Targeting Cellular Stress Mechanisms and Metabolic Homeostasis by Chinese Herbal Drugs for Neuroprotection
Abstract
:1. Introduction
2. Chinese Herbal Drugs for Neuroprotection via Cytokine Regulation
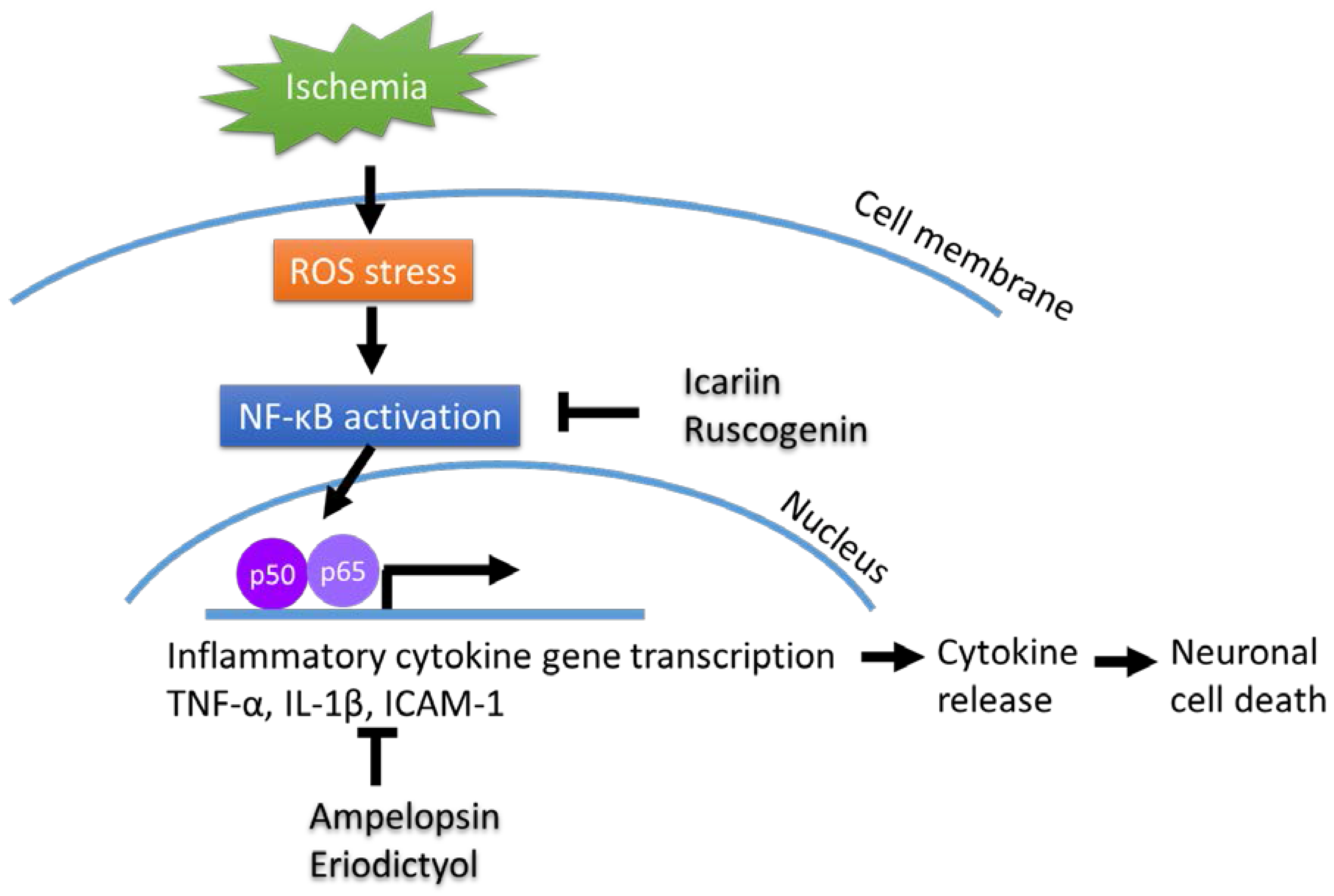
2.1. Neuroinflammation in the Nervous System
2.2. Cytokines and Neuronal Diseases
2.3. Chinese Herbaldrugs that Benefit Neuronal Diseases via Cytokine Regulation
2.4. Brief Summary of Chinese Herbal Compounds on Neuroinflammation
3. Chinese Herbal Drugs for Neuroprotection via Autophagy
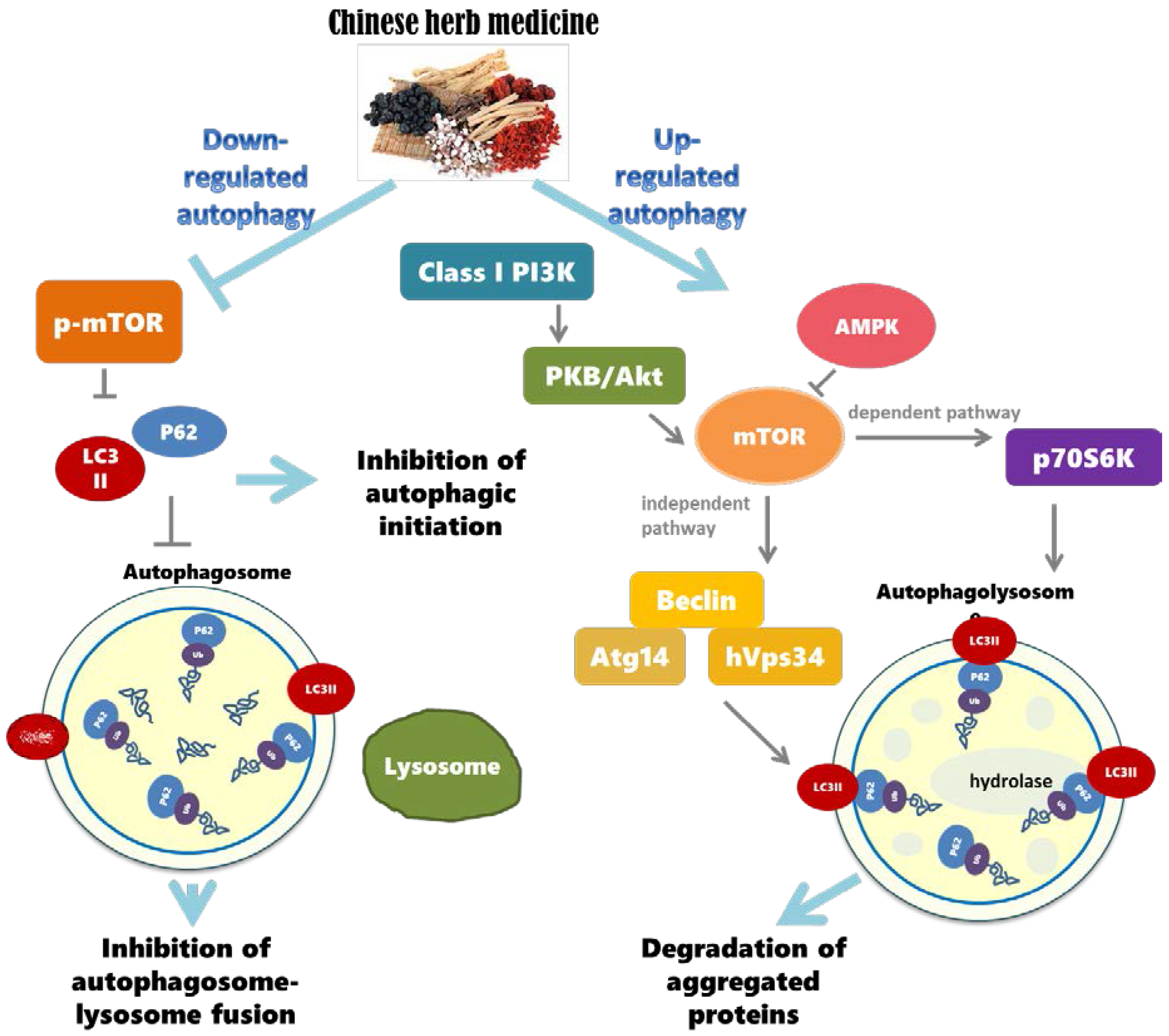
3.1. The Importance of Regulating Autophagy in the Nervous System
3.2. Autophagy Dysregulation and Neurodegenerative Diseases
3.3. Chinese Herbal Compounds Promote the Regulation of Autophagy and Improve Neurodegenerative Diseases
3.4. Brief Summary of Chinese Herbal Compounds Involved in Regulating Autophagy
4. Chinese Herbal Drugs for Neuroprotection via Reduced ER Stress
4.1. ER Function and Maintenance of Neurons
4.2. The Role of ER Stress in Neuronal Diseases
4.3. Chinese Herbal Drugs Reduce ER Stress and Benefit Nervous System
4.4. Brief Summary of Chinese Herbal Drugs Involved in ER Stress
5. Chinese Herbal Drugs for Neuroprotection via Glucose Metabolism
5.1. Effects of Glucose Metabolism on Neurophysiology
5.2. Dysregulation of Glucose Metabolism in Neurodegenerative Diseases
5.3. Potential of Chinese Herbal Drugs to Improve Neural Metabolism
6. Chinese Herbal Drugs for Neuroprotection via Neurotransmitters and Synaptic Function
6.1. Neurotransmitters on Synapse and Neural Function
6.2. Neurotransmitters and Synaptic Function in Neurodegenerative Diseases
6.3. Chinese Herbal Drugs Improve Neurotransmitter Production and Synaptic Function
7. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ross, C.A.; Poirier, M.A. Protein aggregation and neurodegenerative disease. Nat. Med. 2004, 10, S10–S17. [Google Scholar] [CrossRef] [PubMed]
- Bloom, G.S. Amyloid-β and tau: The trigger and bullet in alzheimer disease pathogenesis. JAMA Neurol. 2014, 71, 505–508. [Google Scholar] [CrossRef] [PubMed]
- Gaugler, M.N.; Genc, O.; Bobela, W.; Mohanna, S.; Ardah, M.T.; El-Agnaf, O.M.; Cantoni, M.; Bensadoun, J.-C.; Schneggenburger, R.; Knott, G.W. Nigrostriatal overabundance of α-synuclein leads to decreased vesicle density and deficits in dopamine release that correlate with reduced motor activity. Acta Neuropathol. 2012, 123, 653–669. [Google Scholar] [CrossRef] [PubMed]
- Braak, H.; Tredici, K.D. Neuroanatomy and Pathology of Sporadic Parkinson's Disease; Springer: Berlin/Heidelberg, Germany, 2009. [Google Scholar]
- Blokhuis, A.M.; Groen, E.J.; Koppers, M.; van den Berg, L.H.; Pasterkamp, R.J. Protein aggregation in amyotrophic lateral sclerosis. Acta Neuropathol. 2013, 125, 777–794. [Google Scholar] [CrossRef] [PubMed]
- Sakahira, H.; Breuer, P.; Hayer-Hartl, M.K.; Hartl, F.U. Molecular chaperones as modulators of polyglutamine protein aggregation and toxicity. Proc. Natl. Acad. Sci. USA 2002, 99, 16412–16418. [Google Scholar] [CrossRef] [PubMed]
- Huie, C.W. A review of modern sample-preparation techniques for the extraction and analysis of medicinal plants. Anal. Bioanal. Chem. 2002, 373, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.F.; Wei, D.Q.; Chou, K.C. Drug candidates from traditional chinese medicines. Curr. Top. Med. Chem. 2008, 8, 1656–1665. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.W.; Wang, Y.Q.; Wei, L.C.; Shi, M.; Chan, Y.S. Chinese herbs and herbal extracts for neuroprotection of dopaminergic neurons and potential therapeutic treatment of parkinson’s disease. CNS Neurol. Disord. 2007, 6, 273–281. [Google Scholar] [CrossRef]
- Sun, Z.K.; Yang, H.Q.; Chen, S.D. Traditional chinese medicine: A promising candidate for the treatment of alzheimer’s disease. Transl. Neurodegener. 2013, 2, 6. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.S.; John, G.R.; Sikora, A.; Lee, S.C.; Brosnan, C.F. Modulation of interleukin-1beta and tumor necrosis factor alpha signaling by p2 purinergic receptors in human fetal astrocytes. J. Neurosci. 2000, 20, 5292–5299. [Google Scholar] [PubMed]
- Gavillet, M.; Allaman, I.; Magistretti, P.J. Modulation of astrocytic metabolic phenotype by proinflammatory cytokines. Glia 2008, 56, 975–989. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Huang, Y.; Zhao, L.; Li, Y.; Sun, L.; Zhou, Y.; Qian, G.; Zheng, J.C. Il-1beta and tnf-alpha induce neurotoxicity through glutamate production: A potential role for neuronal glutaminase. J. Neurochem. 2013, 125, 897–908. [Google Scholar] [CrossRef] [PubMed]
