Immunomodulatory Effect of Tremella Polysaccharides against Cyclophosphamide-Induced Immunosuppression in Mice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Chemicals
2.2. Animal Experiments
2.3. Histological Observations
2.4. Measurement of Biochemical Parameters in the Serum
2.5. qRT-PCR Analysis
2.6. Statistical Analysis
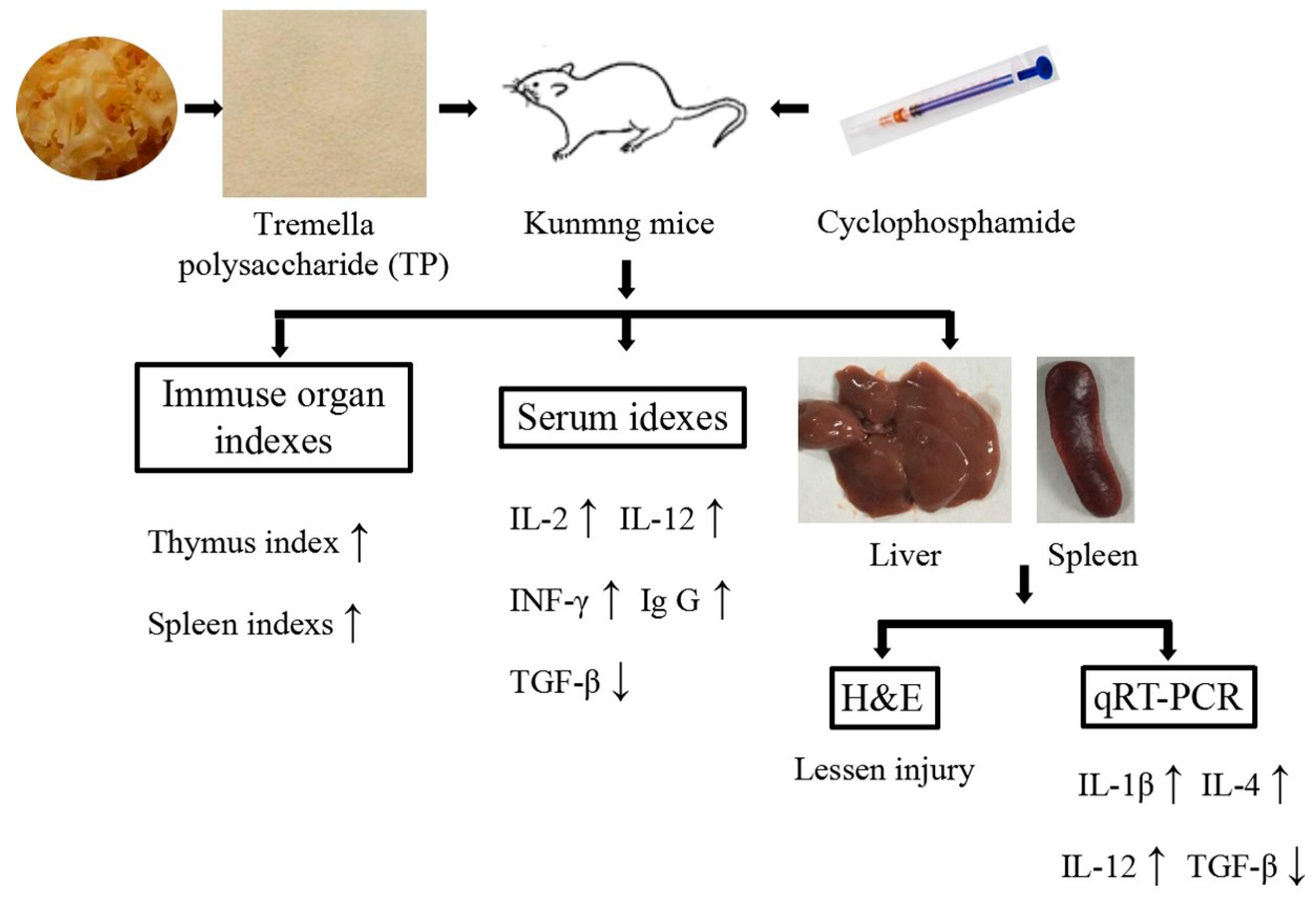
3. Results
3.1. Immune Organ Index
3.2. Histological Observations of Liver and Spleen
3.3. Effect of TP on Biochemical Parameters in the Serum
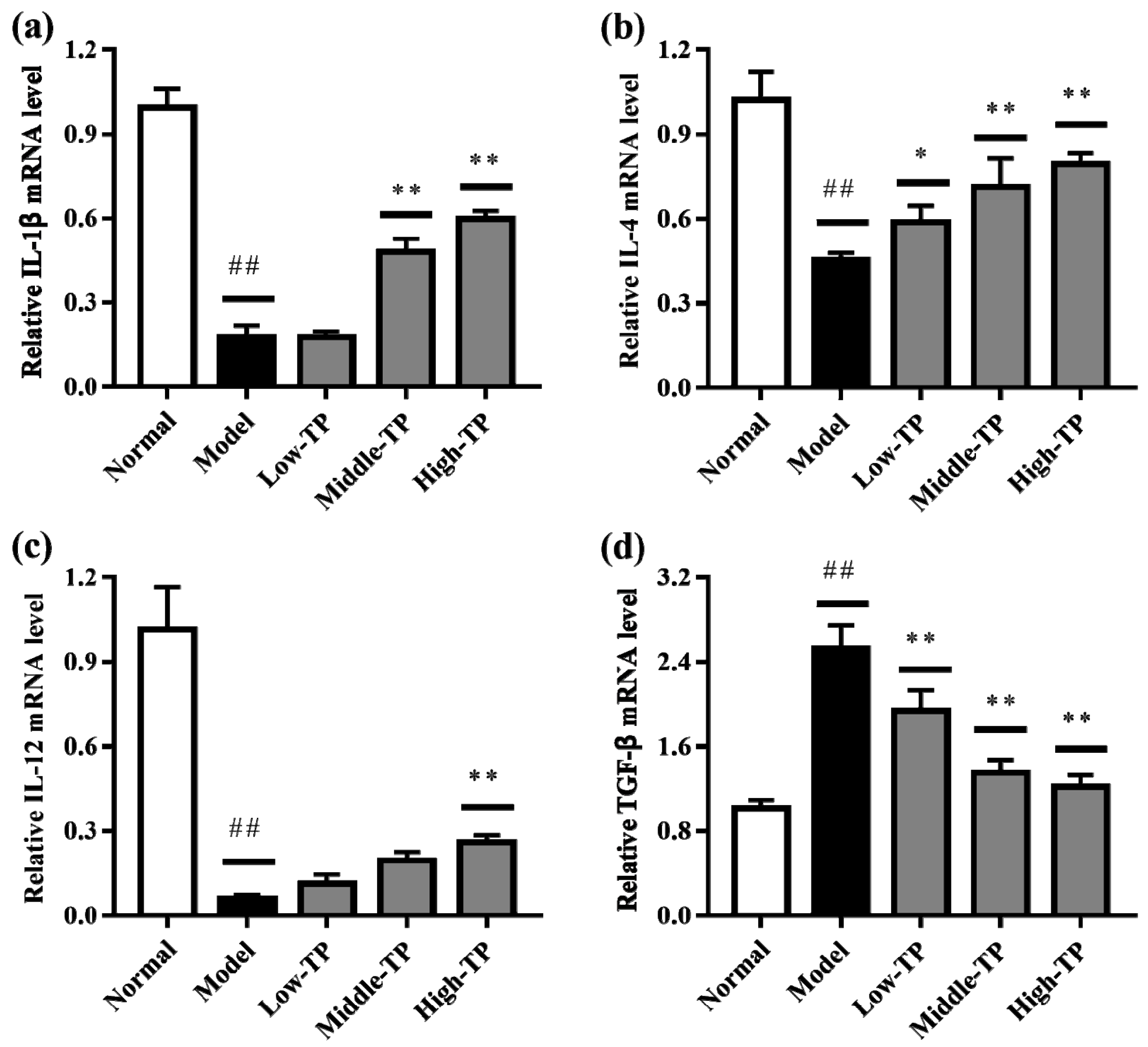
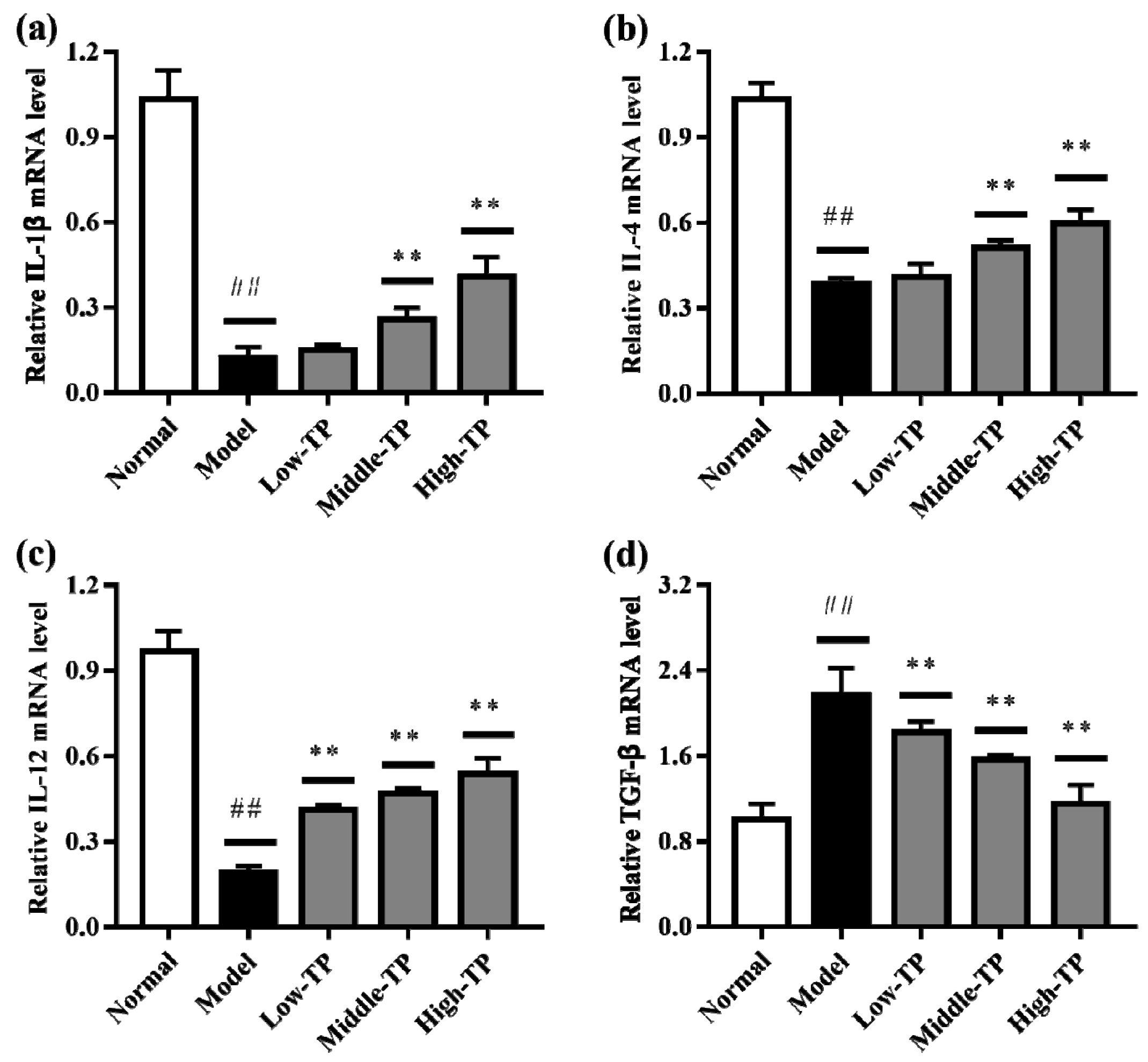
3.4. The Levels of Relative Gene Expression
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Du, X.; Zhang, Y.; Mu, H.; Lv, Z.; Yang, Y.; Zhang, J. Structural elucidation and antioxidant activity of a novel polysaccharide (TAPB1) from Tremella aurantialba. Food Hydrocolloid. 2015, 43, 459–464. [Google Scholar] [CrossRef]
- Yu, Q.; Nie, S.; Wang, J.; Liu, X.; Yin, P.; Huang, D.; Li, W.; Gong, D.; Xie, M. Chemoprotective effects of Ganoderma atrum polysaccharide in cyclophosphamide-induced mice. Int. J. Biol. Macromol. 2014, 64, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.; He, C.; Fan, Y.; Si, H.; Wang, Y.; Shi, Z.; Zhao, X.; Zheng, Y.; Liu, Q.; Zhang, H. Immune-enhancing activity of polysaccharides from Cyrtomium macrophyllum. Int. J. Biol. Macromol. 2014, 70, 590–595. [Google Scholar] [CrossRef] [PubMed]
- Cho, C.; Ahn, S.; Lim, T.; Hong, H.; Rhee, Y.K.; Yang, D.; Jang, M. Cynanchum wilfordii Polysaccharides Suppress Dextran Sulfate Sodium-Induced Acute Colitis in Mice and the Production of Inflammatory Mediators from Macrophages. Mediat. Inflamm. 2017, 2017, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.; Hwang, S.; Yoon, T.; Kim, S.H.; Shin, K. Polysaccharides from ginseng leaves inhibit tumor metastasis via macrophage and NK cell activation. Int. J. Biol. Macromol. 2017, 103, 1327–1333. