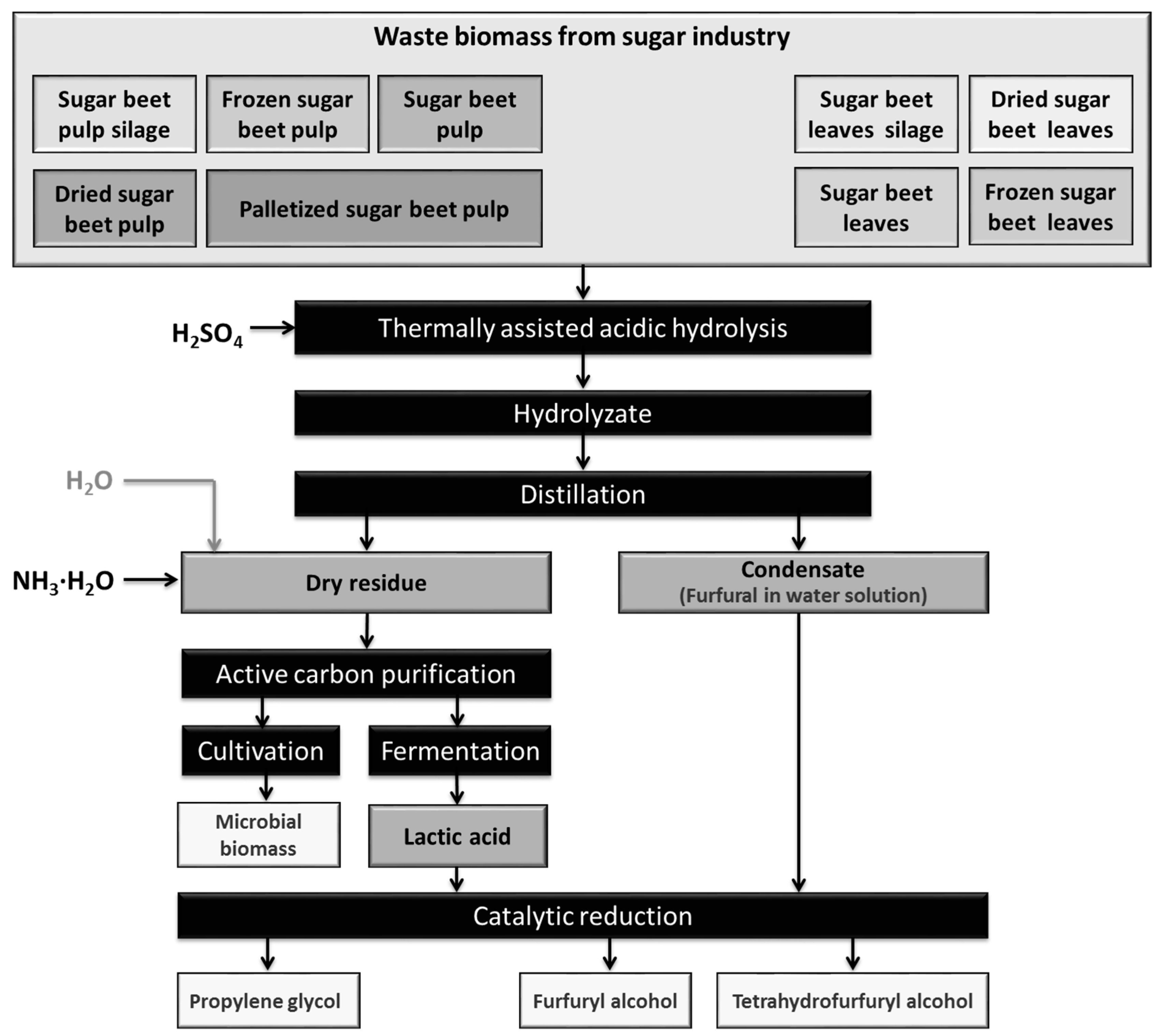
3.1. Dehydration of Pentoses into Furfural and Catalytic Reduction into Furfural Alcohol and Tetrahydrofurfural
Biomass is considered by some to be the only sustainable source of energy and organic carbon for industry, with the potential to displace petroleum in the production of chemicals and liquid transportation fuels [
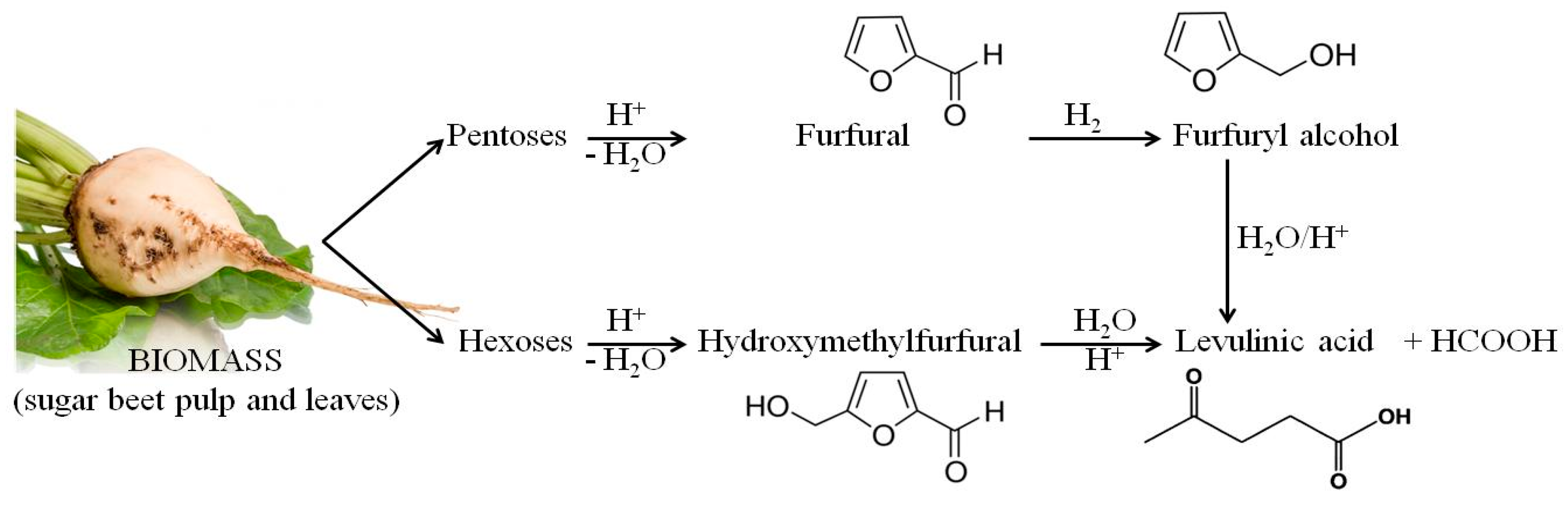
37]. Economical and energy-efficient processes are required for the sustainable production of so-called platform chemical compounds—chemicals that can be easily converted into other valuable chemicals. Furfural, one of the main products of the acid hydrolysis of biomass, is often cited as a chemical platform compound for the production of chemicals and fuels [
38,
39,
40,
41]. Furfural contains both a carbonyl group, which can undergo reduction, oxidation, acylation, acetylation, aldolization, Knoevenagel condensation, decarboxylation and Grignard reactions, and a furan ring, which can undergo hydrogenation, oxidation, alkylation, halogenation, ring opening and nitration [
