A Systems-Level Analysis of Mechanisms of Platycodon grandiflorum Based on A Network Pharmacological Approach
Abstract
:1. Introduction
2. Compound Analysis
3. Construction of PG Compound–Target–Disease (CTD) Network
4. Target Analysis
5. Pathway Analysis
6. Disease Analysis
7. Concluding Remarks and Future Directions
Author Contributions
Funding
Conflicts of Interest
References
- Choi, J.H.; Hwang, Y.P.; Lee, H.S.; Jeong, H.G. Inhibitory effect of Platycodi Radix on ovalbumin-induced airway inflammation in a murine model of asthma. Food Chem. Toxicol. 2009, 47, 1272–1279. [Google Scholar] [CrossRef] [PubMed]
- Jang, K.J.; Kim, H.K.; Han, M.H.; Oh, Y.N.; Yoon, H.M.; Chung, Y.H.; Kim, G.Y.; Hwang, H.J.; Kim, B.W.; Choi, Y.H. Anti-inflammatory effects of saponins derived from the roots of Platycodon grandiflorus in lipopolysaccharidestimulated BV2 microglial cells. Int. J. Mol. Med. 2013, 31, 1357–1366. [Google Scholar] [CrossRef] [PubMed]
- Ahn, K.S.; Noh, E.J.; Zhao, H.L.; Jung, S.H.; Kang, S.S.; Kim, Y.S. Inhibition of inducible nitric oxide synthase and cyclooxygenase II by Platycodon grandiflorum saponins via suppression of nuclear factor-kappaB activation in RAW 264.7 cells. Life Sci. 2005, 76, 2315–2328. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.H.; Hwang, Y.P.; Kim, H.G.; Khanal, T.; Do, M.T.; Jin, S.W.; Han, H.J.; Lee, H.S.; Lee, Y.C.; Chung, Y.C.; et al. Saponins from the roots of Platycodon grandiflorum suppresses TGFbeta1-induced epithelial-mesenchymal transition via repression of PI3K/Akt, ERK1/2 and Smad2/3 pathway in human lung carcinoma A549 cells. Nutr. Cancer 2014, 66, 140–151. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Park, K.W.; Moon, K.D.; Lee, M.K.; Choi, J.; Yee, S.T.; Shim, K.H.; Seo, K.I. Induction of apoptosis in HT-29 colon cancer cells by crude saponin from Platycodi Radix. Food Chem. Toxicol. 2008, 46, 3753–3758. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.H.; Hwang, Y.P.; Han, E.H.; Kim, H.G.; Park, B.H.; Lee, H.S.; Park, B.K.; Lee, Y.C.; Chung, Y.C.; Jeong, H.G. Inhibition of acrolein-stimulated MUC5AC expression by Platycodon grandiflorum root-derived saponin in A549 cells. Food Chem. Toxicol. 2011, 49, 2157–2166. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.J.; Liu, H.B.; Wang, P.; Guan, H.S. A study on the antioxidant activity and tissues selective inhibition of lipid peroxidation by saponins from the roots of Platycodon grandiflorum. Am. J. Chin. Med. 2009, 37, 967–975. [Google Scholar] [CrossRef] [PubMed]
- Chung, M.J.; Kim, S.H.; Park, J.W.; Lee, Y.J.; Ham, S.S. Platycodon grandiflorum root attenuates vascular endothelial cell injury by oxidized low-density lipoprotein and prevents high-fat diet-induced dyslipidemia in mice by up-regulating antioxidant proteins. Nutr. Res. 2012, 32, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Ahn, K.S.; Hahn, B.S.; Kwack, K.; Lee, E.B.; Kim, Y.S. Platycodin D-induced apoptosis through nuclear factor-kappaB activation in immortalized keratinocytes. Eur. J. Pharmacol. 2006, 537, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Chen, X.; Chen, X.; Ma, D.L.; Leung, C.H.; Lu, J.J. Platycodin D potentiates proliferation inhibition and apoptosis induction upon AKT inhibition via feedback blockade in non-small cell lung cancer cells. Sci. Rep. 2016, 6, 37997. