Nigella damascena L. Essential Oil—A Valuable Source of β-Elemene for Antimicrobial Testing
Abstract
:1. Introduction
2. Results
2.1. Chemical Composition of N. damascena Essential Oil
2.2. Isolation of β-Elemene
2.3. Identification of Isolated β-Elemene
2.4. Antimicrobial Testing
3. Discussion
3.1. N. damascena Essential Oil Is a New, Rich Source for β-Elemene Isolation
3.2. β-Elemene and N. damascena Essential Oil Exert Action against M. tuberculosis H37Ra
4. Materials and Methods
4.1. Plant Material
4.2. GC-MS Analysis
4.3. Isolation and Identification of β-Elemene
4.4. Antimicrobial Assay
4.4.1. Tested Organism and Storing Conditions
4.4.2. Inoculum Preparation
4.4.3. Dilutions of Tested Substances
4.4.4. References Antimicrobials
4.4.5. Plates Preparation
4.4.6. Determination of Minimal Inhibitory Concentration (MIC)
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, S.; Zhao, Z.; Yun-Ting, S.; Zeng, Z.; Zhan, X.; Li, C.; Xie, T. A review of medicinal plant species with elemene in China. Afr. J. Pharm. Pharmacol. 2012, 6, 3032–3040. [Google Scholar] [CrossRef]
- Guo, Y.T.; Wu, X.Y.; Chen, Y.R. Determination of elemene from the volatile oil of Curcuma wenyujing. China J. Chin. Mater. Med. 1983, 3, 3–5. [Google Scholar]
- Huang, H.C.; Zhu, H.J.; JIiang, Z.F. Isolation of β-elemene from citronella oil by silver ion coordination chromatography. Chin. J. Inorg. Chem. 2009, 25, 433–438. [Google Scholar]
- Sun, M.; Li, S. Chemical Complexing Directional Separation and Purification Method for Preparing High-Purity Beta-Elemene Raw Material Medicament. Patent WO2010091629 A1, 19 August 2010. [Google Scholar]
- Wei, H.P.; Zhao, M.; Li, Y.; Liu, W.Q. Extraction and isolation of β-elemene from Eupatorium adenophorum. J. Chem. Pharm. Res. 2014, 6, 161–165. [Google Scholar]
- Adio, A.M. (−)-Trans-β-elemene and related compounds: Occurrence, synthesis, and anticancer activity. Tetrahedron 2009, 65, 5145–5159. [Google Scholar] [CrossRef]
- Dang, Y.Y.; Li, X.C.; Zhang, X.W.; Li, S.P.; Wang, Y.T. Preparative isolation and purification of six volatile compounds from essential oil of Curcuma wenyujin using high-performance centrifugal partition chromatography. J. Sep. Sci. 2010, 33, 1658–1664. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Wang, X.; Bu, Z.; Lv, L.; Tong, S. Silver ion coordination countercurrent chromatography: Separation of β-elemene from the volatile oil of Curcumae rhizoma. J. Sep. Sci. 2017, 40, 3740–3747. [Google Scholar] [CrossRef] [PubMed]
- Guzlek, H.; Wood, P.L.; Janaway, L. Performance comparison using the guess mixture to evaluate counter-current chromatography instruments. J. Chromatogr. A 2009, 1216, 4181–4186. [Google Scholar] [CrossRef] [PubMed]