38,
39,
40,
41]. However, if furfural is to be used as raw material, it must first be separated from the water phase and purified. These processes have a great impact on both the price of furfural and (because they require organic solvents) also on the environment. The development of new aqueous phase processes, such as that presented in this article, is therefore very desirable from both an economic and environmental standpoint. Liquid-phase hydrogenation of furfural over catalysts based on Cu [
42,
43], Ni [
44], on noble metals such as Ru and Rh, or on Pt [
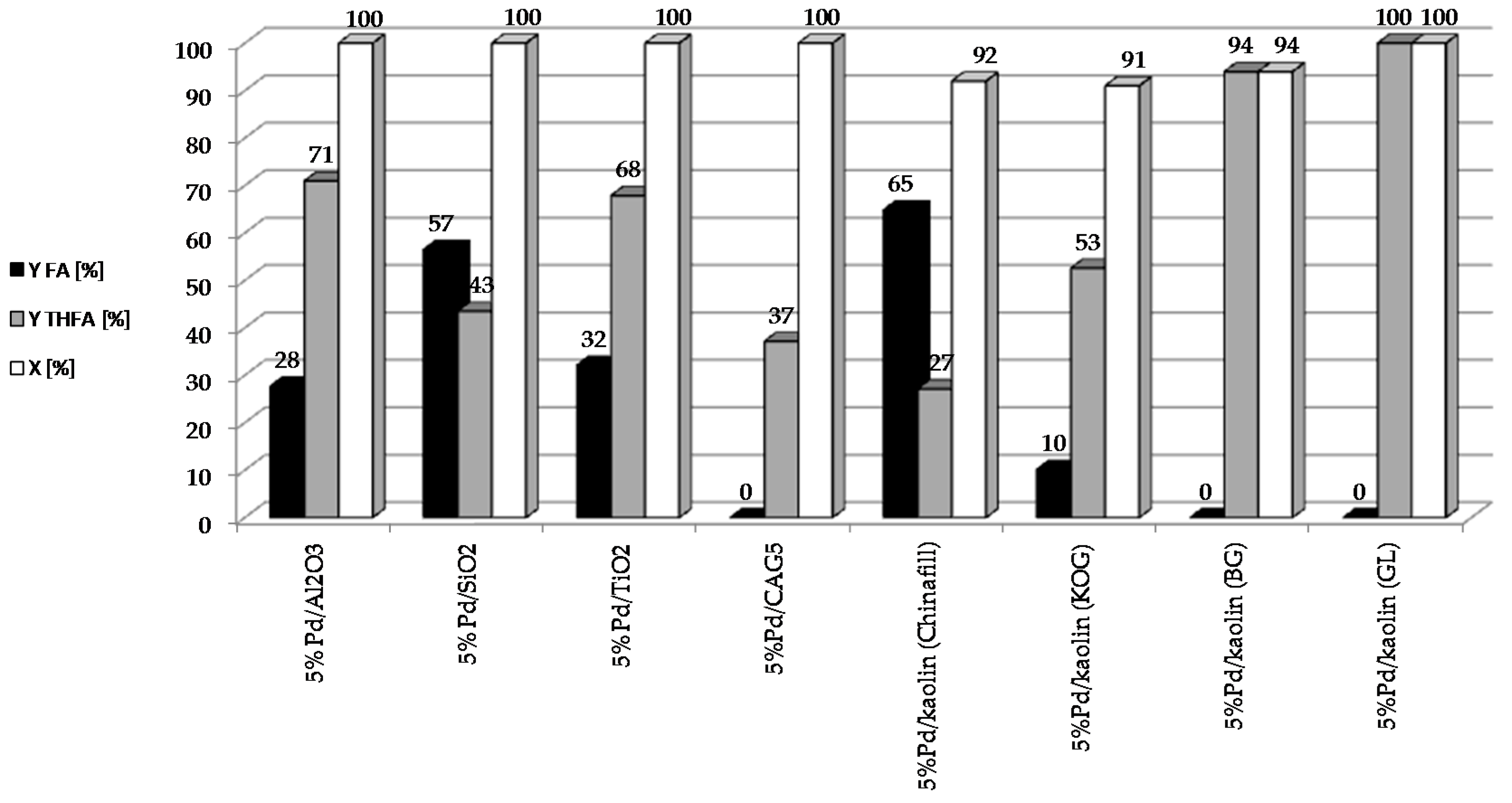
45], can yield valuable products including FA, THFA, 2-MF and 2-MTHF. The influence of the type of support on the catalytic properties of metallic systems for the reduction of furfural requires more