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, T.; Xu, W.S.; Wu, G.S.; Chen, X.P.; Wang, Y.T.; Lu, J.J. Platycodin D induces apoptosis, and inhibits adhesion, migration and invasion in HepG2 hepatocellular carcinoma cells. Asian Pac. J. Cancer Prev. 2014, 15, 1745–1749. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Xu, X.H.; Tang, Z.H.; Wang, Y.F.; Leung, C.H.; Ma, D.L.; Chen, X.P.; Wang, Y.T.; Chen, Y.; Lu, J.J. Platycodin D induces apoptosis and triggers ERK- and JNK-mediated autophagy in human hepatocellular carcinoma BEL-7402 cells. Acta Pharmacol. Sin. 2015, 36, 1503–1513. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, J.S.; Kim, A.K. Platycodin D induces apoptosis in MCF-7 human breast cancer cells. J. Med. Food 2010, 13, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.J.; Kang, M.; Kim, Y.S. Platycodin D inhibits lipogenesis through AMPKalpha-PPARgamma2 in 3T3-L1 cells and modulates fat accumulation in obese mice. Planta Med. 2012, 78, 1536–1542. [Google Scholar] [PubMed]
- Lee, H.; Bae, S.; Kim, Y.S.; Yoon, Y. WNT/beta-catenin pathway mediates the anti-adipogenic effect of platycodin D, a natural compound found in Platycodon grandiflorum. Life Sci. 2011, 89, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.W.; Noh, E.J.; Zhao, H.L.; Sim, J.S.; Ha, Y.W.; Shin, E.M.; Lee, E.B.; Cheong, C.S.; Kim, Y.S. Anti-inflammatory activity of prosapogenin methyl ester of platycodin D via nuclear factor-kappaB pathway inhibition. Biol. Pharm. Bull. 2008, 31, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.P.; Lee, E.B.; Kim, S.Y.; Li, D.; Ban, H.S.; Lim, S.S.; Shin, K.H.; Ohuchi, K. Inhibition of prostaglandin E2 production by platycodin D isolated from the root of Platycodon grandiflorum. Planta Med. 2001, 67, 362–364. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.T.; Yang, G.W.; Qi, C.H.; Zhou, L.; Hu, J.G.; Wang, M.S. Anti-inflammatory activity of platycodin D on alcohol-induced fatty liver rats via TLR4-MYD88-NF-kappaB signal path. Afr. J. Tradit. Complement. Altern. Med. 2016, 13, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Ryu, J.; Lee, H.J.; Park, S.H.; Kim, J.; Lee, D.; Lee, S.K.; Kim, Y.S.; Hong, J.H.; Seok, J.H.; Lee, C.J. Effects of the root of Platycodon grandiflorum on airway mucin hypersecretion in vivo and platycodin D(3) and deapi-platycodin on production and secretion of airway mucin in vitro. Phytomedicine 2014, 21, 529–533. [Google Scholar] [CrossRef] [PubMed]
- Shin, C.Y.; Lee, W.J.; Lee, E.B.; Choi, E.Y.; Ko, K.H. Platycodin D and D3 increase airway mucin release in vivo and in vitro in rats and hamsters. Planta Med. 2002, 68, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Liu, Y.; Wang, Z.; Han, Y.; Tian, Y.H.; Zhang, G.S.; Sun, Y.S.; Wang, Y.P. Platycodin D isolated from the aerial parts of Platycodon grandiflorum protects alcohol-induced liver injury in mice. Food Funct. 2015, 6, 1418–1427. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; He, S.W.; Sun, H.X.; Li, D. Platycodin D2 improves specific cellular and humoral responses to hepatitis B surface antigen in mice. Chem. Biodivers. 