- Skalicka-Woźniak, K.; Garrard, I.A. Comprehensive classification of solvent systems used for natural product purifications in countercurrent and centrifugal partition chromatography. Nat. Prod. Rep. 2015, 32, 1556–1561. [Google Scholar] [CrossRef] [PubMed]
- Skalicka-Woźniak, K.; Garrard, I. Counter-current chromatography for the separation of terpenoids: A comprehensive review with respect to the solvent systems employed. Phytochem. Rev. 2014, 13, 547–572. [Google Scholar] [CrossRef] [PubMed]
- Sieniawska, E.; Swiatek, L.; Rajtar, B.; Kozioł, E.; Polz-Dacewicz, M.; Skalicka-Wozniak, K. Carrot seed essential oil—Source of carotol and cytotoxicity study. Ind. Crops Prod. 2016, 92, 109–115. [Google Scholar] [CrossRef]
- Skalicka-Woźniak, K.; Walasek, M.; Ludwiczuk, A.; Głowniak, K. Isolation of terpenoids from Pimpinella anisum essential oil by high-performance counter-current chromatography. J. Sep. Sci. 2013, 13, 2611–2614. [Google Scholar] [CrossRef] [PubMed]
- Skalicka-Woźniak, K.; Walasek, M. Preparative separation of menthol and pulegone from peppermint oil (Mentha piperita L.) by high-performance counter-current chromatography. Phytochem. Lett. 2014, 10, 94–98. [Google Scholar] [CrossRef]
- Soothill, G.; Hu, Y.; Coates, A. Can we prevent antimicrobial resistance by csing antimicrobials better? Pathogens 2013, 2, 422–435. [Google Scholar] [CrossRef] [PubMed]
- Barrero, A.F.; Quilez del Moral, J.F.; Lara, A.; Herrador, M.M. Antimicrobial activity of sesquiterpenes from the essential oil of Juniperus thurifera Wood. Planta Med. 2005, 71, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Drage, S.; Mitter, B.; Tröls, C.; Muchugi, A.; Jamnadass, R.H.; Sessitsch, A.; Hadacek, F. Antimicrobial drimane sesquiterpenes and their effect on endophyte communities in the medical tree Warburgiau gandensis. Front. Microbiol. 2014, 5, 13. [Google Scholar] [CrossRef]
- O’Neill, T.E.; Li, H.; Colquhoun, C.D.; Johnson, J.A.; Webster, D.; Gray, C.A. Optimisation of the microplate resazurin assay for screening and bioassay-guided fractionation of phytochemical extracts against Mycobacterium tuberculosis. Phytochem. Anal. 2017, 25, 461–467. [Google Scholar] [CrossRef] [PubMed]
- Wadhwani, T.; Desai, K.; Patel, D.; Lawani, D.; Bahaley, P.; Joshi, P.; Kothari, V. Effect of various solvents on bacterial growth in context of determining MIC of various antimicrobials. Internet J. Microbiol. 2008, 7, 1–6. [Google Scholar]
- D’Antuono, L.F.; Moretti, A.; Lovato, A.F.S. Seed yield, yield components, oil content and essential oil content and composition of Nigella sativa L. and Nigella damascena L. Ind. Crops Prod. 2002, 15, 59–69. [Google Scholar] [CrossRef]
- Moretti, A.; Antuono, D.; Filippo, L.; Elementi, S. Essential oil of Nigella sativa L. and Nigella damascena L. Seed. J. Essent. Oil Res. 2004, 16, 182–183. [Google Scholar] [CrossRef]
