Zeamide, a Glycosylinositol Phosphorylceramide with the Novel Core Arap(1β→6)Ins Motif from the Marine Sponge Svenzea zeai
Abstract
:1. Introduction
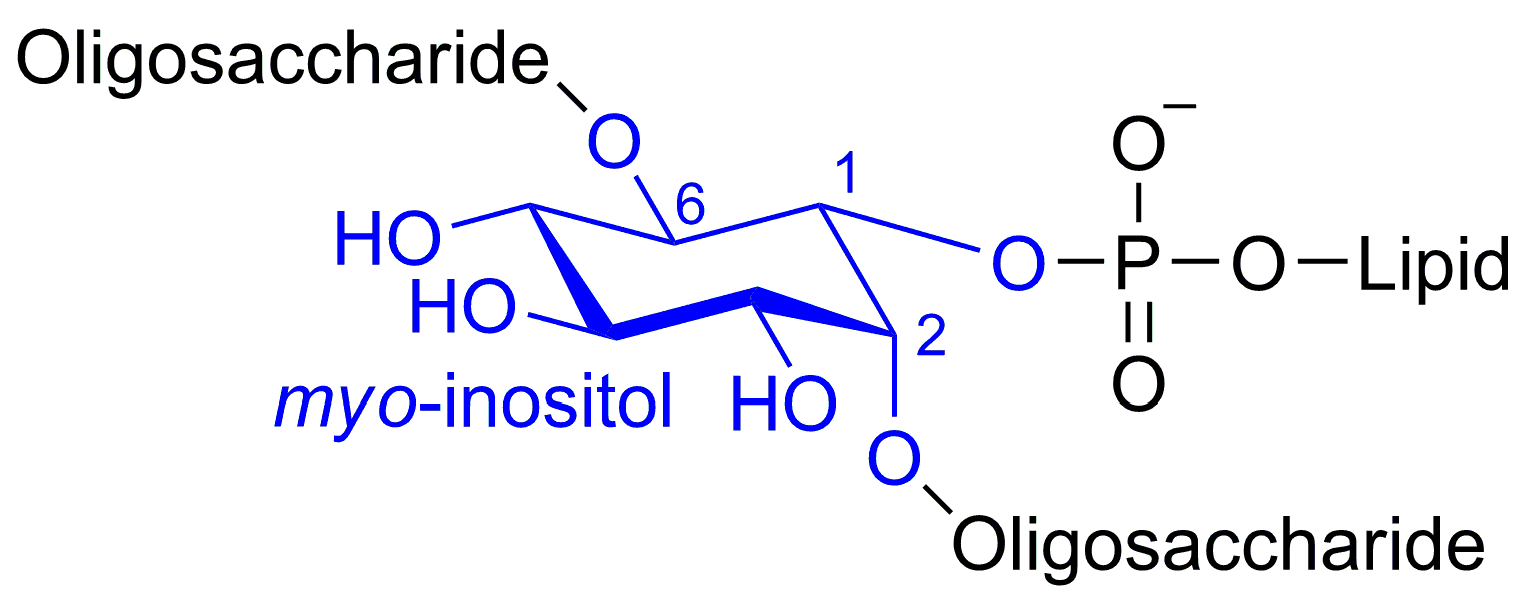
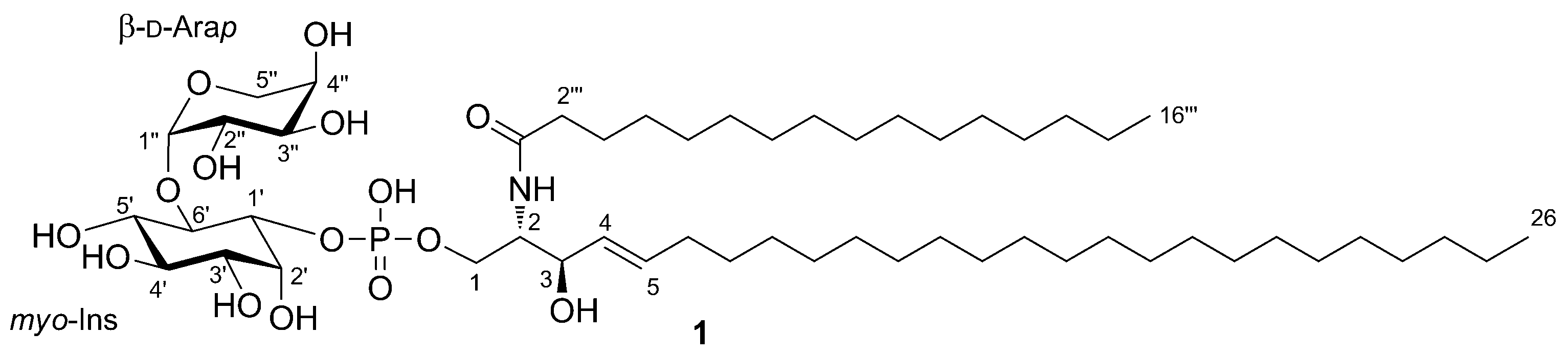
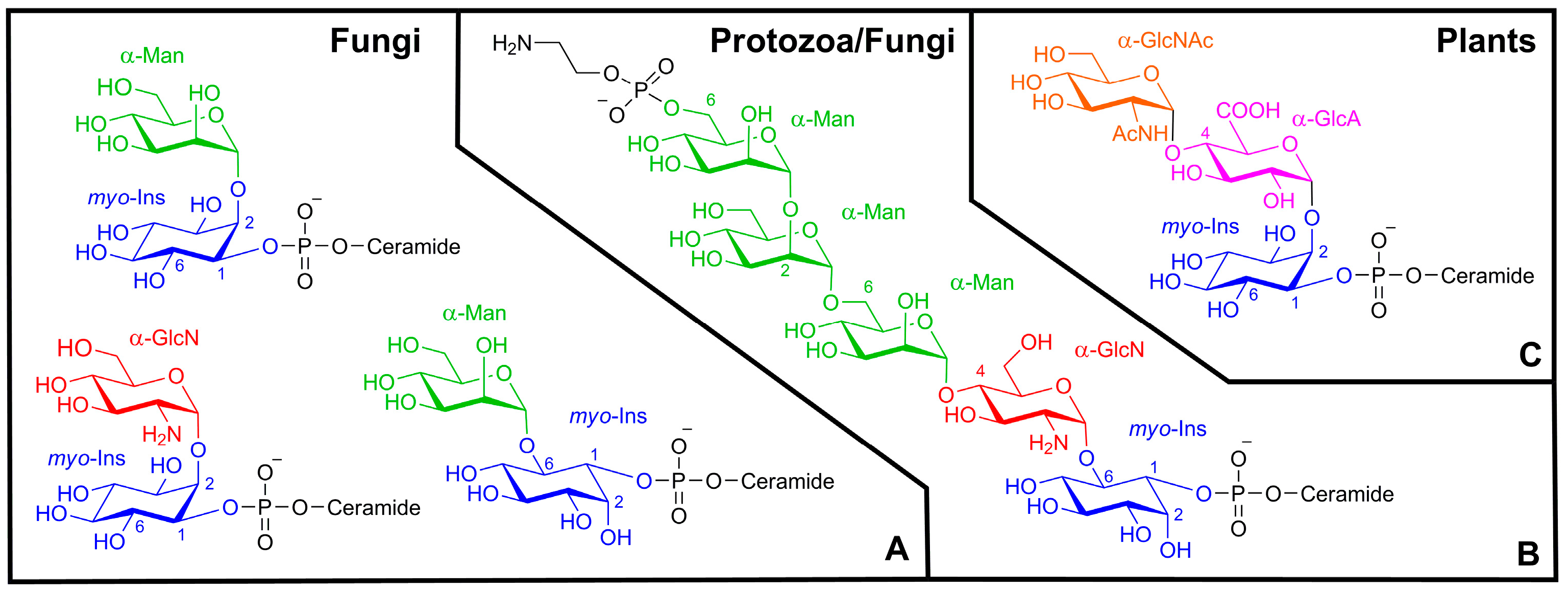
2. Results and Discussion
3. Materials and Methods
3.1. General Experimental Procedure
3.2. Collection, Extraction, and Isolation
3.3. Zeamide (1)
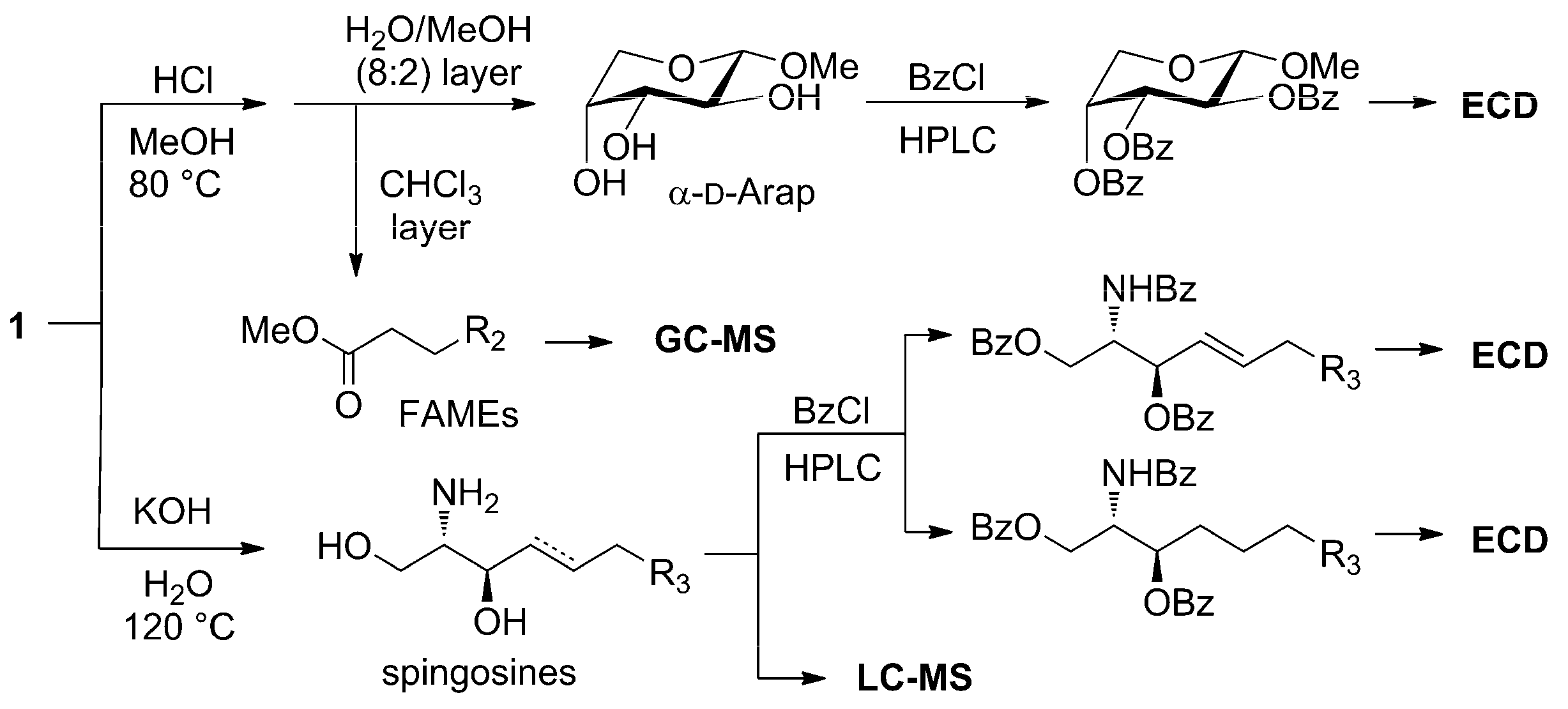
3.4. Methanolysis of Zeamide (1)
3.5. Hydrolysis of Zeamide (1)
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Paulick, M.G.; Bertozzi, C.R. The Glycosylphosphatidylinositol Anchor: A Complex Membrane-Anchoring Structure for Proteins. Biochemistry 2008, 47, 6991–7000. [Google Scholar] [CrossRef] [PubMed]