2010, 7, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Goh, K.I.; Cusick, M.E.; Valle, D.; Childs, B.; Vidal, M.; Barabasi, A.L. The human disease network. Proc. Natl. Acad. Sci. USA 2007, 104, 8685–8690. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hopkins, A.L. Network pharmacology: The next paradigm in drug discovery. Nat. Chem. Biol. 2008, 4, 682–690. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Park, J.H.; Kim, H.S.; Lee, C.Y.; Lee, H.J.; Kang, K.S.; Kim, C.E. Systems-level mechanisms of action of Panax ginseng: A network pharmacological approach. J. Ginseng Res. 2018, 42, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Tao, W.; Xu, X.; Wang, X.; Li, B.; Wang, Y.; Li, Y.; Yang, L. Network pharmacology-based prediction of the active ingredients and potential targets of Chinese herbal Radix Curcumae formula for application to cardiovascular disease. J. Ethnopharmacol. 2013, 145, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Li, H.; Li, S. A novel network pharmacology approach to analyse traditional herbal formulae: The Liu-Wei-Di-Huang pill as a case study. Mol. Biosyst. 2014, 10, 1014–1022. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Wang, X.; Li, S. An Integrative Platform of TCM Network Pharmacology and Its Application on a Herbal Formula, Qing-Luo-Yin. Evid. Based Complement. Alternat. Med. 2013, 2013, 456747. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wu, L.; Liu, W.; Jin, Y.; Chen, Q.; Wang, L.; Fan, X.; Li, Z.; Cheng, Y. A network pharmacology study of Chinese medicine QiShenYiQi to reveal its underlying multi-compound, multi-target, multi-pathway mode of action. PLoS ONE 2014, 9, e95004. [Google Scholar] [CrossRef] [PubMed]
- Ru, J.; Li, P.; Wang, J.; Zhou, W.; Li, B.; Huang, C.; Li, P.; Guo, Z.; Tao, W.; Yang, Y.; et al. TCMSP: A database of systems pharmacology for drug discovery from herbal medicines. J. Cheminform. 2014, 6, 13. [Google Scholar] [CrossRef] [PubMed]
- Mi, H.; Huang, X.; Muruganujan, A.; Tang, H.; Mills, C.; Kang, D.; Thomas, P.D. PANTHER version 11: Expanded annotation data from Gene Ontology and Reactome pathways, and data analysis tool enhancements. Nucleic Acids Res. 2017, 45, D183–D189. [Google Scholar] [CrossRef] [PubMed]
- Mi, H.; Muruganujan, A.; Casagrande, J.T.; Thomas, P.D. Large-scale gene function analysis with the PANTHER classification system. Nat. Protoc. 2013, 8, 1551–1566. [Google Scholar] [CrossRef] [PubMed]
- Chen, E.Y.; Tan, C.M.; Kou, Y.; Duan, Q.; Wang, Z.; Meirelles, G.V.; Clark, N.R.; Ma’ayan, A. Enrichr: Interactive and collaborative HTML5 gene list enrichment analysis tool. BMC Bioinform. 2013, 14, 128. [Google Scholar] [CrossRef] [PubMed]
- Kuleshov, M.V.; Jones, M.R.; Rouillard, A.D.; Fernandez, N.F.; Duan, Q.; Wang, Z.; Koplev, S.; Jenkins, S.L.; Jagodnik, K.M.; Lachmann, A.; et al. Enrichr: A comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 2016, 44, W90–W97. [Google Scholar] [CrossRef] [PubMed]
- Schonlau, M. Visualizing non-hierarchical and hierarchical cluster analyses with clustergrams. Comput. Stat. 2004, 19, 95–111. [Google Scholar] [CrossRef]