- Borsini, A.; Zunszain, P.A.; Thuret, S.; Pariante, C.M. The role of inflammatory cytokines as key modulators of neurogenesis. Trends Neurosci. 2015, 38, 145–157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vom Berg, J.; Prokop, S.; Miller, K.R.; Obst, J.; Kalin, R.E.; Lopategui-Cabezas, I.; Wegner, A.; Mair, F.; Schipke, C.G.; Peters, O.; et al. Inhibition of il-12/il-23 signaling reduces alzheimer’s disease-like pathology and cognitive decline. Nat. Med. 2012, 18, 1812–1819. [Google Scholar] [CrossRef] [PubMed]
- Sampson, T.R.; Debelius, J.W.; Thron, T.; Janssen, S.; Shastri, G.G.; Ilhan, Z.E.; Challis, C.; Schretter, C.E.; Rocha, S.; Gradinaru, V.; et al. Gut microbiota regulate motor deficits and neuroinflammation in a model of parkinson’s disease. Cell 2016, 167, 1469–1480. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.Y.; Zhang, S.P.; Cao, C.; Loh, Y.P.; Cheng, Y. Aberrations in peripheral inflammatory cytokine levels in parkinson disease: A systematic review and meta-analysis. JAMA Neurol. 2016, 73, 1316–1324. [Google Scholar] [CrossRef] [PubMed]
- Heneka, M.T.; Golenbock, D.T.; Latz, E. Innate immunity in alzheimer’s disease. Nat. Immunol. 2015, 16, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Lambertsen, K.L.; Biber, K.; Finsen, B. Inflammatory cytokines in experimental and human stroke. J. Cereb. Blood Flow Metab. 2012, 32, 1677–1698. [Google Scholar] [CrossRef] [PubMed]
- Dowlati, Y.; Herrmann, N.; Swardfager, W.; Liu, H.; Sham, L.; Reim, E.K.; Lanctot, K.L. A meta-analysis of cytokines in major depression. Biol. Psychiatry 2010, 67, 446–457. [Google Scholar] [CrossRef] [PubMed]
- Maes, M. Major depression and activation of the inflammatory response system. Adv. Exp. Med. Biol. 1999, 461, 25–46. [Google Scholar] [PubMed]
- Martinez, J.M.; Garakani, A.; Yehuda, R.; Gorman, J.M. Proinflammatory and “resiliency” proteins in the csf of patients with major depression. Depression Anxiety 2012, 29, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Chen, Y.H.; Liu, H.; Qu, H.D. Therapeutic effects of paeonol on methyl-4-phenyl-1,2,3,6-tetrahydropyridine/probenecid-induced parkinson’s disease in mice. Mol. Med. Rep. 2016, 14, 2397–2404. [Google Scholar] [CrossRef] [PubMed]
- Jing, H.; Wang, S.; Wang, M.; Fu, W.; Zhang, C.; Xu, D. Isobavachalcone attenuates MPTP-induced parkinson’s disease in mice by inhibition of microglial activation through NF-kappab pathway. PLoS ONE 2017, 12, e0169560. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.Y.; Li, C.; Zug, C.; Schluesener, H.J. Icariin ameliorates neuropathological changes, tgf-beta1 accumulation and behavioral deficits in a mouse model of cerebral amyloidosis. PLoS ONE 2014, 9, e104616. [Google Scholar] [CrossRef]
- He, F.Q.; Qiu, B.Y.; Zhang, X.H.; Li, T.K.; Xie, Q.; Cui, D.J.; Huang, X.L.; Gan, H.T. Tetrandrine attenuates spatial memory impairment and hippocampal neuroinflammation via inhibiting NF-kappab activation in a rat model of alzheimer’s disease induced by amyloid-beta(1-42). Brain Res. 2011, 1384, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Yin, C.; Deng, Y.; Gao, J.; Li, X.; Liu, Y.; Gong, Q. Icariside ii, a novel phosphodiesterase-5 inhibitor, attenuates streptozotocin-induced cognitive deficits in rats. Neuroscience 2016, 328, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Xiong, D.; Deng, Y.; Huang, B.; Yin, C.; Liu, B.; Shi, J.; Gong, Q. Icariin attenuates cerebral ischemia-reperfusion injury through inhibition of inflammatory response mediated by nf-kappab, pparalpha and ppargamma in rats. Int. Immunopharmacol. 2016, 30, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.L.; Lu, L.Q.; Li, W.; Lou, Q.; Guo, H.G.; Shi, Q.J. Oral administration of ampelopsin protects against acute brain injury in rats following focal cerebral ischemia. Exp. Ther. Med. 2017, 13, 1725–1734. [Google Scholar] [CrossRef] [PubMed]
- Ferreira Ede, O.; Fernandes, M.Y.; Lima, N.M.; Neves, K.R.; Carmo, M.R.; Lima, F.A.; Fonteles, A.A.; Menezes, A.P.; Andrade, G.M. Neuroinflammatory response to experimental stroke is inhibited by eriodictyol. Behav. Brain Res. 2016, 312, 321–332. [Google Scholar] [CrossRef] [PubMed]
- Guan, T.; Liu, Q.; Qian, Y.; Yang, H.; Kong, J.; Kou, J.; Yu, B. Ruscogenin reduces cerebral ischemic injury via nf-kappab-mediated inflammatory pathway in the mouse model of experimental stroke. Eur. J. Pharmacol. 2013, 714, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Shih, Y.T.; Hsu, Y.Y.; Chang, F.R.; Wu, Y.C.; Lo, Y.C. 6-hydroxycleroda-3,13-dien-15,16-olide protects neuronal cells from lipopolysaccharide-induced neurotoxicity through the inhibition of microglia-mediated inflammation. Planta Medica 2010, 76, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.M.; Niu, L.; Wang, L.L.; Bai, L.; Fang, X.Y.; Li, Y.C.; Yi, L.T. Berberine attenuates depressive-like behaviors by suppressing neuro-inflammation in stressed mice. Brain Res. Bull. 2017, 134, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Xu, C.; Wu, X.; Liu, F.; Du, Y.; Sun, J.; Tao, J.; Dong, J. Icariin exerts an antidepressant effect in an unpredictable chronic mild stress model of depression in rats and is associated with the regulation of hippocampal neuroinflammation. Neuroscience 2015, 294, 193–205. [Google Scholar] [CrossRef] [PubMed]