- Fico, G.; Bader, A.; Flamini, G.; Luigi Cioni, P.; Morelli, I. Essential oil of Nigella damascena L. (Ranunculaceae) seeds. J. Essent. Oil Res. 2003, 15, 56–57. [Google Scholar] [CrossRef]
- Wajs, A.; Bonikowski, R.; Kalemba, D. Different isolation methods for determination of composition of volatiles from Nigella damascena L. Seeds. Nat. Prod. Commun. 2009, 4, 1577–1580. [Google Scholar] [PubMed]
- Benckiser, G.; Schnell, S. Biodiversity in Agricultural Production Systems; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Yan, J.; Chen, G.; Tong, S.; Feng, Y.; Sheng, L.; Lou, J. Preparative isolation and purification of germacrone and curdione from the essential oil of the rhizomes of Curcuma wenyujinby high-speed counter-current chromatography. J. Chromatogr. A 2005, 1070, 207–210. [Google Scholar]
- Cao, J.; Qi, M.; Fang, L.; Zhou, S.; Fu, R.; Zhang, P. Solid-phase microextraction–gas chromatographic–mass spectrometricanalysis of volatile compounds from IY.H. Chen et C. Ling. J. Pharm. Biomed. Anal. 2006, 40, 552–558. [Google Scholar] [CrossRef] [PubMed]
- Sajfrtova, M.; Sovova, H.; Karban, J. Enrichment of Nigella damascena extract with volatile compoundsusing supercritical fluid extraction. J. Supercrit. Fluids 2014, 94, 160–164. [Google Scholar] [CrossRef]
- Wang, S.; Zhao, Z.; Xie, T.; Zeng, Z.; Zhan, X.; Wang, A. Recent advances in the study of elemene on cancer. J. Med. Plants Res. 2012, 6, 5720–5729. [Google Scholar]
- Hashim, S.E.; Sirat, H.M.; Yen, K.H. Chemical compositions and antimicrobial activity of the essential oils of Hornstiedtia havilandii (Zingiberaceae). Nat. Prod. Commun. 2014, 9, 119–120. [Google Scholar] [PubMed]
- Rasoarivelo, S.T.; Grougnet, R.; Vérité, P.; Lecsö, M.; Butel, M.J.; Tillequin, F.; Guillou, C.R.; Deguin, B. Chemical composition and antimicrobial activity of the essential oils of Anthospermumemirnense and Anthospermumperrieri (Rubiaceae). Chem. Biodivers. 2011, 8, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Lower-Nedza, A.D.; Hong, M.; Jie, S.; Wang, Z.; Yingmao, D.; Tschiggerl, C.; Bucar, F.; Brantner, A.H. Chemical composition and antimicrobial activity of three essential oils from Curcuma wenyujin. Nat. Prod. Commun. 2013, 8, 523–526. [Google Scholar] [PubMed]
- Sieniawska, E.; Swatko-Osso, M.; Sawicki, R.; Skalicka-Woźniak, K.; Ginalska, G. Natural terpenes influence the activity of antibiotics against isolated Mycobacterium tuberculosis. Med. Princ. Pract. 2017, 26, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Landa, P.; Marsik, P.; Vanek, T.; Rada, V.; Kokoska, L. In vitro anti-microbial activity of extracts from the callus cultures of some Nigella species. Biologia 2006, 62, 285–288. [Google Scholar] [CrossRef]
- Landa, P.; Marsik, P.; Havlik, J.; Kloucek, P.; Vanek, T.; Kokoska, L. Evaluation of antimicrobial and anti-inflammatory activities of seed extracts from six Nigella species. J. Med. Food 2009, 12, 408–415. [Google Scholar] [CrossRef] [PubMed]