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Cao, J.; Chen, G.; Xu, Y.; Lu, J.; Fang, F.; Chen, K. Anti-tumor and immunomodulatory activities of an exopolysaccharide from Rhizopus nigricans on CT26 tumor-bearing mice. Int. Immunopharmacol. 2016, 36, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Hao, L.; Zhao, X. Immunomodulatory potentials of the water-soluble yam (Dioscorea opposita Thunb) polysaccharides for the normal and cyclophosphamide-suppressed mice. Food Agric. Immunol. 2016, 27, 667–677. [Google Scholar] [CrossRef]
- Shen, C.; Mao, J.; Chen, Y.; Meng, X.; AISIKAER, A. Effect of Polysaccharides from Chinese Rice Wine on Immunity-Related Cytokines in Immunodefi cient Mice. Food Chem. 2015, 36, 158–162, (Chinese). [Google Scholar]
- Wang, Z.; Zeng, Y.; Luo, D. Structure elucidation of a non-branched and entangled heteropolysaccharide from Tremella sanguinea Peng and its antioxidant activity. Carbohyd. Polym. 2016, 152, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Deng, C.; Sun, Y.; Fu, H.; Zhang, S.; Chen, J.; Xu, X. Antioxidant and immunostimulatory activities of polysaccharides extracted from Tremella aurantialba mycelia. Mol. Med. Rep. 2016, 14, 4857–4864. [Google Scholar] [CrossRef] [PubMed]
- De Baets, S.; Vandamme, E.J. Extracellular Tremella polysaccharides: Structure, properties and applications. Biotechnol. Lett. 2001, 23, 1361–1366. [Google Scholar] [CrossRef]
- Chen, B. Optimization of extraction of Tremella fuciformis polysaccharides and its antioxidant and antitumour activities in vitro. Carbohyd. Polym. 2010, 81, 420–424. [Google Scholar] [CrossRef]
- Liu, H.; He, L. Comparison of the moisture retention capacity of Tremella polysaccharides and hyaluronic acid. J. Anhui Agric. Sci. 2012, 40, 13093–13094. [Google Scholar]