- Burman, C.; Ktistakis, N.T. Autophagosome formation in mammalian cells. Semin. Immunopathol. 2010, 32, 397–413. [Google Scholar] [CrossRef] [PubMed]
- Cherra, S.J., 3rd.; Chu, C.T. Autophagy in neuroprotection and neurodegeneration: A question of balance. Future Neurol. 2008, 3, 309–323. [Google Scholar] [PubMed]
- Marino, G.; Madeo, F.; Kroemer, G. Autophagy for tissue homeostasis and neuroprotection. Curr. Opin. Cell Biol. 2011, 23, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Nah, J.; Yuan, J.; Jung, Y.K. Autophagy in neurodegenerative diseases: From mechanism to therapeutic approach. Mol. Cells 2015, 38, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Satriano, J.; Sharma, K. Autophagy and metabolic changes in obesity-related chronic kidney disease. Nephrol. Dial. Transplant. 2013, 28 (Suppl. 4), iv29–iv36. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.L.; Zhang, H.H.; Lai, M.D. Role of autophagy and apoptosis in tumor. Zhonghua Bing Li Xue Za Zhi 2012, 41, 573–576. [Google Scholar] [PubMed]
- Ren, S.Y.; Xu, X. Role of autophagy in metabolic syndrome-associated heart disease. Biochim. Biophys. Acta 2015, 1852, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Ryter, S.W.; Nakahira, K.; Haspel, J.A.; Choi, A.M. Autophagy in pulmonary diseases. Annu. Rev. Physiol. 2012, 74, 377–401. [Google Scholar] [CrossRef] [PubMed]
- Shastry, B.S. Neurodegenerative disorders of protein aggregation. Neurochem. Int. 2003, 43, 1–7. [Google Scholar] [CrossRef]
- Wang, S.F.; Wu, M.Y.; Cai, C.Z.; Li, M.; Lu, J.H. Autophagy modulators from traditional chinese medicine: Mechanisms and therapeutic potentials for cancer and neurodegenerative diseases. J. Ethnopharmacol. 2016, 194, 861–876. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, T.; Javed, S.; Javed, S.; Tariq, A.; Samec, D.; Tejada, S.; Nabavi, S.F.; Braidy, N.; Nabavi, S.M. Resveratrol and alzheimer’s disease: Mechanistic insights. Mol. Neurobiol. 2017, 54, 2622–2635. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Chang, L.; Zhang, X.; Pei, S.; Yu, M.; Gao, J. Ginsenoside compound k promotes beta-amyloid peptide clearance in primary astrocytes via autophagy enhancement. Exp. Ther. Med. 2014, 8, 1271–1274. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Jiang, X.; Liang, Y.; Liu, Q.; Chen, S.; Guo, Y. Berberine improves cognitive impairment by promoting autophagic clearance and inhibiting production of beta-amyloid in app/tau/ps1 mouse model of alzheimer’s disease. Exp. Gerontol. 2017, 91, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Kwon, K.; Kwon, Y.S.; Kim, S.W.; Yu, K.; Lee, K.H.; Kwon, O.Y. Luteolin-induced apoptosis through activation of endoplasmic reticulum stress sensors in pheochromocytoma cells. Mol. Med. Rep. 2017, 16, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Guo, X.D.; Lei, M.; Wu, J.Y.; Jin, J.Z.; Shi, X.F.; Zhu, Z.Y.; Rukachaisirikul, V.; Hu, L.H.; Wen, T.Q.; et al. Thamnolia vermicularis extract improves learning ability in app/ps1 transgenic mice by ameliorating both abeta and tau pathologies. Acta Pharmacol. Sin. 2017, 38, 9–28. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Su, H.; Qu, Q.M. Carnosic acid prevents beta-amyloid-induced injury in human neuroblastoma sh-sy5y cells via the induction of autophagy. Neurochem. Res. 2016, 41, 2311–2323. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.; Li, Z.; Wang, H.; Zhang, X.; Yang, Z. Triptolide inhibited cytotoxicity of differentiated pc12 cells induced by amyloid-beta(2)(5)(-)(3)(5) via the autophagy pathway. PLoS ONE 2015, 10, e0142719. [Google Scholar] [CrossRef]
- Hu, G.; Gong, X.; Wang, L.; Liu, M.; Liu, Y.; Fu, X.; Wang, W.; Zhang, T.; Wang, X. Triptolide promotes the clearance of alpha-synuclein by enhancing autophagy in neuronal cells. Mol. Neurobiol. 2017, 54, 2361–2372. [Google Scholar] [CrossRef] [PubMed]