- Murata, D.; Nomura, K.H.; Dejima, K.; Mizuguchi, S.; Kawasaki, N.; Matsuishi-Nakajima, Y.; Ito, S.; Gengyo-Ando, K.; Kage-Nakadai, E.; Mitani, S.; et al. GPI-anchor synthesis is indispensable for the germline development of the nematode Caenorhabditis elegans. Mol. Biol. Cell 2012, 23, 982–995. [Google Scholar] [CrossRef] [PubMed]
- Bundy, M.G.R.; Kosentka, P.Z.; Willet, A.H.; Zhang, L.; Miller, E.; Shpak, E.D. A Mutation in the Catalytic Subunit of the Glycosylphosphatidylinositol Transamidase Disrupts Growth, Fertility, and Stomata Formation. Plant Physiol. 2016, 171, 974–985. [Google Scholar] [CrossRef] [PubMed]
- Martin, K.L.; Smith, T.K. Phosphatidylinositol synthesis is essential in bloodstream form Trypanosoma brucei. Biochem. J. 2006, 396, 287–295. [Google Scholar] [CrossRef] [PubMed]
- It must be noted in this respect that GIPCs are quite difficult to study because of their unfavorable solubility properties, and generally require specific extraction procedures (see also ref. [10].). Therefore, it is likely that out present knowledge on the distribution of GIPCs in living organisms is biased by the methods used for their isolation and the interests of the researchers performing the studies.
- Gronnier, J.; Germain, V.; Gouguet, P.; Cacas, J.-L.; Mongrand, S. GIPC: Glycosyl Inositol Phospho Ceramides, the major sphingolipids on earth. Plant Signal. Behav. 2016, 11, e1152438. [Google Scholar] [CrossRef] [PubMed]
- Mayor, S.; Riezman, H. Sorting GPI-anchored proteins. Nat. Rev. Mol. Cell Biol. 2004, 5, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, M.A.J.; Kinoshita, T.; Hart, G.W. Glycosyl Phosphatidylinositol Anchors. In Essentials of Glycobiology, 2nd ed.; Varki, A., Cummings, R.D., Esko, J.D., Eds.; Cold Spring Harbor Laboratory Press: Laurel Hollow, NY, USA, 2009; Chapter 11; Available online: https://www.ncbi.nlm.nih.gov/books/NBK1966/ (accessed on 1 September 2017).