- Manente, F.A.; Quinello, C.; Ferreira, L.S.; de Andrade, C.R.; Jellmayer, J.A.; Portuondo, D.L.; Batista-Duharte, A.; Carlos, I.Z. Experimental sporotrichosis in a cyclophosphamide-induced immunosuppressed mice model. Med. Mycol. 2017, 0, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Zhao, Q.; Wang, L.; Xu, W.; Zhang, J. Protective effect of extract of Mauremys mutica suppression of immune function in mice. Food Agric. Immunol. 2016, 27, 577–588. [Google Scholar] [CrossRef]
- Deng, J.; Zhong, Y.; Wu, Y.; Luo, Z.; Sun, Y.; Wang, G.; Kurihara, H.; Li, Y.; He, R. Carnosine attenuates cyclophosphamide-induced bone marrow suppression by reducing oxidative DNA damage. Redox Biol. 2018, 14, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Martello, M.D.; David, N.; Matuo, R.; Carvalho, P.C.; Navarro, S.D.; Monreal, A.C.D.; Cunha-Laura, A.L.; Cardoso, C.A.L.; Kassuya, C.A.L.; Oliveira, R.J. Campomanesia adamantium extract induces DNA damage, apoptosis, and affects cyclophosphamide metabolism. Genet. Mol. Res. 2016, 15, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Yang, J.; Pan, L.; Guo, N.; Li, B.; Yao, J.; Wang, M.; Qi, C.; Zhang, G.; Liu, Z. Improvement of icaritin on hematopoietic function in cyclophosphamide-induced myelosuppression mice. Immunopharmacol Immunotoxicol. 2017, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Logani, M.K.; Anga, A.; Szabo, I.; Agelan, A.; Irizarry, A.R.; Ziskin, M.C. Effect of millimeter waves on cyclophosphamide induced suppression of the immune system. Bioelectromagnetics 2002, 23, 614–621. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Nie, S.; Yu, Q.; Yin, J.; Xiong, T.; Gong, D.; Xie, M. Lactobacillus plantarum NCU116 Attenuates Cyclophosphamide-Induced Immunosuppression and Regulates Th17/Treg Cell Immune Responses in Mice. J. Agric. Food Chem. 2016, 64, 1291–1297. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.Y.; Meng, M.; Sun, H.; Li, Y.; Ren, Y.Y.; Zhang, Y. Immunostimulatory activity of glycopeptides from Paecilomyces sinensis under normal and cyclophosphamide induced immunosuppressive conditions in mice models. Food Funct. 2016, 7, 3566–3576. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.F.; Mao, S.M.; Qin, H.Y.; Liu, Y.H. Study on the Extraction Technology of Polysaccharide from Tremella fuciformis. J. Anhui Agric. 2013, 41, 11148–11149. [Google Scholar]