- Zhou, W.; Wang, J.; Wu, Z.; Huang, C.; Lu, A.; Wang, Y. Systems pharmacology exploration of botanic drug pairs reveals the mechanism for treating different diseases. Sci. Rep. 2016, 6, 36985. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, X.; Zhang, W.; Huang, C.; Li, Y.; Yu, H.; Wang, Y.; Duan, J.; Ling, Y. A novel chemometric method for the prediction of human oral bioavailability. Int. J. Mol. Sci. 2012, 13, 6964–6982. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Wang, J.; Li, Y.; Li, D.; Xu, L.; Hou, T. The application of in silico drug-likeness predictions in pharmaceutical research. Adv. Drug Deliv. Rev. 2015, 86, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Shim, H.Y.; Park, J.H.; Paik, H.D.; Nah, S.Y.; Kim, D.S.; Han, Y.S. Acacetin-induced apoptosis of human breast cancer MCF-7 cells involves caspase cascade, mitochondria-mediated death signaling and SAPK/JNK1/2-c-Jun activation. Mol. Cells 2007, 24, 95–104. [Google Scholar] [PubMed]
- Hsu, Y.L.; Kuo, P.L.; Liu, C.F.; Lin, C.C. Acacetin-induced cell cycle arrest and apoptosis in human non-small cell lung cancer A549 cells. Cancer Lett. 2004, 212, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.L.; Kuo, P.L.; Lin, C.C. Acacetin inhibits the proliferation of Hep G2 by blocking cell cycle progression and inducing apoptosis. Biochem. Pharmacol. 2004, 67, 823–829. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shen, K.H.; Hung, S.H.; Yin, L.T.; Huang, C.S.; Chao, C.H.; Liu, C.L.; Shih, Y.W. Acacetin, a flavonoid, inhibits the invasion and migration of human prostate cancer DU145 cells via inactivation of the p38 MAPK signaling pathway. Mol. Cell. Biochem. 2010, 333, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Dok-Go, H.; Lee, K.H.; Kim, H.J.; Lee, E.H.; Lee, J.; Song, Y.S.; Lee, Y.H.; Jin, C.; Lee, Y.S.; Cho, J. Neuroprotective effects of antioxidative flavonoids, quercetin, (+)-dihydroquercetin and quercetin 3-methyl ether, isolated from Opuntia ficus-indica var. saboten. Brain Res. 2003, 965, 130–136. [Google Scholar] [CrossRef]
- Teselkin, Y.O.; Babenkova, I.V.; Kolhir, V.K.; Baginskaya, A.I.; Tjukavkina, N.A.; Kolesnik, Y.A.; Selivanova, I.A.; Eichholz, A.A. Dihydroquercetin as a means of antioxidative defence in rats with tetrachloromethane hepatitis. Phytother. Res. 2000, 14, 160–162. [Google Scholar] [CrossRef]
- Jeong, C.-H.; Choi, G.N.; Kim, J.H.; Kwak, J.H.; Kim, D.O.; Kim, Y.J.; Heo, H.J. Antioxidant activities from the aerial parts of Platycodon grandiflorum. Food Chem. 2010, 118, 278–282. [Google Scholar] [CrossRef]
- Park, S.W.; Cho, C.S.; Ryu, N.H.; Kim, J.H.; Kim, J.S.; Kim, J.H. Luteolin extracted from Platycodon grandiflorum protects retinal pigment epithelial cells from oxidative stress-induced caspase-3 dependent apoptosis. Biomed. Prev. Nutr. 2012, 2, 77–80. [Google Scholar] [CrossRef]
- Kuppusamy, U.R.; Das, N.P. Effects of flavonoids on cyclic AMP phosphodiesterase and lipid mobilization in rat adipocytes. Biochem. Pharmacol. 1992, 44, 1307–1315. [Google Scholar] [CrossRef]
- Park, S.W.; Cho, C.S.; Jun, H.O.; Ryu, N.H.; Kim, J.H.; Yu, Y.S.; Kim, J.S.; Kim, J.H. Anti-angiogenic effect of luteolin on retinal neovascularization via blockade of reactive oxygen species production. Investig. Ophthalmol. Vis. Sci. 2012, 53, 7718–7726. [Google Scholar] [CrossRef] [PubMed]
- Villasenor, I.M.; Domingo, A.P. Anticarcinogenicity potential of spinasterol isolated from squash flowers. Teratog. Carcinog. Mutagen. 2000, 20, 99–105. [Google Scholar] [CrossRef]