- Hung, K.C.; Huang, H.J.; Wang, Y.T.; Lin, A.M. Baicalein attenuates alpha-synuclein aggregation, inflammasome activation and autophagy in the mpp(+)-treated nigrostriatal dopaminergic system in vivo. J. Ethnopharmacol. 2016, 194, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Kuang, L.; Cao, X.; Lu, Z. Baicalein protects against rotenone-induced neurotoxicity through induction of autophagy. Biol. Pharm. Bull. 2017, 40, 1537–1543. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.Y.; Tsai, C.W. Carnosic acid attenuates 6-hydroxydopamine-induced neurotoxicity in SH-SY5Y cells by inducing autophagy through an enhanced interaction of parkin and beclin1. Mol. Neurobiol. 2017, 54, 2813–2822. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.K.; Chen, S.D.; Chuang, Y.C.; Lin, H.Y.; Huang, C.R.; Chuang, J.H.; Wang, P.W.; Huang, S.T.; Tiao, M.M.; Chen, J.B.; et al. Resveratrol partially prevents rotenone-induced neurotoxicity in dopaminergic SH-SY5Y cells through induction of heme oxygenase-1 dependent autophagy. Int. J. Mol. Sci. 2014, 15, 1625–1646. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Chen, M.; Wang, X.; Wang, Y.; Duan, C.; Gao, G.; Lu, L.; Wu, X.; Wang, X.; Yang, H. Piperine induces autophagy by enhancing protein phosphotase 2a activity in a rotenone-induced parkinson’s disease model. Oncotarget 2016, 7, 60823–60843. [Google Scholar] [PubMed]
- Sasazawa, Y.; Sato, N.; Umezawa, K.; Simizu, S. Conophylline protects cells in cellular models of neurodegenerative diseases by inducing mammalian target of rapamycin (mTOR)-independent autophagy. J. Biol. Chem. 2015, 290, 6168–6178. [Google Scholar] [CrossRef] [PubMed]
- Wu, A.G.; Zeng, W.; Wong, V.K.; Zhu, Y.Z.; Lo, A.C.; Liu, L.; Law, B.Y. Hederagenin and alpha-hederin promote degradation of proteins in neurodegenerative diseases and improve motor deficits in MPTP-mice. Pharmacol. Res. 2017, 115, 25–44. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Chen, B.; Wang, X.; Wu, L.; Yang, Y.; Cheng, X.; Hu, Z.; Cai, X.; Yang, J.; Sun, X.; et al. Sulforaphane protects against rotenone-induced neurotoxicity in vivo: Involvement of the mTOR, NRF2, and autophagy pathways. Sci. Rep. 2016, 6, 32206. [Google Scholar] [CrossRef] [PubMed]
- Achour, I.; Arel-Dubeau, A.M.; Renaud, J.; Legrand, M.; Attard, E.; Germain, M.; Martinoli, M.G. Oleuropein prevents neuronal death, mitigates mitochondrial superoxide production and modulates autophagy in a dopaminergic cellular model. Int. J. Mol. Sci. 2016, 17, e1293. [Google Scholar] [CrossRef] [PubMed]
- Arel-Dubeau, A.M.; Longpre, F.; Bournival, J.; Tremblay, C.; Demers-Lamarche, J.; Haskova, P.; Attard, E.; Germain, M.; Martinoli, M.G. Cucurbitacin e has neuroprotective properties and autophagic modulating activities on dopaminergic neurons. Oxid. Med. Cell. Longev. 2014, 2014, 425496. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.F.; Lee, Y.C.; Lee, K.H.; Lin, H.C.; Chen, C.L.; Shen, C.J.; Huang, C.C. Therapeutic effect of berberine on TDP-43-related pathogenesis in FTLD and ALS. J. Biomed. Sci. 2016, 23, 72. [Google Scholar] [CrossRef] [PubMed]
- Hsueh, K.W.; Chiou, T.W.; Chiang, S.F.; Yamashita, T.; Abe, K.; Borlongan, C.V.; Sanberg, P.R.; Huang, A.Y.; Lin, S.Z.; Harn, H.J. Autophagic down-regulation in motor neurons remarkably prolongs the survival of ALS mice. Neuropharmacology 2016, 108, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.M.; Zhang, J.J.; Li, S.; Chen, S.; Le, W.D. N-butylidenephthalide treatment prolongs life span and attenuates motor neuron loss in SOD1(G93A) mouse model of amyotrophic lateral sclerosis. CNS Neurosci. Ther. 2017, 23, 375–385. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Hettinger, C.L.; Zhang, D.; Rezvani, K.; Wang, X.; Wang, H. Sulforaphane enhances proteasomal and autophagic activities in mice and is a potential therapeutic reagent for huntington’s disease. J. Neurochem. 2014, 129, 539–547. [Google Scholar] [CrossRef] [PubMed]
- Vidoni, C.; Secomandi, E.; Castiglioni, A.; Melone, M.A.B.; Isidoro, C. Resveratrol protects neuronal-like cells expressing mutant huntingtin from dopamine toxicity by rescuing ATG4-mediated autophagosome formation. Neurochem. Int. 2017. S0197-0186(17)30243-7. [Google Scholar] [CrossRef] [PubMed]
- Wong, V.K.; Wu, A.G.; Wang, J.R.; Liu, L.; Law, B.Y. Neferine attenuates the protein level and toxicity of mutant huntingtin in PC-12 cells via induction of autophagy. Molecules 2015, 20, 3496–3514. [Google Scholar] [CrossRef] [PubMed]
- Pengyue, Z.; Tao, G.; Hongyun, H.; Liqiang, Y.; Yihao, D. Breviscapine confers a neuroprotective efficacy against transient focal cerebral ischemia by attenuating neuronal and astrocytic autophagy in the penumbra. Biomed. Pharmacother. 2017, 90, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Lin, C.; Wu, S.; Huang, K.; Wang, Y.; Bao, X.; Zhang, F.; Huang, Z.; Teng, H. Inhibition of autophagy is involved in the protective effects of ginsenoside RB1 on spinal cord injury. Cell. Mol. Neurobiol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Kou, X.; Liu, X.; Chen, X.; Li, J.; Yang, X.; Fan, J.; Yang, Y.; Chen, N. Ampelopsin attenuates brain aging of D-gal-induced rats through miR-34a-mediated SIRT1/mTOR signal pathway. Oncotarget 2016, 7, 74484–74495. [Google Scholar] [CrossRef] [PubMed]
