattering detector. J. Agric. Food Chem. 2008, 56, 757–760. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 1–9 are available from the authors. |
| The IC50 of ATBS+ Free Radical Scavenging Activity (μg/mL) | The IC50 of DPPH Free Radical Scavenging Activity (μg/mL) | |
|---|---|---|
| VC | 70.00 | 39.06 |
| compound 1 | 25.20 | 53.00 |
| compound 2 | 69.75 | 73.00 |
| compound 3 | 166.00 | 143.7 |
| compound 4 | 148.00 | 254.1 |
| compound 5 | 87.23 | 176.4 |
| compound 6 | 79.83 | >450 |
| compound 7 | 18.00 | 23.04 |
| compound 8 | 13.60 | 23.89 |
| compound 9 | <10 | <10 |
| Concentration/(μg·mL−1) | NO/(μmol·mL−1) | TNF-α/(ng·mL−1) | IL-6/(ng·mL−1) | |
|---|---|---|---|---|
| Control | 0.6045 ± 0.0098 | 0.0173 ± 0.0025 | 0.0005 ± 0.0000 | |
| LPS | 6.7458 ± 0.3428 ## | 45.3633 ± 0.2559 ## | 0.6046 ± 0.0045 ## | |
| LPS + compound 1 | 50 | 4.0904 ± 0.4424 ** | 29.8130 ± 0.1658 ** | 0.5232 ± 0.0030 ** |
| LPS + compound 2 | 50 | 5.0565 ± 0.2452 ** | 27.5663 ± 0.1122 ** | 0.3047 ± 0.0040 ** |
| LPS + compound 3 | 50 | 0.1751 ± 0.0353 ** | 26.0250 ± 0.2000 ** | 0.2144 ± 0.0026 ** |
| LPS + compound 4 | 50 | 3.6667 ± 0.2301 ** | 30.8603 ± 0.1000 ** | 0.2771 ± 0.0050 ** |
| LPS + compound 5 | 50 | 3.0282 ± 0.4208 ** | 28.5656 ± 0.1000 ** | 0.2166 ± 0.0035 ** |
| LPS + compound 6 | 50 | 2.0452 ± 0.3327 ** | 31.8536 ± 0.1000 ** | 0.3136 ± 0.0025 ** |
| LPS + compound 7 | 50 | 0.1243 ± 0.1461 ** | 22.6661 ± 0.1528 ** | 0.2065 ± 0.0021 ** |
| LPS + compound 8 | 50 | 3.5865 ± 0.2452 ** | 28.4363 ± 0.1721 ** | 0.3757 ± 0.0034 ** |
| LPS + compound 9 | 50 | 4.2881 ± 0.2691 ** | 32.4133 ± 0.0577 ** | 0.2881 ± 0.0066 ** |
| Compound | IC50 (μg/mL) |
|---|---|
| MCF-7 | |
| compound 1 | 4.83 |
| compound 2 | 5.18 |
| compound 3 | 8.20 |
| compound 4 | >15 |
| compound 5 | 7.85 |
| compound 6 | >15 |
| compound 7 | 7.53 |
| compound 8 | 8.40 |
| compound 9 | 9.24 |
| 5-FU | 3.50 |
| Position | 1 | 2 | ||
|---|---|---|---|---|
| δC | δH | δC | δH | |
| 1 | 121.1 (s) | 121.1 (s) | ||
| 2 | 130.6 (d) | 8.00 (d, 2H, 7.8) | 130.7 (d) | 7.97 (d, 2H, 7.8) |
| 3 | 115.9 (d) | 6.73 (d, 2H, 7.8) | 116.0 (d) | 6.69 (d, 2H, 7.8) |
| 4 | 161.2 (d) | 161.1 (d) | ||
| 5 | 115.9 (d) | 6.73 (d, 2H, 7.8) | 116.0 (d) | 6.69 (d, 2H, 7.8) |