comprehensive studies. The most common supports that have been considered for palladium catalysts used in this reaction include simple oxide supports such as SiO
2 [
40], Al
2O
3 [
43] and MgO [
42], or activated carbons [
42]. In our investigation, the process of furfural hydrogenation performed over palladium catalyst supported on various mineral carriers (Al
2O
3, SiO
2, TiO
2, C
AG5, Kaolins). Natural kaolins provide particularly interesting supports. The chemical composition of these clay minerals is Al
2Si
2O
5(OH)
4, in which one tetrahedral sheet of silica (SiO
4) is linked through oxygen atoms to one octahedral sheet of alumina octahedra (AlO
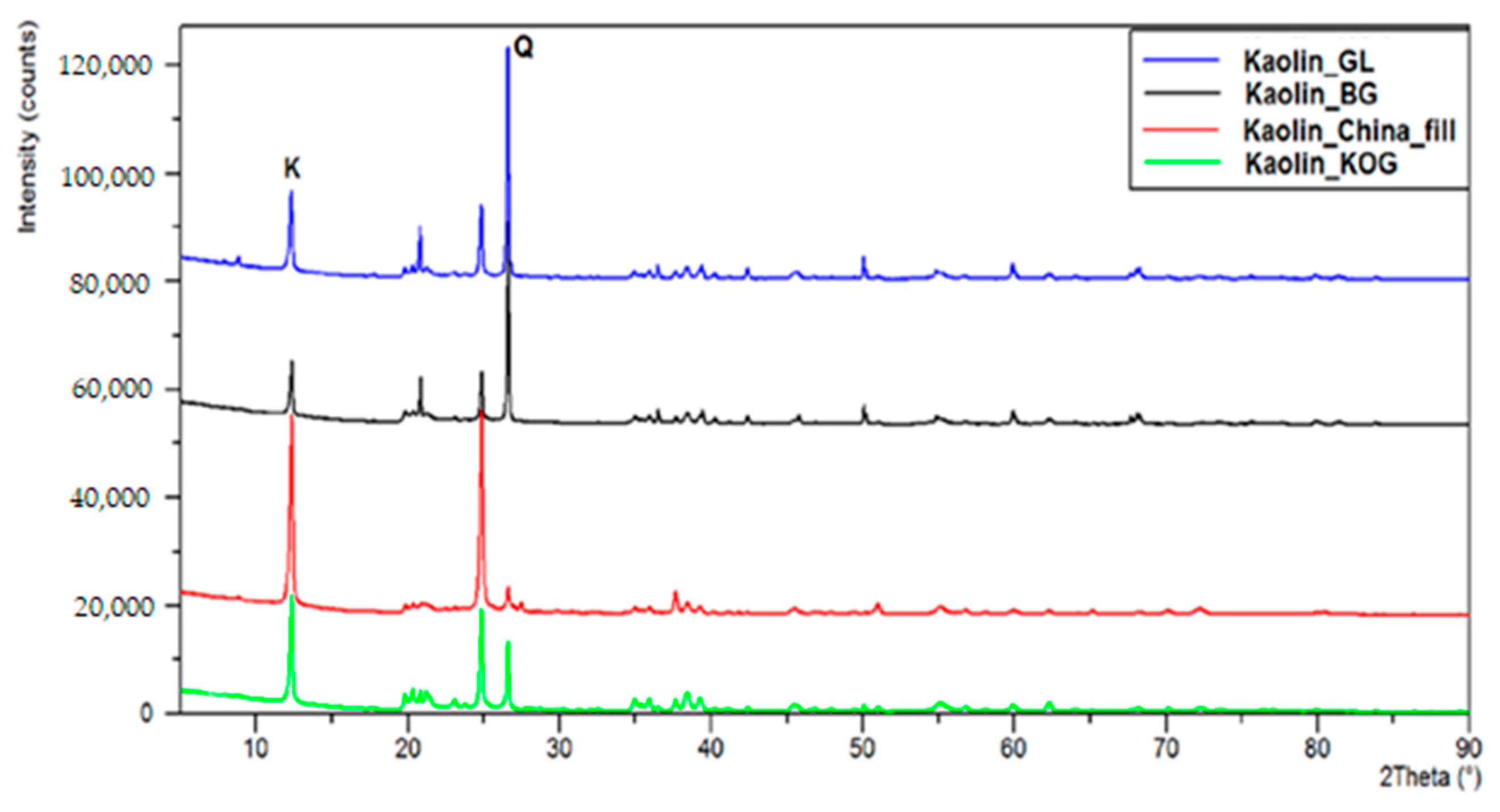
6). We used four kaolin carriers extracted from various mines: Grudzień Las (GL); Biała Góra (BG); the Surmin Mine (KOG); and Chinafill. The carriers differed significantly in terms of phase composition (
Figure 9) as well as surfaces area (
Table 8).