- Xie, Y.; Zhang, H.; Dang, B.; Bing, T.; Hao, J.; Guo, H.; Wang, X. Early effects of carbon-ion irradiation on murine lymphocytes and thymocytes. Adv Space Res. 2007, 40, 563–567. [Google Scholar] [CrossRef]
- Wang, M.; Meng, X.Y.; Yang, R.L.; Qin, T.; Wang, X.Y.; Zhang, K.Y.; Fei, C.Z.; Li, Y.; Hu, Y.L.; Xue, F.Q. Cordyceps militaris polysaccharides can enhance the immunity and antioxidation activity in immunosuppressed mice. Carbohyd. Polym. 2012, 89, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Jiang, L.; Zhao, W.; Xi-nan, M.; Huang, S.; Yang, J.; Hu, T.; Chen, H. Immunomodulatory and antioxidant effects of total flavonoids of Spatholobus suberectus Dunn on PCV2 infected mice. Sci. Rep. UK 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Hu, T.; Su, Z.; Zeng, Y.; Wei, X.; Zhang, S. Immunomodulatory and antioxidant effects of carboxymethylpachymaran on the mice infected with PCV2. Int. J. Biol. Macromol. 2012, 50, 713–719. [Google Scholar] [CrossRef] [PubMed]
- Ruan, G.; Yao, X.; Liu, J.; He, J.; Li, Z.; Yang, J.; Pang, R.; Pan, X. Establishing a tree shrew model of systemic lupus erythematosus and cell transplantation treatment. Stem Cell Res. Ther. 2016, 7, 121. [Google Scholar] [CrossRef] [PubMed]
- Duggina, P.; Kalla, C.M.; Varikasuvu, S.R.; Bukke, S.; Tartte, V. Protective effect of centella triterpene saponins against cyclophosphamide-induced immune and hepatic system dysfunction in rats: Its possible mechanisms of action. J. Physiol. Biochem. 2015, 71, 435–454. [Google Scholar] [CrossRef] [PubMed]
- Cheng, K.; Song, Z.H.; Zheng, X.C.; Zhang, H.; Zhang, J.F.; Zhang, L.L.; Zhou, Y.M.; Wang, T. Effects of dietary vitamin E type on the growth performance and antioxidant capacity in cyclophosphamide immunosuppressed broilers. Poult. Sci. 2019, 96, 1159–1166. [Google Scholar] [CrossRef] [PubMed]
