Anticonvulsant Activity of Halogen-Substituted Cinnamic Acid Derivatives and Their Effects on Glycosylation of PTZ-Induced Chronic Epilepsy in Mice
Abstract
:1. Introduction
2. Results
2.1. Anticonvulsant Activities
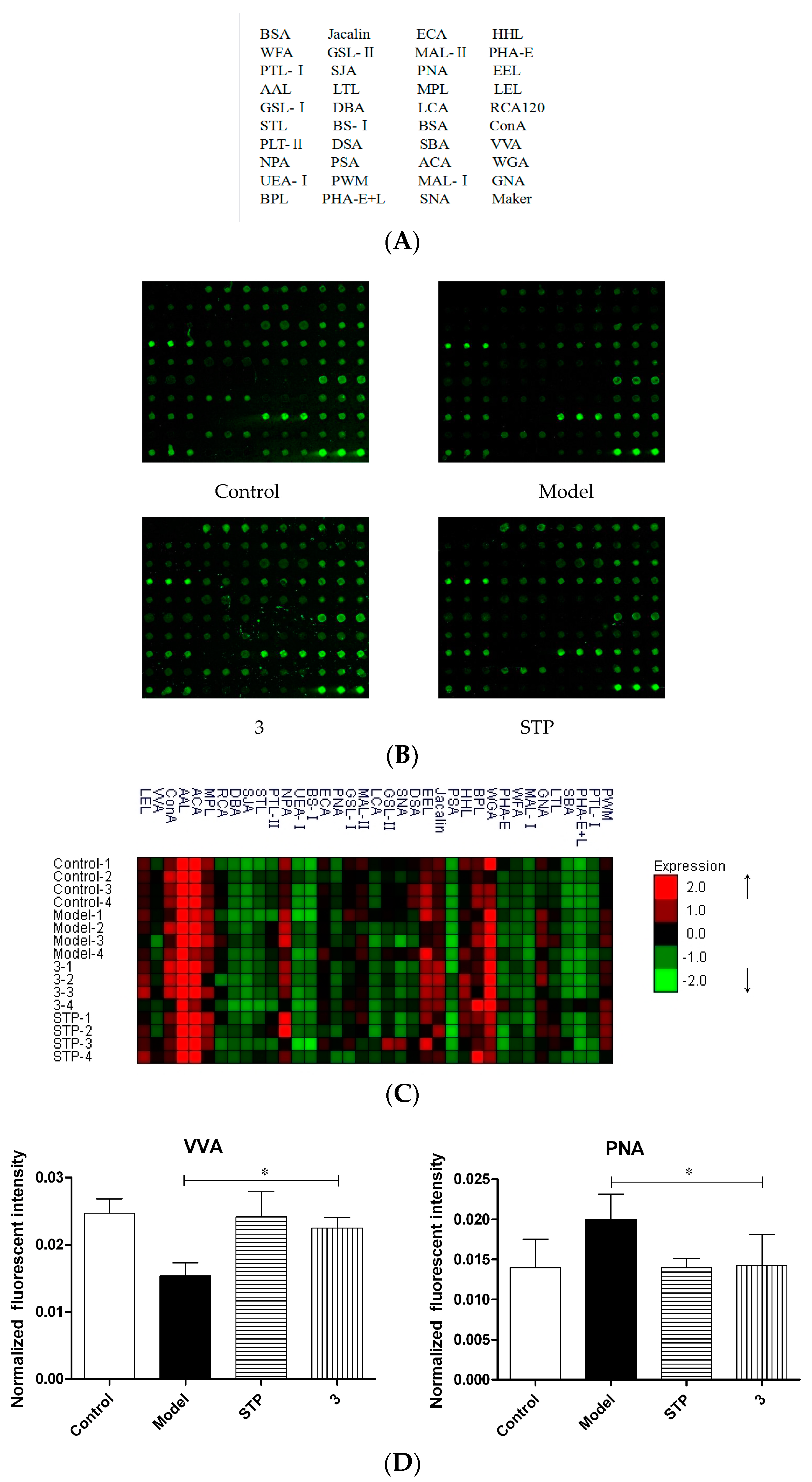
2.2. Lectin Microarrays
3. Discussion
4. Materials and Methods
4.1. Drugs
4.2. Animals
4.3. MES-Induced Seizures in Mice
4.4. Rotarod Test
4.5. PTZ-Induced Acute Seizures in Mice
4.6. PTZ-Induced Chronic Seizures in Mice
4.7. Brain Processing
4.8. Lectin Microarrays
4.9. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fisher, R.S.; Acevedo, C.; Arzimanoglou, A.; Bogacz, A.; Cross, J.H.; Elger, C.E.; Engel, J.J.; Forsgren, L.; French, J.A.; Glynn, M.; et al. ILAE official report: A practical clinical definition of epilepsy. Epilepsia 2014, 55, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Gitai, D.L.; Romcy-Pereira, R.N.; Gitai, L.L.; Leite, J.P.; Garcia-Cairasco, N.; Paço-Larson, M.L. Genes and epilepsy I: Epilepsy and genetic alterations. Rev. Assoc. Med. Bras. 2008, 54, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Chitsaz, A.; Mehvari, J.; Salari, M.; Gholami, F.; Najafi, M.R. A comparative assessment the efficacy of intravenous infusion of sodium valproate and phenytion in the treatment of status epilepticus. Int. J. Prev. Med. 2013, 4, 16–21. [Google Scholar]
- Chbili, C.; Hassine, A.; Laouani, A.; Amor, S.B.; Nouira, M.; Ammou, S.B.; Saguem, S. The relationship between pharmacokinetic parameters of carbamazepine and therapeutic response in epileptic patients. Arch. Med. Sci. 2017, 13, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Park, B.K.; Kitteringham, N.R.; Neill, P.M.O. Metabolism of fluorine-containing drugs. Annu. Rev. Pharmacol. Toxicol. 2001, 41, 443–470. [Google Scholar] [CrossRef] [PubMed]
- Hagmann, W.K. The Many Roles for Fluorine in Medicinal Chemistry. J. Med. Chem. 2008, 51, 4360–4369. [Google Scholar] [CrossRef] [PubMed]