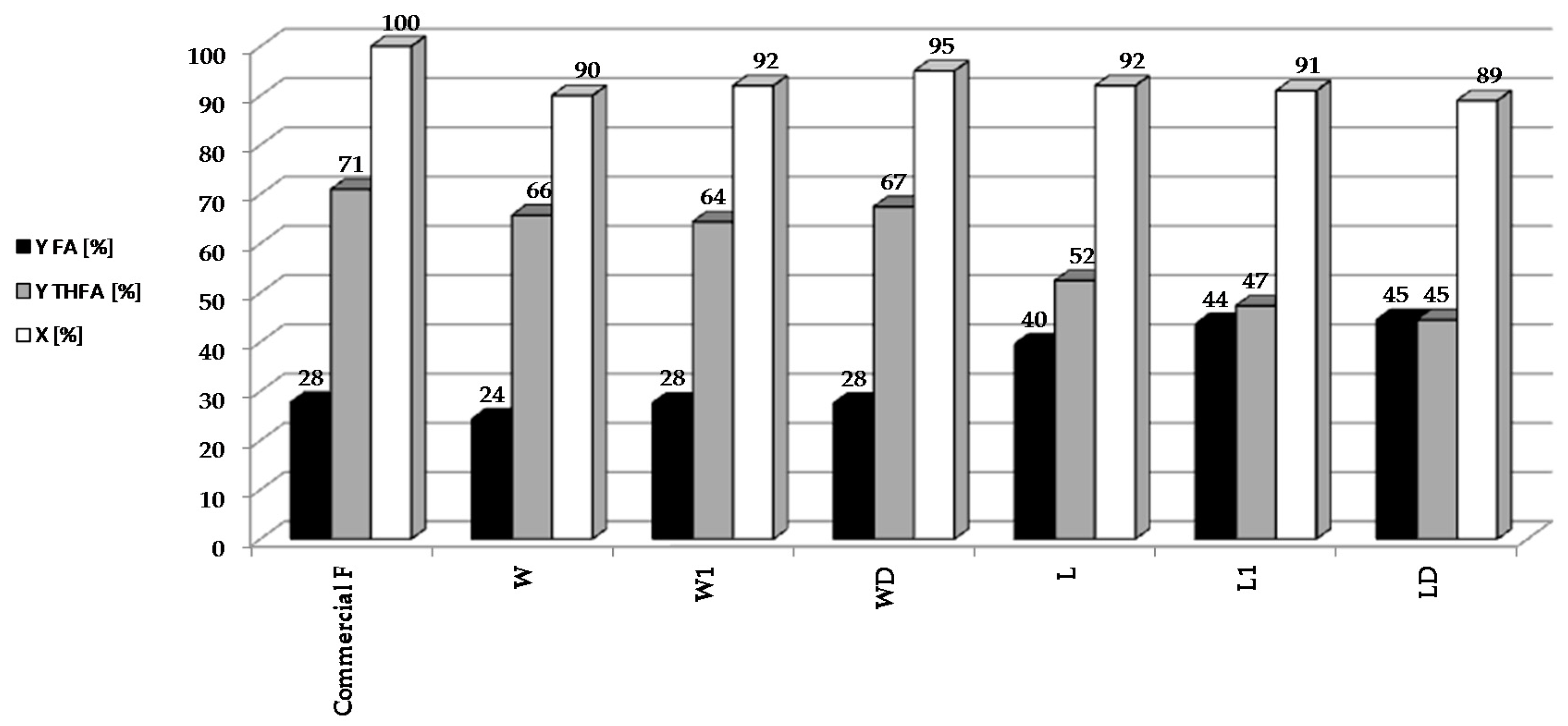
Therefore, it was assumed that the palladium deposited on these systems would be characterized by various degrees of dispersion, and that these systems would exhibit variable catalytic properties. The highest yield of FA was observed in the case of 5%Pd/SiO2, in which the Pd crystallites are larger (diameter approximately 40 nm). In the case of palladium catalysts with smaller Pd crystallites (7–10 nm), THFA was the main product of the reaction. Understanding the nature of this phenomenon is important for the creation of new and stable Pd-based catalysts for the reduction of furfural.