- Jeon, G.C.; Park, M.S.; Yoon, D.Y.; Shin, C.H.; Sin, H.S.; Um, S.J. Antitumor activity of spinasterol isolated from Pueraria roots. Exp. Mol. Med. 2005, 37, 111–120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trevisan, G.; Rossato, M.F.; Walker, C.I.; Klafke, J.Z.; Rosa, F.; Oliveira, S.M.; Tonello, R.; Guerra, G.P.; Boligon, A.A.; Zanon, R.B.; et al. Identification of the plant steroid alpha-spinasterol as a novel transient receptor potential vanilloid 1 antagonist with antinociceptive properties. J. Pharmacol. Exp. Ther. 2012, 343, 258–269. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Sun, G.; Yuan, G.; Wang, R.; Sun, X. Effects of platycodin D on proliferation, apoptosis and PI3K/Akt signal pathway of human glioma U251 cells. Molecules 2014, 19, 21411–21423. [Google Scholar] [CrossRef] [PubMed]
- Yoo, D.S.; Choi, Y.H.; Cha, M.R.; Lee, B.H.; Kim, S.-J.; Yon, G.H.; Hong, K.S.; Jang, Y.S.; Lee, H.S.; Kim, Y.S.; et al. HPLC-ELSD analysis of 18 platycosides from balloon flower roots (Platycodi Radix) sourced from various regions in Korea and geographical clustering of the cultivation areas. Food Chem. 2011, 129, 645–651. [Google Scholar] [CrossRef]
- Yu, H.; Chen, J.; Xu, X.; Li, Y.; Zhao, H.; Fang, Y.; Li, X.; Zhou, W.; Wang, W.; Wang, Y. A systematic prediction of multiple drug-target interactions from chemical, genomic, and pharmacological data. PLoS ONE 2012, 7, e37608. [Google Scholar] [CrossRef] [PubMed]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, Y.; Yang, D.; Zhang, C.; Zhang, N.; Li, M.; Liu, Y. Platycodon grandiflorus–An ethnopharmacological, phytochemical and pharmacological review. J. Ethnopharmacol. 2015, 164, 147–161. [Google Scholar] [CrossRef] [PubMed]
- Nyakudya, E.; Jeong, J.H.; Lee, N.K.; Jeong, Y.S. Platycosides from the Roots of Platycodon grandiflorum and Their Health Benefits. Prev. Nutr. Food Sci. 2014, 19, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Ciruelos Gil, E.M. Targeting the PI3K/AKT/mTOR pathway in estrogen receptor-positive breast cancer. Cancer Treat. Rev. 2014, 40, 862–871. [Google Scholar] [CrossRef] [PubMed]
- Edlind, M.P.; Hsieh, A.C. PI3K-AKT-mTOR signaling in prostate cancer progression and androgen deprivation therapy resistance. Asian J. Androl. 2014, 16, 378–386. [Google Scholar] [PubMed]
- Kargman, S.L.; O’Neill, G.P.; Vickers, P.J.; Evans, J.F.; Mancini, J.A.; Jothy, S. Expression of prostaglandin G/H synthase-1 and -2 protein in human colon cancer. Cancer Res. 1995, 55, 2556–2559. [Google Scholar] [PubMed]
- Seibert, K.; Zhang, Y.; Leahy, K.; Hauser, S.; Masferrer, J.; Perkins, W.; Lee, L.; Isakson, P. Pharmacological and biochemical demonstration of the role of cyclooxygenase 2 in inflammation and pain. Proc. Natl. Acad. Sci. USA 1994, 91, 12013–12017. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.F.; Huri, D.A.; Snyder, S.H. Inducible nitric oxide synthase binds, S-nitrosylates, and activates cyclooxygenase-2. Science 2005, 310, 1966–1970. [Google Scholar] [CrossRef] [PubMed]