- Kubanik, M.; Holtkamp, H.; Sohnel, T.; Jamieson, S.M.F.; Hartinger, C.G. Impact of the Halogen Substitution Pattern on the Biological Activity of Organoruthenium 8-Hydroxyquinoline Anticancer Agents. Organometallics 2015, 34, 5658–5668. [Google Scholar] [CrossRef]
- Regina, G.L.; Coluccia, A.; Piscitelli, F.; Bergamini, A.; Sinistro, A.; Cavazza, A.; Maga, G.; Samuele, A.; Zanoli, S.; Novellino, E.; et al. Indolyl aryl Sulfones as HIV-1 non-nucleoside reverse transcriptase inhibitors: Role of twohalogen atoms at the indole ring in developing new analogues with improved antiviral activity. J. Med. Chem. 2007, 50, 5034–5038. [Google Scholar] [CrossRef] [PubMed]
- He, X.R.; Bai, Y.J.; Zeng, M.; Zhao, Z.F.; Zhang, Q.; Xu, N.; Qin, F.G.; Wei, X.Y.; Zhao, M.M.; Wu, N.; et al. Anticonvulsant activities of α-asaronol ((E)-3′-hydroxyasarone), an active constituent derived from α-asarone. Pharmacol. Rep. 2017, in press. [Google Scholar] [CrossRef]
- Jeschke, U.; Mylonas, I.; Shabani, N.; Kunert-Keil, C.; Schindlbeck, C.; Gerber, B.; Friese, K. Expression of sialyl lewis X, sialyl lewis A, E-cadherin and cathepsin-D in human breast cancer: Immunohistochemical analysis in mammary carcinoma in situ invasive carcinoma and their lymph node metastasis. Anticancer Res. 2005, 25, 1615–1622. [Google Scholar] [PubMed]
- Yan, M.X.; Mao, H.T.; Liu, Q.; Wang, W.Q.; Li, Y.Q. Elevated levels of serum soluble E-selectin in patients with chronic hepatitis B: Correlation with T lymphocyte subsets, NK cells and liver inflammation. Hepatol. Res. 2006, 35, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Kodera, H.; Nakamura, K.; Osaka, H.; Maegaki, Y.; Haginoya, K.; Mizumoto, S.; Kato, M.; Okamoto, N.; Iai, M.; Kondo, Y.; et al. De Novo Mutations in SLC35A2 Encoding a UDP-Galactose Transporter Cause Early-Onset Epileptic Encephalopathy. Hum. Mutat. 