3.2. Utilization of Carbohydrates by Selected Microorganisms
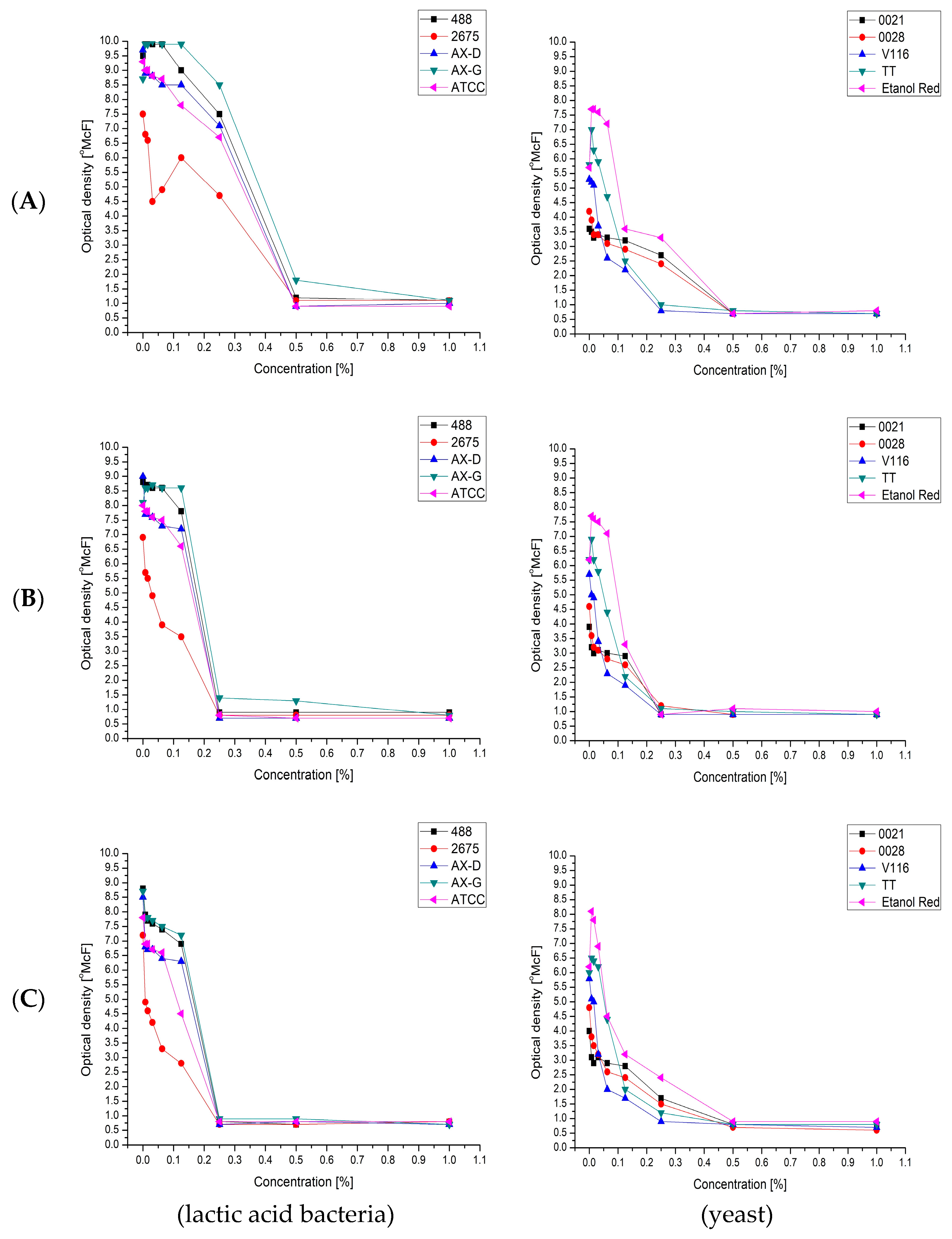
Usually, classical yeasts from the genus Saccharomyces spp. are unsuitable for the assimilation of carbon sources from hydrolysates, since they are unable to ferment pentoses and exhibit low tolerance to alcohols, acids and solvents. Moreover, they are characterized by high sensitivity to both pH changes and cytotoxic compounds, including furfural, 5-(hydroxymethyl)-2-furfural and other organic compounds. The limitations of S. cerevisiae make the development of new industrial fermentation processes very difficult. An additional problem for the simultaneous consumption of pentoses and hexoses is that pentose uptake is inhibited by d-glucose. Progress in improving the utilization of pentose sugars has been slow, as there are few yeasts known to be capable of metabolizing pentose. Therefore, our interest was directed towards using both industrial strains and yeasts belonging to genera other than Saccharomyces, commonly referred to as “non-conventional” yeasts.