- Fico, G.; Panizzi, L.; Flamini, G.; Braca, A.; Morelli, I.; Tomè, F.; Cioni, P.L. Biological screening of Nigella damascena for antimicrobial and molluscicidal activities. Phytother. Res. 2004, 18, 468–470. [Google Scholar] [CrossRef] [PubMed]
- Bourgou, S.; Pichette, A.; Marzouk, B.; Legault, J. Bioactivities of black cumin essential oil and its main terpenes from tunisia. South Afr. J. Bot. 2010, 76, 210–216. [Google Scholar] [CrossRef]
- Sieniawska, E.; Los, R.; Baj, T.; Malm, A.; Glowniak, K. Antimicrobial efficacy of Mutellina purpurea essential oil and α-pinene against Staphylococcus epidermidis grown in planktonic and biofilm cultures. Ind. Crops Prod. 2013, 51, 152–157. [Google Scholar] [CrossRef]
- Baj, T.; Sieniawska, E.; Kowalski, R.; Wesolowski, M.; Ulewicz-Magulska, B. Effectiveness of the deryng and clevenger-type apparatus in isolation of various types of components of essential oil from the Mutelina purpurea Thell. flowers. Acta Pol. Pharm. 2015, 72, 507–515. [Google Scholar] [PubMed]
- The Clinical & Laboratory Standards Institute (CLSI). Methods for determining bactericidal activity of antimicrobial agents. Approved guideline. In CLSI Document M26-A; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 1999. [Google Scholar]
- The Clinical & Laboratory Standards Institute (CLSI). Reference ethod for broth dilution antifungal susceptibility testing of yeasts. Fourth informational supplement. In CLSI Document M27S; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2012. [Google Scholar]
- Palomino, J.C.; Martin, A.; Camacho, M.; Guerra, H.; Swings, J.; Portaels, F. Resazurin microtiter assay plate: Simple and inexpensive method for detection of drug resistance in Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 2002, 46, 2720–2722. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of Nigella damascena seeds are available from the authors. |
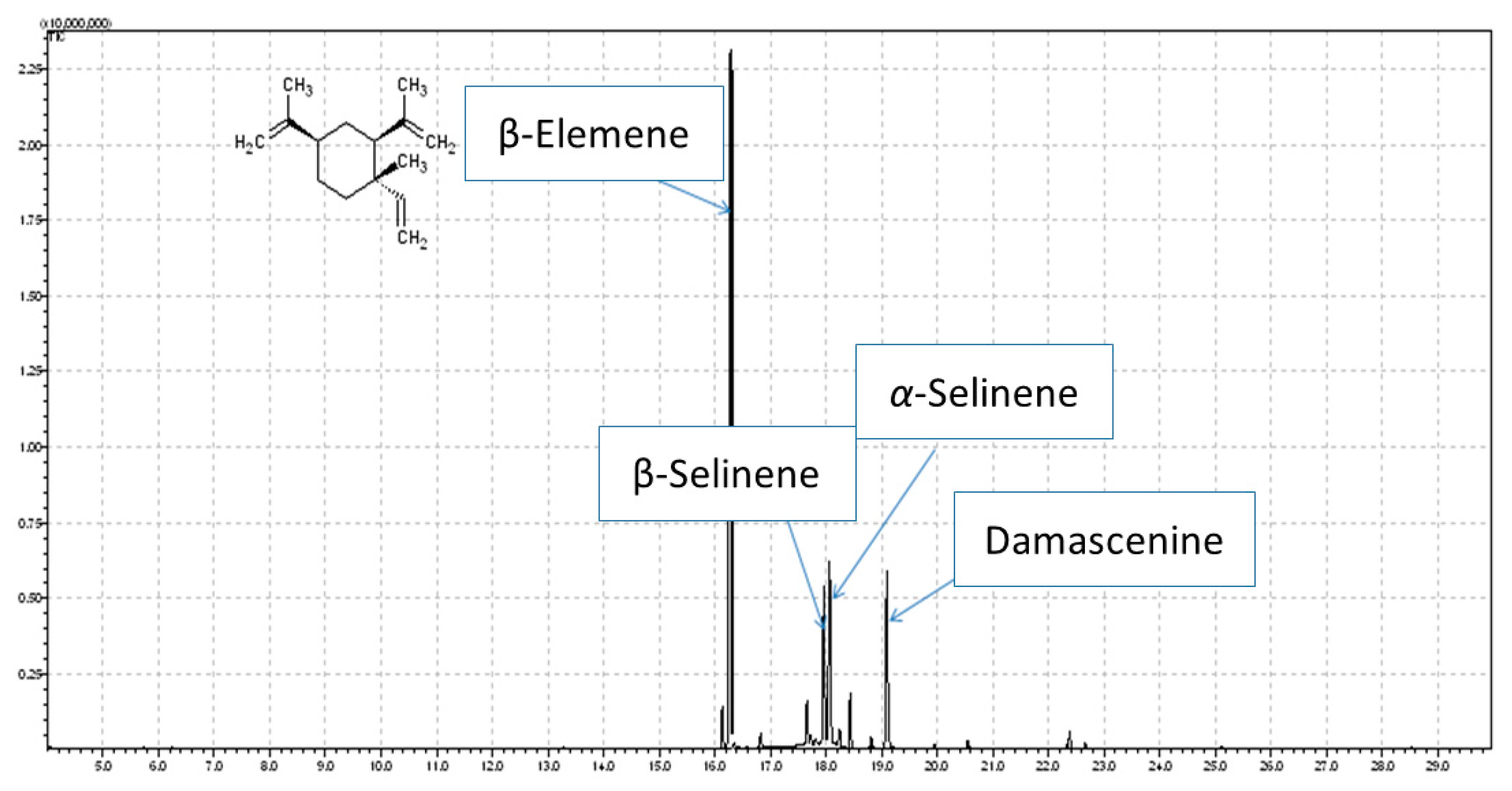
| No | Compound | tr | RI | Area (%) |
|---|---|---|---|---|
| 1 | 2,4-Dimethyl-heptane | 4.057 | 850 | 0.16 |
| 2 | Hexanoic acid, izopropyl ester | 9.838 | 1020 | 0.13 |
| 3 | 2-Methoxyl-6-antranilate | 13.270 | 1227 | 0.10 |
| 4 | Sativen | 15.500 | 1352 | 0.07 |
| 5 | Longifolene | 15.910 | 1376 | 0.09 |
| 6 | Iso-β-elemene | 16.120 | 1383 | 0.13 |
| 7 | β-Bourbonene | 16.187 | 1388 | 2.38 |
| 8 | β-Elemene | 16.270 | 1398 | 47.37 |
| 9 | Isocaryophyllene | 16.570 | 1415 | 0.08 |
| 10 | 7-Isoprenyl-1-methyl-4-methylenedecahydroazulene | 16.707 | 1428 | 0.91 |
| 11 | β-Caryophyllene | 16.803 | 1430 | 0.06 |
| 12 | Selina 4,11-diene | 17.640 | 1482 | 2.68 |
| 13 | γ-Gurjunene | 17.703 | 1486 | 0.69 |
| 14 | Valencene | 17.787 | 1488 | 0.69 |
| 15 | β-Selinene | 17.940 | 1495 | 10.10 |
| 16 | α-Selinene | 18.043 | 1506 | 13.52 |
| 17 | α-Bulnesne | 18.143 | 1514 | 0.43 |