- Hokari, A.; Zeniya, M.; Esumi, H. Cloning and functional expression of human inducible nitric oxide synthase (NOS) cDNA from a glioblastoma cell line A-172. J. Biochem. 1994, 116, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.Y.; Whittington, D.A.; Chang, J.S.; Liao, J.; May, J.A.; Christianson, D.W. Structural aspects of isozyme selectivity in the binding of inhibitors to carbonic anhydrases II and IV. J. Med. Chem. 2002, 45, 888–893. [Google Scholar] [CrossRef] [PubMed]
- Lehenkari, P.; Hentunen, T.A.; Laitala-Leinonen, T.; Tuukkanen, J.; Vaananen, H.K. Carbonic anhydrase II plays a major role in osteoclast differentiation and bone resorption by effecting the steady state intracellular pH and Ca2+. Exp. Cell. Res. 1998, 242, 128–137. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, A.; Tamayo, P.; Mootha, V.K.; Mukherjee, S.; Ebert, B.L.; Gillette, M.A.; Paulovich, A.; Pomeroy, S.L.; Golub, T.R.; Lander, E.S.; et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 2005, 102, 15545–15550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ogata, H.; Goto, S.; Sato, K.; Fujibuchi, W.; Bono, H.; Kanehisa, M. KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res. 1999, 27, 29–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shin, D.Y.; Kim, G.Y.; Li, W.; Choi, B.T.; Kim, N.D.; Kang, H.S.; Choi, Y.H. Implication of intracellular ROS formation, caspase-3 activation and Egr-1 induction in platycodon D-induced apoptosis of U937 human leukemia cells. Biomed. Pharmacother. 2009, 63, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Yim, N.H.; Hwang, Y.H.; Liang, C.; Ma, J.Y. A platycoside-rich fraction from the root of Platycodon grandiflorum enhances cell death in A549 human lung carcinoma cells via mainly AMPK/mTOR/AKT signal-mediated autophagy induction. J. Ethnopharmacol. 2016, 194, 1060–1068. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Chen, X.; Dai, X.-Y.; Wei, B.; Weng, Q.-J.; Chen, X.; Ouyang, D.-F.; Yan, R.; Huang, Z.-J.; Jiang, H.-L.; et al. Novel Hsp90 inhibitor platycodin D disrupts Hsp90/Cdc37 complex and enhances the anticancer effect of mTOR inhibitor. Toxicol. Appl. Pharmacol. 2017, 330, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Choi, C.Y.; Kim, J.Y.; Kim, Y.S.; Chung, Y.C.; Seo, J.K.; Jeong, H.G. Aqueous extract isolated from Platycodon grandiflorum elicits the release of nitric oxide and tumor necrosis factor-α from murine macrophages. Int. Immunopharmacol. 2001, 1, 1141–1151. [Google Scholar] [CrossRef]
- Kim, J.Y.; Kim, D.H.; Kim, H.G.; Song, G.-Y.; Chung, Y.C.; Roh, S.H.; Jeong, H.G. Inhibition of tumor necrosis factor-α-induced expression of adhesion molecules in human endothelial cells by the saponins derived from roots of Platycodon grandiflorum. Toxicol. Appl. Pharmacol. 2006, 210, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Klein, T.E.; Chang, J.T.; Cho, M.K.; Easton, K.L.; Fergerson, R.; Hewett, M.; Lin, Z.; Liu, Y.; Liu, S.; Oliver, D.E.; et al. Integrating genotype and phenotype information: An overview of the PharmGKB project. Pharmacogenomics J. 2001, 1, 167–170. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Qin, C.; Li, Y.H.; Tao, L.; Zhou, J.; Yu, C.Y.; Xu, F.; Chen, Z.; Zhu, F.; Chen, Y.Z. Therapeutic target database update 2016: Enriched resource for bench to clinical drug target and targeted pathway information. Nucleic Acids Res. 2016, 44, D1069–D1074. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.-H.; Li, T.; Gao, H.-W.; Sun, W.; Chen, X.-P.; Wang, Y.