- Baumann, O.; Walz, B. Endoplasmic reticulum of animal cells and its organization into structural and functional domains. Int. Rev. Cytol. 2001, 205, 149–214. [Google Scholar] [PubMed]
- Verkhratsky, A. The endoplasmic reticulum and neuronal calcium signalling. Cell Calcium 2002, 32, 393–404. [Google Scholar] [CrossRef] [PubMed]
- Bola, B.; Allan, V. How and why does the endoplasmic reticulum Move? Biochem. Soc. Trans. 2009, 37, 961–965. [Google Scholar] [CrossRef] [PubMed]
- Bannai, H.; Inoue, T.; Nakayama, T.; Hattori, M.; Mikoshiba, K. Kinesin dependent, rapid, bi-directional transport of er sub-compartment in dendrites of hippocampal neurons. J. Cell Sci. 2004, 117, 163–175. [Google Scholar] [CrossRef] [PubMed]
- Broadwell, R.; Cataldo, A. The neuronal endoplasmic reticulum: Its cytochemistry and contribution to the endomembrane system. I. Cell bodies and dendrites. J. Histochem. Cytochem. 1983, 31, 1077–1088. [Google Scholar] [CrossRef] [PubMed]
- Banno, T.; Kohno, K. Conformational changes of the smooth endoplasmic reticulum are facilitated by l-glutamate and its receptors in rat purkinje cells. J. Comp. Neurol. 1998, 402, 252–263. [Google Scholar] [CrossRef]
- Aihara, Y.; Inoue, T.; Tashiro, T.; Okamoto, K.; Komiya, Y.; Mikoshiba, K. Movement of endoplasmic reticulum in the living axon is distinct from other membranous vesicles in its rate, form, and sensitivity to microtubule inhibitors. J. Neurosci. Res. 2001, 65, 236–246. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Bailly-Maitre, B.; Reed, J.C. Endoplasmic reticulum stress: Cell life and death decisions. J. Clin. Investig. 2005, 115, 2656–2664. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.S. The glucose-regulated proteins: Stress induction and clinical applications. Trends Biochem. Sci. 2001, 26, 504–510. [Google Scholar] [CrossRef]
- Ledoux, S.; Yang, R.; Friedlander, G.; Laouari, D. Glucose depletion enhances p-glycoprotein expression in hepatoma cells. Cancer Res. 2003, 63, 7284–7290. [Google Scholar] [PubMed]
- Remondelli, P.; Renna, M. The endoplasmic reticulum unfolded protein response in neurodegenerative disorders and its potential therapeutic significance. Front. Mol. Neurosci. 2017, 10, 187. [Google Scholar] [CrossRef] [PubMed]
- Imaizumi, K.; Miyoshi, K.; Katayama, T.; Yoneda, T.; Taniguchi, M.; Kudo, T.; Tohyama, M. The unfolded protein response and alzheimer’s disease. Biochim. Biophys. Acta 2001, 1536, 85–96. [Google Scholar] [CrossRef]
- Gouras, G.K.; Tsai, J.; Naslund, J.; Vincent, B.; Edgar, M.; Checler, F.; Greenfield, J.P.; Haroutunian, V.; Buxbaum, J.D.; Xu, H. Intraneuronal aβ42 accumulation in human brain. Am. J. Pathol. 2000, 156, 15–20. [Google Scholar] [CrossRef]
- Fernandez-Vizarra, P.; Fernandez, A.; Castro-Blanco, S.; Serrano, J.; Bentura, M.; Martinez-Murillo, R.; Martinez, A.; Rodrigo, J. Intra-and extracellular aß and phf in clinically evaluated cases of alzheimer’s disease. Histol. Histopathol. 2004, 19, 823–844. [Google Scholar] [PubMed]
- Ho, Y.-S.; Yang, X.; Lau, J.C.-F.; Hung, C.H.-L.; Wuwongse, S.; Zhang, Q.; Wang, J.; Baum, L.; So, K.-F.; Chang, R.C.-C. Endoplasmic reticulum stress induces tau pathology and forms a vicious cycle: Implication in alzheimer’s disease pathogenesis. J. Alzheimers Dis. 2012, 28, 839–854. [Google Scholar] [PubMed]
- Bellucci, A.; Navarria, L.; Zaltieri, M.; Falarti, E.; Bodei, S.; Sigala, S.; Battistin, L.; Spillantini, M.; Missale, C.; Spano, P. Induction of the unfolded protein response by α-synuclein in experimental models of parkinson’s disease. J. Neurochem. 2011, 116, 588–605. [Google Scholar] [CrossRef] [PubMed]
- Cooper, A.A.; Gitler, A.D.; Cashikar, A.; Haynes, C.M.; Hill, K.J.; Bhullar, B.; Liu, K.; Xu, K.; Strathearn, K.E.; Liu, F. Α-synuclein blocks er-golgi traffic and rab1 rescues neuron loss in parkinson’s models. Science 2006, 313, 324–328. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, H.; Almer, G.; Yamashita, S.; Guégan, C.; Nagai, M.; Xu, Z.; Sosunov, A.A.; McKhann, G.M.; Przedborski, S. Spinal cord endoplasmic reticulum stress associated with a microsomal accumulation of mutant superoxide dismutase-1 in an als model. Proc. Natl. Acad. Sci. USA 2006, 103, 6025–6030. [Google Scholar] [CrossRef] [PubMed]
- Nishitoh, H.; Kadowaki, H.; Nagai, A.; Maruyama, T.; Yokota, T.; Fukutomi, H.; Noguchi, T.; Matsuzawa, A.; Takeda, K.; Ichijo, H. Als-linked mutant sod1 induces er stress-and ask1-dependent motor neuron death by targeting derlin-1. Genes Dev. 2008, 22, 1451–1464. [Google Scholar] [CrossRef] [PubMed]
- Urushitani, M.; Ezzi, S.A.; Matsuo, A.; Tooyama, I.; Julien, J.-P. The endoplasmic reticulum-golgi pathway is a target for translocation and aggregation of mutant superoxide dismutase linked to als. FASEB J. 2008, 22, 2476–2487. [Google Scholar] [CrossRef] [PubMed]