| 18 | 8-Isopropenyl-1,5 dimethyl 1,5-cyclodecadiene | 18.223 | 1519 | 1.18 |
| 19 | δ-Cadinene | 18.320 | 1524 | 0.16 |
| 20 | 7-Epi-α-selinene | 18.403 | 1532 | 3.26 |
| 21 | Metyl 2-amino-3-methoxyl benzoate | 18.780 | 1553 | 0.06 |
| 22 | Elemol | 18.807 | 1557 | 0.93 |
| 23 | Damascenine | 19.073 | 1575 | 11.97 |
| 24 | Elema-1,3-dien-6-α-ol | 19.837 | 1630 | 0.24 |
| 25 | Selina-6-en-4-ol | 20.523 | 1673 | 0.67 |
| 26 | Longifolenaldehyde | 20.670 | 1680 | 0.07 |
| 27 | Oleylalkohol, metyl ether | 22.300 | 1802 | 0.22 |
| 28 | Unknown | 22.353 | 1807 | 1.07 |
| 29 | 1-Octadecanol, metyl ether | 22.623 | 1828 | 0.32 |
| Total | 99.2 |
| No | Solvent Systems (v/v) | K |
|---|---|---|
| 1 | Heptane/Acetonitrile/Ethyl acetate (2:1:1) * | 1.54 |
| 2 | Petroleum ether/Acetonitrile/Acetone (7:6:1) | 4.46 |
| 3 | Petroleum ether/Acetonitrile/Acetone (2:1:1) * | 1.74 |
| 4 | Petroleum ether/Acetonitrile/Acetone (2:1.5:0.5) | 2.58 |
| Mass of Sample | Fraction (min) | Purity (%) | Yield (mg) |
|---|---|---|---|
| 200 mg | 49–56 | 87 | 90 |
| 57–61 | 92 | 22 | |
| 165 mg | 50–58 | 86 | 78 |
| 59–64 | 92 | 25 | |
| 100 mg | 55–62 | 89 | 47 |
| 63–68 | 96 | 22 | |
| 69–70 | 92 | 11 |
| CIP | GEN | FOX | ETB | AMP | ND | E | |
|---|---|---|---|---|---|---|---|
| Staphylococcus aureus ATCC 25923 | 0.25 | 0.25 | - | - | - | >1000 | >1000 |
| Staphylococcus epidermidis ATCC 12228 | 0.06 | 0.125 | - | - | - | >1000 | >1000 |
| Escherichia coli ATCC 25922 | 0.008 | 0.5 | - | - | - | >1000 | >1000 |
| Pseudomonas aeruginosa ATCC 27853 | 0.25 | 2 | - | - | - | >1000 | >1000 |
| Candida albicans ATCC 10231 | - | - | - | - | 0.03 | >1000 | - |
| Candida parapsilosis ATCC 22019 | - | - | - | - | 0.25 | >1000 | - |
| Mycobacterium smegmatis ATCC 607 | 0.25 | - | 16 | - | - | >1000 | >1000 |
| Mycobacterium smegmatis ATCC 19420 | 0.25 | - | 16 | - | - | >1000 | >1000 |
| Mycobacterium tuberculosis H37Ra ATCC 25177 | 0.125 | - | - | 2 | - | 256 | 128 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sieniawska, E.; Sawicki, R.; Golus, J.; Swatko-Ossor, M.; Ginalska, G.; Skalicka-Wozniak, K. Nigella damascena L. Essential Oil—A Valuable Source of β-Elemene for Antimicrobial Testing. Molecules 2018, 23, 256. https://doi.org/10.3390/molecules23020256
Sieniawska E, Sawicki R, Golus J, Swatko-Ossor M, Ginalska G, Skalicka-Wozniak K. Nigella damascena L. Essential Oil—A Valuable Source of β-Elemene for Antimicrobial Testing. Molecules. 2018; 23(2):256. https://doi.org/10.3390/molecules23020256
Chicago/Turabian StyleSieniawska, Elwira, Rafal Sawicki, Joanna Golus, Marta Swatko-Ossor, Grazyna Ginalska, and Krystyna Skalicka-Wozniak. 2018. "Nigella damascena L. Essential Oil—A Valuable Source of β-Elemene for Antimicrobial Testing" Molecules 23, no. 2: 256. https://doi.org/10.3390/molecules23020256