-T.; Lu, J.-J. Platycodin D from Platycodonis Radix enhances the anti-proliferative effects of doxorubicin on breast cancer MCF-7 and MDA-MB-231 cells. Chin. Med. 2014, 9, 16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hwang, Y.P.; Choi, J.H.; Kim, H.G.; Lee, H.-S.; Chung, Y.C.; Jeong, H.G. Saponins from Platycodon grandiflorum inhibit hepatic lipogenesis through induction of SIRT1 and activation of AMP-activated protein kinase in high-glucose-induced HepG2 cells. Food Chem. 2013, 140, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-Y.; Moon, K.-D.; Seo, K.-I.; Park, K.-W.; Choi, M.-S.; Do, G.-M.; Jeong, Y.-K.; Cho, Y.-S.; Lee, M.-K. Supplementation of SK1 from Platycodi Radix Ameliorates Obesity and Glucose Intolerance in Mice Fed a High-Fat Diet. J. Med. Food 2009, 12, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Son, I.; Park, Y.; Lee, S.; Yang, H.; Moon, H.-I. Neuroprotective Activity of Triterpenoid Saponins from Platycodi radix Against Glutamate-induced Toxicity in Primary Cultured Rat Cortical Cells. Molecules 2007, 12, 1147. [Google Scholar] [CrossRef] [PubMed]
- Han, S.B.; Park, S.H.; Lee, K.H.; Lee, C.W.; Lee, S.H.; Kim, H.C.; Kim, Y.S.; Lee, H.S.; Kim, H.M. Polysaccharide isolated from the radix of Platycodon grandiflorum selectively activates B cells and macrophages but not T cells. Int. Immunopharmacol. 2001, 1, 1969–1978. [Google Scholar] [CrossRef]
- Ouyang, K.; Chen, L.; Sun, H.; Du, J.; Shi, M. Screening and appraisal for immunological adjuvant-active fractions from Platycodon grandiflorum total saponins. Immunopharmacol. Immunotoxicol. 2012, 34, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Nie, Q.; MacLean, A.L.; Li, Y.; Lei, J.; Li, S. Multiscale Modeling of Inflammation-Induced Tumorigenesis Reveals Competing Oncogenic and Oncoprotective Roles for Inflammation. Cancer Res. 2017, 77, 6429–6441. [Google Scholar] [CrossRef] [PubMed]
- Lv, C.; Wu, X.; Wang, X.; Su, J.; Zeng, H.; Zhao, J.; Lin, S.; Liu, R.; Li, H.; Li, X.; et al. The gene expression profiles in response to 102 traditional Chinese medicine (TCM) components: A general template for research on TCMs. Sci. Rep. 2017, 7, 352. [Google Scholar] [CrossRef] [PubMed]






| Gene Name | Protein Name | Target Degrees | Pharmacological Activities in Uniprot |
|---|---|---|---|
| PRSS1 | Trypsin-1 | 17 | - |
| DPP4 | Dipeptidyl peptidase IV | 16 | Immune activitiesAnti-diabetic activitiesAnti-microbial activities |
| ESR1 | Estrogen receptor | 12 | Anti-inflammatory activitiesAnti-cancer activitiesAnti-tumor activities |
| PTGS2 | Prostaglandin G/H synthase 2 | 12 | Anti-inflammatory activitiesAnti-cancer activitiesAnti-tumor activities |
| PTGS1 | Prostaglandin G/H synthase 1 | 12 | Anti-inflammatory activities |
| ADH1C | Alcohol dehydrogenase 1C | 12 | - |
| AR | Androgen receptor | 10 | Anti-cancer activities |
| NOS2 | Nitric oxide synthase, inducible | 9 | Anti-tumor activitiesAnti-microbial activitiesAnti-inflammatory activities |
| CA2 | Carbonic anhydrase II | 9 | Anti-cancer activities |
| F2 | Prothrombin | 9 | Cardiovascular activities |
| Cluster No. | Enriched Pathway |
|---|---|
| Cluster 1 | Pathways in cancer |
| Cluster 2 | PI3K signaling pathway |
| Cluster 3 | HIF-1 signaling pathway |