- Guimarães, L.L.; Toledo, M.S.; Ferreira, F.A.S.; Straus, A.H.; Takahashi, H.K. Structural diversity and biological significance of glycosphingolipids in pathogenic and opportunistic fungi. Front. Cell. Infect. Microbiol. 2014, 4, 138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cacas, J.L.; Buré, C.; Furt, F.; Maalouf, L.P.; Badoc, A.; Cluzet, S.; Schmitter, J.M.; Antajan, E.; Mongrand, S. Biochemical survey of the polar head of plant glycosylinositolphosphoceramides unravels broad diversity. Phytochemistry 2013, 96, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, T.C.Y.; Kaul, K.; Laine, R.A.; Lester, R.L. Structure of a major glycophosphoceramide from tobacco leaves, PSL-I: 2-deoxy-2-acetamido-d-glucopyranosyl(α1→4)-d-glucuronopyranosyl (α1→2)myoinositol-1-O-phosphoceramide. Biochemistry 1978, 17, 3575–3581. [Google Scholar] [CrossRef] [PubMed]
- Costantino, V.; Fattorusso, E.; Imperatore, C.; Mangoni, A. J-Coupling Analysis for Stereochemical Assignments in Furanosides: Structure Elucidation of Vesparioside B, a Glycosphingolipid from the Marine Sponge Spheciospongia Vesparia. J. Org. Chem. 2008, 73, 6158–6165. [Google Scholar] [CrossRef] [PubMed]
- Costantino, V.; Fattorusso, E.; Mangoni, A.; Di Rosa, M.; Ianaro, A. Glycolipids from Sponges. 6. Plakoside A and B, Two Unique Prenylated Glycosphingolipids with Immunosuppressive Activity from the Marine Sponge Plakortis Simplex. J. Am. Chem. Soc. 1997, 119, 12465–12470. [Google Scholar] [CrossRef]
- Costantino, V.; Fattorusso, E.; Imperatore, C.; Mangoni, A. Ectyoceramide, the First Natural Hexofuranosylceramide from the Marine Sponge Ectyoplasia Ferox. Eur. J. Org. Chem. 2003, 1433–1437. [Google Scholar] [CrossRef]
- Costantino, V.; Fattorusso, E.; Imperatore, C.; Mangoni, A. Isocrasserides, Novel Glycolipids with a Five-Membered Cyclitol Widely Distributed in Marine Sponges. J. Nat. Prod. 2002, 65, 883–886. [Google Scholar] [CrossRef] [PubMed]
- Costantino, V.; Fattorusso, E.; Imperatore, C.; Mangoni, A. Plaxyloside from the Marine Sponge Plakortis Simplex: An Improved Strategy for NMR Structural Studies of Carbohydrate Chains. Eur. J. Org. Chem. 2001, 4457–4462. [Google Scholar] [CrossRef]
- Costantino, V.; Fattorusso, E.; Mangoni, A.; Di Rosa, M.; Ianaro, A. Glycolipids from Sponges. VII.1 Simplexides, Novel Immunosuppressive Glycolipids from the Caribbean Sponge Plakortis Simplex. Bioorg. Med. Chem. Lett. 1999, 9, 271–276. [Google Scholar] [CrossRef]
- Barbieri, L.; Costantino, V.; Fattorusso, E.; Mangoni, A. Glycolipids from Sponges. Part 16. 1 Discoside, a Rare myo-Inositol-Containing Glycolipid from the Caribbean Sponge Discodermia Dissoluta. J. Nat. Prod. 2005, 68, 1527–1530. [Google Scholar] [CrossRef] [PubMed]
- Nigou, J.; Gilleron, M.; Puzo, G. Lipoarabinomannans: From structure to biosynthesis. Biochimie 2003, 85, 153–166. [Google Scholar] [CrossRef]
- Costantino, V.; Della Sala, G.; Mangoni, A.; Perinu, C.; Teta, R. Blurring the Boundary between Bio- and Geohopanoids: Plakohopanoid, a C 32 Biohopanoid Ester from Plakortis Cf. Lita. Eur. J. Org. Chem. 2012, 5171–5176. [Google Scholar] [CrossRef]