- Atkin, J.D.; Farg, M.A.; Turner, B.J.; Tomas, D.; Lysaght, J.A.; Nunan, J.; Rembach, A.; Nagley, P.; Beart, P.M.; Cheema, S.S. Induction of the unfolded protein response in familial amyotrophic lateral sclerosis and association of protein-disulfide isomerase with superoxide dismutase 1. J. Biol. Chem. 2006, 281, 30152–30165. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Gui, D.; Chen, J.; He, D.; Luo, Y.; Wang, N. Down-regulation of PERK-ATF4-chop pathway by astragaloside iv is associated with the inhibition of endoplasmic reticulum stress-induced podocyte apoptosis in diabetic rats. Cell. Physiol. Biochem. 2014, 33, 1975–1987. [Google Scholar] [CrossRef] [PubMed]
- Chan, W.-S.; Durairajan, S.S.K.; Lu, J.-H.; Wang, Y.; Xie, L.-X.; Kum, W.-F.; Koo, I.; Yung, K.K.L.; Li, M. Neuroprotective effects of astragaloside iv in 6-hydroxydopamine-treated primary nigral cell culture. Neurochem. Int. 2009, 55, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.H.; Choi, A.Y.; Yoon, H.; Choe, W.; Yoon, K.-S.; Ha, J.; Yeo, E.-J.; Kang, I. Baicalein protects HT22 murine hippocampal neuronal cells against endoplasmic reticulum stress-induced apoptosis through inhibition of reactive oxygen species production and chop induction. Exp. Mol. Med. 2010, 42, 811–822. [Google Scholar] [CrossRef] [PubMed]
- Song, J.-X.; Choi, M.Y.-M.; Wong, K.C.-K.; Chung, W.W.-Y.; Sze, S.C.-W.; Ng, T.-B.; Zhang, K.Y.-B. Baicalein antagonizes rotenone-induced apoptosis in dopaminergic SH-SY5Y cells related to parkinsonism. Chin. Med. 2012, 7, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, G.F.; Zhang, Y.; Zhao, G. Crocin protects PC12 cells against MPP(+)-induced injury through inhibition of mitochondrial dysfunction and er stress. Neurochem. Int. 2015, 89, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Karakani, A.M.; Riazi, G.; Mahmood Ghaffari, S.; Ahmadian, S.; Mokhtari, F.; Jalili Firuzi, M.; Zahra Bathaie, S. Inhibitory effect of corcin on aggregation of 1N/4R human tau protein in vitro. Iran. J. Basic Med. Sci. 2015, 18, 485–492. [Google Scholar] [PubMed]
- Gao, Y.L.; Zhang, Y.; Cao, J.P.; Wu, S.B.; Cai, X.H.; Zhang, Y.C.; Zhang, R.J.; Song, X.G.; Zhang, L.D. Regulation of the endoplasmic reticulum stress response and neuroprotective effects of acupuncture on brain injury caused by heroin addiction. Acupunct. Med. 2017, 35, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Drubach, D. The Brain Explained; Pearson Prentice Hall: Upper Saddle River, NJ, USA, 2000. [Google Scholar]
- Belanger, M.; Allaman, I.; Magistretti, P.J. Brain energy metabolism: Focus on astrocyte-neuron metabolic cooperation. Cell Metab. 2011, 14, 724–738. [Google Scholar] [CrossRef] [PubMed]
- Ott, A.; Stolk, R.P.; van Harskamp, F.; Pols, H.A.; Hofman, A.; Breteler, M.M. Diabetes mellitus and the risk of dementia: The rotterdam study. Neurology 1999, 53, 1937–1942. [Google Scholar] [CrossRef] [PubMed]
- Akter, K.; Lanza, E.A.; Martin, S.A.; Myronyuk, N.; Rua, M.; Raffa, R.B. Diabetes mellitus and alzheimer's disease: Shared pathology and treatment? Br. J. Clin. Pharmacol. 2011, 71, 365–376. [Google Scholar] [CrossRef] [PubMed]
- De la Monte, S.M.; Wands, J.R. Alzheimer's disease is type 3 diabetes-evidence reviewed. J. Diabetes Sci. Technol. 2008, 2, 1101–1113. [Google Scholar] [CrossRef] [PubMed]
- Mosconi, L. Glucose metabolism in normal aging and alzheimer’s disease: Methodological and physiological considerations for pet studies. Clin. Transl. Imaging 2013, 1. [Google Scholar] [CrossRef] [PubMed]
- Roberts, R.O.; Knopman, D.S.; Cha, R.H.; Mielke, M.M.; Pankratz, V.S.; Boeve, B.F.; Kantarci, K.; Geda, Y.E.; Jack, C.R., Jr.; Petersen, R.C.; et al. Diabetes and elevated hemoglobin a1c levels are associated with brain hypometabolism but not amyloid accumulation. J. Nucl. Med. 2014, 55, 759–764. [Google Scholar] [CrossRef] [PubMed]
- Soucek, T.; Cumming, R.; Dargusch, R.; Maher, P.; Schubert, D. The regulation of glucose metabolism by hif-1 mediates a neuroprotective response to amyloid beta peptide. Neuron 2003, 39, 43–56. [Google Scholar] [CrossRef]
- Horwood, N.; Davies, D.C. Immunolabelling of hippocampal microvessel glucose transporter protein is reduced in alzheimer’s disease. Virchows Arch. 1994, 425, 69–72. [Google Scholar] [CrossRef] [PubMed]