| 6 | 130.6 (s) | 8.00 (d, 2H, 7.8) | 130.7 (s) | 7.97 (d, 2H, 7.8) |
| Rha-1′ | 97.9 (d) | 5.41 (brs,1H,) | 97.8 (d) | 5.40 (brs,1H) |
| 2′ | 70.0 (d) | 3.81 (brs,1H) | 70.3 (d) | 3.81 (brs,1H) |
| 3′ | 75.7 (d) | 3.15 (m,1H) | 73.3 (d) | 3.12 (m,1H) |
| 4′ | 71.8 (d) | 3.58 (m,1H) | 71.8 (d) | 3.50 (m,1H) |
| 5′ | 69.7 (d) | 3.47 (m,1H) | 69.7 (d) | 3.45 (m,1H) |
| 6′ | 17.9 (q) | 1.11(d, 3H, 6.0) | 17.9 (q) | 1.15 (d, 3H, 6.0) |
| Man-1″ | 102.9 (d) | 5.09 (brs,1H) | 103.7 (d) | 4.99 (brs,1H) |
| 2″ | 71.4 (d) | 3.57 (m,1H) | 70.6 (d) | 3.58 (m,1H) |
| 3″ | 70.5 (d) | 3.62 (m,1H) | 71.2 (d) | 3.61 (m,1H) |
| 4″ | 67.6 (d) | 3.69 (brs,1H) | 68.1 (d) | 3.63 (brs,1H) |
| 5″ | 73.4 (d) | 3.27 (m,1H) | 73.5 (d) | 3.13 (m,1H) |
| 6″-A | 59.8 (t) | 3.16 (m,1H) | 65.7 (t) | 3.20 (m,1H) |
| 6″-B | 3.47 (brs,1H) | 3.57 (m,1H) | ||
| Rha-1″′ | 100.2 (d) | 4.40 (brs,1H) | ||
| 2″′ | 70.0 (d) | 3.80 (brs,1H) | ||
| 3″′ | 70.4 (d) | 3.43 (m,1H) | ||
| 4″′ | 72.0 (d) | 3.49 (m,1H) | ||
| 5″′ | 68.2 (d) | 3.39 (m,1H) | ||
| 6″′ | 17.9 (q) | 1.08 (d, 3H, 6.0) | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.-T.; Gong, P.-H.; Xiao, F.-Q.; Shao, S.; Zhao, D.-Q.; Yan, M.-M.; Yang, X.-W. Chemical Constituents and Antioxidant, Anti-Inflammatory and Anti-Tumor Activities of Melilotus officinalis (Linn.) Pall. Molecules 2018, 23, 271. https://doi.org/10.3390/molecules23020271
Liu Y-T, Gong P-H, Xiao F-Q, Shao S, Zhao D-Q, Yan M-M, Yang X-W. Chemical Constituents and Antioxidant, Anti-Inflammatory and Anti-Tumor Activities of Melilotus officinalis (Linn.) Pall. Molecules. 2018; 23(2):271. https://doi.org/10.3390/molecules23020271
Chicago/Turabian StyleLiu, Yu-Ting, Pei-Han Gong, Feng-Qin Xiao, Shuai Shao, Da-Qing Zhao, Ming-Ming Yan, and Xiu-Wei Yang. 2018. "Chemical Constituents and Antioxidant, Anti-Inflammatory and Anti-Tumor Activities of Melilotus officinalis (Linn.) Pall" Molecules 23, no. 2: 271. https://doi.org/10.3390/molecules23020271
APA StyleLiu, Y.-T., Gong, P.-H., Xiao, F.-Q., Shao, S., Zhao, D.-Q., Yan, M.-M., & Yang, X.-W. (2018). Chemical Constituents and Antioxidant, Anti-Inflammatory and Anti-Tumor Activities of Melilotus officinalis (Linn.) Pall. Molecules, 23(2), 271. https://doi.org/10.3390/molecules23020271