2013, 34, 1708–1714. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.V.; Gan, S.H. Cinnamon: A Multifaceted Medicinal Plant. Evid. Based Complement. Altern. Med. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Masic, A.; Hernandez, A.M.V.; Hazra, S.; Glaser, J.; Holzgrabe, U.; Hazra, B.; Schurigt, U. Cinnamic Acid Bornyl Ester Derivatives from Valeriana wallichii Exhibit Antileishmanial In Vivo Activity in Leishmania major-Infected BALB/c Mice. PLoS ONE 2015, 10, e0142386. [Google Scholar] [CrossRef] [PubMed]
- De, P.; Baltas, M.; Bedos-Belval, F. Cinnamic Acid Derivatives as Anticancer Agends—A Review. Curr. Med. Chem. 2011, 18, 1672–1703. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.Y.; Shang, B.Y.; Li, Y.; Zhen, Y.S. Inhibition of histone deacetylases by trans-cinnamic acid and its antitumor effect against colon cancer xenografts in athymic mice. Mol. Med. Rep. 2016, 13, 4159–4166. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.L.; Wang, Z.H.; Yen, H.F.; Lee, Y.J.; Yin, M.C. Reversal of ethanol-induced hepatotoxicity by cinnamic and syringic acids in mice. Food Chem. Toxicol. 2017, 99, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Zhou, K.; Chen, D.D.; Li, B.; Zhang, B.; Miao, F.; Zhou, L. Bioactivity and structure-activity relationship of cinnamic acid esters and their derivatives as potential antifungal agents for plant protection. PLoS ONE 2017, 12, e0176189. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.; Wei, X.D.; Chen, C.R. 3,4,5-Trimethoxycinnamic acid, one of the constituents of Polygalae Radix exerts anti-seizure effects by modulating GABAAergic systems in mice. J. Pharmcol. Sci. 2015, 131, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Rees, C.R.; Costin, J.M.; Fink, R.C.; McMichael, M.; Fontaine, K.A.; Isern, S.; Michael, S.F. In vitro inhibition of dengue virus entry by p-sulfoxy-cinnamic acid and structurally related combinatorial chemistries. Antivir. Res. 2008, 80, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Gunia, A.; Waszkielewicz, A.M.; Cegla, M.; Marona, H. Preliminary Evaluation of Anticonvulsant Activity of Some Aminoalkanol and Amino Acid Cinnamic Acid Derivatives. Lett. Drug Des. Discov. 