The use of non-conventional yeasts may overcome many problems related to the narrow spectrum of carbon sources assimilated by conventional
S. cerevisiae [
46]. Some non-conventional yeast show many uncommon, metabolic features of potential interest to biotechnologists. In
S. cerevisiae, monosaccharides such as glucose, fructose or mannose are transported in a facilitated diffusion process. However, the process of transportation may be different in other yeasts. For example, in
Kluyveromyces spp., glucose transport appears to proceed by facilitated diffusion. When
Candida utilis, the popular “fodder yeast”, is grown at low glucose concentrations, the glucose appears to be transported by proton transport [
47]. Non-conventional yeasts comprise the vast majority of genera and species so far described. Several yeast species have evolved from
S. cerevisiae but possess several unique genes and growth characteristics that make them capable of withstanding different stress conditions [
48]. These exceptional strains are able to utilize various sources of carbon, such as starch, cellulose, raffinose, arabinose, xylose and sugar alcohols. In our study, hydrolysates rich in mannose, arabinose, galactose and raffinose were found to be suitable for use as cultivation media for
Kluyveromyces marxianus and
Candida utilis. These yeasts showed good growth on both beet leaf and beet pulp hydrolysates. The yeast
Candida sp. has been used for many years as a microbe of choice in the production of SCP from media containing carbohydrates.
C. utilis is used with a wide variety of substrates, such as sucrose, ethanol and sulfite-spent liquor. Because of its ability to assimilate pentoses, it can also grow on wood hydrolysates [
49].
K. marxianus can grow on lactose, inulin (a fructose polymer found in some plants) and other simple sugars such as glucose, fructose and sucrose. It is sometimes therefore also grown on molasses [
50].
Dilute-acid hydrolysis is fast and easy to perform, but is hampered by non-selectivity and the formation of by-products. The composition and concentration of inhibitory compounds varies depending on the type of lignocellulosic raw material and how it is pre-treated [
51]. Generally, after pre-treatment of the hemicellulose fraction, hexoses (
d-glucose,
d-galactose,
d-mannose and
d-rhamnose) and pentoses (
d-xylose and
l-arabinose) are obtained [
52,
53]. However, under high temperature and pressure conditions, hexoses and pentoses may be degraded to 5-(hydroxymethyl)-2-furfural and furfural. In general, while furfural and 5-hydroxymethylfurfural can originate from the dehydration of pentoses and hexoses, vanillin originates from degradation of lignin polymers. Thus, in the case of relatively sugar-rich hydrolysates obtained from sugar beet pulp, the potential inhibitors of microbial growth are mainly furfural, HMF and levulinic acid, while vanillin tends to be present mainly in beet leaves pulp. However, due to the fact that in both cases it is possible to obtain a mixture of compounds inhibiting microbial growth it is justified to examine the influence of these inhibitors on the tested microorganisms [
54]. Furthermore, furans and phenol derivatives such as vanillin can be formed in low concentration, what with the presence of other compounds makes them difficult to analyze. Although their presence is low in the medium, they may be highly toxic for the growth and fermentation of microorganisms [
55]. Therefore, it seems to be very important to determine the susceptibility of the microorganisms to these compounds.