- Mayhew, E.; Bennett, M. Metabolic and physiologic studies of nonimmune lymphoid cells cytotoxic for fibroblastic cells in vitro. Cell Immunol. 1974, 13, 41–51. [Google Scholar] [CrossRef]
- Schoenaker, M.H.D.; Henriet, S.S.; Zonderland, J.; van Deuren, M.; Pan-Hammarström, Q.; Posthumus-van Sluijs, S.J.; Pico-Knijnenburg, I.; Weemaes, C.M.R.; IJspeert, H. Immunodeficiency in Bloom’s Syndrome. J. Clin. Immunol. 2018, 38, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Yamagishi, H.; Naito, K.; Ueda, Y.; Itoi, S.; Kohnosu, H.; Kobayashi, M.; Kubo, H.; Matsuda, A.; Oka, T. Bestatin administration and the change in cellular immunity. Biomed. Pharmacother. 1991, 45, 117–120. [Google Scholar] [CrossRef]
- Tsiligianni, I.; Antoniou, K.M.; Kyriakou, D.; Tzanakis, N.; Chrysofakis, G.; Siafakas, N.M.; Bouros, D. Th1/Th2 cytokine pattern in bronchoalveolar lavage fluid and induced sputum in pulmonary sarcoidosis. BMC Pulm. Med. 2005, 5, 8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flavell, R.A.; Gorelik, L. Immune-mediated eradication of tumors through the blockade of transforming growth factor-β signaling in T cells. Nat. Med. 2001, 7, 1118–1122. [Google Scholar]
- Wei, H.; Liu, P.; Swisher, E.; Yip, Y.Y.; Tse, J.H.; Agnew, K.; Hellström, K.E.; Hellström, I. Silencing of the TGF-β1 Gene Increases the Immunogenicity of Cells From Human Ovarian Carcinoma. J. Immunother. 2012, 35, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Morris, S.C.; Orekhova, T.; Meadows, M.J.; Heidorn, S.M.; Yang, J.; Finkelman, F.D. IL-4 induces in vivo production of IFN-gamma by NK and NKT cells. J. Immunol. 2006, 176, 5299–5305. [Google Scholar] [CrossRef] [PubMed]
- Feng, T.; Qin, H.; Wang, L.; Benveniste, E.N.; Elson, C.O.; Cong, Y. Th17 cells induce colitis and promote Th1 cell responses through IL-17 induction of innate IL-12 and IL-23 production. J. Immunol. 2011, 186, 6313–6318. [Google Scholar] [CrossRef] [PubMed]