- Guan, T.; Li, Y.; Sun, H.; Tang, X.; Qian, Y. Effects of maslinic acid, a natural triterpene, on glycogen metabolism in cultured cortical astrocytes. Planta Med. 2009, 75, 1141–1143. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Guan, T.; Tang, X.; Huang, L.; Huang, M.; Li, Y.; Sun, H. Maslinic acid, a natural triterpenoid compound from olea europaea, protects cortical neurons against oxygen-glucose deprivation-induced injury. Eur. J. Pharmacol. 2011, 670, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.H.; Yang, L.L.; Wang, C.C.; Hu, S.Y.; Chang, S.F.; Lee, Y.H. Differential effects of natural polyphenols on neuronal survival in primary cultured central neurons against glutamate- and glucose deprivation-induced neuronal death. Brain Res. 2003, 986, 103–113. [Google Scholar] [CrossRef]
- Sun, A.Y.; Wang, Q.; Simonyi, A.; Sun, G.Y. Botanical phenolics and neurodegeneration. In Herbal Medicine: Biomolecular and Clinical Aspects, 2nd ed.; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2011. [Google Scholar]
- Witte, A.V.; Kerti, L.; Margulies, D.S.; Floel, A. Effects of resveratrol on memory performance, hippocampal functional connectivity, and glucose metabolism in healthy older adults. J. Neurosci. 2014, 34, 7862–7870. [Google Scholar] [CrossRef] [PubMed]
- Edwards, R.H. The neurotransmitter cycle and quantal size. Neuron 2007, 55, 835–858. [Google Scholar] [CrossRef] [PubMed]
- Meldrum, B.S. Glutamate as a neurotransmitter in the brain: Review of physiology and pathology. J. Nutr. 2000, 130, 1007S–1015S. [Google Scholar] [CrossRef] [PubMed]
- Bowery, N.G.; Smart, T.G. Gaba and glycine as neurotransmitters: A brief history. Br. J. Pharmacol. 2006, 147 (Suppl. 1), S109–S119. [Google Scholar] [CrossRef] [PubMed]
- Bak, L.K.; Schousboe, A.; Waagepetersen, H.S. The glutamate/gaba-glutamine cycle: Aspects of transport, neurotransmitter homeostasis and ammonia transfer. J. Neurochem. 2006, 98, 641–653. [Google Scholar] [CrossRef] [PubMed]
- Prado, M.A.; Reis, R.A.; Prado, V.F.; de Mello, M.C.; Gomez, M.V.; de Mello, F.G. Regulation of acetylcholine synthesis and storage. Neurochem. Int. 2002, 41, 291–299. [Google Scholar] [CrossRef]
- Barone, P. Neurotransmission in parkinson’s disease: Beyond dopamine. Eur. J. Neurol. 2010, 17, 364–376. [Google Scholar] [CrossRef] [PubMed]
- Foerster, B.R.; Pomper, M.G.; Callaghan, B.C.; Petrou, M.; Edden, R.A.; Mohamed, M.A.; Welsh, R.C.; Carlos, R.C.; Barker, P.B.; Feldman, E.L. An imbalance between excitatory and inhibitory neurotransmitters in amyotrophic lateral sclerosis revealed by use of 3-t proton magnetic resonance spectroscopy. JAMA Neurol. 2013, 70, 1009–1016. [Google Scholar] [CrossRef] [PubMed]
- Newington, J.T.; Harris, R.A.; Cumming, R.C. Reevaluating metabolism in alzheimer’s disease from the perspective of the astrocyte-neuron lactate shuttle model. J. Neurodegener. Dis. 2013, 2013, 234572. [Google Scholar] [CrossRef] [PubMed]
- Mucke, L.; Selkoe, D.J. Neurotoxicity of amyloid beta-protein: Synaptic and network dysfunction. Cold Spring Harb. Perspect. Med. 2012, 2, a006338. [Google Scholar] [CrossRef] [PubMed]
- Palop, J.J.; Chin, J.; Roberson, E.D.; Wang, J.; Thwin, M.T.; Bien-Ly, N.; Yoo, J.; Ho, K.O.; Yu, G.Q.; Kreitzer, A.; et al. Aberrant excitatory neuronal activity and compensatory remodeling of inhibitory hippocampal circuits in mouse models of alzheimer’s disease. Neuron 2007, 55, 697–711. [Google Scholar] [CrossRef] [PubMed]
- Shaw, P.J.; Ince, P.G. Glutamate, excitotoxicity and amyotrophic lateral sclerosis. J. Neurol. 1997, 244 (Suppl. 2), S3–S14. [Google Scholar] [CrossRef] [PubMed]
- Marczynski, T.J. Gabaergic deafferentation hypothesis of brain aging and alzheimer’s disease revisited. Brain Res. Bull. 1998, 45, 341–379. [Google Scholar] [CrossRef]
- Nava-Mesa, M.O.; Jimenez-Diaz, L.; Yajeya, J.; Navarro-Lopez, J.D. Gabaergic neurotransmission and new strategies of neuromodulation to compensate synaptic dysfunction in early stages of alzheimer’s disease. Front. Cell. Neurosci. 2014, 8, 167. [Google Scholar] [CrossRef] [PubMed]
- Pimlott, S.L.; Piggott, M.; Owens, J.; Greally, E.; Court, J.A.; Jaros, E.; Perry, R.H.; Perry, E.K.; Wyper, D. Nicotinic acetylcholine receptor distribution in alzheimer’s disease, dementia with lewy bodies, parkinson’s disease, and vascular dementia: In vitro binding study using 5-[(125)i]-a-85380. Neuropsychopharmacology 2004, 29, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Rajamani, K.; Liu, J.W.; Wu, C.H.; Chiang, I.T.; You, D.H.; Lin, S.Y.; Hsieh, D.K.; Lin, S.Z.; Harn, H.J.; Chiou, T.W. N-butylidenephthalide exhibits protection against neurotoxicity through regulation of tryptophan 2, 3 dioxygenase in spinocerebellar ataxia type 3. Neuropharmacology 2017, 117, 434–446. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.Y.; Chen, S.M.; Lu, H.E.; Lai, S.M.; Lai, P.S.; Shen, P.W.; Chen, P.Y.; Shen, C.I.; Harn, H.J.; Lin, S.Z.; et al. N-butylidenephthalide attenuates alzheimer’s disease-like cytopathy in down syndrome induced pluripotent stem cell-derived neurons. Sci. Rep. 2015, 5, 8744. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.M.; Hu, G.Y. Huperzine a, a nootropic alkaloid, inhibits n-methyl-d-aspartate-induced current in rat dissociated hippocampal neurons. Neuroscience 2001, 105, 663–669. [Google Scholar] [CrossRef]