| Cluster 4 | Proteoglycans in cancer |
| Cluster 5 | Estrogen signaling pathway |
| Cluster 6 | AGE/RAGE signaling pathway in diabetic complications, apoptosis, T-cell receptor signaling pathway, Chagas disease (American trypanosomiasis), TNF signaling pathway, influenza A |
| Cluster 7 | Insulin signaling pathway, thyroid hormone signaling pathway, non-alcoholic fatty liver disease (NAFLD), insulin resistance |
| Cluster 8 | Prostate cancer, bladder cancer, pancreatic cancer, glioma, melanoma |
| Cluster 9 | Measles, Epstein–Barr virus infection, small-cell lung cancer, hepatitis B, viral carcinogenesis |
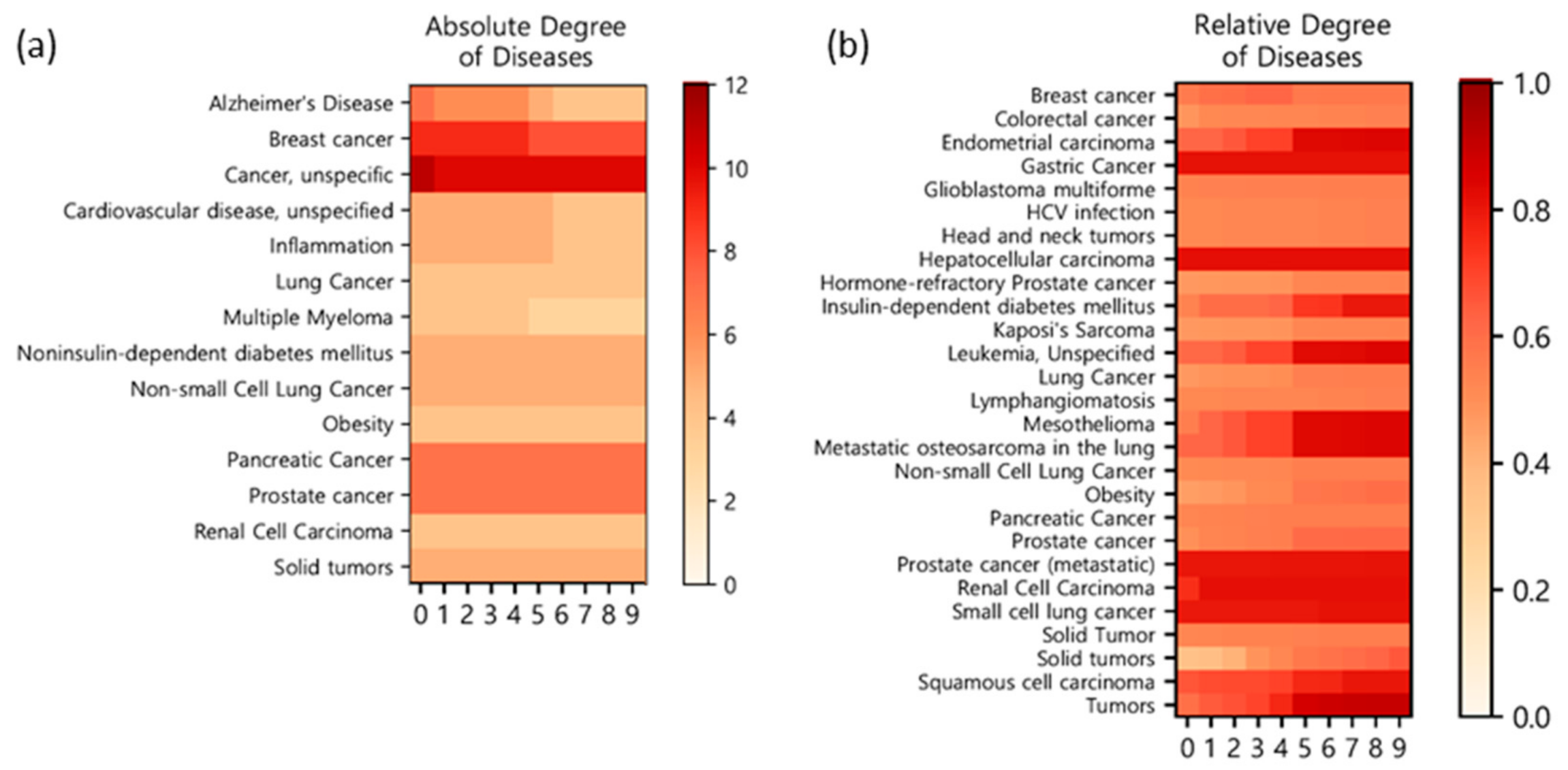
| Disease Name | Degree (From the 1st Level Threshold) | Disease Name | Degree (From the 1st Level Threshold) |
|---|---|---|---|
| Cancer, unspecific | 11 | Alcoholism | 3 |
| Breast cancer | 9 | Bladder cancer | 3 |
| Pancreatic cancer | 7 | Neurodegenerative diseases | 3 |
| Alzheimer’s disease | 7 | Brain injury | 3 |
| Prostate cancer | 7 | Schizophrenia | 3 |
| Inflammation | 5 | Hepatocellular carcinoma | 3 |
| Cardiovascular disease, unspecified | 5 | Multiple sclerosis | 3 |
| Non-small-cell lung cancer | 5 | Chronic inflammatory diseases | 3 |
| Non-insulin-dependent diabetes mellitus | 5 | Diabetes mellitus | 3 |
| Solid tumors | 5 | Osteoarthritis | 3 |
| Renal cell carcinoma | 4 | Anxiety disorder, unspecified | 3 |
| Obesity | 4 | Rheumatoid arthritis, unspecified | 3 |
| Multiple myeloma | 4 | Crohn’s disease, unspecified | 3 |
| Lung cancer | 4 | Asthma | 3 |
| Disease Name | Relative Degree (From the 1st Level Threshold) | Disease Name | Relative Degree (From the 1st Level Threshold) |
|---|---|---|---|
| Hepatocellular carcinoma | 0.82 | Pancreatic cancer | 0.53 |
| Gastric cancer | 0.81 | Solid tumor | 0.53 |
| Prostate cancer (metastatic) | 0.80 | HCV infection | 0.52 |
| Small-cell lung cancer | 0.79 | Head and neck tumors | 0.52 |
| Renal cell carcinoma | 0.75 | Lymphangiomatosis | 0.52 |
| Squamous cell carcinoma | 0.66 | Non-small cell lung cancer | 0.51 |
| Endometrial carcinoma | 0.62 | Prostate cancer | 0.49 |
| Metastatic osteosarcoma in the lung | 0.62 | Colorectal cancer | 0.48 |
| Leukemia, unspecified | 0.61 | Hormone-refractory prostate cancer | 0.47 |
| Tumors | 0.59 | Kaposi’s sarcoma | 0.47 |
| Breast cancer | 0.56 | Lung cancer | 0.47 |
| Mesothelioma | 0.55 | Obesity | 0.45 |
| Glioblastoma multiforme | 0.54 | Solid tumors | 0.34 |
| Insulin-dependent diabetes mellitus | 0.53 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, M.; Park, S.-Y.; Lee, H.-J.; Kim, C.-E. A Systems-Level Analysis of Mechanisms of Platycodon grandiflorum Based on A Network Pharmacological Approach. Molecules 2018, 23, 2841. https://doi.org/10.3390/molecules23112841
Park M, Park S-Y, Lee H-J, Kim C-E. A Systems-Level Analysis of Mechanisms of Platycodon grandiflorum Based on A Network Pharmacological Approach. Molecules. 2018; 23(11):2841. https://doi.org/10.3390/molecules23112841
Chicago/Turabian StylePark, Musun, Sa-Yoon Park, Hae-Jeung Lee, and Chang-Eop Kim. 2018. "A Systems-Level Analysis of Mechanisms of Platycodon grandiflorum Based on A Network Pharmacological Approach" Molecules 23, no. 11: 2841. https://doi.org/10.3390/molecules23112841