2012, 9, 37–43. [Google Scholar] [CrossRef]
- Stevelink, R.; Sanders, M.W.C.B.; Tuinman, M.; Koeleman, B.P.C.; Brilstra, E.H. Epilepsy surgery in patients with genetic refractory epilepsy: A systematic review. Eur. J. Paediatr. Neurol. 2017, 21, e17. [Google Scholar] [CrossRef]
- Ngoh, A.; Parker, A.P.J. New developments in epilepsy management. Paediatr. Child Health 2017, 27, 281–286. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 1–12 are available from the author Yajun Bai. |
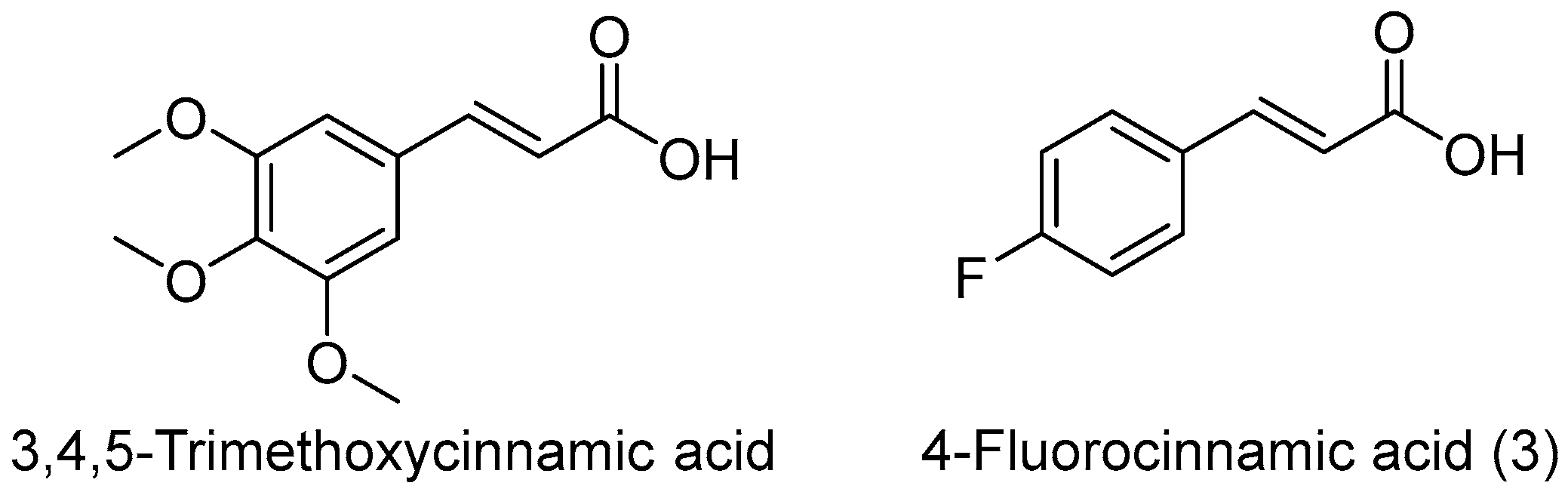

| Compd. | R | m | Sub. Site | MES b | Tox c | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| 0.5 h | 1 h | 2 h | 3 h | 0.5 h | 1 h | 2 h | ||||
| Saline | --- | --- | --- | 0 d/4 e | 0/4 | 0/4 | 0/4 | 0 f/4 e | 0/4 | 0/4 |
| STP g | --- | --- | --- | 3/4 | 2/4 | 1/4 | 0/4 | 1/4 | 0/4 | 0/4 |
| 1 | F | 1 | ortho | 2/4 | 2/4 | 0/4 | 0/4 | 0/4 | 0/4 | 0/4 |
| 2 | F | 1 | meta | 3/4 | 3/4 | 0/4 | 0/4 | 0/4 | 0/4 | 0/4 |
| 3 | F | 1 | para | 4/4 | 3/4 | 1/4 | 0/4 | 0/4 | 0/4 | 0/4 |
| 4 | Cl | 1 | ortho | 2/4 | 2/4 | 0/4 | 0/4 | 0/4 | 0/4 | 0/4 |
| 5 | Cl | 1 | meta | 2/4 | 1/4 | 0/4 | 0/4 | 0/4 | 0/4 | 0/4 |
| 6 | Cl | 1 | para | 3/4 | 2/4 | 1/4 | 0/4 | 0/4 | 0/4 | 0/4 |
| 7 | Br | 1 | ortho | 0/4 | 0/4 | 0/4 | 0/4 | 0/4 | 0/4 | 0/4 |
| 8 | Br | 1 | meta | 0/4 | 0/4 | 0/4 | 0/4 | 0/4 | 0/4 | 0/4 |
| 9 | Br | 1 | para | 0/4 | 0/4 | 0/4 | 0/4 | 0/4 | 0/4 | 0/4 |
| 10 | CF3 | 1 | ortho | 2/4 | 1/4 | 1/4 | 0/4 | 0/4 | 0/4 | 0/4 |
| 11 | CF3 | 1 | meta | 3/4 | 2/4 | 1/4 | 0/4 | 0/4 | 0/4 | 0/4 |
| 12 | CF3 | 1 | para | 3/4 | 3/4 | 1/4 | 0/4 | 0/4 | 0/4 | 0/4 |