Phenolic compounds are generated from the partial breakdown of lignin and have also been reported as forming during the degradation of carbohydrates [
52]. The harmful effects of these compounds, even at low concentrations, have been confirmed. The RNA and DNA, proteins and membranes of yeast cells are particularly sensitive [
56,
57]. Phenolic compounds partition into biological membranes, causing loss of integrity and affecting their ability to serve as selective barriers and enzyme matrices. Vanillin constitutes a large fraction of the phenolic monomers in biomass hydrolysates. Furfural has been shown to reduce the specific growth rate and the cell-mass yield of yeasts [
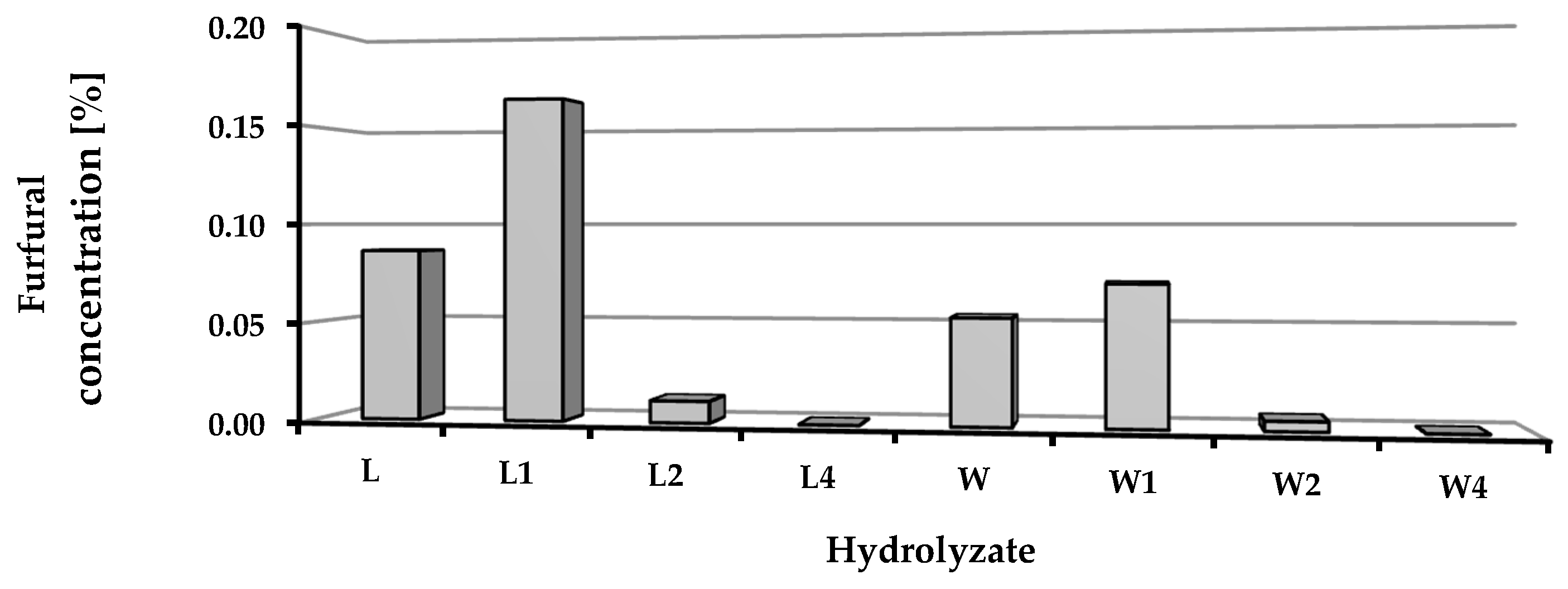
52]. In our research, the level of furfural in the hydrolysates following distillation did not exceed 0.16%. Despite this fact, an inhibitory effect was observed in some cultures. This can be explained by the synergistic action of several inhibitors. Combinations of different by-products can have synergistic inhibitory effects on the growth of yeast strains [
58]. Each of the tested strains was found to be sensitive to all three investigated compounds. Furfural has been shown to have a negative impact on yeast growth (observed as a decrease in the specific growth rate). Its action in the presence of mixed microbial inhibitors can be greater than that of any of the individual compounds [
52]. Most studies on the toxicity of by-products have focused on ethanol-producing yeasts. The toxicity of by-products on lactic acid producing strains has received less attention. In research conducted by van der Pol et al., the inhibition of lactic acid bacteria growth showed large inter-species variation. This variation was confirmed in our study [
58].
Removing toxic compounds from the fermentation medium is usually very expensive. It is therefore more practical to use furan-tolerant yeast strains. According to the literature, some non-conventional yeast species, especially
Pichia spp. and
Candida spp., show good tolerance to furfural and its derivatives. For example, the resistance of
P. kudriavzevii to hydroxymethyl can be up to 7 g/L [
59].
Candida utilis was confirmed in our study to exhibit good growth in sugar beet hydrolysates.
3.3. Waste Biomass from the Sugar Industry as a Potential Source of Platform Chemicals (Furfural, Lactic Acid) and Biotechnological Media
Waste lignocellulose residues, including waste biomass from the sugar beet industry, is usually hydrolyzed for the purposes of the production of second generation fuel, mainly bioethanol [
60,
61]. Here, we present an alternative solution, providing as end products furfural, furfuryl alcohol, tetrafurfuryl alcohol, lactic acid and propylene glycol.
Hydrolysis involves the treatment of lignocellulose at high temperatures, under acidic conditions. It leads to the formation and liberation of a range of compounds. When hemicellulose is degraded, xylose, mannose, acetic acid, galactose and glucose are liberated. Cellulose is hydrolyzed to glucose. At high temperatures and under pressure, xylose is further degraded to furfural [
52]. The conditions of hydrolysis—residence time, temperature and acid concentration—have a strong effect on the spectrum of products. As the values for these parameters increase, the yield of fermentable sugars also increases, to a maximum point above which it starts to decrease. The decrease in the yield of monosaccharides coincides with the maximum concentrations of furfural [
62]. This requires a proper balance to be found between the production of monosaccharides and of furfural.