- Lamoral-Theys, D.; Fattorusso, E.; Mangoni, A.; Perinu, C.; Kiss, R.; Costantino, V. Evaluation of the Antiproliferative Activity of Diterpene Isonitriles from the Sponge Pseudoaxinella Flava in Apoptosis-Sensitive and Apoptosis-Resistant Cancer Cell Lines. J. Nat. Prod. 2011, 74, 2299–2303. [Google Scholar] [CrossRef] [PubMed]
- Costantino, V.; Fattorusso, E.; Mangoni, A. Bioactive Compounds from Natural Sources; Tringali, C., Ed.; Taylor & Francis: London, UK, 2001; Chapter 14; pp. 555–576. [Google Scholar]
- Costantino, V.; Fattorusso, E.; Imperatore, C.; Mangoni, A. Vesparioside from the Marine Sponge Spheciospongia vesparia, the First Diglycosylceramide with a Pentose Sugar Residue. Eur. J. Org. Chem. 2005, 2, 368–373. [Google Scholar] [CrossRef]
- Munesada, K.; Yuasa, M.; Suga, T. Cerebrosides of frog brain. Structure of the ceramide part of the cerebrosides. J. Chem. Soc. Perkin Trans. 1 1991, 189–194. [Google Scholar] [CrossRef]
- Lee, O.O.; Chui, P.Y.; Wong, Y.H.; Pawlik, J.R.; Qian, P.-Y. Evidence for Vertical Transmission of Bacterial Symbionts from Adult to Embryo in the Caribbean Sponge Svenzea zeai. Appl. Environ. Microbiol. 2009, 75, 6147–6156. [Google Scholar] [CrossRef] [PubMed]
- Costantino, V.; Fattorusso, E.; Imperatore, C.; Mangoni, A.; Teta, R. Amphiceramide A and B, novel glycosphingolipids from the marine sponge Amphimedon compressa. Eur. J. Org. Chem. 2009, 13, 2112–2119. [Google Scholar] [CrossRef]
- Wilson, M.C.; Mori, T.; Rückert, C.; Uria, A.R.; Helf, M.J.; Takada, K.; Gernert, C.; Steffens, U.A.E.; Heycke, N.; Schmitt, S.; et al. An environmental bacterial taxon with a large and distinct metabolic repertoire. Nature 2014, 506, 58–62. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hochmuth, T.; Niederkruger, H.; Gernert, C.; Siegl, A.; Taudien, S.; Platzer, M.; Crews, P.; Hentschel, U.; Piel, J. Linking chemical and microbial diversity in marine sponges: Possible role for poribacteria as producers of methyl-branched fatty acids. ChemBioChem 2010, 11, 2572–2578. [Google Scholar] [CrossRef] [PubMed]
- Della Sala, G.; Hochmuth, T.; Teta, R.; Costantino, V.; Mangoni, A. Polyketide synthases in the microbiome of the marine sponge Plakortis halichondrioides: A metagenomic update. Mar. Drugs 2014, 12, 5425–5440. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Sample of compound 1 is available from the authors. |
| Position | δC, Type b | δH, mult ( J in Hz) c | HMBC d | |
|---|---|---|---|---|
| 1 | 65.7, CH2 | a | 4.24, ddd (10.9, 7.2, 4.2) | |
| b | 3.96, ddd (10.9, 6.4, 2.7) | |||
| 2 | 55.5, CH | 3.91, ddd (9.0, 4.2, 2.7) | 1‴ | |
| 3 | 72.5, CH | 4.10, t (8.3) | 1, 2, 4, 5 | |
| 4 | 131.2, CH | 5.45, dd (15.3, 7.7) | 3, 6 | |
| 5 | 135.0, CH | 5.71, ddd (15.3, 6.9, 6.9) | 3, 6, 7 | |
| 6 | 33.5, CH2 | 2.02, m | 4, 5, 7 | |
| 7 | 30.5, CH2 | 1.37, m | ||
| 1′ | 76.5, CH | 4.01, ddd (9.9, 8.0, 2.7) | 6′ | |