| Compd. | Dose (mg/kg) | Latent Time (s) b | CSR (%) c | TSR (%) c | MR (%) c |
|---|---|---|---|---|---|
| Saline | --- | 131.1 ± 21.4 | 100 | 100 | 75 |
| STP | 100 | 219.3 ± 23.75 ** | 100 | 50 | 50 |
| 3 | 100 | 140.4 ± 14.5 | 100 | 100 | 75 |
| 200 | 196.0 ± 58.2 ** | 100 | 75 | 75 | |
| 300 | 280.5 ± 59.2 ** | 100 | 25 | 25 | |
| 6 | 100 | 128.6 ± 17.3 | 100 | 100 | 75 |
| 300 | 160.5 ± 54.4 * | 100 | 50 | 75 | |
| 300 | 214.0 ± 44.7 ** | 100 | 25 | 50 | |
| 12 | 100 | 145.2 ± 18.8 | 100 | 100 | 75 |
| 200 | 181.0 ± 43.4 * | 100 | 75 | 75 | |
| 300 | 206 ± 49.0 ** | 100 | 25 | 75 |
| Compd. | TPE (h) b | MES, ED50 (mg/kg) | PTZ, ED50 (mg/kg) | Tox, TD50 (mg/kg) |
|---|---|---|---|---|
| STP | --- | 240 [9] | 115 [9] | <500 |
| 3 | 0.5 | 47.36 (17.08–67.95) | 245.2 | >500 |
| 6 | 0.5 | 75.72 (56.85–104.67) | >300 | >500 |
| 12 | 0.5 | 70.65 (50.74–121.59) | 285.2 | >500 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cuan, Y.; He, X.; Zhao, Y.; Yang, J.; Bai, Y.; Sun, Y.; Zhang, Q.; Zhao, Z.; Wei, X.; Zheng, X. Anticonvulsant Activity of Halogen-Substituted Cinnamic Acid Derivatives and Their Effects on Glycosylation of PTZ-Induced Chronic Epilepsy in Mice. Molecules 2018, 23, 76. https://doi.org/10.3390/molecules23010076
Cuan Y, He X, Zhao Y, Yang J, Bai Y, Sun Y, Zhang Q, Zhao Z, Wei X, Zheng X. Anticonvulsant Activity of Halogen-Substituted Cinnamic Acid Derivatives and Their Effects on Glycosylation of PTZ-Induced Chronic Epilepsy in Mice. Molecules. 2018; 23(1):76. https://doi.org/10.3390/molecules23010076
Chicago/Turabian StyleCuan, Ye, Xirui He, Yuhui Zhao, Jiajun Yang, Yajun Bai, Yin Sun, Qiang Zhang, Zefeng Zhao, Xiaoyang Wei, and Xiaohui Zheng. 2018. "Anticonvulsant Activity of Halogen-Substituted Cinnamic Acid Derivatives and Their Effects on Glycosylation of PTZ-Induced Chronic Epilepsy in Mice" Molecules 23, no. 1: 76. https://doi.org/10.3390/molecules23010076