The bio-based media were next used for the production of propylene glycol from the lactic acid biosynthesized in the hydrolysate. The process of bioconversion of sugar beet pulp carbohydrates into lactic acid has been described in several previous studies [
33,
34,
36]. Lactic acid is one of the primary platform chemicals, and can be used to synthesize a wide variety of useful products. Its reduced derivatives, such as propylene glycol, can be used as green chemicals in industrial applications [
34]. The reduction of lactic acid to propylene glycol was conducted over commercial ruthenium catalysts under mild temperature conditions (130 °C) and H
2 pressure (35 atm). In our earlier studies, these conditions had been found to be optimal [
33,
36]. As can be seen in
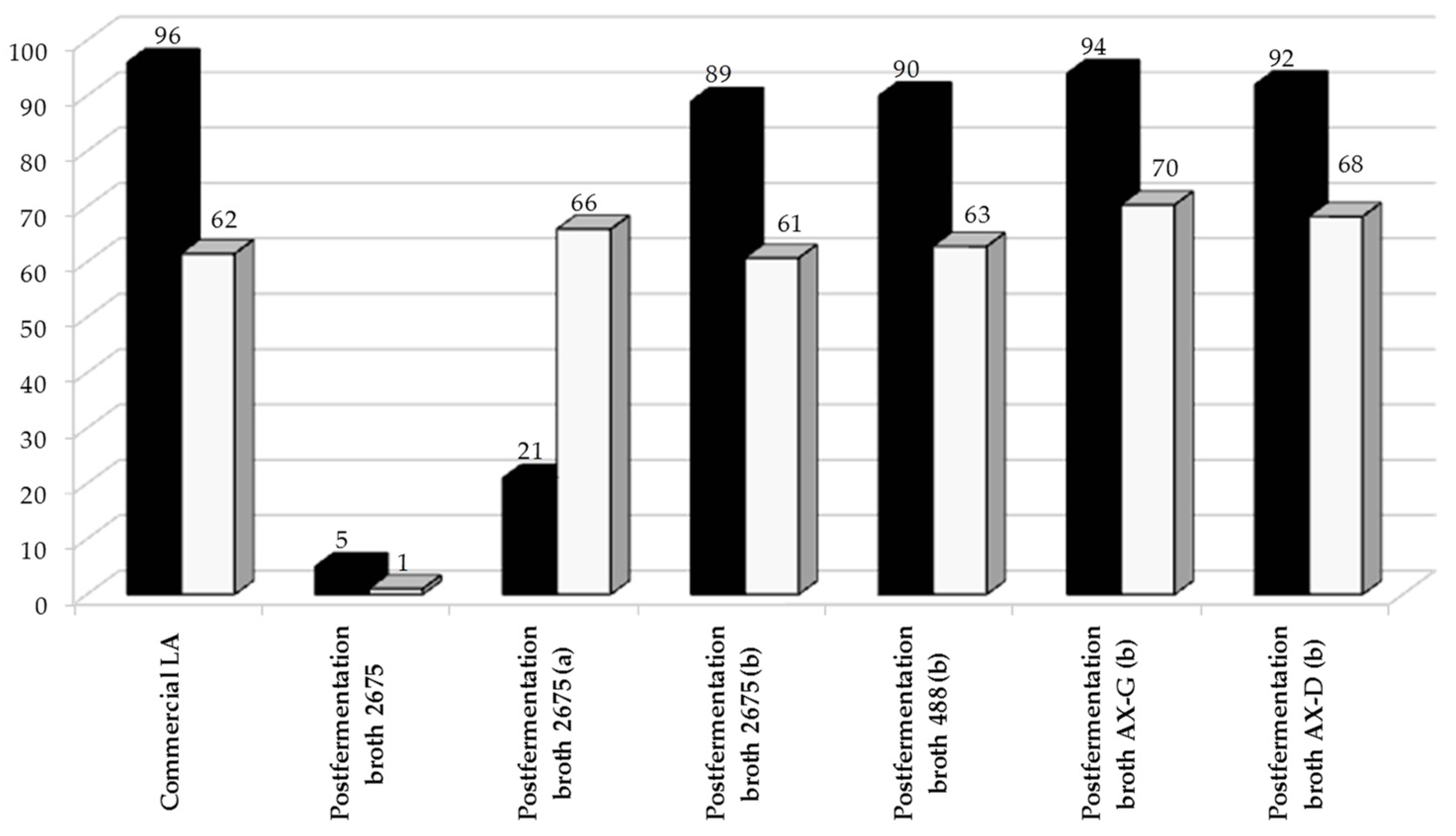
Figure 7, the lactic acid obtained from the sugar beet pulp hydrolysates can be converted with good yields into propylene glycol. However, this requires initial purification of the media, since the sulfide-containing amino acids present in post-fermentation broths irreversibly poison the ruthenium catalysts, whereas non-sulfurous amino acids only partially and reversibly poison the catalysts [
33]. In the conventional biological method of pure lactic acid production, the separation and purification stages account for up to 50% of production costs. In our study, the post-fermentation broth was purified using active carbon. A diluted water solution of the lactic acid was then reduced over a Ru-based supported catalyst. The yield of propylene glycol obtained in this way was satisfactory, and the water solutions of propylene glycol obtained could be used in concentrated form as a component in anti-freeze. The process of propylene glycol production described here could be of interest to industry, but its disadvantage is that it is material intensive (requiring 1.25 kg LA/1.00 kg PG). It is therefore necessary to the reduce costs and increase efficiency. The use of less pure LA as a feedstock is one possibility.
Our research clearly shows that the hydrolysates obtained from waste products of the sugar industry provide a good carbon source for the selected yeasts, especially the non-conventional yeast strains. Production systems exploiting some non-
Saccharomyces yeasts have a distinct advantage, in that they are non-pathogenic and have received the “generally recognized as safe” (GRAS) designation from the FDA [
35,
63]. Yeast species other than
Saccharomyces spp. use complex substrates, and exhibit features such as thermotolerance and tolerance to the presence of chemical inhibitors, making them particularly useful in industrial processes. For example, its broad enzymatic activity and fermentation ability in the presence of high concentrations of saccharides makes the yeast
K. marxianus a good material for various biotechnological processes [
64,
65].
K. marxianus cultures have also been grown on sugar beet leaf hydrolysates, and
Candida tropicalis on media derived from both sugar beet pulp and sugar beet leaf hydrolysates, with positive results [
66,
67]. The advantages of the solution presented here include not only the value of the end products but also the fact it enables virtually “zero-waste” production. Currently, the sugar industry produces large amounts of waste biomass, only part of which can be only used without processing, as components in animal feed [
68,
69].