- Hebel, K.; Rudolph, M.; Kosak, B.; Chang, H.D.; Butzmann, J.; Brunner-Weinzierl, M.C. IL-1beta and TGF-beta act antagonistically in induction and differentially in propagation of human proinflammatory precursor CD4+ T cells. J. Immunol. 2011, 187, 5627–5635. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.W.; Liu, Y.; Xu, Y.; Hong, Y.R.; Liu, Q.; Li, X.L.; Wang, Z.G. Tremella Polysaccharides attenuated sepsis through inhibiting abnormal CD4⁺CD25(high) regulatory T cells in mice. Cell. Immunol. 2014, 288, 60–65. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not available. |




| Gene | Sequence (5′-3′) * | Size (bp) |
|---|---|---|
| IL-1β | GAAATGCCACCTTTTGACAGTG | 116 |
| TGGATGCTCTCATCAGGACAG | ||
| IL-4 | GGTCTCAACCCCCAGCTAGT | 102 |
| GCCGATGATCTCTCTCAAGTGAT | ||
| 1L-12 | CTGGAGCACTCCCCATTCCTA | 160 |
| GCAGACATTCCCGCCTTTG | ||
| TGF-β | CTTCAATACGTCAGACATTCGGG | 142 |
| GTAACGCCAGGAATTGTTGCTA | ||
| β-actin | GAGAAAATCTGGCACCACACCT | 172 |
| GCACAGCCTGGATAGCAACGTA |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, Y.; Chen, X.; Yi, R.; Li, G.; Sun, P.; Qian, Y.; Zhao, X. Immunomodulatory Effect of Tremella Polysaccharides against Cyclophosphamide-Induced Immunosuppression in Mice. Molecules 2018, 23, 239. https://doi.org/10.3390/molecules23020239
Zhou Y, Chen X, Yi R, Li G, Sun P, Qian Y, Zhao X. Immunomodulatory Effect of Tremella Polysaccharides against Cyclophosphamide-Induced Immunosuppression in Mice. Molecules. 2018; 23(2):239. https://doi.org/10.3390/molecules23020239
Chicago/Turabian StyleZhou, Yalin, Xiaoyong Chen, Ruokun Yi, Guijie Li, Peng Sun, Yu Qian, and Xin Zhao. 2018. "Immunomodulatory Effect of Tremella Polysaccharides against Cyclophosphamide-Induced Immunosuppression in Mice" Molecules 23, no. 2: 239. https://doi.org/10.3390/molecules23020239