- Yang, G.; Wang, Y.; Tian, J.; Liu, J.P. Huperzine a for alzheimer’s disease: A systematic review and meta-analysis of randomized clinical trials. PLoS ONE 2013, 8, e74916. [Google Scholar] [CrossRef] [PubMed]
- Tzeng, Y.M.; Lee, M.J. Neuroprotective properties of kavalactones. Neural Regen. Res. 2015, 10, 875–877. [Google Scholar] [PubMed]
- Breyer, A.; Elstner, M.; Gillessen, T.; Weiser, D.; Elstner, E. Glutamate-induced cell death in neuronal HT22 cells is attenuated by extracts from st. John’s wort (hypericum perforatum l.). Phytomedicine 2007, 14, 250–255. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |

| Herbal Drugs | Plant Sources | Structure | Involved Mechanism (s) | Treated Model | Main Citations |
|---|---|---|---|---|---|
| 6-hydroxycleroda-3,13-dien-15,16-olide (PL3) | Polyalthia longifolia var. pendula |  | cytokine regulation | LPS-induced model | [32] |
| Ampelopsin | Ampelopsis grossedentata |  | cytokine regulation, autophagy regulation | Stroke, brain aging | [29,71] |
| Astragaloside IV | Astragalus membranaceus |  | ER stress | PD | [94] |
| Baicalein | Scutellaria baicalensis and S. lateriflora |  | autophagy regulation, ER stress | PD | [53,54,96] |
| Berberine | Coptis chinensis |  | cytokine regulation, autophagy regulation | depression, AD, and ALS | [33,47,63] |
| Breviscapine | Erigerin breviscapus |  | autophagy regulation | cerebral ischemic | [69] |
| n-butylidenephthalide | Angelica sinensis |  | autophagy regulation, neurotransmitters and synaptic function | ALS, SCA3, and down syndrome model | [64,65,128,129] |
| Carnosic acid | Rosmarinus officinalis |  | autophagy regulation | AD and PD | [50,55] |
| Conophylline | Ervatamia microphylla |  | autophagy regulation | PD and HD | [58] |
| Crocin | Crocus sativus |  | ER stress | PD and AD | [97,98] |
| Cucurbitacin E | Wilbrandia ebracteata |  | autophagy regulation | PD | [62] |
| Eriodictyol | Dracocephalum rupestre |  | cytokine regulation | stroke | [30] |
| Ginsenoside compound K | Panax ginseng |  | autophagy regulation | AD | [46] |
| Ginsenoside Rb1 | Panax ginseng |  | autophagy regulation | spinal cord injury | [70] |
| Hederagenin | Hedera helix |  | autophagy regulation | PD | [59] |
| Huperzine A | Huperzia serrata |  | neurotransmitters and synaptic function | AD | [130,131] |
| Icariin | Epimedium segittatum |  | cytokine regulation | AD, stroke, depression | [25,28,34] |
| Icariside II | Epimedium brevicornum |  | cytokine regulation | AD | [26] |
| Isobavachalcone | Psoralea corylifolia |  | cytokine regulation | PD | [24] |
| Luteolin | Lonicera japonica |  | autophagy regulation | AD | [48] |
| Maslinic acid | Olea europaea |  | glucose metabolism | oxygen-glucose deprivation-induced injury model | [110] |
| Neferine | Nelumbo nucifera |  | autophagy regulation | HD | [68] |
| Oleuropein | Olea europaea |  | autophagy regulation | PD | [61] |
| Paeonol | Cortex Moutan |  | cytokine regulation | PD | [23] |
| Piperine | Piper longum L. |  | autophagy regulation | PD | [57] |
| Resveratrol | red wine |  | autophagy regulation, glucose metabolism | AD, PD, HD, and healthy older adults | [45,56,67,113] |
| Ruscogenin | Ophiopogon japonicas |  | cytokine regulation | Ischemic stroke | [31] |
| Sulforaphane | cruciferous vegetables |  | autophagy regulation | PD, HD | [60,66] |
| Tetrandrine | Stephania tetrandra |  | cytokine regulation | AD | [27] |
| Thamnolia vermicularis extract | Thamnolia vermicularis |  | autophagy regulation | AD | [49] |
| Triptolide | Tripterygium wilfordii |  | autophagy regulation | AD, PD | [51,52] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ting, H.-C.; Chang, C.-Y.; Lu, K.-Y.; Chuang, H.-M.; Tsai, S.-F.; Huang, M.-H.; Liu, C.-A.; Lin, S.-Z.; Harn, H.-J. Targeting Cellular Stress Mechanisms and Metabolic Homeostasis by Chinese Herbal Drugs for Neuroprotection. Molecules 2018, 23, 259. https://doi.org/10.3390/molecules23020259
Ting H-C, Chang C-Y, Lu K-Y, Chuang H-M, Tsai S-F, Huang M-H, Liu C-A, Lin S-Z, Harn H-J. Targeting Cellular Stress Mechanisms and Metabolic Homeostasis by Chinese Herbal Drugs for Neuroprotection. Molecules. 2018; 23(2):259. https://doi.org/10.3390/molecules23020259
Chicago/Turabian StyleTing, Hsiao-Chien, Chia-Yu Chang, Kang-Yun Lu, Hong-Meng Chuang, Sheng-Feng Tsai, Mao-Hsuan Huang, Ching-Ann Liu, Shinn-Zong Lin, and Horng-Jyh Harn. 2018. "Targeting Cellular Stress Mechanisms and Metabolic Homeostasis by Chinese Herbal Drugs for Neuroprotection" Molecules 23, no. 2: 259. https://doi.org/10.3390/molecules23020259