| 2′ | 72.9, CH | 4.27, t (2.7) | 1′, 3′, 4′, 6′ | |
| 3′ | 72.9, CH | 3.36, dd (9.8, 2.7) | 4′ | |
| 4′ | 74.1, CH | 3.63, t (9.5) | 2′, 3′, 5′ | |
| 5′ | 77.3, CH | 3.41, t (9.3) | 4′, 6′ | |
| 6′ | 78.9, CH | 3.91, t (9.6) | 4′, 5′, 1″ | |
| 1′′ | 101.5, CH | 5.36, d (3.7) | 6′, 3″, 5″ | |
| 2″ | 71.1, CH | 3.76, dd (9.9, 3.7) | ||
| 3″ | 70.8, CH | 3.91, dd (9.9, 3.7) | ||
| 4″ | 71.1, CH | 3.88, br. s | 3″ | |
| 5″ | 64.9, CH | a | 4.36, br. d (12.7) | 1″, 3″ |
| b | 3.54, dd (12.7, 2.2) | 1″, 3″ | ||
| 1‴ | 175.9, C | |||
| 2‴ | 37.4, CH2 | 2.21, t (7.6) | 1‴, 3‴ | |
| 3‴ | 27.2, CH2 | 1.58 | 1‴, 2‴ |
| Fatty Acid Methyl Ester | |
|---|---|
| methyl 13-methyltetradecanoate ( iso-C15) | 22.6% |
| methyl 14-methylpentadecanoate ( iso-C16) | 21.0% |
| methyl hexadecanoate ( n-C16) | 25.4% |
| methyl 10-methylhexadecanoate (10Me-C17) | 12.9% |
| methyl 15-methylhexadecanoate ( iso-C17) | 14.2% |
| methyl octadecanoate ( n-C18) | 3.9% |
| Sphingosine | |
| (2 S,3R,4E)-2-amino-4-tetracosene-1,3-diol (C24 d-erythro-sphingosine, d24:1) | 3.5% |
| (2 S,3R,4E)-2-amino-4-pentacosene-1,3-diol (C25 d-erythro-sphingosine, d25:1) | 2.7% |
| (2 S,3R,4E)-2-amino-4-hexacosene-1,3-diol (C26 d-erythro-sphingosine, d26:1) | 62.1% |
| (2 S,3R,4E)-X-methyl-2-amino-4-hexacosene-1,3-diol (branched d27:1) | 3.4% |
| (2 S,3R,4E)-2-amino-4-heptacosene-1,3-diol (C27 d-erythro-sphingosine, d27:1) | 4.5% |
| (2 S,3R,4E)-2-amino-4-octacosene-1,3-diol (C28 d-erythro-sphingosine, d28:1) | 5.2% |
| Total unsaturated sphingosines | 81.4% |
| (2 S,3R)-2-amino-1,3-tetracosenediol (C24 d-erythro-dihydrosphingosine, d24:0) | 1.8% |
| (2 S,3R)-X-methyl-2-amino-1,3-tetracosenediol (branched d25:0) | 1.1% |
| (2 S,3R)-2-amino-1,3-pentacosenediol (C25 d-erythro-dihydrosphingosine, d25:0) | 1.3% |
| (2 S,3R)-2-amino-1,3-hexacosenediol (C26 d-erythro-dihydrosphingosine, d26:0) | 11.4% |
| (2 S,3R)-2-amino-1,3-heptacosenediol (C27 d-erythro-dihydrosphingosine, d27:0) | 1.8% |
| (2 S,3R)-2-amino-1,3-octacosenediol (C28 d-erythro-dihydrosphingosine, d28:0) | 1.2% |
| Total saturated sphingosines | 18.6% |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Della Sala, G.; Teta, R.; Esposito, G.; Pawlik, J.R.; Mangoni, A.; Costantino, V. Zeamide, a Glycosylinositol Phosphorylceramide with the Novel Core Arap(1β→6)Ins Motif from the Marine Sponge Svenzea zeai. Molecules 2017, 22, 1455. https://doi.org/10.3390/molecules22091455
Della Sala G, Teta R, Esposito G, Pawlik JR, Mangoni A, Costantino V. Zeamide, a Glycosylinositol Phosphorylceramide with the Novel Core Arap(1β→6)Ins Motif from the Marine Sponge Svenzea zeai. Molecules. 2017; 22(9):1455. https://doi.org/10.3390/molecules22091455
Chicago/Turabian StyleDella Sala, Gerardo, Roberta Teta, Germana Esposito, Joseph R. Pawlik, Alfonso Mangoni, and Valeria Costantino. 2017. "Zeamide, a Glycosylinositol Phosphorylceramide with the Novel Core Arap(1β→6)Ins Motif from the Marine Sponge Svenzea zeai" Molecules 22, no. 9: 1455. https://doi.org/10.3390/molecules22091455







